Abstract
A novel approach of making a biomimetic nerve conduit was established by seeding adipose-derived adult stem cells (ADSCs) on the external wall of porous poly(D, L-lactic acid) (PLA) nerve conduits. The PLA conduits were fabricated using gas foaming salt and solvent–nonsolvent phase conversion. We examined the effect of two different porous structures (GS and GL) on ADSC growth and proliferation. The GS conduits had better structural stability, permeability, and porosity, as well as better cell viability at 4, 7, and 10 days. The epineuriallike tissue was grown from ADSC-seeded conduits cultured for 7 days in vitro and then implanted into 10-mm rat sciatic nerve defects for evaluation. The regeneration capacity and functional recovery were evaluated by histological staining, electrophysiology, walking track, and functional gait analysis after 6 weeks of implantation. Experimental data indicated that the autograft and ADSC-seeded GS conduits had better functional recovery than the blank conduits and ADSC-seeded GL conduits. The area of regenerated nerve and number of myelinated axons quantified based on the histology also indicated that the autograft and AGS groups performed better than the other two groups. We suggested that ADSCs may interact with endogenous Schwann cells and release neurotrophic factors to promote peripheral nerve regeneration. The design of the conduit may be critical for producing a biohybrid nerve conduit and to provide an epineurial-like support.
Keywords
Introduction
The connective tissue of the peripheral nervous system contains epineurium, perineurium, endoneurium, and blood vessels. The nerve is packed by the connective tissue to form a bundle. Epineurium is the outer layer of the connective tissue surrounding the peripheral nerve, which is composed of the adipose tissue, collagen fibrils, and fibroblasts (11,36,39). After peripheral nerve injury, a nerve can be reconstructed at the proximal end. Proliferation of Schwann cells (SCs), macrophages, and monocytes works to remove myelin debris, release neurotrophic factors, and lead axons toward their synaptic targets, resulting in neuronal reconstruction (18). Collagen III and IV are found around the epineurium and perineurium in the regenerated nerve. The production and distribution of collagen III and IV have influence on the formation of scar tissue and regeneration of damaged peripheral nerve fibers (19).
Surgical treatments for peripheral nerve injury, such as end-to-end and fascicular suturing, are often applied for nerve gaps smaller than 5 mm. For a nerve gap greater than 5 mm, a graft or a nerve conduit is required for growth orientation (14). At present, nerve autografts, epineurial sheath, arteries, veins, and skeletal muscle tissue have served as the grafts for bridging peripheral nerve defects (9,26,34). Among them, autografts have yielded favorable results (21,34). The epineurial sheath as a conduit was reported to have similar in vivo results to that of nerve autografts at 28 weeks with 80% successful nerve regeneration (21). However, grafts for peripheral nerve reconstruction may cause donor site morbidity, and it may be difficult to find a suitable donor site in patients with diabetes or other chronic diseases. Artificial, biodegradable nerve conduits can segregate the ingrowth of fibroblasts. Bioactive conduits functionalized with neurotrophic factors or extracellular components can promote nerve regeneration. Common biodegradable polymers such as poly(glycolic acid) (PGA), poly(D, L-lactic acid) (PLA), poly(D, L-lactide-
Nerve conduits can be further combined with supportive cells like stem cells, such as neural stem cells (NSCs), mesenchymal stem cells (MSCs), and adipose-derived stem cells (ADSCs) (17,18,23,32,37). ADSCs are fibroblast-like in shape, express MSC markers (CD73, CD90, CD105, etc.), and have multilineage differentiation capacity [cardiomyocytes, adipocytes, chondrocytes, osteocytes, and neuron-like cells (2,22,37,44,45)]. ADSCs can be differentiated to a SC phenotype or be induced to form glia-like cells when cultured with neural induction medium (2). Following transplantation into sciatic nerves of laminin-deficient mutant mice, ADSCs expressed laminin and induced the myelination of axons from endogenous SCs (2,11,22). In addition, ADSCs could secrete paracrine factors such as vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) to stimulate collagen synthesis and migration of fibroblasts (31).
Schwann-like cells derived from ADSCs combined with nerve conduits could promote nerve regeneration, an effect similar to that of SCs (13,42,46). In some studies, ADSCs were directly seeded in the nerve conduits with the intent of soliciting their paracrine effects. The experimental results from conduits seeded with ADSCs, SCs, or Schwann-like cells showed similar efficacy in nerve regeneration (25,30,33,43).
In the literature on peripheral nerve regeneration, ADSCs are often introduced into the lumen of the conduits by direct injection (7,13,25,42), embedding in gels (gelatin, collagen, fibrin) (6,12,31), or dynamic seeding (5,17,20,24,29). In this study, we used a novel approach of making a biomimetic nerve conduit by seeding ADSCs on the external wall of porous PLA nerve conduits. The conduits were fabricated by phase inversion and gas foaming reaction. We predicted that the ADSC-seeded nerve conduit would mimic the function of an epineurial sheath and prove beneficial for nerve regeneration. The novel design was first optimized in vitro and then implanted into 10-mm rat sciatic nerve defects for evaluation.
Materials and Methods
Fabrication of PLA Substrates and Conduits
PLA conduits were fabricated using gas foaming salt and solvent–nonsolvent phase conversion. A 10% solution (w/w) of PLA with an average molecular weight about 180 kDa (8300D; Cargill Dow, Minnetonka, MN, USA) was prepared in 1,4-dioxane (Sigma-Aldrich, St. Louis, MO, USA). Ammonium bicarbonate (NH4HCO3; Choneye Pure Chemicals Ltd. Co., Taipei, Taiwan) was sieved and collected in two different particle sizes (< 44 μm or 44~125 μm). Each was added to the solution so the weight ratio of NH4HCO3 to PLA was 1:2. The polymer/salt solution was stirred for 2 h and then cast into the glass plate and processed differently to form porous substrates on the plate. First, the polymer/salt plate was placed in 95% alcohol at 4°C for 2 days, which served as the nonsolvent for solvent–nonsolvent phase conversion. The substrates were then immersed into 10% citric acid solution (Choneye Pure Chemicals) at room temperature to induce gas foaming within the polymer/salt matrices. After the completion of gas foaming, the substrates were taken out from the plate, washed with distilled water several times, and then dried in an oven at 37°C for 24 h. These substrates were abbreviated as GS and GL, each indicating the use of smaller salt particles (< 44 μm) and larger salt particles (44~125 μm). The PLA substrates (GS and GL) were sectioned into ~22 mm in length and rolled into conduits using a 1.5-mm-diameter mandrel with the edges adhered tightly using PLA solution.
Characterization of the PLA Substrates
The cross-sectional and surface structures of the PLA substrates were examined by a scanning electron microscope (SEM) (JSM-6360/LV; JOEL, Tokyo, Japan). The surface contact angle was determined by a contact angle meter (FTA175; First Ten Angstroms, St. Portsmouth, VA, USA). The dynamic stretch modulus of these substrates was measured using a dynamic mechanical analyzer (DMA2980; TA Instruments, New Castle, DE, USA) after the samples were rinsed in the phosphate-buffered saline (PBS) for 30 min. The analysis was performed at 1 Hz frequency under 37°C. The permeability of different substrates was characterized as described in the literature (2). A 1% solution of bovine serum albumin (BSA, molecular weight 62 kDa) was prepared in PBS. BSA solution (1.5 ml) was added to a tube, the top of which was sealed with the PLA substrate. The sealed tube and 3 ml of PBS were placed in a vial. To measure the BSA diffused through the PLA substrate at 3, 22, 48, 72, and 96 h, the solution was retrieved from the PBS vial and analyzed using a UV/Vis spectrophotometer at 280 nm (UV-160A, Shimadzu Co., Kyoto, Japan). The procedure was repeated on the reverse side of the PLA substrate to determine the directional permeability (inflow and outflow rates of diffusion).
Isolation and Cell Culture of Rat ADSCs
ADSCs were extracted from the subcutaneous fat positioned at the hind leg and side abdominal region of six male Sprague–Dawley rats purchased from the National Laboratory Animal Center (Taipei, Taiwan). Cells were enzymatically isolated as described in the literature (4,5). The adipose tissues were cut into several pieces and treated with 200 U/ml collagenase (type I; Sigma-Aldrich) in Hank's balanced salt solution (HBSS; Gibco, Grand Island, NY, USA) for 30 min at 37°C with gentle agitation. Cells were isolated by centrifugation at 500 ×
Seeding Rat ADSCs on the External Wall of the Conduit
The protocol of seeding rat ADSCs on the external wall of the conduit is shown in Figure 1. A hole was drilled into the cap of a 1.5-ml Eppendorf tube that was sealed with a 0.22-μm filter membrane (Millipore, Billerica, MA, USA) to allow for gas exchange. The substrates were rolled into conduits with the assistance of a 1.5-mm-diameter mandrel, and the ends of conduits were sealed to prevent entry of ADSCs.

The protocol for seeding ADSCs from the external surface of the nerve conduit. This protocol was intended for producing an epineurial-like hybrid for transplantation.
The Eppendorf tube was sterilized in an autoclave. The conduits were sterilized by 70% ethanol for 30 min and rinsed with PBS. Rat ADSCs (with 1 × 106 cells in 0.5 ml medium) were suspended in an Eppendorf tube containing a conduit, which was placed on a horizontal shaker (150 rpm) for 2 h in the incubator and followed by static culture for 4 h. The seeded conduits were then transferred to a 15-ml conical tube, and 8 ml medium was added. Half of the volume of medium was changed every day. The metabolic activities of rat ADSCs on the conduit was estimated using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT; Sigma-Aldrich) assay. After 4, 7, and 10 days of incubation, the seeded conduits were sectioned into a few pieces and placed in a 24-well tissue culture plate. MTT solution (300 ml) was added to each well. After 4 h, the insoluble purple formazan crystals were dissolved with 600 ml of dimethyl sulfoxide (DMSO; Sigma-Aldrich). The optical density (OD) was read by a microplate reader (Emax; Molecular Devices, Sunnyvale, CA, USA) at 550 nm. Cell attachment and proliferation were observed by a SEM at 4 and 10 days.
Gene Analysis
Sterilized PLA substrates were placed in 24-well tissue culture plates (Corning, Corning, NY, USA). Rat SCs (RSC96, ATCC: CRL-2765, 1 × 104 cells/well; ATTC, Manassas, VA, USA) were grown on the PLA substrates for 1 day. The substrates were then taken out, reversed, and positioned on a silicon O-ring so that the SCs faced down and ADSCs (1 × 104 cells/well) were subsequently seeded on the top side. ADSCs and SCs were cocultured on the two sides of the PLA substrate for 3 days. Cells were analyzed for neurotrophic gene expression by RT-PCR as previously described (4). For RT-PCR, total RNA was extracted from cells using TRIzol reagent (15596-018; Invitrogen, Bangalore, India) after the cells were trypsinized by 0.05% trypsin/EDTA (Gibco). Total RNA was used for reverse transcription with a first-strand cDNA synthesis kit (Fermentas, Thermo Fisher Scientific, Waltham, MA, USA). The cDNA reaction mixture was applied in each PCR reaction. The PCR reaction was performed using selective forward and reverse primers for glyceraldehyde 3-phosphate dehydrogenase (GAPDH, used as an internal standard), nerve growth factor (NGF), glial cell-derived neurotrophic factor (GDNF), brain-derived neurotrophic factor (BDNF), and collagen type I. The sequences of the primers are shown in Supplemental Table S1 (supplementary material available at: https://www.dropbox.com/s/tw0ptakda9j1qet/CT-1439.R1-Supplemental%20data.doc?dl=0).
Surgery
For tracking cells, rat ADSCs were labeled by DiI (CellTracker™ CM-DiI, C7000; Invitrogen) at a concentration of 2.5 mg/ml in medium for 5 min. Before implantation, two experimental groups were seeded with ADSCs from the outside of the conduit (abbreviated as AGS and AGL) and were incubated for 7 days. The other two groups received reversed autografts and nonseeded GS conduits. Twenty-four male Sprague–Dawley rats weighing around 250–300 g (aged about 56–70 days) were used for the animal test (
Animals were deeply anesthetized with sodium pentobarbital (30 mg/kg, IP; TCI, Tokyo, Japan) throughout the surgical procedure. Surgery was performed on the left leg of the rat under aseptic conditions. A 10-mm-long segment of the sciatic nerve was transected, and the nerve stumps were anchored in a conduit (12 mm long), using 9-0 nylon microsutures to join the perineurium of the two stumps, and were sutured into the conduit wall. After nerve surgery, the wound was closed in layers with 4-0 Dexon sutures. The animals were housed in temperature-(22°C) and humidity (45%)-controlled rooms with 12-h light cycles. All procedures followed the ethical guidelines of and were approved by the Animal Care and Use Committee of the Central Taiwan University of Science and Technology (Taichung, Taiwan).
Functional Assessment of Nerve Regeneration
The walking track was recorded on all animals weekly before the animals were sacrificed at 6 weeks as previously reported (1,8). The rats' hind feet were dipped in ink, and the rats were allowed to walk across a plastic tunnel so that the footprints were recorded on paper positioned on the bottom of the tunnel. For the calculation of sciatic functional index (SFI), the following footprint parameters were measured: print length (PL) is the distance from the heel to the top of the third toe, toe spread (TS) is the distance of the one to five toe, and intermedian toe spread (IT) is the distance of the two to four toe. Data were collected for both the control group (N, normal rat,
Electrophysiological Study
Rats were anesthetized by sodium pentobarbital (4%, 40 mg/kg; Sigma-Aldrich), and the regenerated sciatic nerves were carefully exposed at 6 weeks postimplantation. The needle electrode, which was connected with a DC electrical stimulator (PowerLab ML866; AD Instrument, Castle Hill, Australia), was placed on the proximal end of the nerve. The compound muscle action potential (CMAP) was recorded in the gastrocnemius muscle with an active monopolar needle electrode 15 mm below the tibia tubercle and with a reference needle 20 mm from the active electrode. The stimulation voltage was generally from 1 to 2 V, 2 Hz pulse, and 0.2 ms duration. The nerve conduction velocity (NCV) was obtained from the CMAPs recorded by computer software (Scope for Windows; AD Instrument). The NCV across the regenerated nerve was calculated by evoking the compound action potential and dividing the distance between electrodes by conduction latency.
Histological Examination
Six weeks after implantation, the rats were euthanatized by CO2 overdose, and the regenerated nerves within the tubes were harvested. The nerve grafts were fixed in 10% neutral buffered formalin, embedded in paraffin wax, and thin sectioned (4 mm). The sections were stained with hematoxylin and eosin (H&E; Muto Pure Chemicals, Tokyo, Japan) for histological analysis and S-100 for the presence of SCs. For immunohistochemical staining of S-100, the sections were stained with S-100 antibody (1:200 dilution, NCL-S100p; Leica Biosystems, Breckland, UK) using an HRP-conjugated compact polymer system and visualized via 3,3′-diaminobenzidine (DAB; Immunotech, Marseille, France) staining. In addition, the nerve grafts were fixed immediately in a cold buffered 3% glutaraldehyde solution (Acros Organics, Geel, Belgium). After fixation, these nerve tissues were washed in PBS, postfixed and stained with 1% osmium tetroxide (Polysciences, Warrington, PA, USA), dehydrated, and embedded in paraffin. The paraffin sections (4 mm) were stained by Heidenhain-Woelck's hematoxylin method (15) for myelin. All nerve sections were observed under a light microscope (BX51; Olympus, Tokyo, Japan), and photographs were taken using a digital camera. The images of the histological sections were digitized, and subsequently the area of the regenerated nerve was analyzed, and the number of myelinated axons and blood vessels were calculated using an image analysis software (Image-Pro Lite).
To track DiI-labeled cells, specimens were immediately embedded in an OCT compound before sectioning (10 μm thick) at −30°C. A fluorescence microscope (Nikon Eclipse 80i, Nikon, Tokyo, Japan) was used to locate the DiI-labeled cells.
Statistical Analysis
Data from the experiments were expressed as mean ± standard deviation. Statistical differences were analyzed by ANOVA (Microsoft Excel; Microsoft, Redmond, WA, USA). The Tukey–Kramer post hoc test was used for multiple group comparisons to determine the statistical significance of the results. A level of
Results
Physicochemical Properties of the PLA Substrates
SEM images of the microstructure of two different porous PLA substrates (GS and GL) are shown in Figure 2. Both substrates showed an asymmetrical porous structure. There were macropores (30~40 mm) and micropores (5~10 mm) in each substrate. GL had more macropores than GS. The top surface of GL and GS had a dense skin layer, but the bottom of GL did not.

SEM images showing the microstructure of the PLA substrates (GL and GS). The substrates GS and GL represent those fabricated by the use of smaller salt particles (< 44 μm) and larger salt particles (44~125 mm), respectively. Both substrates showed asymmetric porous structure. The top surface of GL and GS had a dense skin layer, but the bottom of GL did not have a dense skin layer. Cross-section (150×, 500×), top, and bottom (1,000×).
The mechanical properties of PLA substrates are listed in Table 1. The storage modulus of GL significantly decreased after wetting. On the other hand, the storage modulus of GS remained similar after wetting, indicating a more stable structure of GS. The permeabilities of GL and GS are summarized in Supplemental Table S2 (supplementary material available at: https://www.dropbox.com/s/tw0ptakda9j1qet/CT-1439.R1-Supplemental%20data.doc?dl=0). GS had better permeability than GL during a period of 96 h. GS also had a higher water content and degradation rate in 14 and 28 days (in Supplemental Table S2). The data suggested that GS may be a better candidate for making a biohybrid nerve conduit.
Mechanical Properties of PLA Substrates

Cell Studies In Vitro
The viability of cells seeded on GS and GL substrates at 4, 7, and 10 days after transplantation is shown in Figure 3A. Cells proliferated from 4 to 7 days in both groups. From 7 to 10 days, the cell viability decreased, possibly because cells had already reached maximal confluence at 7 days. As evident from the cell images at 10 days, cells on GS substrates had already stacked together (Fig. 3B).

(A) MTT analysis for the viability of ADSCs cultured on GS and GL conduits after 4, 7, and 10 days. *
ADSCs and SCs were cocultured on the two sides of the PLA substrates to simulate the environment inhabited by ADSCs after conduit implantation. The gene expression of cocultured ADSCs and SCs at 3 days is demonstrated in Supplemental Table S3 (supplementary material available at: https://www.dropbox.com/s/tw0ptakda9j1qet/CT-1439.R1-Supplemental%20data.doc?dl=0). The gene expression of coculture groups was similar to that of monocultured SCs. The results seem to indicate that ADSCs seeded on the conduit may produce extracellular matrix (ECM) proteins, such as collagen type I, and neurotrophic factors. The former may help form epineurial-like tissue, and the latter may be conducive to nerve regeneration.
For the aforementioned reasons, we selected ADSC-seeded GS and ADSC-seeded GL conduits (7 days) as the tissue engineering conduits for further evaluation (abbreviated as AGS and AGL, respectively). Autografts and nonseeded GS conduits were used for comparison.
In Vivo Animal Implantation
Results from gait analysis are shown in Figure 4. At 2 weeks, the group receiving AGS had higher SFI than those receiving AGL and GS. At 4, 5, and 6 weeks, the group receiving AGS had higher SFI than those receiving GS. The knee motion angle revealed significant increases from 2 to 6 weeks only in the autograft group and the group receiving AGS. In the groups receiving AGL and GS, the increase of the angle from 2 to 6 weeks was less evident. Considering the fact that the normal rats had a value ~26°, the animals receiving autogafts and AGS had better functional recovery in general.

Functional recovery of the experimental animals by the walking track analysis (sciatic function index SFI and knee motion angle) during a period of 6 weeks. *
The electrophysiological data collected at 6 weeks (before sacrifice) are demonstrated in Figure 5A. The results in Figure 5B showed that the autograft group had higher amplitudes than the AGS group, followed by the AGL and GS groups. Higher amplitudes are indicative of motor nerve conduction gradually recovering. The NCV value (Fig. 5C) in the autograft and AGS groups was greater than that in the AGL and GS groups. These data indicated that the autograft and AGS groups had better functional recovery than the other two groups.

Electrophysiological measurement of the experimental animals after the conduit implantation in vivo for 6 weeks: the corresponding (A) electrophysiological recordings of compound muscle action potential (CMAP) and the (B) amplitude and (C) nerve conduction velocity (NCV) obtained from CMAP. *
Tracking of DiI-labeled ADSCs in the AGS and AGL groups after 6 weeks in vivo is demonstrated in Figure 6. It was apparent that ADSCs had already migrated from the external surface to the inner porous structure of the conduit and further into the lumen of the conduit (~250 μm inward from the inner border of the tube). In particular, cells in AGS were more abundant and uniformly distributed near the conduit, that is, forming an epineurial-like tissue.

The presence of DiI-labeled ADSCs in vitro after 7 days and in the histological longitudinal sections after 6 weeks of implantation. DiI-labeled ADSCs had migrated from the external surface of the conduit to the inner porous structure of the conduit and further into the lumen of the conduit (~250 μm further from the border of the conduit). A: ADSCs; conduit: the nerve conduit; lumen: the lumen of the conduit.
The H&E-stained cross-sections of the regenerated nerve are shown in Figure 7A. The area of regenerated nerve, number of myelinated axons, and number of blood vessels quantified based on the histology are demonstrated in Figure 7B. The area of the regenerated nerve was the largest in autografts followed by AGS and finally AGL and GS. The number of myelinated axons was the greatest in autografts followed by AGS, AGL, and GS. On the other hand, the number of blood vessels was the smallest in the GS group. Larger images for the histology of the regenerated nerve are shown in Figure 8. The myelinated axons were self-assembled in the autograft and AGS groups, forming endoneurial-like tissue. The S-100 staining (for SCs) is displayed in Figure 9. SCs were distributed close to the central axis of the lumen (i.e., regenerated nerve) and a distance away from the conduit. AGS had the darkest stain as evident by the longitudinal sections as well as the cross-sections.

Histology of the nerve section at the midconduit after 6 weeks in vivo. (A) Images of H&E stained sections where areas inside the smaller inner circles indicate regenerated nerve while areas between the inner and outer circles indicate the connective tissue. (B) Quantitative data for the area of regenerated nerve, number of myelinated axons, and number of blood vessels. Scale bars: 200 μm, *
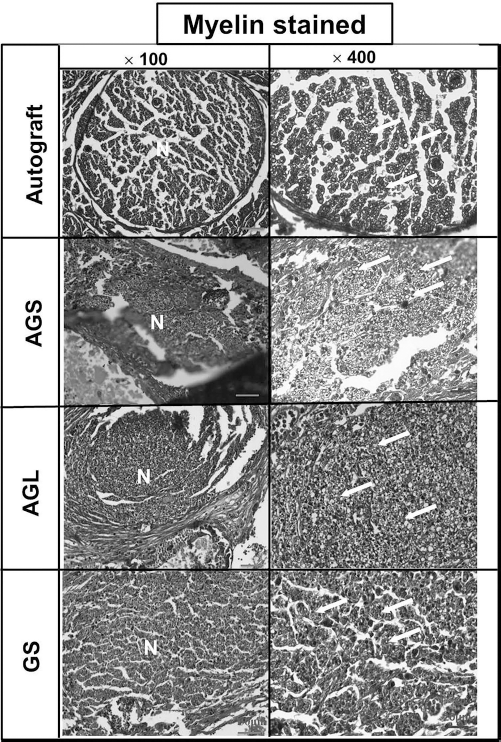
Histology of the nerve section at the midconduit after 6 weeks in vivo, stained with osmium tetroxide and Heidenhain-Woelck's hematoxylin for myelins (100×, 400×). N: area of regenerated nerve. Arrows: myelinated axons.

Histology of the nerve section at the midconduit after 6 weeks in vivo, stained with S-100 for SCs. It was noted that S-100-positive SCs were distributed close to the central axis of the lumen (longitudinal sections for AGS, AGL, and GS, 40×) and still a distance (~350 μm) away from the border of the conduit. The cross-section (100×, 200×) for AGS was also demonstrated. C: conduit; arrows: myelinated axons.
Taken together, the data suggest that seeding ADSCs on the external surface of AGS could provide an epineurial-like support that promotes nerve regeneration as compared to a conduit (GS) without cells. The conduit structure (GS instead of GL) may determine if the ADSCs successfully colonize on the conduit and form an epineurial-like structure (in Supplemental Fig. S1; supplementary material available at: https://www.dropbox.com/s/tw0ptakda9j1qet/CT-1439.R1-Supplemental%20data.doc?dl=0).
Discussion
Previous studies have shown that peripheral nerve repair can be enhanced by SC transplantation (10,27,35), but clinical application is limited because of the difficulty in obtaining a sufficient number of cells. MSCs are an attractive cell source for nerve regeneration. They are able to self-renew with a high growth rate and possess multipotent differentiation properties (6,7,23,42,45). MSCs, including ADSCs, have been used to repair peripheral nerves. In some studies, MSCs were differentiated into Schwann-like cells and combined with nerve conduits (6,7,42,46). In the other studies, MSCs were either directly seeded inside nerve conduits (25,30,33,43) or cocultured with SCs in nerve conduits (5), with the hope that they would secrete paracrine factors that would stimulate nerve regeneration.
It has been shown that Schwann-like cell differentiation of MSCs could be induced by using chemicals combined with a mixture of glial growth factors (7,13,42,46). Differentiated bone marrow-derived mesenchymal stem cells (BM-MSCs) or ADSCs seeded in conduits have been shown to promote nerve regeneration, in a manner similar to that attained by using SCs (13,42,46). Nerve conduits seeded with Schwann-like cells were used to bridge 15-mm rat sciatic nerves, and at 12 weeks, the number of myelinated axons was about 2,700–3,100 (42). However, the aforementioned induction methods for obtaining differentiated cells are rather complicated and time-consuming.
Many studies have confirmed the regenerative potential of nondifferentiated ADSCs in peripheral nerve repair and functional recovery of exercise behavior. For a 10-mm gap, differentiated and nondifferentiated ADSC-seeded conduits produced regenerative effects similar to those attained using SCs after 24 weeks (30,43). Nondifferentiated ADSCs directly introduced into the nerve conduits could promote sciatic nerve repair across a 10-mm gap after 6–10 weeks (25,33). In our study, the surface markers for ADSCs were identified by flow cytometric analysis of cluster of differentiation antibodies (CD29+, CD31-, CD34-, CD44+, CD45-, CD73+, CD90+, and CD105+). The surface marker expression of ADSCs was consistent with that of MSCs in the literature (5,40). Compared to the work of Shen et al. (33), who seeded ADSCs inside nerve gelatin–tricalcium phosphate conduits, our epineurial-like conduit (AGS) had a greater number of myelinated axons (>6,000 at 6 weeks vs. ~4,000 at 8 weeks). Moreover, our epineurial-like conduit (AGS) resulted in a higher nerve conduction velocity (42.7 m/s, 6 weeks) than that reported for acellular nerve allografts containing nondifferentiated ADSCs in the lumen (12.45 m/s, 12 weeks) (25).
In all of the previously published methods, cells were introduced into the lumen of the conduits by direct injection (7,13,25,33,42), embedding in gels (gelatin, collagen, fibrin) (5,12,30), or dynamic seeding (5,18,24,28,29). Cells were either filled inside the nerve conduit or attached on the inner surface. Unlike the previous approaches, we seeded ADSCs from the outside of the conduit, intending to produce an epineurial-like tissue for nerve regeneration. Compared to earlier studies (5,33) in which ADSCs were introduced into the lumen of the conduits by direct injection, our study showed higher NCVs (33) and better morphology of regenerated nerves manifested by more myelinated axons and a larger cross-sectional area (5). For ADSC-seeded conduits cultured for 7 days in vitro, a portion of the cells moved into the porous wall, while other ADSCs attached on the outer surface. Since the porous structure and surface dense skin layer may significantly affect the cell ingrowth, we examined the effect of two different porous structures (GS and GL) on ADSC growth and proliferation. GS had better structural stability, permeability, and porosity. When ADSCs were seeded on GS, the cell growth and cell viability were greater than that observed on GL. The AGS conduits may have favorable effects on the metabolic exchange, which may also enhance the diffusion of growth-promoting factors. In a previous study (5), coculture of SCs and ADSCs resulted in an upregulation in the expression of BDNF, GDNF, and NGF genes as time over time. Cocultured SCs and ADSCs at 7 days showed greater NGF gene expression than ADSCs or SCs alone. Based on the results of the present study, it appears that SCs secreted more neurotrophic factors than ADSCs. Moreover, Kingham et al. (22) indicated that ADSCs could differentiate into Schwann-like cells in vitro. Others found that ADSCs did not differentiate into SCs after implantation (38). In another study, transplanted ADSCs were differentiated into nonmyelinating SCs after 31 days (40). In our study, DiI-labeled ADSCs were found close to the nerve conduits, while S-100-positive SCs were predominantly observed in the neural tissue close to the center of the lumen but not in the epineurial-like structure of the conduit (Figs. 6 and 9). This observation indicated that ADSCs did not or had not differentiated into SCs after 6 weeks. Based on the literature (5,40), we assumed that ADSCs might interact with endogenous SCs and release neurotrophic factors. This assumption was substantiated by the better efficacy of AGS versus AGL. It was also evident that the design of a nerve conduit was important for producing a biohybrid nerve conduit that could provide epineurial-like support.
In summary, this study established a new concept in peripheral nerve regeneration. Nondifferentiated ADSCs were seeded from the outside of the conduit to produce an epineurial-like biohybrid nerve conduit. Investigation remains to be conducted regarding the interaction of ADSCs and endogenous SCs, the possible cytokine effect, the feasibility in bridging a longer gap, and the long-term results for future clinical applications.
Footnotes
Acknowledgments
