Abstract
Traumatic nerve injuries are a major clinical challenge. Tissue engineering using a combination of nerve conduits and cell-based therapies represents a promising approach to nerve repair. The aim of this study was to examine the regeneration potential of human adipose-derived stem cells (hASCs) after transplantation in a nonautogenous setting and to compare them with autogenous rat ASCs (rASCs) for early peripheral nerve regeneration. Furthermore, the use of MRI to assess the continuous process of nerve regeneration was elaborated. The sciatic nerve injury model in female Sprague-Dawley rats was applied, and a 10-mm gap created by using a fibrin conduit seeded with the following cell types: rASCs, Schwann cell (SC)-like cells from rASC, rat SCs (rSCs), hASCs from the superficial and deep abdominal layer, as well as human stromal vascular fraction (1 × 106 cells). As a negative control group, culture medium only was used. After 2 weeks, nerve regeneration was assessed by immunocytochemistry. Furthermore, MRI was performed after 2 and 4 weeks to monitor nerve regeneration. Autogenous ASCs and SC-like cells led to accelerated peripheral nerve regeneration, whereas the human stem cell groups displayed inferior results. Nevertheless, positive tends could be observed for hASCs from the deep abdominal layer. By using a clinical 3T MRI scanner, we were able to visualize the graft as a small black outline and small hyperintensity indicating the regenerating axon front. Furthermore, a strong correlation was found between the length of the regenerating axon front measured by MRI and the length measured by immunocytochemistry (
Introduction
Nerve lesions due to traumatic injuries affect several hundreds of thousands of patients every year in Europe and the US alone (1). Despite recent advancements in microsurgical techniques, the outcome of peripheral nerve injury often remains poor (2).
Tissue engineering using a combination of nerve conduits and cell-based therapies represents a promising approach for nerve repair (18). Schwann cells (SCs) are the key drivers of endogenous regeneration, and experimentally, a combination of SCs and artificial nerve conduits enhances regeneration (4,31). However, the clinical benefits of SCs are limited by their inability to generate sufficient cell numbers quickly. Therefore, identification of novel cell-based therapies is required to develop tissue-engineered strategies for nerve repair (13).
Adipose tissue has received a great deal of attention in tissue engineering over the last years due to its extensive availability because of the ease of its withdrawal from the same patient and the fact that it possesses stem cells (23,31). Pretreatment of adipose tissue aimed at isolating adipose-derived stem/precursor cells (ASCs) and/or stromal vascular fraction (SVF) are considered to be a fundamental requisite for nerve repair (23).
We have recently demonstrated that human (h)ASCs express a range of neurotrophic factors, and cells isolated from the superficial abdominal fat layer promote neurite process outgrowth in vitro (12). Together with their favorable proliferation kinetics and immunosuppressive properties, the potential of hASCs for nonautogenous application has to be demonstrated separately in vivo (15,33). Thus, since no allogenic human model is available for ethical concerns, the current research uses human stem cells in a nonautogenous setting for peripheral nerve regeneration as a maximal HLA-mismatch condition.
MRI is considered to be the most attractive imaging modality to monitor the maximum efficacy of cellular therapies (2). However, only a few investigations have been conducted to date, whereas immunocytochemistry has been shown to be an accepted standard of peripheral neural regeneration measurement (2,30).
In the present project, the ability of human stem cells to improve peripheral nerve regeneration was investigated and compared with autologous stem cell transplantation in a rat sciatic nerve injury model. Evaluation was performed using immunocytochemistry and MRI.
Materials and Methods
Study Design
In a first step, a sciatic nerve lesion creating a 10-mm gap was used in female Sprague–Dawley rats (Harlan Laboratories B.V., The Netherlands). In group 1 (negative control group), a fibrin conduit was inserted with culture medium (CM) alone, containing DMEM/F-12 (Cat. No. 10565–018; Gibco, Invitrogen, Carlsbad, CA, USA), 1% penicillin/streptomycin (Cat. No. 15140122; Gibco, Invitrogen), and 10% FBS (Cat. No. 10091–148; Gibco, Invitrogen). All study animals (groups 2–7, seven per group) were treated with either rat (r)ASCs, differentiated rASCs (SC-like cells), hASCs from the superficial and deep layer, SVF, or SCs only (positive control group) (Fig. 1). After 2 weeks, nerve regeneration was assessed by immunocytochemistry. Furthermore, imaging analysis (MRI) was performed after 2 and 4 weeks to monitor nerve regeneration. Ethical approval was obtained for all experiments by the local veterinary physician and local ethical committee [Ethikkommission beider Basel (EKBB), No. 2455].
Overall study design. ASC/SVF, adipose-derived stem cell/stromal vascular fraction; SC, Schwann cell; FSK, forskolin; FGF, fibroblast growth factor; GGF, glial growth factor (neuregulin-1β1); PDGF: platelet-derived growth factor.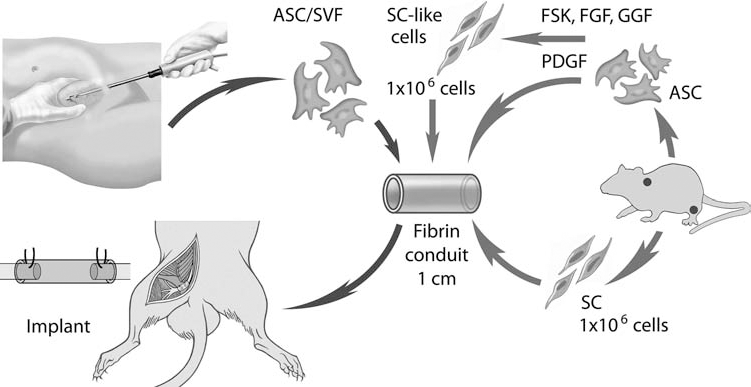
Isolation of ASCs, SVF, SC-Like Cells, and rSCs In Vitro
For ASC isolation and transdifferentiation of rASCs into SC-like cells, established protocols from our group were used (4,8,13,29). To isolate ASCs from rats, subcutaneous adipose tissue from the neck of adult female Sprague–Dawley rats (8 weeks old, weighing 250–300 g) was excised. After written informed consent was obtained, hASCs from the deep and superficial abdominal layer as well as human SVF (mixed population from both deep and superficial abdominal layer) were harvested from a total of six female patients (mean age 45 years, range 28–74 years) undergoing either dermolipectomy or liposuction. Ethical approval for the use of human-derived samples was obtained from the local ethical committee (EKBB). For the isolation of human SVF, floating adipocytes and supernatant were removed after centrifugation from the SVF pellet, washed, and resuspended in sterilized phosphate-buffered saline (PBS, AM 9625; Invitrogen). The singlecell suspension was filtered through a 70-μm cell strainer (Ref. 352350; BD Falcon, Franklin Lakes, NJ, USA). Total cell number was assessed by crystal violet stain (Sigma-Aldrich Chemie GmbH, Buchs, Switzerland), and the nucleated cells were counted in a Neubauer chamber (Marienfeld, Germany). Rat SCs were isolated as described previously by our group (4). All of the experiments were carried out using cells that were between passages 2 and 4.
Fluorescence-Activated Cell Sorting (FACS) and Immunocytochemistry
Cytofluorimetry analysis of SVF/ASCs for standard markers [cluster of differentiation 105 (CD105)], mesenchymal markers (CD90 and CD73), endothelial markers (CD31 and CD34), and the pan-hematopoietic marker (CD45) was performed as described previously (8,29). The suspensions were incubated for 30 min at 4°C with fluorochrome-conjugated antibodies against the indicated proteins or an isotype control. The antibodies used were from Becton, Dickinson and Company (BD, Franklin Lakes, NJ, USA); anti-CD105 antibody was obtained from AbD Serotec (Oxon, UK). Cells were washed, resuspended in PBS (Invitrogen), and analyzed with a FACSCalibur flow cytometer (BD). SCs and SC-like cells were plated onto a four-well slide chamber (Thermo Scientific, Nunc, Fisher Scientific AG, Wohlen, Switzerland) and fixed for 10 min in 4% paraformaldehyde (PFA, 76240 Fluka; Sigma, Buchs, Switzerland). The cells were washed for 2 min in PBS and blocked for 1 h in immunofluorescent blocking buffer (IBB) containing 1% bovine serum albumin (BSA, A7906; Sigma), 1× PBS, and 0.1% Triton X-100 (Sigma-Aldrich). The cells were subsequently incubated for 1 h in IBB containing the following monoclonal antibodies: glial fibrillary acidic protein (GFAP, 1:400, ab7260; Abcam, Cambridge, UK), S100 (1:500, Z0311; Dako, Baar, Switzerland), and low-affinity nerve growth factor receptor (p75, 1:100, ab8874; Abcam). After incubating for 1 h at room temperature or overnight at 4°C, the cells were washed three times with PBS and incubated with cyanine 3 (Cy3) sheep anti-rabbit IgG (1:500, C2306; Sigma) or Cy3 sheep anti-mouse IgG (1:500, C2181; Sigma). Cell nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI, 1:500, D9542; Sigma).
Harvested conduits were fixed by PFA for 16 h, then rinsed for 24 h at 4°C in PBS containing 30% sucrose (S7903; Sigma) and 0.1% sodium azide (No. 822335; Merck-Schuchardt, Darmstadt, Germany), with PBS changes after 12 h to eliminate any PFA remnants. The specimens were then embedded in OCT freezing media (Tissue-tek, Sakura, Japan), and longitudinal cryosections (12 μm) were prepared onto slides (Superfrost plus; Menzel-Gläser, Braunschweig, Germany). After 3 × 5-min PBS washes, an appropriate normal serum (BSA; Sigma) was applied for 1 h to block nonspecific antibody binding. Slides were incubated overnight at 4°C with primary antibodies rabbit anti-S100 (1:500, Z0311; Dako) or rabbit anti-protein gene product 9.5 (PGP 9.5, 1:500, Z5116; Dako). To identify human cells after 2 weeks, a mouse anti-human mitochondria monoclonal antibody was used in PKH26-negative cells (1:20, MAB1273; Milliport, Schaffhausen, Switzerland). Subsequently, slides were washed 3 × 5 min in PBS and incubated with secondary Cy3 sheep anti-rabbit IgG (1:500, C2306; Sigma) or Cy3 sheep anti-mouse IgG (1:500, C2181; Sigma) antibodies at room temperature in the dark for 40 min. After 3 × 5-min PBS washes, the slides were finally mounted with Vectashield with DAPI (Vector Labs, Peterborough, UK) and examined under the fluorescence microscope (10× magnification, IX81; Olympus, Center Valley, PA, USA). Axonal regeneration distance (PGP 9.5) and S100-positive cell distribution inside the conduit were evaluated. Histomorphometric analysis was determined by ImageJ software (NIH, Bethesda, MD, USA).
Fibrin Conduit Preparation and PKH Labeling
The fibrin conduit was constructed according to earlier protocols from two compound fibrin glues (Tisseel Kit VH 1.0; Baxter SA, Volketswil, Switzerland) (11). Tisseel contains fibrinogen, 70 to 110 mg/ml; plasma fibronectin, 2 to 9 mg/ml; factor XIII, 10 to 50 U/ml; plasminogen, 40 to 120 mg/ml; aprotinin solution, 3,000 KIU/ml; thrombin, 4 IU/ml; and calcium chlorides, 40 mmol/L. Fibrin glue was filled into a specially designed silicone inlay (Dublisil 20®; Dreve-Dentamid GMBH, Unna, Germany) of a developed medical compressor (Jade-Stone®; Whip Mix Corp. Louisville, KY, USA) around a stainless steel core and pressed into shape using locking screws. This allows the generation of uniform conduits with both materials of a 14-mm length, 1-mm wall thickness, and 2-mm lumen. Cells, 1 × 106, were suspended in 20 μl of 10% fibrinogen solution/CM and injected into the conduit by using a 20-μl pipette tip. Target cells were labeled by PKH26 (PKH26GL; Sigma) according to the manufacturer's recommendation.
Surgical Procedure and Experimental Groups
The animal care and experimental procedures were carried out in accordance with the standards established by the NIH Guide for Care and Use of Laboratory Animals (National Institutes of Health Publications No. 86–23, revised 1985) and the European Communities Council Directive (86/609/EEC). All surgical procedures were performed under general anesthesia with 3% isoflurane. Routinely, buprenorphin (Temgesic®, 2 ml Amp, 0.3 mg/ml, 0.1–0.2 mg/kg KG; Reckitt Benckiser, Zürich, Switzerland) was administered prior to surgery. The left sciatic nerve was approached dorsally using a gentle spreading technique of the gluteus muscle. After resection of the nerve, it was replaced by the tissue-engineered fibrin conduit. Surgical implantation of the structures was undertaken with an operating microscope (Carl Zeiss, Oberkochen, Germany), and the nerve ends were fixed to the conduit by a single epineural suture (9/0 nylon; Ethicon, Somerville, NJ, USA): proximal and distal nerve stumps were inserted 2 mm into the nerve conduits, thus leaving a 10-mm gap. Muscles and fascia layers were closed with single resorbable stitches (4/0 Vicryl; Ethicon) and the skin by using a continuous running suture (4/0 Prolen; Ethicon).
MRI Scanning
After euthanasia with CO2, animals were examined on a 3T human whole body scanner (Magnetom Verio; Siemens, Forchheim, Germany) using an eight-channel wrist coil. After scout images in three directions, we performed imaging perpendicular to the calves and to the thighs using T1 weighted and T2 short tau inversion recovery (STIR), as well as sagittal images of the thigh using T2 weighted and T2 STIR sequences.
Image Evaluation
Images from 13 animals were evaluated by a radiologist blinded to the treatment groups using open-source imaging software (OsiriX; Pixmeo, Geneva, Switzerland). To quantify muscle atrophy, ROIs were drawn on the axial images encompassing all muscles of the calves but excluding the tibial bone. Muscle volume was calculated for each leg separately. Length of nerve growth was evaluated on sagittal and axial images of the thighs and correlated with histological images.
Statistical Analysis
Data are presented as mean ± standard deviations (SD) from at least three animals per group, and all experimental samples were conducted in triplicate. One-way ANOVA tests with corresponding post hoc tests (Bonferroni or Tukey; GraphPad Prism version 5.00 for Windows; GraphPad Software, San Diego, CA, USA) were used where appropriate to determine statistical differences between the experimental groups;
Results
Cell Characterization and Histomorphometric Analysis
Cytofluorimetry analysis of SVF/ASCs for standard markers (hematopoietic CD45; endothelial CD31/CD34; stromal CD105, CD73, and CD90) was consistent with previous characterization of those cells (8,29). Similar observations were found for SCs, showing a similar lineagespecific pattern (S100, p75, and GFAP) as previously reported (4). After transduction of rASCs into SC-like cells, a bipolar-shaped morphology was observed expressing S100, P75, and GFAP (Fig. 2).
Characterization of SC-like cells. (a) Light microscopy showed a bipolar-shaped morphology similar to SCs. SC-like cells of passage 3 were stained using antibodies against GFAP (b), S100 (c), and low-affinity nerve growth factor receptor (p75) (d). Nuclei were stained with DAPI (4′-6-diamidino-2-phenylindole, arrowhead and stars). Scale bar: 250 μm.
In Vivo Nerve Regeneration
In accordance with the animal protocol, a total of two animals were terminated early due to surgical site infections. At 2 weeks, the fibrin matrix grid had almost completely dissolved. On macroscopic and microscopic examination, no signs of tumor formation or hematoma were noticed.
Sciatic nerve axonal regeneration was different depending on the type of cells that were injected in the fibrin conduits (Fig. 3). Cell viability was confirmed after 14 days where PKH26-labeled ASCs were arranged in densely arranged clusters at the tip of the regenerating axon front coexpressing S100 (Fig. 3h). No fluorescent labeling was detected from the negative control group. Human-specific antimitochondrial staining showed intense perinuclear fluorescence (Fig. 3i).
Representative S100 staining after 2 weeks with axonal regeneration in the fibrin conduits either with CM or seeded with different cell types. (a) Negative control group with CM only. (b) Fibrin conduit + rASCs. (c) Fibrin conduit + hASCs deep layer. (d) Fibrin conduit + hASCs superficial layer. (e) Fibrin conduit + rat SC-like cells. (f) Fibrin conduit + human SVF. (g) Fibrin conduit + rat SCs. (h) Representative image of PKH26-labeled cells at the tip of the regenerating axon front 2 weeks after rASC injection arranged in dense clusters (arrowhead) costaining S100 (stars). (i) The human-specific antimitochondrial staining 2 weeks after implantation of PKH26-negative SVF shows typical perinuclear localization (arrowheads). Nuclei were stained with DAPI (stars). Scale bars: 100 μm.
We observed a significant longer regeneration distance in the SC fibrin conduit group ( Overview from the results of histomorphometric analysis with PGP 9.5 (
MRI Analysis
Two weeks after implantation, we were able to visualize the graft as a small black outline, distal and proximal sutures, and small hyperintensity indicating the regenerating axon front of all cell types (Fig. 5). Taking all the groups pooled together, nerve growth as evaluated by radiology was significantly higher in animals 4 weeks (6.7 ± 1.5 mm, CI 2.9–10.6 mm) after implantation ( MRI showing the graft and the regenerating axon front. Representative sagittal (a) image of the femur in one rat 2 weeks after insertion of a fibrin conduit filled with hASCs from the superficial layer (T2 weighted with fat saturation = T2wFS). Axial image at the level of the regenerating axon front (b). Relevant structures magnified in the insets. The graft is visible (arrowheads) as a small black outline. The distal nerve stem shows typical hyperintensity of denervation (arrow). A small hyperintensity indicates the regenerating axon front (arrow outline). The circular outline of the graft in (b) is surrounded by minimal tissue edema. Scale bar: 1 cm (a). MRI analysis. (a) After pooling all the available data together, the mean length of the nerve-regenerating axon front significantly increased after 4 weeks (

Discussion
In this study, we successfully transplanted human stem cells in a nonautogenous setting for peripheral nerve regeneration and compared them with autogenous stem cells. Although human stem cells displayed inferior results compared with the autogenous treatment groups, positive trends could be observed from transplantation of hASCs from the deep abdominal layer. No significant improvement was found in the animals in which an empty fibrin conduit with CM only was used. Furthermore, we were able to implement the clinical 3T MRI scanner to monitor the efficacy of cellular therapy for all cell types, showing a strong correlation with the immunocytochemistry findings.
Our MRI analysis is in line with recent findings (2,5, 16,17). In a study by Liao et al., the longitudinal changes of nerve repair in rats after tissue-engineered construct implantation was monitored with MRI (17). They were able to show that nerves implanted with mesenchymal stem cell (MSC)-seeded tubes achieved better functional recovery and enhanced nerve regeneration, while showing a slower return to baseline T2 relaxation time and a more rapid decline in gadofluorine M enhancement compared with nerves implanted with chitosan tubes alone (17). Nevertheless, there is a paucity of data about tracking the efficacy of transplanted stem cells to treat acute peripheral nerve injury.
It has been shown that in vitro expanded ASCs retain a low immunogenicity and immunosuppressive effect, and prostaglandin E2 might be the major soluble factor involved in the in vivo inhibition of allogeneic lymphocyte reaction (3). Further in vivo studies suggested that ASCs have immunosuppressive properties of inhibiting mixed lymphocyte proliferation, which may share the same immunosuppressive mechanism with MSCs (19,32). Thus, in our study, it was decided to perform xenogenic transplantation experiments as a model of maximal HLA mismatch, since no allogenic model is available for human cells due to ethical reasons (22). From a clinical point of view, no severe adverse events were observed during the course of the experiment related to the transplantation of hASCs using an immunocompetent host with a functional immune system. This observation goes in line with recent published studies, which describe the absence of immunological rejection toward xenograft stem cells (22). Furthermore, no tumor formation was observed in the area of the transplanted cells in histology and radiology.
Owing to the technically demanding procedure and analysis of cryosections, the three best arithmetic mean values from three animals per group could be included in the histomorphometric analysis. Taking our results together, human ASCs seem to lead to inferior results when compared to autogenous ASCs. Despite the fact that better results were observed in the human stem cell groups compared with empty controls, this effect seems also related to the fibrin conduit, since biodegradable fibrin conduits have been shown to promote neuronal regeneration following peripheral nerve injury (24,25). Importantly, cell viability has been maintained after 72-h culture in a fibrin matrix (10).
PKH26-positive cells were observed after 2 weeks at the tip of the regenerating axon front coexpressing S100, which suggests transdifferentiation of our transplanted cells. However, due to the “leaky” nature of this dye, caution should be applied (14,28). Nevertheless, only little microenvironmental contamination can be expected after 2 weeks (21). In our study, human cells were identified by human-specific antimitochondrial immunofluorescence, indicating cell viability. It is suggested that the injected cells may have a bystander effect, including the production of in situ molecules, which, directly or indirectly through a cross-talk with local glial cells, may modulate the local environment with the downregulation of inflammation and the promotion of axonal regeneration (19).
In our study, rASCs were harvested from the neck because they may be the ideal stem cell sources for tissue engineering approaches for the regeneration of nervous tissue (6). Biopsies for the isolation of hASCs were selected from the superficial and deep abdominal layer because ASCs from both layers express high levels of the neurotrophic factors nerve growth factor, brain-derived neurotrophic factor, glial cell-derived neurotrophic factor, and neurotrophin-3 (12). Furthermore, it has been reported in vitro that hASCs from the superficial layer significantly induced enhanced neurite outgrowth from NG108–15 motor neuron-like cells when compared with the deep layer cells (12). However, this was not confirmed in our study. ASCs from the deep layer showed a more favorable outcome as evidenced by a longer regeneration distance. The reason for the different outcomes might be the use of hASCs in rats.
Our results with the SVF group are not in line with the current literature (20). SVF is not a fully defined cell population and is known to consist of various types of cells, including fibroblasts, immune cells, pericytes, endothelial cells, adipocyte progenitors, undefined stem cells, as well as stromal cells (7). It has the advantage of reduction in the interval from tissue collection until cell injection and simplicity of laboratory procedure, especially where a traumatic injury is concerned (20). The stromal cells have a potential to differentiate into several lineages and are characterized by their specific markers (26,27). Nevertheless, SVF contains other nonimmunoprivileged cells, the frequency of stromal cells is much less than hematopoietic stem and progenitor cells in the bone marrow, and there is a lack of sufficient neurotrophic factors (9).
Conclusion
We successfully transplanted human stem cells in a nonautogenous setting and compared them with autogenous rASCs for early peripheral nerve regeneration. Furthermore, we were able to implement the clinical 3T MRI scanner to monitor the efficacy of cellular therapy over time. A strong correlation was found comparing radiographic and histological results.
Footnotes
Acknowledgments
