Abstract
Cell transplantation is a useful therapy for treating peripheral nerve injuries. The clinical use of Schwann cells (SCs), however, is limited because of their limited availability. An emerging solution to promote nerve regeneration is to apply injured nerves with stem cells derived from various tissues. In this study, different types of allogeneic cells including SCs, adipose-derived adult stem cells (ASCs), dental pulp stem cells (DPSCs), and the combination of SCs with ASCs or DPSCs were seeded on nerve conduits to test their efficacy in repairing a 15-mm-long critical gap defect of rat sciatic nerve. The regeneration capacity and functional recovery were evaluated by the histological staining, electrophysiology, walking track, and functional gait analysis after 8 weeks of implantation. An in vitro study was also performed to verify if the combination of cells led to syner-gistic neurotrophic effects (NGF, BDNF, and GDNF). Experimental rats receiving conduits seeded with a combination of SCs and ASCs had the greatest functional recovery, as evaluated by the walking track, functional gait, nerve conduction velocity (NCV), and histological analysis. Conduits seeded with cells were always superior to the blank conduits without cells. Regarding NCV and the number of blood vessels, conduits seeded with SCs and DPSCs exhibited better values than those seeded with DPSCs only. Results from the in vitro study confirmed the synergistic NGF production from the coculture of SCs and ASCs. It was concluded that coculture of SCs with ASCs or DPSCs in a conduit promoted peripheral nerve regeneration over a critical gap defect.
Keywords
Introduction
Peripheral nerve injuries, often caused by trauma, are treated extensively under surgical intervention. The surgical technique named as “gold standard” refers to the use of autologous nerve in bridging a critical gap defect but still has some drawbacks such as the secondary surgery, painful neuroma formation, and difficulty in obtaining nerve of suitable size from patients (2,4,36). Besides, the nerve functionality may not be completely recovered due to several factors, including the loss of targeted tissue or the longer nerve gap.
Schwann cells (SCs) can participate in remyelination of the bridged nerves (5). These cells have been demonstrated to promote regrowth of injured axons and to cause the functional recovery based on animal models. The addition of exogenous SCs can also guide the axonal regrowth across the nerve gap (11,31). Although transplantation of SCs has been considered as a biologically effective strategy (16,17), it also has disadvantages such as the limited expansion potential and lengthy culture times of SCs as well as the possible morbidity associated with nerve harvested to obtain a sufficient number of SCs for transplantation. Therefore, a number of stem cells have been explored as alternatives for use in nerve regeneration (27). Adipose-derived stem cells (ASCs) have been shown to successfully differentiate into neuronal lineages and SCs in vitro and may be of benefit for treatment of peripheral nerve injuries (13,24,51). This may be also related to the fact that a number of nerve growth factors, including insulinlike growth factor (IGF) and fibroblast growth factor (FGF), are secreted by ASCs (37,55). Dental pulp consists of ecto-mesenchymal components, containing neural crest-derived cells, which display plasticity and multipotential capabilities (43). Dental pulp stem cells (DPSCs) also have the ability to differentiate into neuronal-like cells, which makes them a potential alternative for the treatment of neurological disease (3,22,29,30). Like ASCs, DPSCs also produce neurotrophic factors, including nerve growth factor (NGF), glial cell line–derived neurotrophic factor (GDNF), and brain-derived neurotrophic factor (BDNF) (32). Recently, the coculture system of SCs and stem cells was also reported. The coculture of SCs and ASCs induced the ASCs to differentiate into Schwann-like cells in vitro, which may help create a microenvironment that promotes nerve regeneration (49). However, there is little study regarding the synergistic effect of SCs and stem cells on peripheral nerve regeneration in vivo.
Artificial nerve conduits have been used to replace autografts in the peripheral nerve surgery. Poly(d,l-lactide) (PLA) is easily handled with proper biodegradation rate and a low inflammatory response (46). Novel microporous PLA conduits were previously developed to promote peripheral nerve regeneration (20,21). In this study, the candidate cells including SCs, ASCs, DPSCs, and the combination of SCs with ASCs or DPSCs were analyzed in vitro for neurotrophic gene expression by RT-PCR at 0, 3, and 7 days and protein expression by ELISA at 7 days. The ability of cell-seeded conduits to regenerate rat sciatic nerve over a 15-mm-long defect was evaluated. Histological staining, functional gait analysis, and electrophysiology were used to contrast the effects on nerve regeneration from different types of cells.
Materials and Methods
Isolation and Culture of ASCs
All protocols involving the use of experimental animals were approved by the institutional review board. Isolation of rat ASCs followed that described in literature (6). ASCs were extracted from the subcutaneous fat positioned at the hind leg and side abdominal region of two female Sprague– Dawley rats (weight from 350 to 500 g) purchased from the National Laboratory Animal Center (Taipei, Taiwan). The adipose tissues were minced and added with 200 U/ml type I collagenase (Sigma-Aldrich, St. Louis, MO, USA) in phosphate-buffered saline (PBS; Invitrogen, Carlsbad, CA, USA) for degradation at 37°C with gentle agitation for 30 min. Following mechanical and enzymatic treatment, the tissues were homogenized in the medium containing low-glucose Dulbecco's modified Eagle's medium/ Ham's F12 (1:1) (Gibco, Gaithersburg, MD, USA) supplemented with 10% (v/v) fetal bovine serum (FBS; Gibco), 100 U/ml penicillin, and 100 mg/ml streptomycin (Gibco). After homogenization, cells were cultured in an incubator (37°C/5% CO2, 95% relative humidity). The culture medium was refreshed two times every week. Cells of the 2nd to 5th passages were used in this study.
Isolation and Culture of DPSCs
Rat DPSCs were isolated according to a method modified from a previous study (54). Rat dental pulp was obtained from the mandibular incisors using two female 2-weekold neonatal Sprague-Dawley rats purchased from the National Laboratory Animal Center and washed two times with calcium- and magnesium-free PBS (Gibco). The teeth were cut into several pieces and incubated with gentle shaking at 37°C for 60 min in 3 ml of a sterile enzyme solution containing 0.3% collagenase type IA (Sigma) and dipase II (Roche, Basel, Switzerland) in PBS. The digested medium containing DPSCs was filtered by a 40-μM mesh (BD Falcon, Becton-Dickinson, Heidelberg, Germany) and washed with PBS three times. The cell pellets were resuspended with Eagle's α-minimal essential medium (α-MEM) (Invitrogen) supplemented with 10% FBS and 100 U/ml penicillin–streptomycin, and cultured in T75 flasks (BD Falcon) in the incubator. The culture medium was refreshed every 2 days. Cells of the 2nd to 5th passages were used in this study.
Culture of SCs
Rat SCs derived from culture of rat primary cells (RSC96, ATCC: CRL-2765, Manassas, VA, USA) were purchased and cultured in T75 flasks with Dulbecco's modified Eagle's medium (containing 4 mM l-glutamine, 1.5 g/L sodium bicarbonate, and 4.5 g/L glucose; Gibco), supplemented with 100 U/ml penicillin, 100 mg/ml streptomycin, and 10% FBS in an incubator. The culture medium was refreshed every 2 days. Cells of the 2nd to 5th passages were used in this study.
Analysis of Surface Markers and Cell Morphology
The surface markers for ASCs and DPSCs were identified by flow cytometric analysis of cluster of differentiation 29 (CD29), CD31, CD34, CD44, CD45, CD105 (all from BioLegend, San Diego, CA, USA), CD73 (BD Pharmingen, San Diego, CA, USA), and CD90 (Serotec, Raleigh, NC, USA) antibodies. A total of 5 × 105 cells were used and washed twice with PBS supplemented with 1% FBS. Cells were then resuspended in 100 ml of PBS/1% FBS and incubated with monoclonal antibodies for 30 min at 4°C, before further washing with PBS. Fluorescein isothiocyanate (FITC)-conjugated CD29 (CD29-FITC), phycoerythrin (PE)-conjugated CD31 (CD31-PE), CD44-FITC, and CD90-PE were operated for direct staining; mouse monoclonal anti-CD34 (Santa Cruz Biotechnology), mouse monoclonal anti-CD45 (Santa Cruz Biotechnology), mouse monoclonal anti-CD73 (BD Biosciences), and mouse monoclonal anti-CD105 (BD Biosciences) were purchased for indirect staining. The corresponding isotypic antibodies including mIgG1-FITC and mIgG1-PE (Serotec) were used for direct staining. Secondary antibodies applied in this study were FITC-conjugated goat-anti-mouse antibody (Chemicon). The final concentration was the same between the isotype controls and test antibodies. Fluorescence intensities were determined by a flow cytometer (FACSCalibur, BD, Franklin Lakes, NJ, USA).
Additionally, the cellular morphology of SCs, ASCs, and DPSCs was observed using an inverted optical microscope (Leica DMIRB, Wetzlar, Germany).
Coculture of SCs with ASCs or DPSCs
SCs and ASCs (or DPSCs) at 1:1 ratio (1 × 104 cells/cm2) were mixed and seeded on six-well culture plates (Nunc, Penfield, NY, USA). SCs, ASCs, and DPSCs (2 × 104 cells/cm2) were also seeded as controls. Cocultured cells were analyzed for neurotrophic gene expression by RT-PCR at 0, 3, and 7 days and protein expression by ELISA at 7 days.
For RT-PCR, total RNA was extracted from cells using Trizol reagent (15596-018, Invitrogen, Bangalore, India) after the cells were trypsinized by 0.05% trypsin/EDTA (Gibco). Five micrograms of total RNA was used for reverse transcription with the first-strand cDNA synthesis kit (Fermentas, Waltham, MA, USA). One microliter of the cDNA reaction mixture was applied in each PCR reaction. The PCR reaction was performed using selective forward and reverse primers for β-actin (as an internal standard), GDNF, BDNF, and NGF. The sequences of the primers used were as follows: GDNF forward, 5′-CCAGAGAAT TCCAGAGGGAAAGGTC-3′; GDNF reverse, 5′-CAG ATACATCCACACCGTTTAGCGG-3′; BDNF forward, 5′-AGTGGGCAAAGGAGCGG-3′; BDNF reverse, 5′-CG CTCATTCATTAGAATCACGT-3′; NGF forward, 5′-TCA CTGTGGACCCCAAACT-3′; NGF reverse, 5′-TCCTGT GAGTCCTGTTGAAGG-3′; β-actin forward, 5′-GTATG CCTCTGGTCGTACCA-3′; β-actin reverse, 5′-CTTCTG CATCCTGTCAGC AA-3′. PCR was performed in a GeneAmp PCR system 2700 thermal cycler (Applied Biosystems, Foster City, CA, USA) for 35 cycles. Cycling parameters were 94°C for 30 s, 60°C (for BDNF and GDNF) or 55°C (for NGF and β-actin) for 30 s, and then 72°C for 30 s, followed by a final extension at 72°C for 7 min. β-Actin was used as an internal control for the semiquantitative analysis and to confirm the fidelity of the PCR reaction. The amplified products were determined by electrophoresis (Gelcompany, San Francisco, CA, USA) on 1.5% agarose-TAE [10 mM Tris (pH 7.5), 5.7% glacial acetic acid, and 1 mM EDTA] gels (Roche) and quantified by ethidium bromide (Sigma) staining.
For ELISA, the medium from each well was collected and analyzed using the ChemiKine sandwich ELISA kits (Chemicon, Chandlers Ford, Hampshire, UK) for NGF and Emax immunoassay systems (Promega, Madison, WI, USA) for GDNF and BDNF, according to the manufacturer's protocols. All samples were analyzed in triplicate, and the absorbance was measured at 450 nm (Hitachi F2500, Tokyo, Japan).
Fabrication of Nerve Conduits
Nerve conduits were fabricated as previously described (20,21). PLA (8300D, Cargill, Minneapolis, MN, USA) was dissolved in 1,4-dioxane solvent (10% concentration; Qiagen, Germantown, MD, USA) and cast on a dish. The dish (Phytotechnology Laboratories, Shawnee Mission, KS, USA) was then put in 40% alcohol for 24 h, replaced in water for 8 h, and dried in a 40°C oven for 24 h to remove any organic solvent residue. The cast membranes were rolled into conduits by a 1.5-mm-diameter mandrel, and the lateral ends were adhered tightly by a small amount of 1,4-dioxane solvent.
Animal Surgery
Prior to surgery, cells were seeded onto the inner wall of the conduits dynamically. A total of 1 × 106 cells were filled into conduits and sealed at both ends with agarose plugs (Roche). The constructs were placed on a roller that rotated at a fixed speed (0.16 rpm) at 37°C for 3 days (12), which enabled uniform attachment of cells on the lumen surface of the conduits. In the cocultured groups, each type of cells at half the number was seeded. Thirty adult male Sprague–Dawley rats (250-300 g), purchased from the National Laboratory Animal Center, were divided into six groups. Group 1 received blank conduits (without cells). Group 2 received conduits with SCs. Group 3 received conduits with ASCs. Group 4 received conduits with DPSCs. Group 5 received conduits with ASCs and SCs. Group 6 received conduits with DPSCs and SCs.
Rats were anesthetized with isoflurane (Baxter, Deerfield, IL, USA) during the surgery. The sciatic nerve was exposed by skin and muscle splitting incision. The left sciatic nerve was cut and removed near the obturator tendon in midthigh with the aid of an operation microscope. A 17-mm nerve conduit (1.53-mm inner diameter, 0.21-mm thickness) was used to bridge the 15-mm-long nerve gap. The conduit ends were left 1 mm for anchoring the proximal and distal nerve stumps by 7-0 nylon microsutures (UNIK Surgical Sutures Mfg. Co., Taipei, Taiwan). The wound was subsequently closed in layers using 2-0 Dexon sutures (UNIK) after surgical treatment. The animals were given food and water ad libitum and sacrificed at 8 weeks after surgery. The time schedule allowed sufficient time for nerve regeneration with respect to the length of the gap.
Walking Track Analysis
The walking track was recorded on all animals weekly before the animals were sacrificed at 8 weeks. Before the experiment, animals were trained to walk down in a 150 × 8-cm track set on a darkened enclosure. The sciatic functional index (SFI) used in evaluating the functional muscle reinnervation was obtained based on the walking track analysis (18). The equation was expressed as SFI = −38.3 (PLF) + 109.5 (TSF) + 13.3 (ITF) – 8.8, where PLF (print length function) = (experimental PL – normal PL)/normal PL; TSF (toe spread function) = (experimental TS – normal TS)/normal TS (first to fifth toe); and ITF (intermedian toe spread function) = (experimental IT – normal IT)/normal IT (second to fourth toe).
Functional Gait Analysis
The procedure for functional gait analysis followed the protocol described previously (34). First, digital videos of experimental rats were captured weekly from 1 to 8 weeks after sciatic nerve surgery. The photographs of gait cycle of walking movement were recorded from a confining walkway until the steady and continuous walking patterns were obtained for tested rats. Each individual frame was further analyzed by the Ulead Video Software (Torrance, CA, USA). The terminal stance phase angle of the rat gait cycle was calculated as the angle (in degrees) of the intersection of the lines extending from the knee to the ankle joint and from the ankle joint to the metatarsal head detected from the images. The angle was used as an index for the functional gait analysis.
Electrophysiological Examination
Rats were anesthetized by sodium pentobarbital (4%, 40 mg/kg; Sigma-Aldrich), and the regenerated sciatic nerves were carefully exposed at 8 weeks postimplantation. The nerve conduction velocity (NCV) was measured using a recording system (ADInstruments Pty. Ltd., Castle Hill, NSW, Australia), where the associated signals were recorded by the software for data capture and analysis. The NCV across the regenerated nerve was calculated by evoking the compound action potential and dividing the distance between electrodes by conduction latency (8).
Histological Analysis
The tissues of regenerative sciatic nerve were harvested after surgery and immediately fixed in cold 3% glutaraldehyde solution (Amresco, Solon, OH, USA). After 2 days, the nerve conduits were transected at medium segment. For successful connection of sciatic nerve in a conduit, a thin white tubular substance that connected the two anastomoses ends was observed. The success rate was determined for each group. Tissue samples with conduits were then washed in PBS. They were then postfixed in 1% osmium tetroxide (Polysciences, Warrington, PA, USA), dehydrated in the gradient concentrations of ethanol solutions, and finally embedded. The embedded samples were sliced into 3-μm-thick sections and then stained with 1% toluidine blue (Amresco), which did not stain the conduits. All sliced sections were observed under an optical microscope, and photographs were taken using a digital camera (Nikon H666L, Tokyo, Japan). The cross-sectional area of the regenerated nerve, as well as the number of myelinated axons and blood vessels, was calculated using an image analysis system (Image-Pro Lite, Media Cybernetics, Silver Spring, MD, USA).
Statistical Analysis
Data from the experiments were revealed as mean±standard deviation. Statistical differences were determined by Tukey–Kramer post hoc test used for multiple comparisons. A value of p < 0.05 was indicated as statistically significant.
Results
Morphology of Cultured Cells and Phenotypic Characterization of Stem Cells
The cells isolated showed typical fibroblast-like morphology. The surface markers for ASCs by flow cytometry were positive for specific antigen markers of mesenchymal stem cells including CD29 (95%), CD44 (93%), CD73 (96%), CD90 (94%), and CD105 (91%), whereas the DPSCs were also positive for CD29 (91%), CD44 (86%), CD73 (95%), CD90 (98%), and CD105 (89%). The expression of the endothelial cell or hematopoietic cell surface markers such as CD31 (7% for ASCs; 15% for DPSCs), CD34 (3% for ASCs; 5% for DPSCs), and CD45 (8% for ASCs; 4% for DPSCs) was considerable low. The expression of CD29, CD44, CD73, CD90, and CD105 was similar between ASCs and DPSCs. The expression of CD31 and CD34 was a little more for DPSCs compared to ASCs, while that of CD45 in DPSCs was a little less versus ASCs.
Expression of NGF, BDNF, and GDNF for Cocultured Cells In Vitro
Results from the RT-PCR analysis of the coculture in vitro are shown in Figure 1. Coculture of SCs and ASCs upregulated the expression of BDNF, GDNF, and NGF genes as time increased, versus ASCs or DPSCs alone. At 7 days, cocultured SCs and ASCs showed the greatest NGF gene expression level among all groups. In addition, the coculture of SCs and ASCs presented a similar level of GDNF gene expression as SCs alone, and both levels were greater than the other groups at 7 days. Coculture of SCs and DPSCs upregulated the expression of neurotrophic genes versus DPSCs alone, especially for BDNF. However, the expression levels of GDNF and NGF genes for cocultured SCs and DPSCs were still lower than those of SCs alone.
Neurotrophic gene expression of the five different cell culture groups. The gene expressions of brain-derived neurotrophic factor (BDNF), glial cell line-derived neurotrophic factor (GDNF), and nerve growth factor (NGF) from five cultures including Schwann cell (SCs), adipose-derived stem cells (ASCs), dental pulp stem cells (DPSCs), SC + ASC, and SC + DPSC determined by PCR amplification after 0, 3, and 7 days of incubation. The experiments were conducted in triplicate. *p < 0.05.
The results were further confirmed by the data from ELISA at 7 days of culture, which are shown in Figure 2. ASCs alone demonstrated the lowest levels of expression for all three neurotrophic factors. The cocultured SCs and ASCs showed a remarkable increase in NGF production, which took it to the highest level among all groups. Moreover, the NGF production from cocultured SCs and ASCs was much greater (~50% greater) than that from SCs alone. On the other hand, the coculture of SCs and DPSCs revealed the highest GDNF production, followed by cocultured SCs and ASCs or SCs alone. The coculture of SCs and DPSCs, as well as SCs alone, had the highest production of BDNF.

Neurotrophic protein expression of the five different cell culture groups. The concentrations of BDNF, GDNF, and NGF proteins produced from five cultures including SC, ASC, DPSC, SC + ASC, and SC + DPSC. The proteins were determined by the ELISA assay from culture medium in a six-well plate seeded with 5 × 105 cells per well after 0, 3, and 7 days of incubation. The experiments were conducted in triplicate. *p < 0.05.
Success Rate of Nerve Connection
The conduits were collected from the experimental rats after anesthetization at 8 weeks postimplantation. The connection of sciatic nerve was confirmed under the optical microscope. Conduits containing cells had a higher success rate (100%) than conduits without cells (80%).
Walking Track Analysis
According to walking track analysis, a slight variation in the SFI was observed from 1 to 4 weeks in all groups (Fig. 3). The gradual increase of SFI from 4 to 8 weeks was observed for all conduits with cells. Conduits seeded with cocultured SCs and ASCs were obviously superior after 5 weeks in the score of SFI to other groups. The mean recovery of sciatic nerve function at 8 weeks expressed as the percent recovery of SFI was as follows: 29.4% for SC + ASC, 24.9% for ASC, 24.3% for SC, 23.3% for DPSC, 23.2% for SC + DPSC, and 4.4% for conduits without cells. Therefore, the functional repair capacity of various groups at 8 weeks was in the order of SC + ASC > ASC ~ SC ~ DPSC ~ SC + DPSC > conduits without cells.

Walking track analysis following cell and conduit transplantation. The walking track analysis expressed as sciatic functional index (SFI) weekly at 1–8 weeks postsurgery for rats implanted with the blank conduits (“Conduit”), conduits seeded with SCs (“SC”), conduits seeded with ASCs (“ASC”), conduits seeded with DPSCs (“DPSC”), conduits seeded with cocultured SCs and ASCs (“SC + ASC”), and conduits seeded with cocultured SCs and DPSCs (“SC + DPSC”). The total number was the same among each group with cells. *p < 0.05 with respect to the other groups.
Functional Gait Analysis
The results from functional gait analysis for each experimental group are shown in Figure 4. The maximal terminal stance phase angle exhibited similar trends of increase for conduits with cells. Rats receiving conduits with cocultured SCs and ASCs had the largest angle values among all groups at 8 weeks. The rank for improvement was SC + ASC > ASC~ SC~ DPSC ~SC + DPSC > conduits without cells.
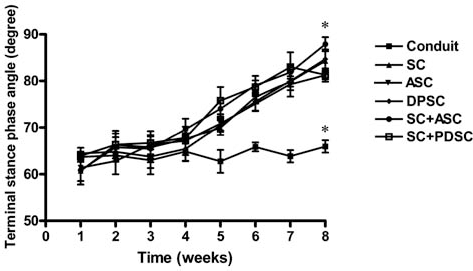
The terminal stance phase angle of functional gait cycle following cell and conduit transplantation. The terminal stance phase angle of functional gait cycle weekly at 1–8 weeks postsurgery for rats implanted with conduits and various types of cells. For abbreviations, refer to Figure 3. *p < 0.05 with respect to the other groups.
Nerve Conduction
The average maximum values of NCV, derived from each group, are demonstrated in Figure 5. The normal sciatic nerve had significantly greater value of maximum NCV compared with the injured nerve (p < 0.05). Conduits seeded with cocultured SCs and ASCs and those seeded with cocultured SCs and DPSCs presented significantly greater values of maximum NCV than the blank conduits and those seeded with DPSCs only.

Electrophysiological evaluation of normal and regenerated sciatic nerves following cell and conduit transplantation. Electrophysiological evaluation by nerve conduction velocity (NCV) at 8 weeks postsurgery for normal sciatic nerve (“Normal”) and the regenerated nerve conduits in rats receiving varieties types of cells. For abbreviations, refer to Figure 3. *p < 0.05 between the indicated groups.
Histological Analysis
The typical histology for the regenerated nerve at the mid-conduit is shown in Figure 6, where the myelin sheaths could be directly observed. Among the different groups, thicker and more deeply colored myelin sheaths were observed for conduits seeded with cocultured SCs and ASCs. The largest regenerated area was also found in this group. Quantification for the area of regenerated nerve, the number of myelinated axons, and the number of blood vessels is shown in Figure 7. The area of regenerated nerve and the number of myelinated axons exhibited the same ranking order: SC + ASC > SC + DPSC ~ SC ~ ASC ~ DPSC > conduits without cells (Fig. 7A, B). The number of blood vessels presented a slightly different tendency: SC + ASC > SC + DPSC ~ ASC > SC ~ DPSC > conduits without cells (Fig. 7C).

Histology of the nerve sections at the mid-conduit after 8 weeks of implantation. (A) Conduit, (B) SC, (C) ASC, (D) DPSC, (E) SC + ASC, and (F) SC +DPSC. For abbreviations, refer to Figure 3. Scale bar: 100 mm.

Quantitative data from histological analyses of the transplant area. (A) Area of regenerated nerve; (B) number of myelinated axons; (C) number of blood vessels. For abbreviations, refer to Figure 3. *p < 0.05 between the indicated groups.
Discussion
The migration and proliferation of SCs play a crucial role in the cellular phase of successful nerve repair (11,53). SCs can secrete neurotrophic factors, which is important for nerve regeneration (1,42). SCs, however, are not easy to obtain for adequate amount of cells, and their efficacy in bridging a longer gap may not be as satisfactory. Therefore, stem cell-based therapies are considered as an alternative for repairing the long nerve defect (44).
We compared in this study two stem cells and two combinations of SCs with stem cells in bridging sciatic nerve injury over a critical gap by the use of nerve conduits. From the success rate of nerve connection, electrophysiology, functional recovery, and histological analysis, conduits seeded with cells had better performance than the blank conduits. The addition of either SCs or combination of SCs with stem cells (half the number each) was beneficial for peripheral nerve repair, although the effect from SCs alone was not as remarkable. SCs and stem cells may help create a microenviroment that promoted axon extension gap. Previous studies showed that ASCs could differentiate into neuron-like cells and SC-like cells in vitro (38,51). ASCs can secrete a few nerve growth factors such as IGF and FGF (55). ASCs can be differentiated into cells with a Schwannlike phenotype that benefited neurite extension (24). ASCs in Matrigel were reported to enhance the regeneration of nerves and induce axon growth in sciatic nerve defects (15,26). On the other hand, the differentiation of DPSCs into neuron-like, SC-like, or endothelial cell-like cells was also reported in literature (14,23,41). DPSCs from both rats and humans expressed NGF, BDNF, and GDNF genes in vitro (32). The paracrine effect as well as the differentiation possibility of the implanted stem cells may contribute to nerve regeneration. DPSCs also promoted facial nerve regeneration in vivo (40).
The positive effect of SCs has often been reported in literature. In our study, conduits cocultured with SCs and ASCs were superior to other groups including SC-seeded conduits. On the other hand, functional recovery based on walking track and functional gait analyses showed that rats receiving conduits seeded with SCs, ASCs, DPSCs alone, or SCs plus DPSCs had similar performance with no statistical difference among the groups. Silicone tubes containing dental pulp cells were previously shown to promote facial nerve repair over the control tubes in rats (41). Conduits seeded with differentiated ASCs also revealed better improvement of axonal and fiber diameter for the regenerated nerve than the blank conduits and those seeded with differentiated bone marrow-derived mesenchymal stem cells (BMSCs) (10). Coculture of BMSCs and SCs without intercellular contact could act synergistically to enhance the differentiation of BMSCs in vitro (56). The indirect coculture of ASCs and SCs could induce ASCs to differentiate into SC-like cells in vitro (49). The in vivo effect of cocultured stem cells and SCs on peripheral nerve regeneration was not reported in literature. Based on our in vivo results, the coculture of SCs and ASCs could promote peripheral nerve regeneration and was superior to the other stem cell groups.
In the developing vertebrate peripheral nervous system, the supply of NGF from tissues they innervate is important to the survival of sympathetic neurons and the majority of sensory neurons (50). BDNF plays prominent roles in learning, memory, and food intake regulation through central mechanisms (9,52). GDNF was originally identified as a survival factor for midbrain dopaminergic neurons and also a very potent trophic factor for spinal motor neurons. (19,25). On the other hand, BDNF and GDNF also appeared to promote peripheral nerve regeneration (7,48). Our in vitro study showed that the coculture of SCs and ASCs increased the gene as well as protein expression levels of neurotrophic factors, especially the level of NGF, which was 50% greater than that for SCs alone. Regarding that only half SCs were used and ASCs alone produced very little NGF, this increase was remarkable. In our in vivo study, rats receiving conduits with cocultured SCs and ASCs had the best functional recovery among all groups at 8 weeks in functional gat analysis. Data from functional gait analysis were comparable to the best data reported in a study involving the use of collagen-chitosan nerve guide to bridge a 10-mm-long nerve gap (35). Conduits seeded with cocultured SCs and ASCs were also obviously superior in the score of SFI to other groups. The recovery of SFI was 20.4–23.4% at 3 months for a silicone tube filled with ASCs to repair a 10-mm gap on the left sciatic nerves of Fischer rats, which was slighter lower than that observed in our study (24.9% in 2 months) (33). The recovery of SFI was 21.6% at 3 months when adipose precursor cells (APCs) in a nerve guide were implanted in a 6-mm sciatic nerve defect in athymic rats (39). The gap defect in the latter study was much shorter than that (15 mm) in our study, implicating that the nerve conduit may also play a critical role in regeneration capacity. This may arise from the synergistic neurotrophic effect of the combination. The upregulation of NGF in vitro may also imply the differentiation of ASCs into Schwann-like cells in cocultured cells. In addition, the data suggested that NGF may be more important than BDNF and GDNF in peripheral nerve regeneration, which deserves further studies. On the other hand, the coculture of SCs and DPSCs did not cause a significant synergistic effect on the expression of neurotrophic factors, although the releases of BDNF and GDNF from the combination appeared to be a little more than those from SCs. In vivo, this combination only significantly increased NCV compared to individual cells. We suspected that NCV may be more sensible to reflect the difference in axonal regeneration. The efficacy of SC/DPSC combination yet requires more investigation.
The same trend with functional analyses was observed on the area of regenerated nerve and the number of myelinated axons in this study. It was found that new myelin sheaths were formed after 8 weeks. The number of blood vessels for conduits seeded with SCs and DPSCs was larger than that for conduits seeded with DPSCs or SCs alone. Neurotrophic factors promote peripheral nerve regeneration by enhancing Schwann cell myelination. They also induce capillary formation (29,45). BDNF can stimulate blood vessel formation (47). NGF can also play a role as an angiogenic regulator (34). BDNF was recently found to be associated with the axon growth promoted by stem cells (26). The addition of NGF and GDNF significantly enhanced the early nerve regeneration including axon growth and Schwann cell migration in a 10-mm rat sciatic nerve defect (28). Therefore, the coculture of SCs and DPSCs may supply more neurotrophic factors than DPSCs or SCs alone for inducing capillary formation. The blood vessels associated with coculture of SCs and ASCs were even more than those associated with coculture of SCs and DPSCs, which was consistent with the larger area of regenerated nerve and number of myelinated axons of the former. The greater number of capillary formation may also contribute to the better regeneration capacity of SC/ASC seeded conduits.
Conclusion
Conduits seeded with cocultured SCs and ASCs showed better performance than those seeded with SCs, DPSCs, ASCs alone, or the combination of SC/DPSC based on histological staining, electrophysiology, walking track, and functional gait measurement. This suggested that coculture techniques may have potential clinical applications in promoting the peripheral nerve regeneration.
Footnotes
Acknowledgment
This work was supported by the Program for Stem Cell and Regeneration Medicine Frontier Research (NSC100-2321-B-002-077) sponsored by the National Science Council, Taiwan, R.O.C. The authors declare no conflict of interest.
