Abstract
Corneal endothelium-associated corneal blindness is the most common indication for corneal transplantation. Restorative corneal transplant surgery is the only option to reverse the blindness, but a global shortage of donor material remains an issue. There are immense clinical interests in the development of alternative treatment strategies to alleviate current reliance on donor materials. For such endeavors, ex vivo propagation of human corneal endothelial cells (hCECs) is required, but current methodology lacks consistency, with expanded hCECs losing cellular morphology to a mesenchymal-like transformation. In this study, we describe a novel dual media culture approach for the in vitro expansion of primary hCECs. Initial characterization included analysis of growth dynamics of hCECs grown in either proliferative (M4) or maintenance (M5) medium. Subsequent comparisons were performed on isolated hCECs cultured in M4 alone against cells expanded using the dual media approach. Further characterizations were performed using immunocytochemistry, quantitative real-time PCR, and gene expression microarray. At the third passage, results showed that hCECs propagated using the dual media approach were homogeneous in appearance, retained their unique polygonal cellular morphology, and expressed higher levels of corneal endothelium-associated markers in comparison to hCECs cultured in M4 alone, which were heterogeneous and fibroblastic in appearance. Finally, for hCECs cultured using the dual media approach, global gene expression and pathway analysis between confluent hCECs before and after 7-day exposure to M5 exhibited differential gene expression associated predominately with cell proliferation and wound healing. These findings showed that the propagation of primary hCECs using the novel dual media approach presented in this study is a consistent method to obtain bona fide hCECs. This, in turn, will elicit greater confidence in facilitating downstream development of alternative corneal endothelium replacement using tissue-engineered graft materials or cell injection therapy.
Keywords
Introduction
The corneal endothelium is the innermost layer of the human cornea. This cellular monolayer is an important “barrier and pump” that regulates corneal hydration to keep the cornea transparent (4). It is known that corneal endothelial cells (CECs) are arrested in the G1 phase of the cell cycle and do not undergo active cellular regeneration within the eye (26,27). Hence, in situations where they become damaged or the cell density falls below a critical threshold, the functional dynamics of the corneal endothelium will be compromised (41). This leads to a cascade of pathological events, beginning with stromal edema, corneal clouding, and a loss of visual acuity, which will eventually result in corneal blindness. Restorative corneal transplant surgery, either by full thickness penetrating keratoplasty or selective replacement of the ineffective corneal endothelium by endothelial keratoplasty (EK) with a functional donor corneal endothelium, is the only option to restore vision (36,53). Selective transplantation of the donor corneal endothelium using less invasive sutureless keyhole EK options, such as Descemet's stripping endothelial keratoplasty (DSEK) (45) and Descemet's membrane endothelial keratoplasty (DMEK) (38), involves the transplantation of a very thin corneal endothelial layer instead of using a full-thickness cornea. With the rapid advancements in EK techniques over the past decade, surgical outcomes of such selective replacement of the dysfunctional corneal endothelial layer have significantly improved (49,53). However, as current corneal endothelial transplant is a one-to-one donor-to-recipient surgery, the shortage of available donor corneal graft tissues remains a problem. This is an increasingly pertinent multifaceted global issue driven further by (a) the process involved in the stringent assessments of donor corneal tissues that may render a potential donor cornea unsuitable for transplantation (41), (b) a potential need for regrafting procedures following graft failures as a result of infection or nonimmunologic/immunological endothelial decompensation (5,37), and (c) a global aging population that reduces the potential donor pool while potentially increasing the demand for corneal transplantation (41).
Although human corneal endothelial cells (hCECs) are not known to be proliferative within the eye, the limited in vitro expansion of isolated CECs has been demonstrated in several laboratories, including ours (2,10,24,34, 44). Consistency in the culture and expansion of primary hCECs is a significant issue, affected by factors such as donor-to-donor variation (63); different isolation protocols, which may affect the overall yield of hCECs; as well as the use of different complex serum-supplemented culture media as reported in various studies (41,44). Although a robust and clearly described culture methodology is still lacking, a great amount of clinical interest has been generated for the development of alternative approaches in the treatment or in reversing the effect of corneal endothelial decompensation using cultivated hCECs. Potentially, thin tissue-engineered constructs of approximately 100 μm developed from expanded hCECs can be used as alternative graft material for selective replacement of the dysfunctional corneal endothelium using advanced EK surgical techniques. The use of a thin tissue-engineered corneal endothelial construct is a very attractive alternative as current DSEK and DMEK surgical approach enables the delivery of such thin graft material into the anterior chamber of the eye. It has also been postulated that cultured hCECs can be injected into the anterior chamber in patients afflicted by corneal endothelial dysfunction as an alternative form of treatment (31). Nevertheless, either approach will require a robust culture system that enables the isolation and propagation of hCECs in vitro with relative consistency despite known donor-to-donor variations.
In our previous study, we showed that two serum-supplemented media, coded in that report as M2 (63) and M4 (15), were able to consistently support the proliferation of primary hCECs isolated from pairs of donor corneas (44). However, the unique cellular polygonal morphology of the cultivated CECs could not be maintained beyond the second passage in a majority of the established cultures, and cells became fibroblast-like (44). This phenomenon has been reported by Zhu and colleagues in cultures of hCECs that were exposed to growth factors such as basic fibroblast growth factor (bFGF), which could activate canonical Wnt signaling, resulting in an endothelial-to-mesenchymal transition (EMT) (64).
We have discovered the use of a serum-supplemented culture medium (referred to as M5 in this study), which is able to preserve the cellular morphology of primary hCECs in vitro (unpublished observation). For this study, in order to prevent EMT of hCECs expanded in proliferative medium M4, we assessed the incorporation of M5 in a dual media culture system as a novel approach for the propagation of isolated hCECs. Expression of key markers indicative of the human corneal endothelium was examined for cells grown to P3 in M4 alone and were compared pairwise to CECs from the same donors expanded to P3 using the dual media approach. Finally, microarray analysis was performed on P3 hCECs that were expanded using the dual media system to compare gene expression profiles of the proliferating cells before the switch to M5 and those exposed to M5 for 7 days.
Materials and Methods
Materials
Ham's F12, Medium 199 (M199), human endothelial serum-free medium (SFM), fetal bovine serum (FBS), Dulbecco's phosphate-buffered saline (PBS), TrypLE” Express (TE), gentamicin, amphotericin B, penicillin G, streptomycin sulfate, TotalPrep” 96 RNA Amplification Kit, and Ambion® proprietary MEGAscript® Kit were purchased from Life Technologies (Carlsbad, CA, USA). Insulin, transferrin, selenium (ITS), ascorbic acid, trypan blue (0.4%), calcium chloride, chondroitin sulfate, paraformaldehyde (PFA), bovine serum albumin (BSA), Triton X-100, normal goat serum, and chloroform were purchased from Sigma (St. Louis, MO, USA). Fibronectin, collagen, and albumin (FNC) coating mix was purchased from United States Biologicals (Swampscott, MA, USA). Collagenase A was obtained from Roche (Mannhein, Germany).
Research-Grade Human Corneoscleral Tissues
Donor Information
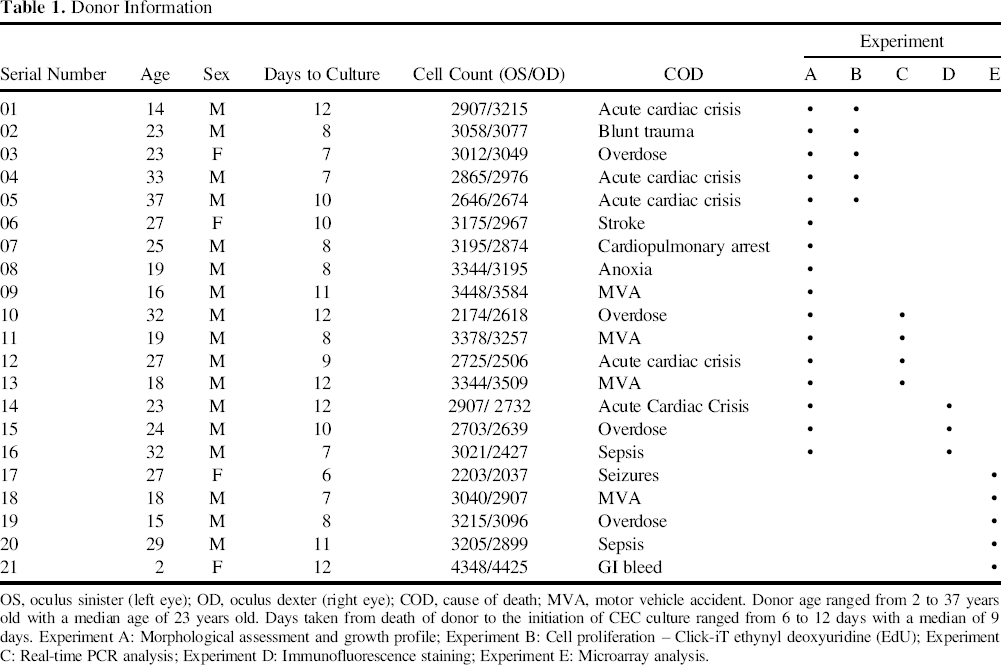
OS, oculus sinister (left eye); OD, oculus dexter (right eye); COD, cause of death; MVA, motor vehicle accident. Donor age ranged from 2 to 37 years old with a median age of 23 years old. Days taken from death of donor to the initiation of CEC culture ranged from 6 to 12 days with a median of 9 days. Experiment A: Morphological assessment and growth profile; Experiment B: Cell proliferation – Click-iT ethynyl deoxyuridine (EdU); Experiment C: Real-time PCR analysis; Experiment D: Immunofluorescence staining; Experiment E: Microarray analysis.
Isolation and Growth of Human Corneal Endothelial Cells
Isolation of hCECs using a two-step peel and digest method was performed as previously described (44). Briefly, after peeling off the Descemet's membrane (DM), the pieces of DM were exposed to 2 mg/ml collagenase (in M5 medium) for 2–4 h to dislodge the CECs from the DM, which resulted in tightly packed CEC clusters. The CEC clusters were further dissociated in 1× TE for 2 min to further dissociate the clusters into smaller clumps and single cells.
For initial comparative studies, isolated CEC clumps were divided equally into two conditions: either M4 alone or M5 alone. The formulation of M4 [Ham's F12/M199, 5% FBS, 20 μg/ml ascorbic acid, 1% ITS, 10 ng/ml bFGF (R&D Systems, Minneapolis, MN, USA), and 1% antibiotic/antimycotic] was reconstituted as previously published (15). The basal medium of M5, human endothelial-SFM, was supplemented with 5% FBS and 1% antibiotic/antimycotic.
In subsequent studies wherein the dual media culture approach was utilized, isolated hCECs were first established in M5 medium overnight to allow for cell adherence and stabilization. The culture medium was subsequently replaced with M4 to promote the proliferation of the adhered CECs. When the growth of CECs reached 80% to 90% confluence (approximately 2 weeks), M4 medium was withdrawn, and M5 medium was reintroduced to the CECs for at least 7 days before passage.
Confluent CECs were passaged via 1× TE dissociation. For cellular expansion, the dissociated CECs were plated at the seeding density of 104 cells per cm2, as described previously (43). A Nikon TS1000 phase contrast microscope with a Nikon DS-Fi1 digital camera (Tokyo, Japan) was used to capture cellular morphology at every passage. All cultures were incubated in a humidified atmosphere at 37°C and 5% CO2.
Cell Proliferation Assay
Proliferation rate of hCECs cultured in M4 alone or M5 alone was determined by Click-iT” ethynyl deoxyuridine (EdU) Alexa Fluor 488 imaging kit (Invitrogen/Life Technologies) as per the manufacturer's instructions. Briefly, passaged hCECs were seeded onto a FNC-coated slide at a lower density of 5 × 103 cells per cm2 and cultured for 24 h in their respective culture condition. The cells were then incubated in the respective medium containing 10 μM EdU for another 24 h. After incubation, the cells were rinsed once with PBS followed by fixation with 4% PFA for 15 min on ice. The cells were then washed twice with 3% BSA in PBS and permeabilized using 0.5% Triton X-100 for 20 min at room temperature and washed twice with 3% BSA. The samples were incubated in a reaction cocktail containing 1× reaction buffer, CuSO4, and Alexa Fluor azide for 30 min in the dark. Samples were rinsed with PBS and mounted in Vectashield containing 4′,6-diamidino-2-phenylindole (DAPI) (Vector Laboratories, Burlingame, CA, USA). A Zeiss Axioplan 2 fluorescence microscope (Carl Zeiss, Jena, Germany) was used to examine the labeled proliferative cells. At least 250 nuclei were analyzed for each experimental set.
Morphometry Analysis of Cellular Circularity
Cells of the healthy human corneal endothelium are generally hexagonal in shape (60,62). Hence, an important morphological characteristic of hCECs expanded in vitro is the maintenance of their polygonal/hexagonal cellular shape in culture. The roundness or circularity of a cell was determined as previously described using the formula: circularity
Gene Expression Analysis
List of Primer Sequences
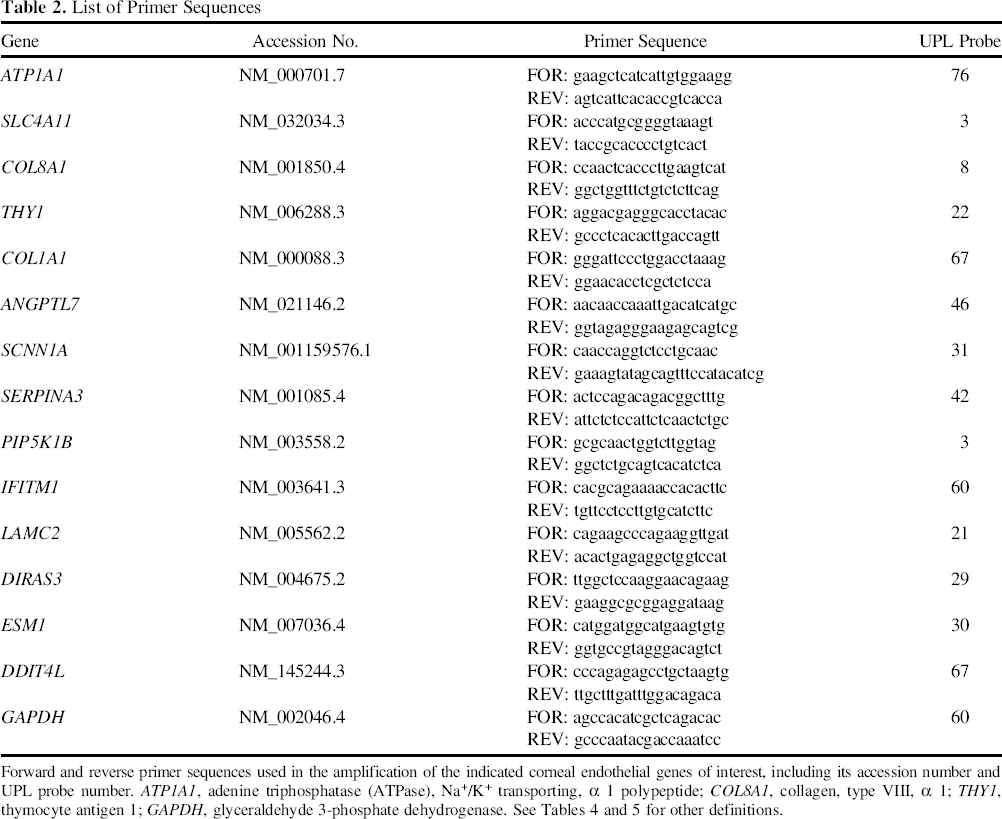
Forward and reverse primer sequences used in the amplification of the indicated corneal endothelial genes of interest, including its accession number and UPL probe number. ATP1A1, adenine triphosphatase (ATPase), Na+/K+ transporting, α 1 polypeptide; COL8A1, collagen, type VIII, α 1; THY1, thymocyte antigen 1; GAPDH, glyceraldehyde 3-phosphate dehydrogenase. See Tables 4 and 5 for other definitions.
Immunocytochemistry
Confluent passage 2 (P2) hCECs were passaged and plated at a high density of 2,000 cells per mm2 on FNC-coated glass coverslips (7 mm in diameter) and maintained for approximately 7 days in M5 medium before fixation with 100% ice-cold ethanol for 5 min or 4% PFA for 15 min at 4°C. Samples were rinsed and blocked in 5% normal goat serum in PBS for 30 min at room temperature. Subsequently, samples were labeled with primary antibodies for 1 h at room temperature. Primary antibodies used in this study were mouse IgG1 anti-sodium-potassium-transporting adenosine triphosphatase (Na+K+/ATPase, 5 μg/ml; Santa Cruz Biotechnology, Dallas, TX, USA), mouse IgG1 antizona occludens 1 (ZO-1, 5 μg/ml; BD Biosciences Pharmingen, Franklin Lakes, NJ, USA), mouse IgG1 anti-cluster of differentiation 200 (CD200, 20 μg/ml; BD Biosciences Pharmingen), and mouse IgG1 anti-glypican 4 (GPC4, 20 μg/ml; Novus Biologicals, Littleton, CO, USA). The samples were then washed twice with PBS, 5 min each, and labeled with AlexaFluor 488-conjugated goat anti-mouse IgG secondary antibody (1:750; Life Technologies) for 1 h at room temperature in dark. After two brief PBS washes, they were mounted in Vectashield containing DAPI and visualized under a fluorescence microscope.
Microarray Analysis
hCECs from two donors (D17 and D18) were cultivated to the third passage using the dual media approach. Confluent cells in the proliferative M4 medium (D17p and D18p) were compared to the same batches of confluent cells that were maintained in M5 medium (D17m and D18m) for an additional 7 days. Samples were extracted in 1 ml TRIzol reagent (Invitrogen). Samples were homogenized using a handheld homogenizer (VWR, Radnor, PA, USA) before the addition of 200 μl chloroform. After vigorous shaking, samples were centrifuged at 12,500 relative centrifugal force (rcf) for 15 min at 4°C. The upper aqueous phase was transferred to a new tube and mixed with an equal volume of 70% ethanol. The resulting solution was transferred to a Qiagen RNeasy column, and the RNA purification procedures were performed as per the manufacturer's protocol with a RNAse-free DNase digestion step incorporated. An Agilent RNA 6000 Nano Kit and Agilent 2100 Bioanalyzer (Santa Clara, CA, USA) were used to determine the quality and integrity of RNA before microarray analysis. Subsequently, total RNA was prepared for microarray analysis using a TotalPrep” 96 RNA Amplification Kit according to the manufacturer's protocol. Briefly, total RNA was reverse transcribed with oligo(dT) primer bearing a T7 promoter using ArrayScript” reverse transcriptase. The cDNA underwent second-strand synthesis and cleanup to become a template for in vitro transcription with T7 RNA Polymerase. Ambion® proprietary MEGAscript® Kit was used to generate biotinylated antisense cRNA. The labeled strands were hybridized in triplicate onto HumanHT-12 v4 Expression BeadChips (Illumina, San Diego, CA, USA) using the Illumina IntelliHyb Seal.
Array data were analyzed using Partek Genomic Suite 6.5 beta software (Partek, Inc., St. Louis, MO, USA). Data was first imported and normalized using robust multiarray averaging. Statistical testing using ANOVA was performed to identify genes that were differentially expressed. The lists of differentially expressed gene transcripts between the two culture conditions were filtered based on a selection criterion of ≥2 relative fold change at a false discovery rate of ≤5%. Raw data of the array have been deposited at Gene Expression Omnibus under accession number GSE50212.
Pathway Analyses
Differentially expressed genes were imported to the Database for Annotation, Visualization and Integrated Discovery (DAVID) Functional Annotation Bioinformatics Microarray Analysis v6.7 (http://david.abcc.ncifcrf.gov). Functional gene clusters in association with biological events were identified. The likelihood of event presentation in Gene Ontology Consortium Annotation Categories (GO biological process, cell component, and molecular functions) was examined.
Statistics
All numeric data obtained were expressed as mean ± standard deviation (SD). Statistical analyses were performed using SPSS Statistics 17.0 (IBM, Chicago, IL, USA) as follows: the analyses of cell proliferation between hCECs cultured in M4 alone or M5 alone using Click-iT EdU assay (Fig. 1A and B); comparisons of cell numbers obtained from culture in M4 alone and M5 alone of the five donors (Fig. 1C); comparisons of cell sizes and cell circularity (Table 3) of hCECs propagated in M4 alone and the dual media approach and between each passage were evaluated using paired Student's t tests. For all gene expression analysis, results were analyzed using independent Student's t tests. Results with a value of p < 0.05 were deemed to be statistically significant.
Human CECs established and propagated in M4 alone and in M5 alone. Representative sets of photomicrographs showing morphology of confluent human corneal endothelial cells (hCECs) (passage 0; P0) established in (A) M4 alone; with percentages of proliferative hCECs (at the first passage; inset) as assessed by the Click-IT ethynyl deoxyuridine (EdU) assay, which is significantly different (*p < 0.05) when compared to (B) percentages of proliferative hCECs cultured in M5 alone (at the first passage; inset). (C) Total cell numbers obtained for the passage of five independent sets of donor-matched hCECs cultured in M4 alone compared to M5 alone, **p < 0.01. (D) Representative photomicrograph showing a typical fibroblast-like morphology of cultivated hCECs at the third passage cultured using M4 alone. Cell Circularity of Cultured hCECs at P1, P2, and P3 at Confluence Cultured hCECs propagated in dual media were found to be larger than their respective counterparts that were cultured in M4 alone at the second passage (P2) p < 0.01, and the third passage p < 0.01. However, cell circularity analysis showed that hCECs expanded using the dual media approach were significantly more polygonal/hexagonal compared to hCECs cultured in M4 alone, which were significantly more heterogeneous in terms of their cellular morphology at the first passage p < 0.01, second passage p < 0.01, as well as the third passage p < 0.01. When P2 hCECs expanded in the dual media were plated at a high seeding density of approximately 3,000 per mm2 (dual mediaH), average cell sizes of 828.06 ± 256.20 μm2 and a cell circularity measurement of 0.74 ± 0.11 were obtained.

Results
Morphology and Proliferation of Human Corneal Endothelial Cells Grown in M4 and M5
A total of five pairs of donor corneas (Table 1; serial numbers 01 to 05) were used for this experiment. hCECs were harvested and cultured in either M4 alone or M5 alone. At confluence (P0), striking morphological differences were observed in CECs that were grown in M4 (Fig. 1A) and M5 (Fig. 1B). Assessment of cell proliferation at the first passage using Click-iT EdU assay showed that hCECs grown in M4 alone (Fig. 1A inset; 21.1 ± 8.8%) were significantly more proliferative (*p < 0.05) than cells grown in M5 alone (Fig. 1B, inset; 6.9 ± 4.5%). As assessed by paired Student's t tests, confluent P0 hCECs grown in M4 alone yielded significantly more cells compared to those grown in M5 alone (**p < 0.01) (Fig. 1C). As such, with the exception of Donor 3, insufficient hCECs from M5 culture were obtainable for continual cell expansion. Although we were able to expand hCECs grown in M4 culture to the third passage and beyond, the unique polygonal/hexagonal cellular morphology was lost, and cells became elongated and fibroblast-like (Fig. 1D). Signs of such transformations were observed in some of the CECs expanded in M4 from as early as the first round of passage (unpublished observation).
Dual Media Culture System to Propagate Human Corneal Endothelial Cells
Based on the above observation, we hypothesized that using M4 for cell growth and proliferation and M5 for cell stabilization and maintenance would aid in an increased capacity to cultivate the hCECs while retaining their cellular morphology. Hence, we developed a dual media culture strategy for the propagation of hCECs as depicted in Figure 2. Subsequent comparative studies were performed on independent sets of hCECs isolated from 11 pairs of donor corneas (Table 1; serial numbers 06 to 16), where isolated CECs were divided equally into two culture conditions, using either M4 alone or via the dual media approach.
Schematic diagram depicting the propagation of hCECs using a dual media approach. (A) Procurement—pairs of research-grade corneas used in this study were procured from Lions Eye Institute for Transplant and Research Inc. (Tampa, FL). Research corneas were preserved and transported in Optisol-GS, and processed within 14 days from preservation. (B) Process and isolation—once received, corneas were processed within 1 day where the hCECs were isolated and plated as passage 0 culture. (C to C′) Stabilization— isolated hCECs were seeded and allowed to attach and stabilize in M5 media overnight. (C′ to C″) Proliferation—to promote the proliferation of the adhered CECs, M4 media was utilized throughout the expansion phase. (C″ to D) Stabilization and maintenance—when expanding hCECs became approximately 80% confluent, M5 media was reintroduced for at least 7 days, which aided in the preservation of the cellular morphology of cultivated hCECs. Thereafter, confluent hCECs were dissociated using TrypLE” Express (TE), seeded at a density of 1 × 104 cells per cm2, and subjected to the same interswitching dual media approach for subsequent passages.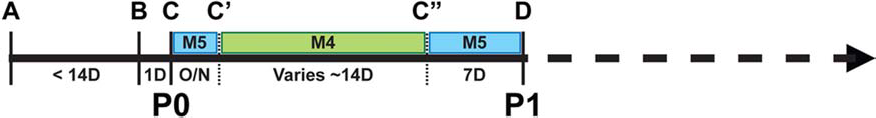
Morphometric Analysis: A Comparison of CECs in M4 Alone Versus CECs in Dual Media
hCECs isolated from four pairs of donor corneas (Table 1; serial numbers 06 to 09) were used for the following analysis and grown for three passages. Over the three passages, it was evident that cells expanded in M4 alone gradually lost their unique hexagonal/polygonal morphology and became vastly heterogeneous by the third passage (Fig. 3). Interestingly, the cellular morphology of hCECs propagated using the dual media was maintained throughout the three passages, and phase contrast microscopy showed that dual media-expanded CECs appeared more homogenous than their respective counterparts propagated in M4 alone (Fig. 3). This observation was confirmed by comparative cell circularity measurements of the two CEC populations from P1 to P3, and CECs grown in M4 alone were significantly more elongated (less polygonal/hexagonal) at confluence across all three passages (Fig. 3, Table 3). It should be noted that cell circularity measurement of CECs from a healthy human corneal endothelium taken from a specular micrograph is 0.82 ± 0.03 (unpublished observation).
Cellular morphology and circularity of hCECs cultured over three passages. Representative sets of photomicrographs showing morphology of confluent hCECs propagated in either M4 alone or using the dual media approach and a bar chart comparing cell circularity for both conditions during (A) the first passage, (B) the second passage, and (C) the third passage.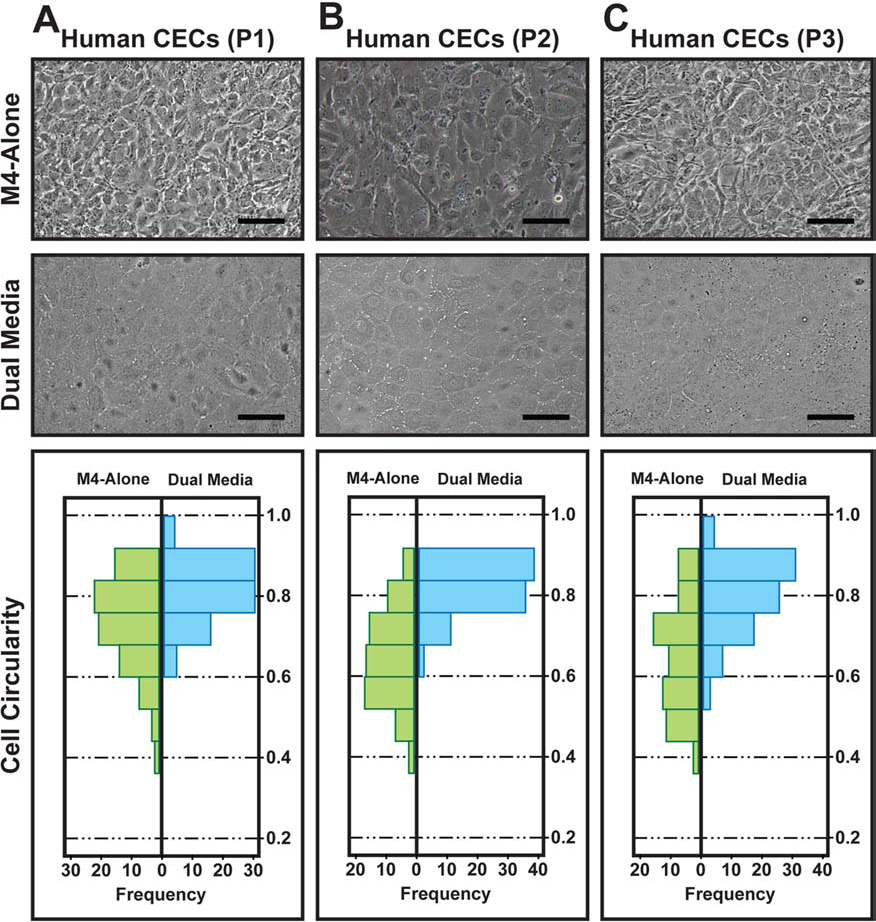
When confluent P0 hCECs were passaged for further cell expansion, based on a seeding density of 1 × 104 cells per cm2, their cellular sizes became significantly larger at confluence—especially CECs propagated under the dual media approach (Table 3). To investigate if this was a reversible phenomenon, we seeded P2 hCECs (average size of 4,954.47 ± 1,227.25 μm2) (Table 3) at a physiological cell density of 2,000 per mm2. Subsequent cell size measurement of these CECs showed average cell sizes of 828.06 ± 256.20 μm2 and a cell circularity of 0.74 ± 0.11 (dual mediaH; Table 3).
Characterization of Cultivated Human Corneal Endothelial Cells at the Third Passage
Expression levels of five genes, three known to be expressed by the corneal endothelium [ATPase, Na+/K+ transporting, α 1 polypeptide (ATP1A1), solute carrier family 4, sodium borate transporter, member 11 (SLC4A11), collagen, type VIII, α 1 (COL8A1)] and two not specifically related to the corneal endothelium [thymocyte antigen 1 (THY1; CD90), collagen, type I, α 1 (COL1A1)], were measured using quantitative real-time PCR in four separate sets of hCECs (P3) exposed to M4 alone or cultivated using the dual medium approach (n = 4) (Table 1; serial numbers 10 to 13). The relative expression of each gene in P3 hCECs cultured using the two different approaches were compared and normalized to obtain a relative fold increase. Results showed that cells cultured using the dual media approach expressed higher levels of ATP1A1 (1.75 ± 0.15-fold*), SLC4A11 (11.94 ± 4.20-fold*), and COL8A1 (4.42 ± 2.10-fold**), and cells grown in M4 alone showed higher levels of THY1 (6.77 ± 2.38-fold**) and COL1A1 (4.76 ± 1.93-fold**) (*p < 0.01, **p < 0.05) (Fig. 4A). In separate experiments, indirect immunofluorescence showed that P3 hCECs propagated using the dual media approach (Table 1; serial numbers 14 to 16), seeded at a density of 2,000 cells per mm2, expressed corneal endothelium-associated pump marker Na+K+/ATPase (Fig. 4B), tight junction marker ZO-1 (Fig. 4C), heparin sulfate proteoglycan GPC-4 (Fig. 4D) (11), and cell membrane glycoprotein CD200 (Fig. 4E) (11). More importantly, the staining patterns clearly showed the polygonal shape of cultivated hCECs at the third passage. These results suggested that hCECs propagated using the dual media system retained their unique cellular morphology, and expressed higher levels of corneal endothelium-specific genes ATP1A1, SLC4A11 and COL8A1, as well as reported cellular markers of the corneal endothelium (11,44).
Characterization of cultivated hCECs of the third passage. (A) Quantitative RT-PCR of mRNA from cultures of confluent human CECs at the third passage expanded using M4 alone (green), compared to CECs from the same sets of donors propagated to the third passage under the dual media approach (blue). Fold increase of each gene was calculated and significantly higher expression of adenine triphosphatase (ATPase), Na+/K+ transporting, α 1 polypeptide (ATP1A1), solute carrier family 4, sodium borate transporter, member 11 (SLC4A11), and collagen, type VIII, α 1 (COL8A1) was observed in CECs propagated using the dual media approach, whereas thymocyte antigen 1 (THY1) and COL1A1 expression was significantly higher in CECs expanded in M4 medium only. n = 4, *p < 0.01, and **p < 0.05. Primary CECs were cultivated using the dual media approach to the third passage and characterized for their expression of known markers indicative of the corneal endothelium such as (B) sodium–potassium-transporting adenosine triphosphatase (Na+K+ATPase), (C) zona occludens 1 (ZO-1), (D) glypican 4 (GPC-4), and (E) cluster of differentiation 200 (CD200), by immunocytochemistry. (F) A representative image of an isotype-matched negative control. (n = 5; scale bar: 50 μm).
Gaining Insights Into Gene Expression Changes of Human Corneal Endothelial Cells Following Exposure to M5 Medium
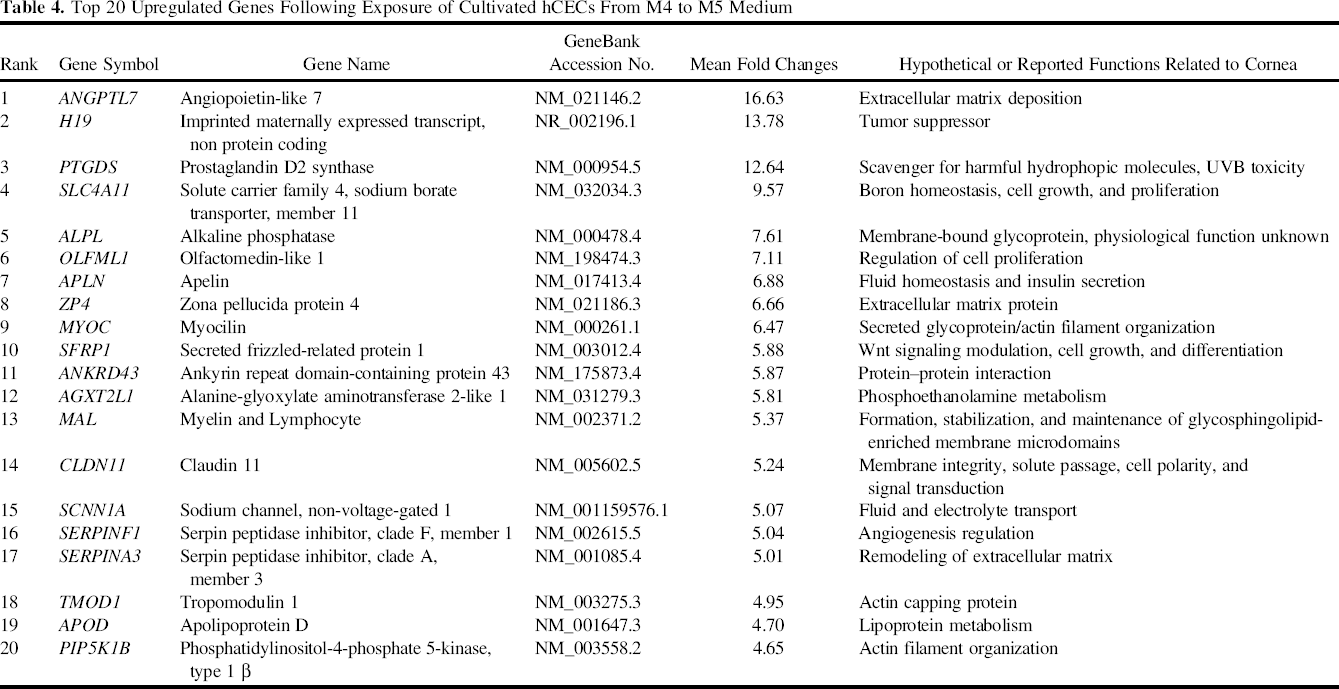
The polygonal morphology of primary hCECs was consistently retained for each of the three rounds of expansion when the confluent cells grown in the proliferative M4 medium were switched to the maintenance M5 medium for 7 days. In order to better understand the observed morphological changes at the molecular level, high throughput microarray analysis was performed on hCECs isolated from two donors (Table 1; serial numbers 17 and 18) cultivated to the third passage. Here, the global gene expression profile of the confluent CECs expanded in M4 for the first (D17p) and second donor (D18p) were compared to their respective counterparts maintained in M5 medium (D17m and D18m) for a further 7 days. Hierarchical clustering showed that propagated hCECs from the two separate donors cultured in M4 had more similarities in their gene expression profiles than cells that were maintained in M5 for an additional 7 days (Fig. 5A). In all, a total of 1,485 upregulated genes (Fig. 5B) and 1,420 downregulated genes (Fig. 5C) were found. Additionally, the top 20 upregulated and downregulated genes are listed in Tables 4 and 5, respectively.
Microarray analysis of hCECs expanded using the dual media approach at the third passage. Microarray analysis was performed to compare gene expression profiles of hCECs cultured using the dual media approach at the end of the proliferative phase in M4 medium and following the switch into M5 for 7 days in the maintenance/stabilizing medium. (A) Hierarchical clustering shows that the samples cluster according to the culture condition, indicating that hCECs cultured in the proliferative M4 medium (D17p and D18p) have more similar gene expression profiles compared to their counterparts after they were exposed and maintained in M5 medium (D17m and D18m). (B) Venn diagram showing that 1,485 genes with fold change ≥2.0 were upregulated in both Donor 1 and Donor 2 when the cultured hCECs were switched from M4 medium to M5 medium. (C) Venn diagram showing that 1,420 genes with fold changes ≥2.0 were downregulated in both donors following the switch in medium from M4 to M5. (D) Relative gene expression levels of hCECs cultivated to the third passage, at confluence in M4 medium before the switch into M5 medium and 7 days after the switch into M5 medium, from three independent donors, were determined using QRT-PCR to validate the microarray data. Relative quantification values of selected upregulated genes (olive green columns) and downregulated genes (azure blue columns) were calculated with glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as the internal control, and fold changes in expression were calculated relative to confluent cultivated cells in M4 medium before the 7-day exposure to M5 medium. ANGPTL7, angiopoietin-like 7; SCNN1A, sodium channel, non-voltage-gated 1; SERPINA3, serpin peptidase inhibitor, clade A, member 3; PIP5K1B, phosphatidylinositol-4-phosphate 5-kinase, type 1 β; IFITM1, interferon-induced transmembrane protein 1; LAMC2, laminin, γ 2; DIRAS3, DIRAS family, GTP-binding RAS-like 3; ESM1, endothelial cell-specific molecule 1; DDIT4L, DNA-damage-inducible transcript 4-like.
Validation of Microarray Data for Selected Differentially Expressed Genes by QPCR
We performed quantitative real-time PCR using three independent sets of cultivated P3 hCECs to validate the microarray results (Table 1; serial numbers 19 to 21). The relative gene expression of five upregulated genes [SLC4A11, angiopoietin-like 7 (ANGPTL7), sodium channel, non-voltage-gated 1 (SCNN1A), serpin peptidase inhibitor, clade A, member 3 (SERPINA3), and phosphatidylinositol-4-phosphate 5-kinase, type 1 β (PIP5K1B)] and five downregulated genes [interferon-induced transmembrane protein 1 (IFITM1), laminin, γ 2 (LAMC2), DIRAS family, GTP-binding RAS-like 3 (DIRAS3), endothelial cell-specific molecule 1 (ESM1), and DNA-damageinducible transcript 4-like (DDIT4L)] selected from the top 20 up- and downregulated list of genes were examined. The fold changes in gene expression were calculated relative to GAPDH, and the results obtained were normalized against cultures in M4 before the 7-day exposure to M5. Results showed that the direction of gene expression of the selected genes was consistent with the microarray results (Fig. 5D).
Pathway Analysis
The sorted gene list (fold difference >2 and p < 0.05) comparing primary hCECs in proliferative M4 medium and maintained an additional 7 days in M5 media was analyzed by DAVID Functional Annotation Bioinformatics Microarray analysis v6.7 for gene ontology and significant pathways. Differentially expressed genes were engaged to calculate the significant gene annotation and enrichment to reveal the potential biological pathways and functions. Table 6 showed the significant ranking of gene ontology terms by p values (<0.05) and the associated genes. The regulation of cell proliferation (GO:0042127) was found to be the most significant pathway (p = 4.3 × 10−10), followed by wound healing (p = 2.9 × 10−8). Pathways associated with endothelial cell function included regeneration (p = 2.2 × 10−5), regulation of developmental growth (p = 1.4 × 10−4), biological adhesion (p = 1.6 × 10−4), vasculature development (p = 3.8 × 10−4), hemostasis (p = 5.2 × 10−4), regulation of cell migration (p = 6.5 × 10−4), and response to nutrients (p = 6.7 × 10−4). Other pathways, like ossification, humoral immune response, regulation of phosphate metabolic process, epithelial cell proliferation, and nervous system development were also highlighted in the analysis.
Discussion
The global shortage of donor corneal tissues available for transplantation is one of the main problems limiting the numbers of corneal transplantation performed yearly. Hence, the potential of alternate approaches such as using tissue-engineered graft materials (12,33,46) or cell injection therapy (31) are attractive. However, facilitating the developments of these approaches requires the ability to expand the numbers of primary hCECs with relative consistency. The expression of important CEC-related markers and cellular morphology of the cultivated CECs must be maintained. Although the isolation and cultivation of primary hCECs have been described by many (41), results have been variable with limited success. We have previously shown that isolated hCECs cultivated in different proliferative media, though able to support their continual expansion, underwent morphological changes and lost their unique polygonal/hexagonal cellular morphology (44). This phenomenon is most likely due to the activation of canonical Wnt signaling pathway, which in the presence of bFGF induces EMT, resulting in their morphological transformation toward a more elongated fibroblast-like morphology (64).
In this study, we first compared and characterized the effect of two culture media on the growth and cellular morphology of cultivated hCECs. When expanded in the proliferative medium M4 alone, the total cell yielded at P0 and the proliferation rate measured at P1 were significantly higher than those cells cultured in M5 alone. Comparatively, with the lower proliferation rate of cells grown in M5 alone, we were not able to efficiently scale up the culture to obtain sufficient numbers of hCECs for downstream studies. Despite the lack of cell growth, primary CECs cultured in M5 alone remained morphologically similar to cells of the corneal endothelium in situ with regards to their unique hexagonal shape. Although the continual expansion of hCECs using M4 alone is achievable, undesirable EMT-like transformation tends to occur, usually by the second passage. In some cultures, these signs can be detected as early as within the first passage (donor dependent; unpublished observations). This is not surprising in light of the study reported by Zhu and colleagues (64) as M4 medium contains bFGF.
Next, we explored the feasibility of using the two culture media, M4 and M5, sequentially for the propagation of isolated hCECs (Fig. 2). Once hCECs were isolated from the DM by two rounds of enzymatic treatments (44), the cells were left in M5 overnight to allow the isolated cells to adhere on FNC-coated plates. The incorporation of an overnight stabilization phase was adapted from a report by Zhu and Joyce, whereby isolated intact corneal endothelium was incubated in a serum-supplemented medium overnight to stabilize the cells before culture (63). This also allowed us a predetermined time point to assess the quality of the isolated hCECs. On the following day, M5 medium was withdrawn, and the adhered hCECs were expanded in the proliferative M4 medium. By withdrawing M4 medium and reintroducing M5 medium to confluent or near-confluent cultures of expanding hCECs, factors involved in driving EMT transformation of CECs were removed. Indeed, a comparative cell circularity analysis showed that cells expanded using the dual media approach were significantly more “circular” in shape when compared to their respective counterparts propagated in M4 alone. Over the three passages, CECs cultured in M4 alone were more heterogeneous in terms of their circularity and contained more elongated cells, indicating the prevalence of cells with fibroblast-like morphology. Further molecular characterization of hCECs expanded in the dual media system showed higher expression of gene transcripts important in the functional homeostasis [ATP1A1 (40) and SLC4A11 (48)] and the cellular component [COL8A1 (28)] with respect to the corneal endothelium in situ. The a 1 subunit of Na+/K+-ATPase is encoded by ATP1A1 and forms part of an essential component of the corneal “pump-leak” mechanism (4). Mutations in SLC4A11, a sodium-coupled borate cotransporter, have been associated with different variants of corneal endothelial dystrophy (25,48). COL8A1 is the a 1 chain of type VIII collagen, a major component of the basement membrane of the corneal endothelium (28). Conversely, we found that cultivated CECs expanded in M4 alone had upregulated noncorneal endothelium gene transcripts such as THY1 (7) and COL1A1 (29). THY1 is widely expressed by different cell types including fibroblasts (7) and COL1A1, the pro-α 1 chains of type I fibril-forming collagen, is found in most connective tissues, bone, and dermis (29).
The healthy corneal endothelium layer of an adult has a central CEC density count between 2,000 and 3,000 cells per mm2 (6,59,62). In this study, the central CEC counts of adult cadaveric donors above 18 years old ranged from 2,037 to 3,509 cells per mm2 (Table 1). This is equivalent to CEC sizes of 491 to 285 μm2, respectively. However, other studies have also reported CEC sizes ranging from between 170 and 976 μm2 (21,59). In the current study, when hCECs were taken out of their in situ environment and propagated in vitro, cell size measurements showed a significant increase in the sizes of CECs expanded over the three rounds of passage to 4,839.94 ± 1,614.22 μm2 (Table 3). However, we showed that CECs from the same donors can be seeded at a “physiologically functional” density of 2,000 cells per mm2 to achieve a calculated cell size of 828.06 ± 256.20 μm2. These results suggested that propagated hCECs were amenable to the dual media approach presented in this study and could be plated at a higher density to achieve a denser monolayer of CECs with compact cellular pattern. Indeed, when seeded at a higher density onto a compressed collagen-based hydrogel (a potential transplantable corneal graft substitute), a cell density of approximately 1,941 cells per mm2 was achieved (33).
Top 20 Upregulated Genes Following Exposure of Cultivated hCECs From M4 to M5 Medium
Top 20 Downregulated Genes Following Exposure of Cultivated hCECs From M4 to M5 Medium

Top 10 GO Pathways Attenuated Following Exposure of Cultivated hCECs From M4 to M5 Medium
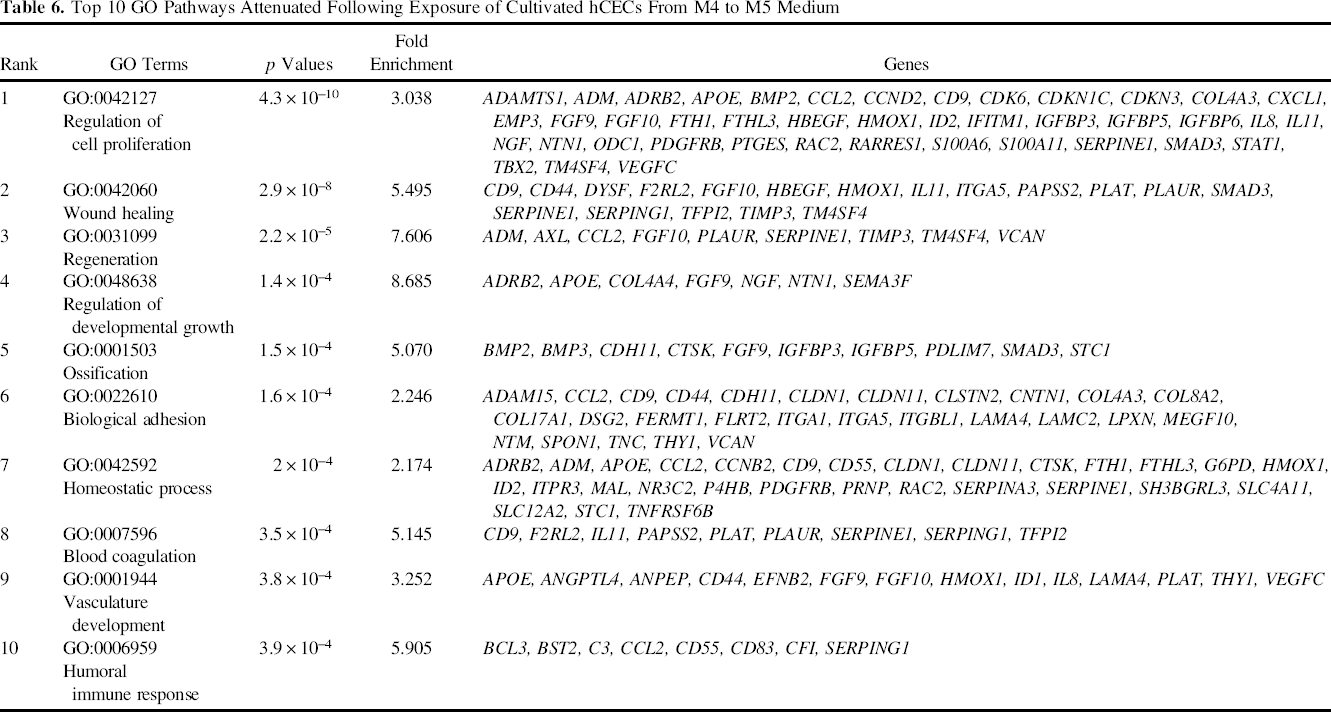
Sensitive and quantitative global gene expression changes in the cultivated hCECs following the switch from M4 to M5 medium provided a wealth of information to better understand the morphological changes observed. It also provided some insights on the signaling pathways that are likely to be involved. For example, SFRP1 and DKK1 are known to regulate the canonical Wnt pathway, and DDIT4L regulates mTOR signaling. Furthermore, the upregulation of function-related genes, such as SLC4A11 and SCNN1A, together with the downregulation of noncollagenous basement membrane glycoproteins LAMC2 provided hints that the switch in culture condition is driving the expression of relevant genes indicative of the corneal endothelium. By DAVID Bioinformatics analysis, we identified that the medium switch significantly affected a variety of GO pathways, predominately in cell growth and regeneration. The regulation of cell proliferation, wound healing, regeneration, and developmental growth were the top four most significant pathways. Further characterization of genes of interest at the protein level will be useful, for example, in validating the signaling pathways involved. Future functional in vitro assays and in vivo animal models will confirm the functionality of the cultivated human CECs expanded using the dual media culture approach.
In conclusion, the expansion of primary hCECs using the novel dual media approach presented in this study provides a robust culture system for the propagation of isolated hCECs. Incorporating the stabilization M5 medium following the proliferation using M4 medium prevented EMT-like transformation and was able to preserve the unique cellular morphology of the cultivated hCECs. Genes and marker expressions indicative of the human corneal endothelium were also maintained. As such, it will elicit greater confidence in utilizing the expanded hCECs to obtain sufficient cell numbers to facilitate downstream development of corneal endothelium replacement using tissue-engineered graft materials or cell injection therapy.
Footnotes
Acknowledgments
We would like to acknowledge Lim Wan'E for thoughtful comments and assistance with the Partek Software. This study was supported by research grants from the National Research Foundation Translational and Clinical Research (TCR) Programme Grant (NMRC/TCR/002-SERI/2008) and from the Biomedical Research Council Translation Clinical Research Partnership (TCRP) Grant (TCR0101673). The funding bodies had no role in study design, data collection, analysis, decision to publish, or preparation of the manuscript. Singapore Patent Application No. 201205413–6 – The Culture of Human Corneal Endothelial Cells Using a Dual Media Approach. The patent is not expected to provide any financial gain to the authors based on the publication of this manuscript.
