Abstract
Corneal transplantation is currently the only effective treatment option for dysfunctional corneal endothelial cells (CEC). In this study, we test in vitro the surgical potential of cultivated human corneal endothelial cells (hCEC) on human anterior lens capsule (HALC), LinkCell™ bioengineered collagen sheets of 20-µm thickness (LK20), and denuded Descemet membrane (dDM) as tissue-engineered grafts for Descemet membrane (DM) endothelial keratoplasty (DMEK) to bypass the problem of donor tissue availability. Primary hCEC cultured on all carriers formed a monolayer of tightly packed cells with a high cell viability rate (96% ± 4%). hCEC on HALC and LK20 showed unremarkable expression of zonula occludens-1 (ZO-1) and Na+/K+-adenosine triphosphatase (ATPase), while Na+/K+-ATPase expression of cells seeded on dDM was mainly cytoplasmic. All hCEC–carrier constructs were evaluated by simulating DMEK surgery in vitro using a human donor cornea without DM mounted on an artificial anterior chamber (AC) and a regular DMEK-graft used as a surgical reference model. During in vitro surgery, hCEC–HALC constructs behaved most similarly to a DMEK-graft during implantation and unfolding, showing good adhesion to the bare stroma. On the other hand, hCEC–LK20 and hCEC–dDM constructs required some additional handling because of challenges related to the surgical procedure, although they were both successfully unfolded and implanted in the artificial AC. The hCEC–dDM constructs showed similar graft adherence as hCEC–HALC constructs, while adherence of hCEC–LK20 constructs was less effective. After the in vitro surgery, the estimated area populated by viable cells on the hCEC–HALC and hCEC–LK20 constructs was ∼83% and ∼67%, respectively. Overall, hCEC–HALC constructs behaved most similarly to a DMEK-graft during in vitro DMEK surgery, while graft adhesion and surgical handling, respectively, are parameters still requiring optimization for hCEC–LK20 and hCEC–dDM constructs.
Keywords
Introduction
Human corneal endothelial cells (hCEC) are vital for maintaining corneal transparency 1 . Loss in hCEC density and functionality due to injury or dystrophic degeneration may lead to corneal edema, haze, and, ultimately if left untreated, to corneal blindness 2,3 . Since hCEC are not thought to replicate in vivo 4,5 , corneal transplantation is currently the only effective method to restore a healthy endothelial barrier 6 . Over the past two decades, corneal transplantation for endothelial disease has evolved from full-thickness penetrating keratoplasty to thinner, more selective surgeries. The thinnest of all is Descemet membrane (DM) endothelial keratoplasty (DMEK), where the damaged corneal endothelial cells (CEC) and the DM of the recipient are removed and replaced with those of a healthy donor 7,8 . This results in faster visual rehabilitation and better visual acuity. Despite the successful outcomes a major limitation for DMEK, and corneal transplantation in general, is the worldwide shortage of suitable donor tissue 9 .
The goal of tissue engineering in corneal grafting is to bypass the problem of donor tissue availability. Endothelial cells can be cultured in vitro and injected as a cell suspension but we expect that by attaching them to a scaffold structure, higher cell densities can be achieved, and implantation can be done in a controlled and reproducible manner, with fewer cells being lost to the aqueous flow through the trabecular meshwork 10 –12 . Potential cell carrier scaffolds may be described as “natural,” that is, derived from the body, like human anterior lens capsules (HALC) or denuded Descemet membrane (dDM), or as bioengineered structures made from collagen, gelatin, or a combination of biopolymers 13 –20 . From a translational point of view, a key requirement is that the engineered cell–carrier constructs can be surgically handled and implanted without excessive tissue manipulation, preferably in a reproducible, standardized manner, like a conventional DMEK.
The aim of this study was to assess the suitability of hCEC–carrier constructs for transplantation in vitro, following the DMEK surgical protocol as a model. For this purpose, we expanded isolated hCEC on three different carriers: HALC, collagen-based bioengineered scaffolds, and DM deprived of their endothelial cells (dDM).
Materials and Methods
Materials
Collagenase from Clostridium histolyticum (type A), Dulbecco’s phosphate-buffered saline (PBS), fetal bovine serum (FBS),
Research-Grade Human Corneoscleral Tissues
Twelve research-grade human corneas ineligible for transplantation but with intact and viable endothelium, obtained from Amnitrans EyeBank Rotterdam and with a mean age of 67 (±12) yr (range 54–83 yr, Table 1), were included in the study. There were no statistically significant differences among the average donor ages of the corneas used to isolate cells to be seeded on HALC, on LinkCell™ bioengineered collagen sheets of 20-µm thickness (LK20), and on dDM, respectively (70 (±9) vs 67 (±13) vs 74 (±13) yr, all P > 0.05). Other baseline donor characteristics are listed in Table 1. All donors of tissue used in this study had stated to have no objection against transplant-related research and the study adhered to the tenets of the Declaration of Helsinki and the Barcelona Principles 21 .
Demographics of Donor Data.

Mean storage time: time between death and culture of first isolated Descemet membrane–endothelial cell tissue; SD: standard deviation.
Isolation and Growth of Human Corneal Endothelial Cells
A two-step, peel-and-digest method was applied for primary hCEC isolation. The DM-EC sheets were first prepared as described previously 22,23 and stored in growth factor–depleted, DMEM-based medium for 4–7 d before isolation and expansion of the cells 24 .
hCEC were isolated from the DM-EC sheets as described previously
24
. Briefly, the DM-EC sheets were exposed to 2 mg/ml collagenase (in DMEM) solution for 3–6 h at 37°C and 5% CO2 to dislodge hCEC from DM, which resulted in tightly packed hCEC clusters. The hCEC clusters were further dissociated into single cells with TrypLETM for 5 min at 37°C and the resulting cell suspension was centrifuged at 500 rpm for 5 min at 37°C. The cell pellet was resuspended in fresh culture medium (DMEM containing 15% FBS, 2 mM
Cell Proliferation and Viability
Immunohistochemical staining of proliferating cells was determined with anti-Ki67 antibody 26 as previously described 27 , followed by incubation with DAPI for staining nuclear deoxyribonucleic acid (DNA). Cell viability was measured using a ReadyProbes™ Cell Viability Imaging Kit. P2-cultured hCEC on FNC-coated substrates were incubated with the premixed solution of live and dead cell stain reagents for 15 min. The number of cell nuclei with compromised plasma was determined and averaged in three areas, each measuring 0.01 mm2, on digital microphotographs. Fixed-frame method was used to manually calculate the percentage of viable cells.
Preparation of Cell Carriers
HALC and dDM were isolated from human donor eyes at Amnitrans EyeBank Rotterdam, as previously described 22,23,27 . The HALC carriers were prepared by gently lifting the anterior capsule with forceps. An incision into the lens equator was made with a surgical blade to accomplish a 360° cut. The obtained HALC were spread epithelial side up, and the lens cortex which was still adherent to the posterior side was removed using a surgical spear (Simovision BV, Diemen, the Netherlands) soaked in 70% ethanol. The HALC were then trephined to the desired size (Ø 8.0 mm) from the interior side. dDM was prepared by first isolating a DM-EC sheet as previously described and then by trephining the carrier to the customized size (Ø 8.0 mm). Both the isolated HALC and the DM-EC sheet were then treated with TE solution for 20 min to obtain a decellularized carrier (supplemental Fig. S1). Remaining, loose lens epithelium or endothelium of the HALC and the DM, respectively, were removed by mechanical sweeping using a sponge soaked in balanced salt solution (BSS, Alcon Nederland BV, Gorinchem, the Netherlands).
For cell seeding, the freshly isolated HALC (with the epithelial side down) 17 and dDM (with the endothelial side up) were spread over FNC-coated glass coverslips in 48-well plates and kept moist in PBS. For both carriers, the orientation was carefully checked before spreading over the FNC-coated glass, in order to ensure the cell seeding on the correct side of the membrane (nonepithelial side for the HALC and endothelial side for the dDM).
LK20 20 were provided by LinkoCare Life Sciences AB (Linköping, Sweden). Diameter of the LK20 carriers for the experiments was 8.0 mm.
Immunofluorescence
For cell characterization, phenotypical markers ZO-1 and Na+/K+-ATPase were used 28,29 . Cells at P2 were cultured for 1 wk directly on the biocompatible carriers and were fixed with 4% PFA in PBS or ice-cold methanol for 15 min at room temperature. Next, samples were permeabilized using 0.1% Triton X-100 and unspecific binding of the antibodies was blocked with 3% BSA in PBS for 30 min. Samples were subsequently incubated with primary antibodies anti-ZO-1/TJP1 (dilution 1:100) and anti-Na+/K+-ATPase (dilution 1:100) for 1 h, followed by secondary antibodies (dilution 1:200) in the dark for 45 min at room temperature. Both primary and secondary antibodies were diluted in blocking buffer and samples were rinsed with PBS before and after each incubation step. Further, the samples were stained with the DNA-specific dye DAPI, and then imaged using an inverted fluorescence microscope.
In Vitro Surgeries
In vitro surgeries were performed using human anterior corneal remnants (donor corneas of which the endothelium and attached DM had been peeled off) mounted onto an artificial anterior chamber (AC; DORC International, Zuidland, the Netherlands) 30 . Surgeries were performed by two experienced DMEK surgeons (ID, SND) who were masked regarding the used carrier to avoid bias.
In vitro surgical procedure was performed similar to a standard DMEK surgery 31 with some modifications as described previously 27 . In brief, three side ports were made at 2, 7, and 10 o’clock limbus-side, the artificial AC was filled with PBS, and a 3-mm main incision was made at 12 o’clock. After staining with 0.04% Trypan Blue, hCEC–carrier constructs were implanted through the main incision into the artificial AC by means of our standard DMEK glass injector (DORC International). Two of the constructs, the HALC and the dDM, were seen to roll with the endothelium facing inwards, which is the opposite direction to a standard DMEK roll. The surgery was therefore modified. The “Moutsouris sign” orientation 31 check was performed behind the roll rather than on top, and a small air bubble for unrolling was placed under the constructs, rather than over the hCEC–carrier constructs. Next, the hCEC–carrier constructs were centered, unfolded, and repositioned against the posterior stroma by using a larger air bubble. The AC was then fully pressurized with air for 1 h in order to enhance the adherence of the hCEC–carrier constructs to the posterior stroma. After 1 h, a partial air–fluid exchange was performed through the paracentesis, leaving a 50% air fill, and the hCEC–carrier construct attachment to the posterior stroma was evaluated by anterior-segment optical coherence tomography (AS-OCT; Heidelberg Engineering GmbH, Heidelberg, Germany). All hCEC–carrier constructs were subjectively scored by the surgeon on a 5-point scale (i.e., 0–5) on the following parameters: (1) ability to roll in BSS, (2) staining intensity with 0.04% Trypan Blue solution, (3) insertion and positioning into the artificial AC, (4) response to surgical graft unfolding techniques, (5) transparency, and (6) ability to adhere to the posterior surface of the donor corneal stroma. Higher scores indicated closer resemblance to the DMEK reference model (i.e., DMEK control grafts).
Cell Viability After In Vitro Surgeries
Calcein-AM was applied to the hCEC–carrier constructs to assess endothelial cell viability after in vitro surgeries. Briefly, the hCEC–carrier constructs were carefully transferred endothelial side up from the human anterior corneal remnants onto silane-coated glass slides and incubated for 45 min in the dark at room temperature with 100 µl of PBS containing 400 µM Calcein-AM. After one more PBS washing step, fluorescence images of the hCEC–carrier constructs were taken, and the level of cellular fluorescence was determined with ImageJ (National Institute of Health, Bethesda, Maryland, USA, https://imagej.nih.gov/ij/) using the thresholding method. In brief, the fluorescence images were converted to 8-bit images to measure the fraction of pixels (i.e., particle area fraction) in the selection that represents fluorescence intensities from viable cells.
Results
hCEC Viability and Proliferation During In Vitro Expansion
The native corneal endothelium is a uniform monolayer of tightly packed, hexagonal CEC (Fig. 1A) that express phenotypical and functional markers such as ZO-1 and Na+/K+-ATPase, respectively (Fig. 1B, C). Primary hCEC cultured and expanded in vitro formed a uniform cell layer with expression of ZO-1 and Na+/K+-ATPase (Fig. 1D–F). Moreover, cultured hCEC retained proliferative capacity (Fig. 2A) and high cell viability (96% ± 4%) (Fig. 2B).
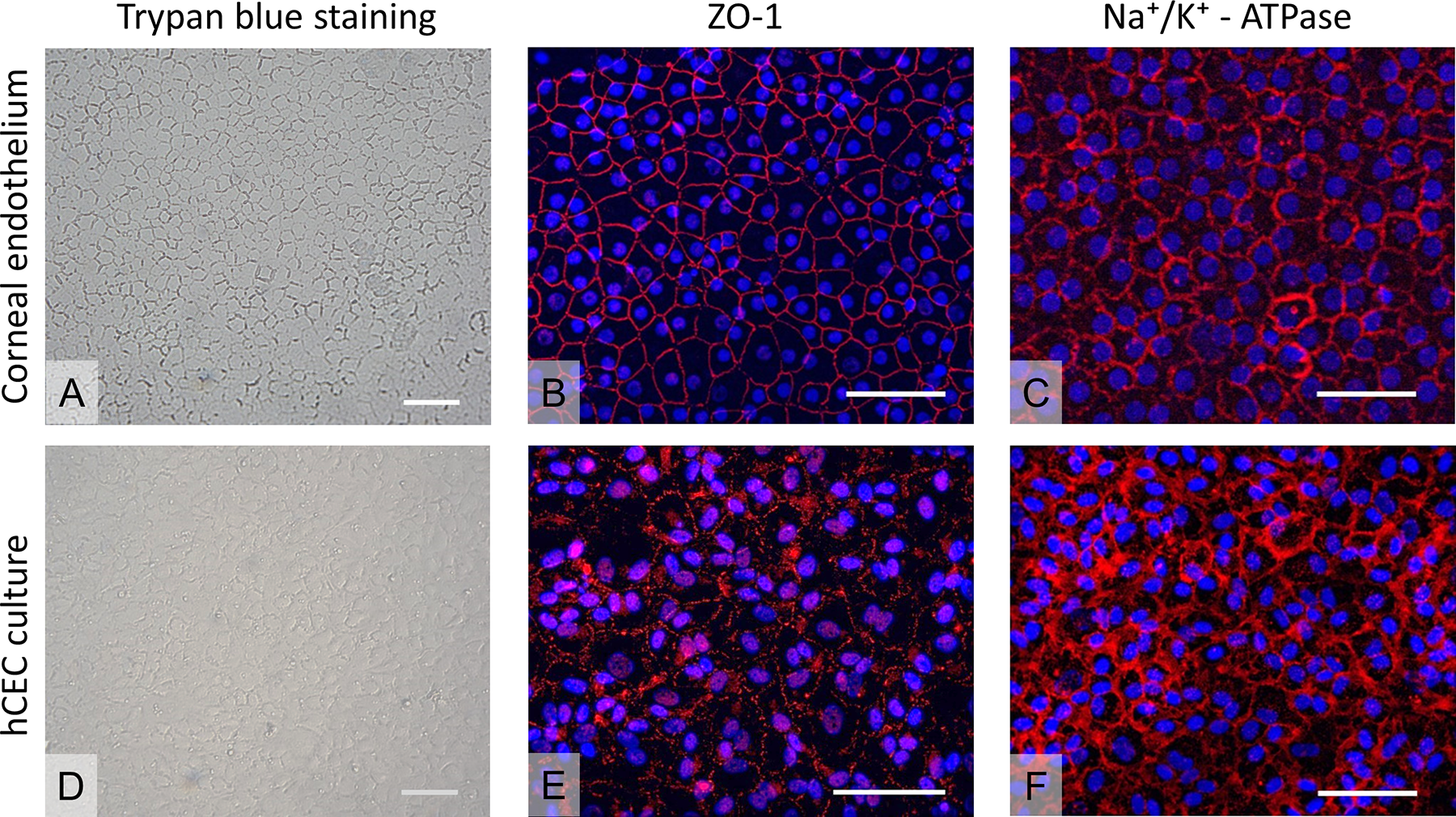
Evaluation of cell morphology on human corneas and of hCEC cultures. (A) Human cornea stained with hypotonic Trypan Blue solution 0.04%. (B) Expression of ZO-1 detected via immunofluorescence on human corneal endothelium. (C) Expression of Na+/K+-ATPase detected via immunofluorescence on human corneal endothelium. (D) hCEC cultured at P2 upon FNC-coated glass coverslips and stained with hypotonic Trypan Blue solution 0.04%. (E) Expression of ZO-1 detected via immunofluorescence on hCEC cultured at P2 upon FNC-coated glass coverslips. (F) Expression of Na+/K+-ATPase detected via immunofluorescence on hCEC cultured at P2 upon FNC-coated glass coverslips. Scale bars: 100 µm. ATP: adenosine triphosphate; FNC: fibronectin, collagen, and albumin; hCEC: human corneal endothelial cells; ZO: zonula occludens.

hCEC proliferation and viability at P2. (A) Evaluation of cell proliferation by expression of Ki67 (red). Nuclei are stained by DAPI in blue. (B) Live–dead assay to determine cell viability (blue: live; green, in circles: dead (= compromised plasma membrane integrity). Scale bars: 100 µm. DAPI: 4′,6-diamidino-2-phenylindole; hCEC: human corneal endothelial cells.
hCEC–Carrier Constructs
Confluent cells at P2 were dissociated and seeded on the selected carriers (HALC, LK20, dDM) with an average cell seeding density of 36,932 ± 15,393 cells/ml of the cell suspension. At confluence, average densities were 2,033 ± 275, 1,911 ± 285, and 1,733 ± 258 cells/mm2 for cells seeded on the HALC, LK20, and dDM carriers, respectively, with no significant difference between the cell density on the HALC and LK20 carriers, but lower densities on the dDM with cells displaying a flat and spread morphology (Fig. 3). hCEC on HALC and LK20 showed unremarkable expression of the markers ZO-1 and Na+/K+-ATPase, while expression of Na+/K+-ATPase for cells seeded on the dDM was mainly cytoplasmic (Fig. 4). After detaching the constructs from the FNC-coated coverslips, the hCEC–HALC and hCEC–dDM constructs, floating freely in culture medium, showed the tendency to spontaneously form a roll, with the hCEC located on the inner surface, unlike a DMEK roll where the endothelium is located on the outer surface. Because of their biomechanical properties, hCEC–LK20 constructs did not form a roll in culture medium.

hCEC morphology at P2 on different biocompatible carriers. Series of illustrative figures showing hCEC cultures at P2 on different carrier. (A) HALC, (B) LK20, (C) denuded DM. Scale bars: 100 µm. DM: Descemet membrane; HALC: human anterior lens capsules; hCEC: human corneal endothelial cells; LK20: LinkCell™ bioengineered collagen sheets of 20-µm thickness.
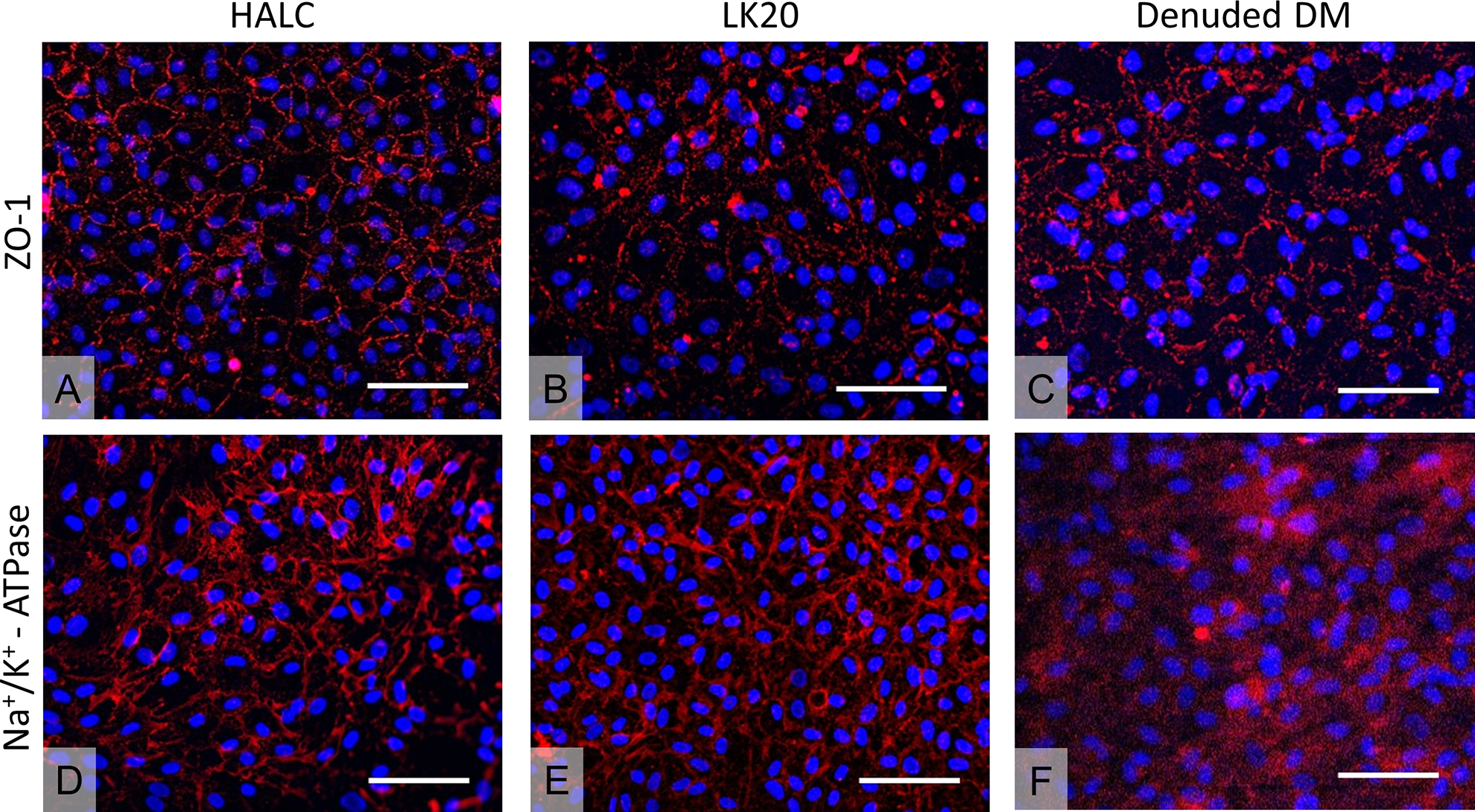
hCEC morphology check at P2 through immunofluorescence on different carriers. (A–C) ZO-1 expression detected in hCEC cultured on HALC, LK20, and denuded DM, respectively. (D–F) Na+/K+ expression detected in hCEC cultured on HALC, LK20, and denuded DM, respectively. Scale bars: 100 µm. ATP: adenosine triphosphate; DM: Descemet membrane; HALC: human anterior lens capsules; hCEC: human corneal endothelial cells; LK20: LinkCell™ bioengineered collagen sheets of 20-µm thickness; ZO: zonula occludens.
In Vitro Surgeries With hCEC–Carrier Constructs
In vitro surgeries were performed for the HALC (n = 5), LK20 (n = 4), and dDM (n = 3) cell–carrier constructs. Additionally, standard DMEK-grafts were used as a positive control (Fig. 5) and served as a reference for the scoring (Table 2). The hCEC–HALC constructs behaved overall very similar to the DMEK reference model. These constructs could be stained with 0.04% Trypan Blue for visualization during surgery and loaded as “double rolls” in a DMEK glass injector (Table 2, Fig. 5A, B). While standard DMEK-grafts roll with their endothelium on the outer surface, hCEC–HALC constructs rolled with the hCEC on the inner side. Before implantation, the graft orientation was carefully checked under a surgical microscope with particular attention to position the endothelium in its correct orientation (Fig. 5B). All hCEC–HALC constructs could be implanted and unfolded by using a surgical approach similar to a DMEK procedure. Unfolding and positioning of the constructs against the posterior surface of the stroma was achieved in a “no-touch” fashion, with some modification to the technique as described earlier (Fig. 5C). One-hour post surgery, OCT measurements showed almost complete adhesion to the bare stroma after a partial air–fluid exchange (Fig. 5D).

In vitro surgeries with hCEC–carrier constructs. The upper three rows show each of the three tested hCEC–carrier constructs after staining with hypotonic Trypan Blue solution 0.04% (A, E, I), loading into the injector (B, F, J), insertion into the artificial anterior chamber and unfolding (C, G, K), and adherence to the bare stroma visualized by anterior segment OCT measurement (D, H, L). The lower row (M–P) shows the corresponding images of a DMEK-graft that served as a positive control and reference point for the scoring. White arrows in image (H) indicate the area without adherence to the bare stroma. DM: Descemet membrane; DMEK: Descemet membrane endothelial keratoplasty; HALC: human anterior lens capsules; hCEC: human corneal endothelial cells; LK20: LinkCell™ bioengineered collagen sheets of 20-µm thickness; OCT: optical coherence tomography.
In Vitro Surgeries With hCEC–Carrier Constructs: Scoring Scheme.
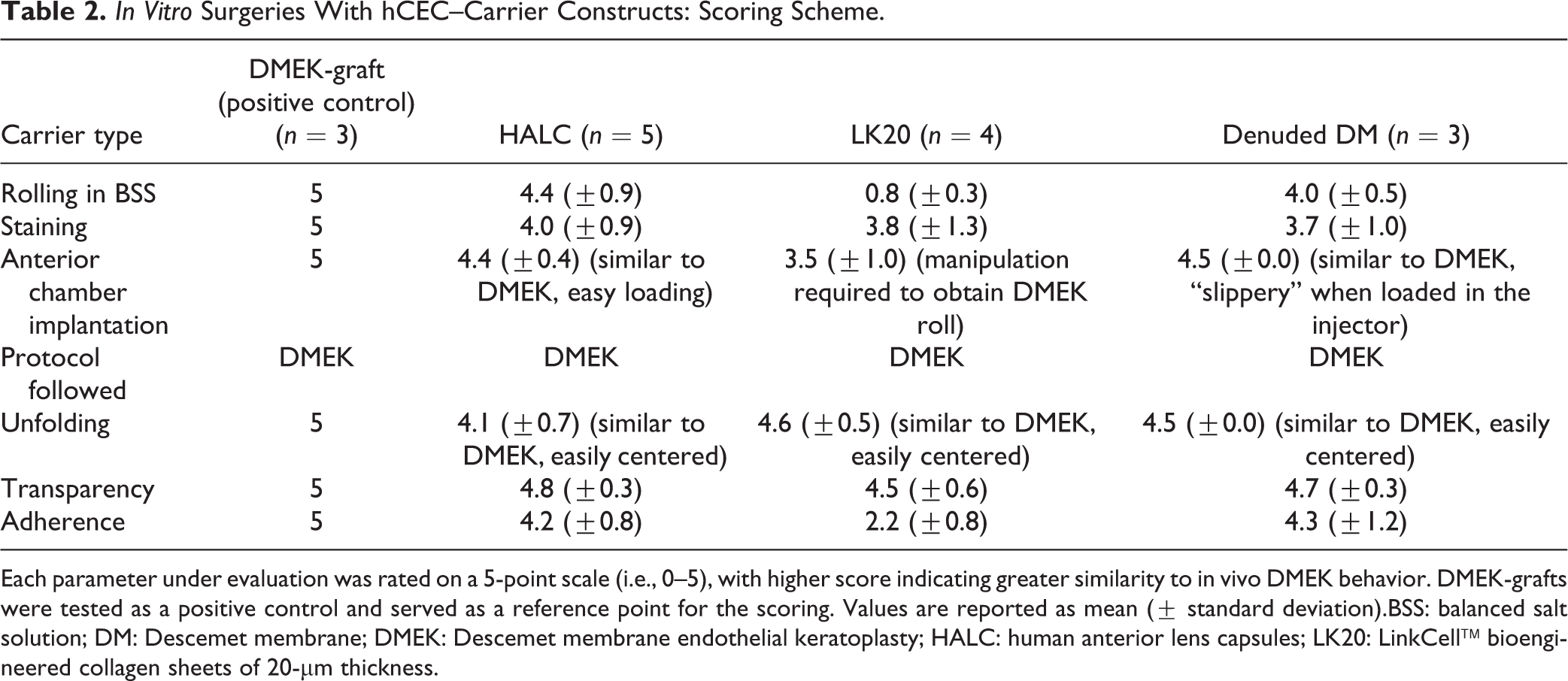
Each parameter under evaluation was rated on a 5-point scale (i.e., 0–5), with higher score indicating greater similarity to in vivo DMEK behavior. DMEK-grafts were tested as a positive control and served as a reference point for the scoring. Values are reported as mean (± standard deviation).BSS: balanced salt solution; DM: Descemet membrane; DMEK: Descemet membrane endothelial keratoplasty; HALC: human anterior lens capsules; LK20: LinkCell™ bioengineered collagen sheets of 20-µm thickness.
The hCEC–LK20 constructs did not roll spontaneously but did show a similar staining capacity with Trypan Blue when compared to the hCEC–HALC constructs and standard DMEK-grafts (Table 2, Fig. 5E). In order to inject the construct, a cannula was used to lift and manually load the hCEC–LK20 construct into the injector, with the endothelium on the external surface (Fig. 5F). The unfolding of the hCEC–LK20 construct in the artificial AC resembled the unrolling of a Descemet stripping automated endothelial keratoplasty graft more than that of a DMEK but the transparency was comparable to the other two tested carriers (Fig. 5G). The graft was lifted to the posterior stroma, but the rigidity of the material resulted in some radial peripheral fold and OCT measurement revealed incomplete adherence of hCEC–LK20 constructs to the bare stroma, 1-h post in vitro surgery (Fig. 5H).
The hCEC–dDM constructs behaved in a manner similar to the hCEC–HALC constructs in both Trypan Blue staining (Fig. 5I) and rolling in BSS (with its endothelium on the inside of the scroll). Although these constructs tended to be fragile and slippery when loaded into the injector, the implantation in the artificial AC and the unfolding were successfully achieved in all cases (Fig. 5J, K). One-hour post in vitro surgery, all hCEC–dDM constructs showed good adherence to the bare stroma (Fig. 5L).
After the OCT measurements, endothelial cell morphology and viability were evaluated on reference DMEK-grafts, hCEC–HALC, and hCEC–LK20 constructs. Calcein-AM staining showed minimal damage to the reference DMEK-graft (viability of 90 (±3)%) except for some areas that were already affected after graft preparation due to the low quality of the cornea that had been discarded for transplantation (Fig. 6A, D). Light microscopy showed centrally an almost intact monolayer of hexagonal hCEC on the HALC (Fig. 6B), while the hCEC layer on the LK20 showed several gaps (Fig. 6C). This finding was also reflected by Calcein-AM staining which resulted in an estimation of ∼ 83 (±5)% and ∼ 67 (±9)% of viable cells present after in vitro surgery on hCEC–HALC constructs and hCEC–LK20 constructs, respectively (Fig. 6E, F).

Cell assessment after in vitro surgeries. (A–C) Central light microscopy images of a DMEK reference graft and the hCEC–HALC and hCEC–LK20 constructs. White arrows in (C) point to gaps in the hCEC monolayer. Scale bars = 100 µm. (D–F) Calcein-AM expression detected on the DMEK reference graft and the hCEC–HALC and hCEC–LK20 constructs, respectively. Scale bars = 1 mm. DMEK: Descemet membrane endothelial keratoplasty; HALC: human anterior lens capsules; hCEC: human corneal endothelial cells; LK20: LinkCell™ bioengineered collagen sheets of 20-µm thickness.
Discussion
In this comparative study, we tested hCEC–carrier constructs in in vitro surgeries comparing them to a DMEK-graft reference model in terms of rolling, staining, unfolding, and adherence to the bare posterior stroma of a human donor anterior remnant.
Tissue-engineering of corneal grafts by in vitro expansion of hCEC relies on the availability of suitable biocompatible carriers. Ideal carrier candidates, which could be of different origins (“biological” and “bioengineered”), composition, and biomechanical properties, should mimic native tissue microenvironment to assure cellular functionality and offer structural and mechanical support for the transplantation using the DMEK technique.
Our study confirmed the potential of HALC as a cell culture substrate as previously shown for different types of ocular cells 32 –34 , including hCEC 15,17 . Its composition is one of the major advantages of the HALC as a scaffold, as it is very similar to the native DM. HALC is mainly formed by the membrane protein collagen IV and other matrix components including collagen I, collagen III, collagen VIII, laminin, and fibronectin 35 . These core molecules self-assemble into a 3D matrix that provides strength and flexibility to the lens. During in vitro surgery, hCEC–HALC constructs behaved comparable to the DMEK reference model in terms of loading into the artificial AC and adherence to the stroma even though the hCEC–HALC constructs showed a “reversed” rolling with the endothelial cells on the inside. After in vitro surgeries, hCEC–HALC constructs also retained a high endothelial cell viability rate (∼82% of the surface area) suggesting that these constructs may be handled surgically with minimal effect on cell viability.
The second biological construct, hCEC–dDM also showed similarities with the DMEK reference model in terms of staining and adherence upon in vitro surgery, which may be related to similar biological properties shared by the two carriers. Seeding of the cells on the dDM carriers induced a “reversed” rolling of the constructs, that is, with the endothelial cells on the inside, as also observed for the hCEC–HALC constructs. Therefore, hCEC–HALC and hCEC–dDM constructs had to be implanted into the artificial chamber by providing small changes compared to the standard DMEK surgical technique.
The preparation of dDM requires an enzymatic treatment in order to remove all natie CEC present on the DM prior to using it as a cell culture carrier. This enzymatic treatment has been reported to also remove parts of the fibronectin-mediated cell–extracellular matrix interaction 36 . Alternatively, usage of a cotton swab to mechanically disrupt cells might create microgrooves subsequently affecting cell alignment and morphology of the freshly seeded CEC 36 . In addition, dDM originating from elderly donors shows a highly structured surface due to imprints left by the removed cells. This structured surface seems to impair the morphology of the cultured cells on these carriers.
In this early stage of developing tissue-engineered cell–carrier constructs as a potential alternative to standard endothelial grafts, both of the tested natural carriers can serve as a benchmark for bioengineered carriers in terms of required surgical handling and robustness upon surgical manipulation in an in vitro setting. For potential future clinical applications, however, using natural carriers such as HALC and dDM as a substrate will still be dependent on donor tissue.
An alternative to the natural carriers is bioengineered substrates such as the LK20 carrier. Bioengineered materials offer distinct advantages over natural carriers: they are donor-independent and could be produced in large quantities and with reproducible quality. Previous studies reported, for example, on the use of silk fibroin-based carriers or plastic compressed type I collagen hydrogel membranes (termed RAFTs) 37,38 as cell carriers and we also reported the feasibility of collagen I–based carrier LK20 for culturing and expanding porcine CEC 27 . Here, we have shown that hCEC could also be successfully cultured on the LK20 carrier. However, upon in vitro surgery, the hCEC–LK20 constructs showed less adherence to the posterior stroma than other carriers after implantation. In addition, an endothelial cell viability rate of ∼67% after in vitro surgery may either reflect cell damage due to the additional handling required during the surgery or may be due to a “compatibility issue” between cells and carrier. In a study involving hCEC seeding on fish scale–derived scaffolds, it has been reported that the stiffness of the underlying matrix (expressed as Young’s modulus) was not sufficient for the cells to properly adhere to the carriers 39 . Improvements on the biophysical (strength of cell-substrate adhesion or membrane permeability) and biomechanical properties (e.g., tensile strength, elasticity) may facilitate cell adherence to the LK20 carriers as well as the surgical handling, which should be further validated in vitro to define the potential of LK20 collagen sheets as suitable carriers for hCEC sheet transplantation.
Some limitations of this study have to be highlighted such as the small sample size. Due to the limited availability of research corneas of sufficient quality, tests could not be performed on a large scale to improve repeatability and reproducibility. In addition, donor age of the available donor cornea, which is mainly derived from elderly donors, may affect the hCEC proliferation capacity. Although there was no statistical difference in donor age for cells seeded onto different carriers, observed hCEC morphology may still have been affected by donor age.
In conclusion, all selected carriers were suitable for in vitro hCEC culture and expansion, and in vitro surgical manipulation showed that the hCEC–carrier constructs behaved mostly like the DMEK reference model, especially the hCEC–HALC construct. However, for potential clinical applications, the use of natural carriers would require reproducible carrier quality while biomechanical properties of the tested bioengineered carrier would need to be further improved to ensure successful graft function.
Supplemental Material
Supplemental Material, 1803_In_vitro_human_cell_sheet_transplantation_-_Supplemental_Figure - In Vitro Evaluation and Transplantation of Human Corneal Endothelial Cells Cultured on Biocompatible Carriers
Supplemental Material, 1803_In_vitro_human_cell_sheet_transplantation_-_Supplemental_Figure for In Vitro Evaluation and Transplantation of Human Corneal Endothelial Cells Cultured on Biocompatible Carriers by Daniele Spinozzi, Alina Miron, Jessica T. Lie, Mehrdad Rafat, Neil Lagali, Gerrit R.J. Melles, Sorcha Ni Dhubhghaill, Isabel Dapena and Silke Oellerich in Cell Transplantation
Supplemental Material
Supplemental_Data_Table - In Vitro Evaluation and Transplantation of Human Corneal Endothelial Cells Cultured on Biocompatible Carriers
Supplemental_Data_Table for In Vitro Evaluation and Transplantation of Human Corneal Endothelial Cells Cultured on Biocompatible Carriers by Daniele Spinozzi, Alina Miron, Jessica T. Lie, Mehrdad Rafat, Neil Lagali, Gerrit R.J. Melles, Sorcha Ni Dhubhghaill, Isabel Dapena and Silke Oellerich in Cell Transplantation
Footnotes
Ethical Approval
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Melles is a consultant for DORC International/Dutch Ophthalmic USA and SurgiCube International. Dr Rafat serves on the Board of Directors of LinkoCare Life Sciences AB, which is a spin-off firm developing products related to the research being reported and holds relevant patents. Dr Rafat’s terms of arrangements have been reviewed and approved by Linköping University in accordance with its policy on objectivity in research. The other authors have no conflicting relationship to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Union’s Horizon 2020 research and innovation programme (grant number 667400—ARREST BLINDNESS Consortium).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
