Abstract
Corneal transplantation is a common transplant procedure used to improve visual acuity by replacing the opaque or distorted host tissue with clear healthy donor tissue. However, its clinical utility is limited due to a lack of donor supply of high-quality corneas. Bioengineered neocorneas, created using an expandable population of human donor-derived corneal endothelial cells (HCECs), could address this shortage. Thus, the objective of this study was to evaluate HCEC sourcing with various isolation methods, including enzymatic digestion, culture medium components, and adhesive proteins. HCECs were obtained from corneas obtained from various aged donors after endothelial keratoplasty. Under a dissection microscope, the Descemet's membrane, including the attached corneal endothelium, was stripped from the stroma, and the cells were isolated and expanded by explant culture or by enzymatic digestion with enzymes such as collagenase II, dispase, or trypsin. In order to improve the initial cell attachment, tissue culture plates were coated with collagen IV, fibronectin, or fibronectin–collagen combination coating mix (FNC) before cell plating. We were able to successfully obtain HCECs from 32% (86/269) of donor corneas. Donor age and isolation method influenced the characteristics of the resulting in vitro HCEC culture. Under all conditions tested, FNC-coated plates showed higher quality cultures than the other coatings tested. These results suggest that donor age and HCEC isolation methodology are the two factors that most directly affect the quality of the resulting HCEC culture in vitro. These factors should guide the methodological development for the clinical expansion of HCECs for the generation of bioengineered neocorneas.
Introduction
The inner layer of the cornea is a single layer of neural crest-derived cells, named corneal endothelial cells (CECs), that form a barrier between the cornea and the aqueous humor. These cells are essential for transport of water from the corneal stroma to the anterior chamber of the eye. Damage or decomposition of the CEC pump results in corneal edema and loss of vision. Currently, Descemet's stripping and endothelial kerotoplasty (DSEK) is the most common surgical technique used to treat irreversible opacification of the cornea (18). In this procedure, CECs and their underlying Descemet's membrane are physically removed from the recipient, and a very thin layer of stroma, with intact CECs from a cadaveric donor cornea, is transferred to the recipient. Although DSEK surgery has an excellent overall safety profile, there are shortcomings associated with the lack of sufficient numbers of corneas for transplantation and the quality of the corneal endothelium available from cadaveric donors (12).
To overcome the limitations of the current standard of care, cultured CECs have been introduced as a potential source of high-quality cells for restoration of damaged corneal endothelium (1,15). Previously, we developed a novel bioengineered neocornea prototype using an expandable population of human donor-derived corneal endothelial cells (HCECs) and decellularized corneal stroma scaffolds (4). We demonstrated that HCECs could be cultured and expanded in vitro, and a prototype corneal endothelium could be fabricated ex vivo by seeding the cultured HCECs onto corneal scaffolds for transplantation. Others have also shown that CECs can be expanded and seeded onto a variety of biological or synthetic corneal scaffolds to replace the damaged corneal endothelium (4,8,11,14).
HCECs are potentially an ideal cell source to build a cell-seeded bioengineered neocornea due to the ease of isolation from cadaveric corneas and their ability to be expanded ex vivo (5,9,17,19). However, there is a lack of understanding of the factors that affect the quality of the resulting HCEC cultures in vitro. Although a variety of isolation and expansion protocols exist, a systematic assessment of how parameters such as enzymatic digestion, culture surface coatings, and media components influence the phenotype and growth characteristics of the resulting cultures has not been performed to date. HCEC cultures have been derived from corneal tissue both by explant and enzymatic digestion (3,13). Similarly, CEC cultures have been expanded on a variety of culture surfaces, some of which have been postulated to enhance HCEC attachment (2,6,15). Lastly, a broad range of complex serum-supplemented culture media has been developed using different basal media containing various growth factors thought to support HCEC growth in vitro (2,3,8,13).
In this study, we evaluated some of the various parameters that have historically been used to isolate and expand HCECs in order to initiate the design of a protocol that could be scaled up into current good manufacturing processes (cGMP) and further incorporated into a construct that would facilitate preclinical and clinical evaluation of bioengineered constructs to address indications for which the DSEK procedure is the current standard of care. Several parameters were investigated, including primary cell isolation (e.g., source corneal tissue region, explant, and enzymatic digestion) and expansion (e.g., culture surface coatings and culture medium components) protocols. Subsequently, we evaluated the quality of the resulting HCEC cultures by evaluating the adherence, morphology, and expansion of the cells in vitro.
Materials and Methods
Human Corneal Endothelial Cell (HCEC) Isolation and Culture
Donor corneas (total 269) were obtained from discarded tissue after endothelial keratoplasty, penetrating keratoplasty, or from research-grade tissue from Ocular Systems, Inc. (Winston-Salem, NC, USA) under an approved protocol of the Wake Forest School of Medicine Institutional Review Board (IRB). HCECs were harvested from three different regions of cornea: whole cornea (11.5 mm diameter), center (8 mm diameter), and corneal scleral rim (or corneal periphery) (Fig. 1). Under a dissection microscope, the Descemet's membrane, including the attached corneal endothelium, was stripped from the stroma.
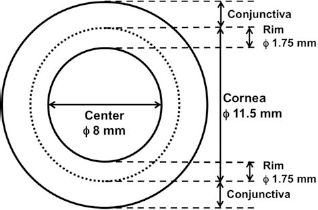
The different cornea regions: whole, center, and rim. A representative drawing of the different regions of the cornea from which human corneal endothelial cells (HCECs) were isolated, as described in the Methods and Materials section. The whole cornea was about 11.5 mm in diameter, the center of about 8 mm in diameter, and the rim was an approximately 1.75-mm-wide ring. The approximate locations of the conjunctiva are indicated.
Primary HCEC cultures were established from the dissected tissue by either explant culture directly onto culture surfaces or enzymatic digestion of the tissue to obtain single-cell suspensions that were subsequently plated. For the explant culture method, the dissected tissue was chopped into approximately 1-mm pieces and placed in a six-well plate. For the enzymatic digestion methods, the dissected tissue was placed in a six-well plate (BD Falcon®, Bedford, MA, USA) containing various enzymes: 0.1% or 0.2% collagenase II (w/v from 300 U/mg, Worthington Biochemical Co., Lakewood, NJ, USA), 0.05% trypsin-ethylenediaminetetraacetic acid (EDTA) (Gibco, Gaithersburg, MD, USA), 0.1% dispase (II) (w/v from 0.87 U/mg, Roche Diagnostics, Indianapolis, IN, USA).
HCECs were cultured in one of two growth media. One culture medium used endothelial growth medium-2 (EGM-2, Bulletkit™; Lonza Group Ltd., Basel, Switzerland) as a base, which consisted of basal culture medium, epidermal growth factor (EGF), vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), insulin-like growth factor (IGF), hydrocortisone, gentamicin, amphotericin-B, and 2% fetal bovine serum (FBS; all components are also from Lonza Group Ltd.). The concentration of FBS was adjusted up to 10%. The second culture medium used Dulbecco's modified Eagle's medium (DMEM; Gibco, Gaithersburg, MD, USA) as a base, which was further supplemented with FGF and 10% FBS. The medium was changed 3 days after primary plating and then every other day. After 2 weeks, some of the cells had reached confluence and were further subcultured (3,500 cells/cm2) by treating the plates with 0.05% trypsin-EDTA.
Tissue culture plates used to culture HCECs were either used uncoated from the manufacturer (Falcon®) or were coated prior to plating with collagen IV (BD Biosciences, San Jose, CA, USA), fibronectin (BD Biosciences), or FNC coating mix® (a mixture of bovine serum albumin, collagen I and fibronectin, 0.2 ml/cm2; Athena Enzyme Systems™, Baltimore, MD, USA).
The quality of the resulting HCEC cultures was evaluated by brightfield microscopy using a Leica DMI400B inverted microscope (Leica, Wetzlar, Germany), and images were captured using a Retiga-2000RV camera (Qimaging, Surrey, BC, Canada). HCEC cultures were scored as “successful” when the primary cell isolates had both attached to the culture dish and showed cuboidal morphology at passage 1 (P1), characteristics that are aligned with anticipated release criteria by which cultures will be evaluated for preclinical and clinical use in future product candidate formulations. In contrast, HCEC cultures were scored as “unsuccessful” when the primary cell isolates had little attachment to the culture dish and elongated morphology at P1.
Statistical Analysis
Initial descriptive statistics for the variables being studied were stratified by HCEC culture as “successful” or “unsuccessful” status. For continuous variables, we examined means and standard deviations, and for categorical variables, we examined counts and percents. The characteristics examined were donor age (continuous and categorized as a three-level ordinal variable with the following values: under 30, 30–60, and over 60 years of age), donor gender, source tissue region (categorized as button, rim, or whole), isolation method (categorized as explant, 0.1% collagenase, 0.2% collagenase, 0.1% dispase, or trypsin-EDTA digestion), culture medium (categorized as DMEM or EGM-2), and culture surface coating (collagen IV, FNC coating, fibronectin, or none).
Next, we performed a series of chi-square or Fisher's exact tests to compare “success” outcomes by categorical measure (chi-square test for variables with more than two levels and Fisher's exact tests for variables with two levels). For continuous measures (donor age), we compared the groups using two-sample t tests. We then fit stepwise logistic regression models to determine which set of predictors best predicted an HCEC isolation success. We fit these models in several different manners. First, we examined the subset of participants where donor gender was known. For this group of data, we fit stepwise logistic regression models forcing gender into the model (regardless of statistical significance in order to be sure to adjust for this characteristic). These stepwise logistic regression models were fit in two manners, the first considering donor age as a continuous variable and then as a three-level ordinal variable. We compared the fit of these models to determine which representation of age (continuous or ordinal) provided the best-predicted fit for HCEC isolation success status. For these models, only variables that were significant at 0.05 were included in the final model. In the best-fitting models, we also examined interactions between gender and the other significant predictors. Next, we fit stepwise logistic regression models using the full data set (without including gender as a covariate in the models since it was not measured on many participants). This approach was justified since we found that the gender effect was not statistically significant and that there were no significant interactions (tested at p = 0.10) in the models that were fit using the subset of participants where gender was measured. We fit two stepwise logistic regression models, using the full data set, with donor age, source tissue region, isolation method, culture medium, and culture surface considered as potential predictors, once with donor age considered as a continuous variable and once considered as a three-level ordinal variable. For each of these models, we used p = 0.05 as the criteria for a variable entering (and remaining) in the final fitted logistic regression models. All analyses were performed using SAS Version 9.2 (Cary, NC, USA).
Results
Overall Success of HCEC Isolation and Expansion
For the current study, 269 donated cornea samples were processed. The overall rate of success of HCEC isolation and culture was 31.97% (86 of 269) (Table 1). Significant differences were found in the mean of the donor age in successful versus unsuccessful cultures (47.21 ± 17.68 vs. 58.11 ± 14.23, p < 0.001). Representative light microscopic images of HCEC cultures, at passages 0 and 1, are shown in Figure 2. Successful HCEC cultures exhibited high initial cell attachment at P0 and a compact monolayer and higher cell density at P1. The unsuccessful HCEC cultures showed low initial cell attachment at P0 and exhibited an elongated morphology at P1, indicating the loss of native HCEC morphology.

Representative microscopic images of the initial culture quality of HCECs. HCECs were isolated and cultured, as described in the Methods and Materials section. Representative light microscopy images of the initial culture (P0) and at passage 1 were taken from successful and unsuccessful samples. Successful samples were defined as having high adherence after isolation (P0) and a cuboidal morphology (P1). Unsuccessful samples were defined as having low adherence after isolation with an elongated morphology.
Overall Success of HCEC Isolation and Culture

Human corneal endothelial cells (HCECs) were isolated and cultured, as described in the Methods and Materials section. The number of successful cultures and the characteristics of the study population are shown.
No, cell attachment, but no cell growth; Yes, cell attachment and cell growth up to passage 3.
Effect of Donor Age on HCEC Isolation and Expansion
In order to characterize specific factors affecting the generation of expandable cultures of HCECs, we analyzed the data shown in Table 1 based on five factors: donor age, source tissue region, isolation method, culture medium, and culture surface (Table 2). When the samples were classified into three categories based on donor age, (A) £30 years old, (B) 31 ≤ 60 years old, and (C) >60 years old (Table 2a), 60.71% of cultures derived from group A were scored a success (17 of 28) compared with 38.84% of cultures derived from group B (47 of 121) and 18.33% of cultures derived from group C (22 of 120) (p < 0.001, chi-square). Representative microscopic images of cultures isolated from each group at passages 0–5 are shown in Figure 3. HCEC cultures derived from each age group were subcultured in vitro for up to 3 months. HCEC cultures derived from group C corneas exhibited relatively slow cell growth during subculture. By passage 3, the cells had completely lost their morphological phenotype and had delayed or no further proliferation. In contrast, HCEC cultures derived from groups A and B grew continuously for five passages and maintained a compact monolayer phenotype. These results indicate that HCEC cultures with more native phenotype and higher expansion potential were obtained with higher frequency from younger corneal donors.

Representative microscopic images of established cultures of HCECs from different donor's age corneas. HCECs were isolated and cultured as described in the Methods and Materials section. Representative light microscopy images of the initial culture (P0) and at passages 1–5 (P1–P5) were taken from samples of different ages, as indicated. HCEC cultures from samples derived from samples of older donors (over 60 years old) could not be passaged beyond passage 3.
Success of HCEC Isolation and Culture With Various Parameters

HCECs were isolated and cultured, using different techniques, as described in the Methods and Materials section. The table describes the number of successful cultures and their statistical values, with regard to donor age, regions of the cornea, isolation method, culture media, and surface coating method. Other methods: combination of trypsin, EDTA, and trypsin-EDTA, dispase. EGM-2: endothelial growth medium 2 and human epidermal growth factor (hEGF), hydrocortisone, vascular endothelial growth factor (VEGF), human fibroblast growth factor-β (hFGF-b0), insulin-like growth factor (IGF-1), ascorbic acid, and heparin. FNC mix: mixture of fibronectin, collagen, and albumin. DMEM, Dulbecco's modified Eagle's medium; FBS, fetal bovine serum.
Effect of Source Tissue Region on HCEC Isolation and Expansion
When the samples were segregated by source tissue region (Fig. 1 and Table 2b), 47.06% of cultures derived from whole cornea (8 of 17) were scored a success, compared to 27.83% of cultures derived from the corneal rim (64 of 230) and 63.64% of cultures derived from the corneal center (14 of 22) (p = 0.001 from chi-square test). Although these data indicate that successful cultures were derived from the corneal rim with a lower frequency than other regions, the sample sizes for each group were not comparable. The vast majority of the samples processed in this study were from the corneal rim region because corneal rims remaining after the corneal center was used for transplantation are more plentiful than whole corneas that can be processed directly or from which the corneal center can be isolated.
Effect of the Isolation Method on HCEC Isolation and Expansion
When the samples were classified into four groups based on isolation/digestion method, explant, 0.1% collagenase, 0.2% collagenase, or other methods (dispase and trypsin-EDTA digestion groups were combined in this analysis) (Table 2c), 30.77% of HCEC cultures derived by explant were scored a success (4 of 13), compared with 31.68% of cultures derived after 0.1% collagenase digestion (32 of 101), 36.64% of cultures derived after 0.2% collagenase digestion (48 of 131), and 8.33% of cultures derived by dispase or trypsin-EDTA digestion (2 of 24). These results indicated that the use of 0.2% collagenase digestion yielded the highest frequency of successful HCEC cultures, although the p value for the chi-square test was borderline nonsignificant at p = 0.058.
Effect of Culture Media on HCEC Isolation and Expansion
When the samples were classified into two groups based on the culture medium (Table 2d), no cultures grown in DMEM were scored a success (0 of 11) compared with 33.33% of cultures grown in EGM-2 (86 of 258), p = 0.019 from the Fisher's exact test. These results indicate that EGM-2, which contains various growth factors in addition to FBS, yielded the highest frequency of successful HCEC cultures.
Effect of Culture Surface on HCEC Isolation and Expansion
When the samples were classified into four groups based on culture surface (Table 2e), 55.74% of HCEC cultures grown on FNC-coated plates were scored a success (34 of 61), compared with 22.58% of cultures grown on fibronectin-coated plates (7 of 31), 7.69% of cultures grown on collagen IV-coated plates (1 of 13), and 26.83% of cultures grown on uncoated plates (44 of 164), p < 0.001 from the chi-square test. These results indicated that FNC coating yielded the highest frequency of HCEC cultures that were scored a success.
Effect of Donor Gender on HCEC Isolation and Expansion
Donor gender data were available for only 123 samples (77 males and 46 females) of the total 269 samples (Table 3). HCEC cultures derived from male donors were scored a success more frequently than cultures derived from female donors (28.6% and 15.2%, respectively); however, this difference was not statistically significant (p = 0.12 from the Fisher's exact test). The average age of the two genders was very similar (58.0 ± 14 and 57.9 ± 13, males and females, respectively, p = NS). When donor age was stratified into the three categories, the frequency of cultures that scored as a success also appeared to be higher from male donors in the 31–60 and >60 age categories, but statistically, this apparent age by gender interaction was nonsignificant (p > 0.10).
Success of HCEC Isolation and Culture With Gender

HCECs were isolated and cultured, as described in the Methods and Materials section. The number of successful cultures with regard to the donor's gender is shown. Fisher's exact text (two-sided): p = NS for both success and age between the genders.
Stepwise Logistic Regression Models
Logistical regression models were built starting from two variables: donor gender and donor age. When we fit the stepwise logistic regression models using the subset of participants that had gender available (n = 121), we found that the best model was considered as a continuous measure included age (odds ratio 0.963, p = 0.029, indicating an inverse relationship between donor age and the success score of the resulting HCEC culture) and isolation method (p = 0.048). Table 4 shows the odds ratios, confidence intervals, and p values for the fitted model. Gender, which was forced into this model, appeared to show that male donor tissue had a higher probability of yielding HCEC cultures that scored as a success than female donor tissue; however, this effect was not statistically significant (p = 0.091). When we examined the model from isolation method, we found that digestion with 0.2% collagenase yielded HCEC cultures that had the highest probability of being scored a success, followed by explant, then 0.1% collagenase, with dispase and trypsin-EDTA having the lowest probability. When we refit the stepwise logistic regression model with age considered as a categorical covariate, only the isolation method entered the model, suggesting that age was a stronger predictor when measured as a continuous variable rather than a categorical variable. In the best model, with age (continuous) and isolation method included, we examined gender by age and gender by isolation method interactions and found these to be nonsignificant (p > 0.10).
Odds Ratio Estimates for Participants With Gender Measured

HCECs were isolated and cultured, as described in the Methods and Materials section. A stepwise logistic regression model was used to create the odds ratio estimates for samples with known gender. Values represent fold odds, 95% confidence intervals, and p values.
When the stepwise logistic regression modeling was started with donor age on the entire 269-sample dataset (omitting donor gender as a variable as it is not known for all samples), we found that age (regardless of whether age was considered as a continuous or categorical variable), coating method, and cornea region were significant predictors of success. Since the logistic regression model predicting success when donor age was considered as a continuous measure had a better fit (higher C-statistic), we show only the results from that model (Table 5).
Odds Ratio Estimates for Full Sample

HCECs were isolated and cultured, as described in the Methods and Materials section. A stepwise logistic regression model was used to create the odds ratio estimates for all samples. Values represent fold odds, 95% confidence intervals, and p values.
From both sets of analyses, those using the subset when gender was available and those using the larger participant group, we found that age is a significant predictor of success; however, depending on which dataset we analyzed, we found different other predictors in the multiple regression models (isolation method for the 121-sample dataset and surface coating and source tissue region in the full 269-sample data set).
Discussion
In 2011, over 40,000 corneal transplants were performed in the US (7). Roughly half of those transplants were for disorders primarily involving the posterior cornea, where the endothelium is located. Worldwide, the availability of posterior corneal transplant tissue is less than the demand. In the US, at present, there is adequate availability of tissue for DSEK procedures; however, since corneal endothelial cells do not regenerate, there is a need for transplant tissue with a higher density of cells, similar to the tissue obtained from younger donors. A high-quality, easily attainable, potentially less expensive, and equally safe, if not safer, alternative source of corneal tissue is desirable in the US and could help fill the more serious global need for tissue. A bioengineered cornea using cultured endothelial cells could potentially fit these requirements. The first steps to manufacturing such an implant are to identify a reliable source of cells and to establish a consistent process for culturing endothelial cells.
In our study, we have attempted to culture endothelial cells from 269 donors with varying characteristics and evaluated several isolation and expansion methods. The relatively large sample size permitted a preliminary analysis of the significance of these various factors with reasonable statistical power. Admittedly, there was a lack of equivalency in the sample sizes for several characteristics (e.g., source tissue region, isolation method, culture medium); therefore, the regression model must be viewed in light of this reality as a preliminary analysis. However, given the variety of published conditions under which primary HCEC cultures have been established, this analysis of the influence of various donor, isolation, and expansion parameters on the ability of the cells to adhere and grow provides a useful starting point for further evaluation to identify culture conditions that can yield cells that are not only capable of the appropriate level of expansion, but that also retain characteristics relevant to biological function (e.g., morphology, gene expression, protein activity).
Donor factors that we found to positively predict successful endothelial cell cultures were young (vs. old) donor age and central cornea (vs. corneal rim) as the cell source. While not statistically significant, our data suggested a trend for male gender of the source cells to yield a successful culture more frequently than cells derived from female donors. An advantage of culturing donor cells for transplantation is that cells from one donor can be expanded to produce transplantable tissue for multiple recipients. Therefore, the possibility exists that very selective donor criteria could still yield sufficient implants to increase supply above current levels. Our study suggests that a potential set of donor criteria might be the central cornea region of a young (≤30 years old) male donor. Factors that were found to positively influence the establishment of HCEC cultures that scored as a success were the use of EGM-2 as culture medium and the use of FNC coating (mixture of fibronectin, collagen I, and bovine serum albumin) as a culture surface. While not statistically significant, we found a trend that cells isolated with 0.2% collagenase digestion were more likely to produce successful cultures than other tested isolation methods.
Our findings are consistent with that of other studies. For example, Zhu et al. compared the effect of several growth-promoting factors on proliferation of HCECs from young donors (<30 years old) and older donors (>50 years old) (19). Their results showed that HCECs from young donors were more sensitive to growth-promoting factors. FBS alone, or combination with EGF, nerve growth factor (NGF), and pituitary extract, stimulated the highest proliferation of HCECs, regardless of donor age. Moreover, FBS consistently yielded higher cell numbers in HCECs obtained from younger donors.
Our results for ideal source tissue region (button, rim, or whole) are different from other previously published studies. For example, Konomi et al. found higher proliferation capacity of peripheral CECs compared with cells isolated from other corneal regions (10). However, our study used a different definition for distinguishing central from rim regions of donor corneas. In our study, peripheral cells were isolated from rim tissue remaining after the corneal button was harvested for transplant. In contrast, Konomi et al.'s definition of the central region of the cornea was smaller than the button harvested for transplant in our study; therefore, cells from a “midperiphery” region were included in the rim tissue of Konomi et al. and in the button region as defined in our study. It is possible that HCECs with the highest proliferative capacity are located in this midperipheral region that was designated differently in each of the two studies.
EGM-2 was used as a starting point for our culture medium, and due to good initial results we continued its use. Further study is currently being conducted to optimize EGM-2 for HCEC in vitro expansion. Peh et al. evaluated various culture media in the isolation and proliferation of HCECs (16); however, in their study, the typical morphological characteristics of cultivated HCECs were not maintained beyond the third passage. Although this study evaluated several factors affecting the frequency with which HCECs adhered and expanded in vitro, there are additional parameters that could affect both the establishment of in vitro cultures and maintenance of a phenotype that is predictive of clinical outcome. Further study will extend the findings presented here by characterizing in vitro culture conditions and evaluating the in vivo function of cultured HCECs via transplant into animal models.
In summary, our large series of HCEC culture attempts and successes has confirmed earlier work by ourselves and others that in vitro HCEC culture is feasible. Furthermore, the dataset generated was large enough to identify some parameters that influence the quality of the resulting in vitro cultures with statistical significance. This preliminary analysis forms a foundation upon which to build an isolation, characterization, and expansion process that can potentially yield in vitro HCEC cultures suitable for use in preclinical and clinical evaluations of bioengineered posterior cornea implants.
Footnotes
Acknowledgments
This research was supported by Ocular Systems, Inc. (Winston-Salem, NC, USA) and a Collaborative Funding Grant (CFG) from the North Carolina Biotechnology Center. Jin-San Choi is an employee of Ocular Systems, Inc., who partially supported the study.
