Abstract
Embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) are considered the most powerful in terms of differentiating into three-germ-layer cells. However, maintaining self-renewing ESCs and iPSCs in vitro requires leukemia-induced factor (LIF), an expensive reagent. Here we describe a less expensive compound that may serve as a LIF substitute—salvianolic acid B (Sal B), a Salvia miltiorrhiza extract. We found that Sal B is capable of upregulating Oct4 and Sox2, two genes considered important for the maintenance of ESC pluripotency. Our MTT data indicate that instead of triggering cell death, Sal B induced cell proliferation, especially at optimum concentrations of 0.01 nM and 0.1 nM. Other results indicate that compared to non-LIF controls, Sal B-treated ESCs expressed higher levels of several stem cell markers while still maintaining differentiation into three-germ-layer cells after six passages. Further, we found that Sal B triggers the Jak2–Stat3 and EGFR–ERK1/2 signaling pathways. Following Sal B treatment, (a) levels of phosphorylated (p)-Jak2, p-Stat3, p-EGFR, and p-ERK proteins all increased; (b) these increases were suppressed by AG490 (a Jak2 inhibitor) and ZD1839 (an EGFR inhibitor); and (c) cytokines associated with the Jak2–Stat3 signaling pathway were upregulated. Our findings suggest that Sal B can be used as a LIF replacement for maintaining ESC pluripotency while increasing cell proliferation.
Introduction
Stem cell therapy has the potential to combat many diseases for which therapies are not currently available. In previous studies, we reported that implanted mesenchymal and bone marrow stem cells promote neuroplasticity in a stroke model (13,15), but these cells are limited in terms of cell type differentiation. Embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) (two types of pluripotent stem cells) are believed to have the greatest potential for cell therapies due to their capability to differentiate into endodermal, mesodermal, and ectodermal cells—also referred to as three-germ-layer cells (1,18). However, pluripotent stem cell cultures require an exceptionally expensive reagent, leukemia-induced factor (LIF), to maintain stemness and pluripotency (20). LIF binds to LIF receptors and glycoprotein 130 (gp130) to activate the Janus kinase 2 (Jak2, a signal transducer) and the signal transducer and activator of transcription 3 (Stat3, a transcription 3 activator) signaling pathways to maintain ESC self-renewal (22). The Jak2 and Stat3 signaling pathways upregulate LIF receptor-related cytokine expression levels, including interleukin (IL)-5, IL-11, epidermal growth factor (EGF), ciliaryneurotrophic factor (CNTF9, erythropoietin (EPO), and IL-6 (8,9,11,19,27).
Known as danshen in Chinese, Salvia miltiorrhiza has long been used by traditional Chinese medicine practitioners to treat cardiovascular and cerebrovascular diseases such as angina pectoris, hyperlipidemia, and acute ischemic stroke (3,4,12). The most abundant and bioactive component of Salvia miltiorrhiza is the water-soluble salvianolic acid B (Sal B), which binds to the epidermal growth factor receptor (EGFR) and triggers downstream gene expression (2,7). In addition to inhibiting platelet aggregation (17), Sal B has been shown to exert cardioprotective (6), anticancer (28), and antiapoptotic (30) effects. However, to our knowledge, the effects of Sal B on stem cells have not been studied; therefore, our goal was to determine whether Sal B is capable of maintaining stem cell pluripotency. Our primary finding is that it not only maintains ESC pluripotency via Jak2 and Stat3 activation but also increases the rate of ESC proliferation via EGFR–extracellular signal-regulated kinase 1/2 (ERK1/2) activation.
Materials and Methods
Mouse Embryonic Fibroblast and ESC Cultures
Mouse embryonic fibroblast (MEF) cell isolation was performed as previously described (14). Cells were isolated from 13.5-day-old C57BL/6 mice embryos (pregnant mouse fromTaiwan National Laboratory Animal Center, Taipei, Taiwan) retrieved by Cesarean section. Internal organs, legs, and heads were removed and remaining embryo parts digested with trypsin (Gibco-BRL, Grand Island, NY, USA). Cells were cultured in Dulbecco's modified Eagle's medium (DMEM; Gibco-BRL) containing 10% heat-inactivated fetal bovine serum (FBS; Gibco-BRL), penicillin (100 U/ml; Gibco-BRL), streptomycin (100 μg/ml; Gibco-BRL), nonessential amino acids (0.1 mM; Gibco-BRL), and l-glutamine (2 mM; Gibco-BRL) in a humidified incubator (37°C) with 5% CO2. Mouse ESCs (from Taiwan Bioresource Collection and Research Center, Hsinchu, Taiwan) were cultured in DMEM with 15% FBS (Hyclone, Logan, UT, USA), nonessential amino acids (0.1 mM), l-glutamine (2 mM), β-mercaptoethanol (0.2 mM; Gibco-BRL), and LIF (4 ng/ml; Millipore, Billerica, MA, USA), in a humidified incubator (37°C) with 5% CO2. All experimental protocols were approved by the Institutional Animal Care and Use Committee of China Medical University.
Sal B Treatment
Salvia miltiorrhiza extract (mol weight 718.61) was purchased from ChromaDex, Inc. (Irvine, CA, USA). Sal B was dissolved in pure water to a concentration of 1 μM and stored at −20°C until used as a stock solution. MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] assays were performed using 0.001, 0.01, 0.1, and 1 nM concentrations (see below). The same concentrations were used for determining gene expression profiles and maintaining stem cell self-renewal.
Cell Viability and BrdU Analysis
To test viability, ESCs were reseeded in six-well plates (Corning, Corning, NY, USA), treated with different concentrations of Sal B (0.001, 0.01, 0.1, and 1 nM), and analyzed using previously described MTT procedures (14). Briefly, culture medium in each well was replaced with 100 μl of fresh medium containing 10 μl of 5 mg/ml MTT stock solution (Sigma-Aldrich, St. Louis, MO, USA). Medium was removed after 4 h and replaced with 100 μl dimethyl sulfoxide (DMSO; Sigma-Aldrich) per well and held for 10 min at 37°C, after which samples were mixed and absorbances read at 540 nm. Bromodeoxyuridine (BrdU) analyses were used to detect cell proliferation rates (i.e., BrdU incorporation into the DNA of proliferating cells). Cells were treated with BrdU (10 μM; Sigma-Aldrich) for 24 h and then fixed for immunofluorescent antibody assays (IFA).
Quantitative Real-Time PCR
TRIzol (Invitrogen, Carlsbad, CA, USA) was used to extract total RNA from MEF and ESCs. RNA concentrations were determined by spectrophotometry. Complementary DNA was produced from mRNA (5 μg) using SuperScript III Reverse Transcriptase Kits (Invitrogen). Real-time PCR was performed as previously described to determine the gene expression levels of octamer-binding transcription factor 4 (Oct4), sex-determining region Y box 2 (Sox2), IL-5, IL-11, EGF, CNTF, EPO, and IL-6 (16). Primer sequences are listed in Table 1.
Real-Time PCR Primers Used in the Study

Oct4, octamer-binding transcription factor 4; Sox2, sex-determining region Y box 2; IL-5, interleukin 5; EGF, epidermal growth factor; CNTF, ciliary neurotrophic factor; EPO, erythropoietin.
Alkaline Phosphatase Staining and IFA
Alkaline phosphatase staining was performed using alkaline phosphatase substrate kits (Vector Lab, Burlingame, CA, USA). IFAs were performed according to steps described in a previous study (14) using anti-Nanog (Novus, Saint Charles, MO, USA), antistage-specific embryonic antigen (SSEA1; Millipore), and anti-BrdU (Sigma-Aldrich), anti-α smooth muscle actin (α-SMA), and antineuron-specific class III β-tubulin (Tuj1 antibody clone; both Chemicon, Temecula, CA, USA) monoclonal antibodies, and anti-α fetoprotein (AFP; Abcam, Cambridge, MA, USA) polyclonal antibodies.
In Vitro and In Vivo ESC Pluripotency
Embryoid body and teratoma formation and differentiation experiments were used to determine the capability of Sal B to maintain ESC pluripotency (six passages of Sal B-containing medium). Embryoid body experiments were performed as described in Liu et al. (14). For teratoma formation, ESCs were suspended (5 × 107 cells/ml) in DMEM containing 10% FBS. Nude male mice (from Taiwan National Laboratory Animal Center, N = 3) were injected subcutaneously in their dorsal flanks with 100 μl (5 × 106) of suspended cells. Four weeks post-treatment, tumors were surgically removed, weighed, fixed in PBS containing 4% formaldehyde (Sigma-Aldrich), and embedded in paraffin. Sections were stained with hematoxylin and eosin (Medical Chemical Corporation, Torrance, CA, USA) to determine cell types.
Antagonist Treatment
AG490 at 5 μM concentrations in medium (Sigma-Aldrich) was used to inhibit Jak2. ZD1839 (also at 5 μM concentrations in medium) (LC Laboratories, Woburn, MA, USA) was used to inhibit EGFR. We dissolved AG490 in methanol (Sigma-Aldrich) to a concentration of 5 mM and stored it at −20°C until its use as a stock solution. ZD1839 was dissolved in PBS to the same concentration and stored at the same temperature.
Western Blot Assays
ESCs treated with different concentrations were lysed in buffer containing 320 mM sucrose (Sigma-Aldrich), 5 mM HEPES (Gibco-BRL), 1 μg/ml leupeptin (Sigma-Aldrich), and 1 μg/ml aprotinin (Sigma-Aldrich). Lysates were centrifuged at 13,000 × g for 15 min. The resulting pellet was resuspended in buffer [62.5 mM Tris-HCl, 10% glycerol, 2% sodium dodecyl sulfate (SDS), 0.1% bromophenol blue (all Sigma-Aldrich), and 50 mM dithiothreitol (DTT; Promega, Madison, WI, USA)] and subjected to 10% SDS-polyacrylamide gel electrophoresis (SDS-PAGE; Sigma-Aldrich). The proteins separated in the gel were then transferred to a polyvinylidene difluoride (PVDF) membrane (Millipore) and incubated with appropriately diluted antibodies to rabbit anti-Jak2 (Cell Signaling Technology, Danvers, MA, USA), mouse anti-phospho-Jak2 (Cell Signaling), rabbit anti-Stat3 (BD Pharmingen, San Diego, CA, USA), rabbit anti-phospho-Stat3 (Cell Signaling), rabbit anti-EGFR (GeneTex, Irvine, CA, USA), rabbit anti-phospho-EGFR (GeneTex), rabbit anti-ERK (GeneTex), rabbit anti-phospho-ERK antibodies (Invitrogen), and mouse anti-β-actin (Millipore). The enhanced chemiluminescent (ECL) Western blotting substrate kit (Thermoscientific, Waltham, MA, USA) was used to stain the membranes, and signals were obtained using UVP BioSpectrum Imaging System (Upland, CA, USA).
Statistical Analysis
Results are expressed as median. Data lacking normal distribution were Kruskal–Wallis and Dunn's post hoc tests; statistical significance was established as p < 0.05.
Results
Viability Assays for Sal B-Treated Cells
We used MTT assays to determine the viability of ESCs treated with Sal B at concentrations of 0.001, 0.01, 0.1, and 1 nM (7). Our results indicate significant (p < 0.05) increases in cell survival rates in the 0.01 and 0.1 nM groups 24 h and 48 h posttreatment (Fig. 1A). This confirmed our decision to use the four Sal B concentrations for additional experiments.
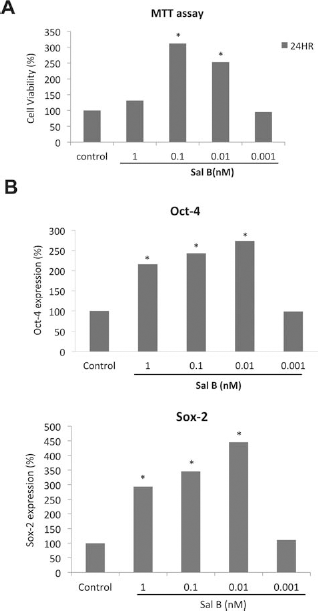
Cell viability and pluripotent marker gene expression levels in embryonic stem cells (ESCs) treated with salvianolic acid B (Sal B). (A) MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] assay of ESCs treated with various concentrations of Sal B. (B) Octamer-binding transcription factor 4 (Oct4) and sex-determining region Y box 2 (Sox2) gene expression levels in ESCs treated with various concentrations of Sal B (real-time PCR). *p < 0.05 versus No LIF group.
Oct4 and Sox2 Expression Levels Following Sal B Treatment
To confirm the ability of Sal B to maintain stem cell pluripotency, we used Q-PCR to determine the presence of two pluripotent marker genes, Oct4 and Sox2, in the four treatment groups at 24 h. Our results indicate significant (p < 0.05) increases in the expression levels of both genes in the 0.01, 0.1, and 1 nM groups (Fig. 1B)—in other words, Sal B maintained ESC pluripotency at those treatment levels.
Maintaining ESC Self-Renewal
LIF was added to mouse ESC cultures to maintain cell self-renewal and pluripotency. However, our goal was to determine whether a much less expensive agent can be used for the same purpose; therefore, our next step was to determine the ability of Sal B to maintain ESC self-renewal and pluripotency at the same level as LIF. Three stem cell markers [alkaline phosphatase (AP), Nanog, and SSEA-1] were used to survey ESC stemness. As shown in Figure 2A, AP staining levels in cells treated with 0.01 or 0.1 nM of Sal B were significantly (p < 0.05) higher than in the NO LIF(medium without LIF) controls. Immunofluorescence staining analysis results indicate that Nanog and SSEA-1 expression levels in ESCs treated with 0.01 or 0.1 nM Sal B were also significantly higher (p < 0.05) than in the NO LIF control cells (Fig. 2B and C). These results suggest that Sal B can be used as an inexpensive substitute for LIF to maintain ESC self-renewal.

Stem cell markers staining in ESCs treated with Sal B. (A) Alkaline phosphatase (AP) staining ESCs treated with various concentrations of Sal B and the quantities of AP-positive clones in ESCs. (B) Nanog immunocytochemistry in ESCs treated with various concentrations of Sal B and the quantities of Nanog expression levels in ESCs. (C) Stage-specific embryonic antigen 1 (SSEA1) immunocytochemistry in ESCs treated with various concentrations of Sal B and the quantities of SSEA1 expression levels in ESCs. Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI). *p < 0.05 versus No LIF group.
ESC Pluripotency Following Sal B Treatment
Embryoid body (EB) and teratoma formation and differentiation experiments were used to determine whether Sal B is capable of maintaining ESC pluripotency (six passages with Sal B-containing medium at 0.01 nM). EB immunofluorescence staining results indicate that ESCs differentiated into cells that tested positive for neuronal β III tubulin (the antibody clone Tuj1; an ectoderm marker), α-SMA (a mesoderm marker), and AFP (an endoderm marker) (Fig. 3A). For the teratoma formation analysis, we injected ESCs that had been passaged six times with Sal B-containing medium (0.01 nM) into the dorsa of nude male mice. As shown in Figure 3B, ESCs differentiated into all three-germ-layer cell types—specifically, respiratory epithelium, adipose tissue, and epidermal cells.

Detection of the pluripotency of ESCs passaged six times in Sal B-containing medium (replaced LIF). (A) Immunofluorescent staining in spontaneous differentiation of ESCs: α-fetoprotein (AFP; endoderm marker), α-smooth muscle actin (α-SMA; mesoderm marker), and neuron-specific class III β-tubulin (Tuj1 antibody clone; ectoderm marker). Nuclei were stained with DAPI (blue). (B) Various kinds of tissue cells were observed in teratomas derived from ESCs 4 weeks after their transplantation into a nude mouse.
Sal B-Induced Cell Proliferation
According to MTT assay results, treatment with Sal B at 0.01 nM and 0.1 nM increased ESC viability (Fig. 1). We used viability assays and immunofluorescence staining with BrdU to determine cell proliferation rates. Viability assay results indicate significantly (p < 0.05) higher total cell numbers in the Sal B-treated ESCs compared to the LIF group at 48 and 72 h (Fig. 4A). Total cell numbers in the 0.1 nM Sal B treatment group were also higher than in the LIF treatment group at 48 and 72 h, but not at statistically significant levels. The immunofluorescence staining results also indicate significantly higher (p < 0.05) levels of BrdU expression in cells treated with 0.01 nM and 0.1 nM Sal B compared to cells treated with LIF (Fig. 4B).

Detection of the cell proliferation rate after Sal B treatment. (A) Cell viability assay of ESCs treated with various concentrations of Sal B. Total cell numbers in the 0.1 nM Sal B treatment group were also higher than in the LIF treatment group at 48 and 72 h. (B) The quantities of bromodeoxyuridine (BrdU) expression levels in ESCs treated with various concentrations of Sal B by immunocytochemical staining. *p < 0.05 versus No LIF group.
Pathway Analyses of Sal B-Treated ESCs
The Jak–Stat signaling pathway is the most important for ESC maintenance and renewal (20). Other researchers have described the ability of Sal B to bind to EGFR and to trigger downstream gene expression via an EGFR signaling pathway (2,7). We therefore used these two signaling pathways to determine whether ESC self-renewal and pluripotency maintenance mechanisms are affected by Sal B treatment.
Jak2 and Stat3 are key Jak–Stat pathway proteins for maintaining stem cell self-renewal. We used Western blotting to measure Jak2 and Stat3 protein levels in Sal B-treated ESCs and did not observe increases in either protein (Fig. 5A). However, we did note increases in phosphorylated Jak2 and Stat3 forms (active forms) following treatment with either 0.1 nM or 0.01 nM Sal B (Fig. 5A). In the EGFR signaling pathway, EGFR phosphorylation has been shown to induce Stat3 phosphorylation (5). Our Western blot results indicate increases in the protein expression of phosphorylated EGFR and phosphorylated Stat3 following treatment with Sal B, especially at concentrations of 0.1 nM or 0.01 nM (Fig. 5A). In other words, the data suggest that Sal B is capable of maintaining ESC self-renewal and pluripotency by activating the Jak2–Stat3 and EGFR–Stat3 signaling pathways.

EGFR, ERK, Jak2, and Stat3 expression levels in Sal B-treated ESCs. (A) Epidermal growth factor receptor (EGFR), phosphorylated (p)-EFGR, extracellular signal-regulated kinase (ERK), phosphorylated-ERK, Janus kinase 2 (Jak2), phosphorylated-Jak2, signal transducer and activator of transcription 3 (Stat3), and phosphorylated-Stat3 protein expression levels in ESCs treated with various concentrations of Sal B (Western blot). (B) mRNA levels of six cytokine genes involved in the Jak2–Stat3 pathway: interleukin (IL)-5, IL-11, EGF, ciliary neurotrophic factor (CNTF), erythropoietin (EPO), and IL-6 in ESCs treated with various concentrations of Sal B. *p < 0.05 versus No LIF group.
Pathway Analyses of Sal B-Treated ESCs and Increased Proliferation
As shown in Figure 4A, we found that Sal B treatment induced cell proliferation, especially at a concentration of 0.01 nM. According to a previous study, EGFR phosphorylation induces ERK1/2 activation, which also results in greater cell proliferation (29). We therefore focused on the EGFR–ERK signaling pathway to identify the mechanism behind cell proliferation induced by Sal B, using Western blot analyses to measure ERK and phosphorylated ERK protein levels in Sal B-treated ESCs. We did not observe increases in EGFR or ERK protein levels but did note increases in phosphorylated EGFR and phosphorylated ERK forms (active forms) following treatment with 0.1 nM or 0.01 nM Sal B (Fig. 5A). These data suggest that Sal B induces activation of the EGFR–ERK signaling pathway.
Cytokine Expression Level and ESC Stemness
To identify the mechanism behind Jak2–Stat3 signal upregulation triggered by Sal B treatment, we used real-time PCR analysis to quantify the mRNA levels of six cytokine genes involved in the Jak2–Stat3 pathway: IL-5, IL-11, EGF, CNTF, EPO, and IL-6. We observed significantly increased (p < 0.05) expression levels for all six genes at a Sal B treatment level of 0.01 nM (Fig. 5B). Combined, they suggest that Sal B is capable of increasing Jak2–Stat3-associated cytokine levels and activating Jak2 and Stat3 proteins to maintain stem cell pluripotency.
To confirm the mechanism underlying the Sal B activation of the Jak2–Stat3, EGFR–Stat3, and EGFR–ERK signaling pathways, we used AG490 (a Jak2 antagonist) and ZD1839 (an EGFR antagonist) to suppress phosphorylated Jak2 and EGFR and to detect downstream protein expression levels. As shown in Figure 6A and B, AG490 suppressed phosphorylated Jak2 and phosphorylated Stat3 expression levels, and ZD1839 suppressed phosphorylated EGFR and phosphorylated ERK expression levels. We therefore concluded that Sal B maintains ESC self-renewal and pluripotency by activating both the Jak2–Stat3 and EGFR–Stat3 signaling pathways (Fig. 7).
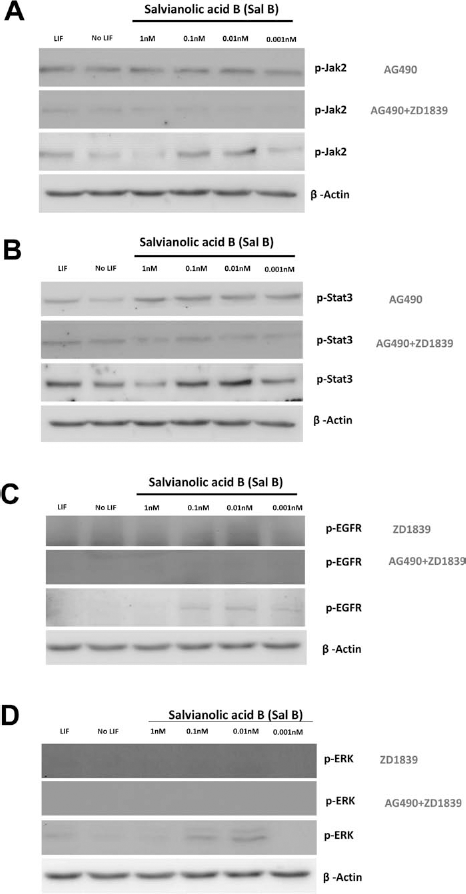
The related phosphorylated protein levels in ESCs treated with Sal B, Jak2 inhibitor, AG490 (Western blot), and EFGR inhibitor, ZD1839. (A) phosphorylated Jak2, (B) phosphorylated Stat3, (C) phosphorylated EFGR, (D) phosphorylated ERK.

Model for the Sal B involved in maintaining ESC pluripotency and increasing cell proliferation.
Discussion
ESCs (pluripotent stem cells derived from the inner cell masses of mammalian blastocysts) are capable of differentiating into embryo endodermal, mesodermal, and ectodermal cells (1). They are believed to have the greatest potential for use in cell therapies, but their cultures require LIF, a very expensive reagent, to maintain stemness. This expense has limited the number of labs that can participate in stem cell research and is encouraging efforts to find another, less expensive agent capable of inhibiting differentiation while also supporting the proliferation of undifferentiated stem cells (20). Our results indicate that (Sal B) is capable of maintaining stem cell pluripotency and increasing cell proliferation by activating the Jak2–Stat3 and EGFR–ERK1/2 pathways.
Sal B is a natural compound with known protective effects against ischemia–reperfusion heart injuries (7). Sal B significantly increases cell viability at concentrations of 0.00001, 0.001, 0.001, and 0.01 nM, but decreases cell viability at 10,000 and 100,000 nM concentrations (7). According to our MTT assay results, Sal B at concentrations of 0.1 and 0.01 nM significantly increased cell viability (Fig. 1A). Similar data have been reported by other researchers for different cell types (7). We also noted that Sal B treatment at concentrations of 0.1 and 0.01 nM increased the expression levels of Oct4 and Sox2—key proteins for maintaining stem cell pluripotency (Fig. 1B) (26). We therefore suggest that Sal B concentrations of 0.1 and 0.01 nM are optimal when used as a LIF replacement.
To further test the maintenance of stem cell pluripotency, we employed several stem cell markers containing AP, Nanog, and SSEA-1 to determine the effects of substituting Sal B for LIF when culturing ESCs (Fig. 2) and used embryoid body and teratoma formation tests (21,23–25) to determine ESC pluripotency by passaging cells six times in culture medium containing 0.01 nM Sal B (Fig. 3A and B). Our results indicate that the ESCs maintained pluripotency as well as their ability to differentiate into three germ layers. In a previous study, we reported that n-butylidenephthalide (BP) was also capable of maintaining stem cell pluripotency by activating the Jak2–Stat3 pathway (14). This capability is similar to that which we found for Sal B; however, Sal B has the added feature of increasing stem cell proliferation, perhaps making it a better substitute for LIF.
According to Tamm et al., Oct4 and Sox2 are the primary proteins for maintaining ESC pluripotency via the Jak2–Stat3 signaling pathway (26). We therefore tested whether Sal B treatment triggered the Jak2–Stat3 signaling pathway, especially in light of our finding that Sal B treatment increased Oct4 and Sox2 gene expression levels (Fig. 1). For an earlier study, we used the same methods to determine the ability of BP to maintain ESC pluripotency via the Jak2–Stat3 signaling pathway (14); for the present project, we looked at the effects of Sal B treatment on the same pathway. Our data indicate higher p-Jak2 and p-Stat3 protein levels in the Sal B treatment group compared to the No LIF control group, especially at concentrations of 0.1 and 0.01 nM (Fig. 5A). LIF-activated Jak2–Stat3 has been described as essential for maintaining ESC self-renewal and pluripotency (20,22). According to our Western blot data, treatment with Sal B exerted the same effect on Jak2 and Stat3 proteins, as well as on ESC self-renewal and pluripotency (Fig. 5A), with concentrations of 0.1 and 0.01 nM identified as optimum.
Other researchers have described the binding of Sal B to EGFR and the consequent triggering of downstream gene expression, including ERK (2,7). As shown in Figure 4, we observed increases in phosphorylated EGFR and phosphorylated ERK protein levels following Sal B treatment. ERK1 signaling has been shown to induce pancreatic β-cell proliferation (10). Our cell viability assay results indicate that Sal B treatment increased cell proliferation, especially at concentrations of 0.1 or 0.01 nM (Fig. 4). In order to confirm that Sal B treatment increased ESC proliferation via the EGFR–ERK pathway, we used the EGFR inhibitor ZD1839 to block EGFR (29) and found that ZD1839 suppressed cell proliferation in ESCs treated with Sal B (Fig. 6).
According to our data, Sal B is capable of maintaining stem cell pluripotency via the Jak2–Stat3 pathway and of increasing cell proliferation via the EGFR–ERK pathway. It is our hope that Sal B can serve as a viable alternative to the much more expensive LIF in ESC research, but additional research is required to confirm its utility.
Footnotes
Acknowledgments
This study was supported in part by the Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH102-TD-B-111-004), China Medical University (CMU100-TS-16), Republic of China National Science Council (NSC 101-2314-B-039-009-MY2), and Gwoxi Stem Cell Applied Technology Co., Ltd. (10242643). The authors declare no conflicts of interest.
