Abstract
Better results have been recently reported in clinical pancreatic islet transplantation (ITX) due mostly to improved isolation techniques and immunosuppression; however, some limitations still exist. It is known that following transplantation, 30% to 60% of the islets are lost. In our study, we have investigated 1) the role of size as a factor affecting islet engraftment and 2) potential procedural manipulations to increase the number of smaller functional islets that can be transplanted. C57/BL10 mice were used as donors and recipients in a syngeneic islet transplant model. Isolated islets were divided by size (large, >300 μm; medium 150–300 μm; small, <150 μm). Each size was transplanted in chemically induced diabetic mice as full (600 IEQ), suboptimal (400 IEQ), and marginal mass (200 IEQ). Control animals received all size islets. Engraftment was defined as reversal of diabetes by day 7 posttransplantation. When the superiority of smaller islets was observed, strategies of overdigestion and fragmentation were adopted during islet isolation in the attempt to reduce islet size and improve engraftment. Smaller islets were significantly superior in engraftment compared to medium, large, and control (all sizes) groups. This was more evident when marginal mass data were compared. In all masses, success decreased as islet size increased. Once islets were engrafted, functionality was not affected by size. When larger islets were fragmented, a significant decrease in islet functionality was observed. On the contrary, if pancreata were slightly overdigested, although not as successful as small naive islets, an increase in engraftment was observed when compared to the control group. In conclusion, smaller islets are superior in engraftment following islet transplantation. Fragmentation has a deleterious effect on islet engraftment. Islet isolations can be performed by reducing islet size with slight overdigestion, and it can be safely adopted to improve clinical outcome.
Keywords
Introduction
Glycemic control is a key factor in the development of chronic complications related to diabetes mellitus. Successful islet transplantation (ITX) allows improved glycemic control and quality of life in diabetic patients. Islet transplantation outcomes have improved over time with better success rates at 1 year posttransplant (3,50,51). Pancreas transplantation has been proven successful, and it is now confirmed that one pancreas is sufficient to treat diabetes regardless of insulin resistance, duration of diabetes, and other preexisting conditions (49,54). On the contrary, most of the time, islets from at least two human pancreata are needed to reverse diabetes in a type 1 patient (6,9,50), evidencing that some major limitations to this procedure and its clinical applicability still exist. A limited number of islets can be isolated from a human pancreas even under optimal circumstances (i.e., optimal donor) (13,16, 19,42). In addition, factors not completely known seem to be responsible for loss of islet functionality in the immediate posttransplant period and/or over time (13). Primary nonfunction may be attributed to transplantation of islets of poor quality (43). Other factors can involve autoimmunity (18,50,59), rejection (48), immunosuppression toxicity (31,36,58), and posttransplant inflammatory conditions (22). Significant attention has been directed toward the development of strategies to improve organ retrieval and islet isolation (2,16,19,20), and although both have been improved over time, protocols and products are not completely standardized. It is known that following transplantation (and/or culture) from 30% to 60% of the islets can be lost (8,15). Investigations have looked at the morphologic characterization of the graft (41) and at islet viability by various techniques (23,25,32,46,60). Some of these techniques have been implemented to predict clinical outcomes and possibly reach standardization of the procedure. Current clinical protocols for transplantation take into account islet size distribution converting the real number of islets to the islet equivalent number (IEQ: islet of an average diameter of 150 μm). In the clinical setting, the total number of islets is calculated as IEQ per kilogram of the recipient body weight with a target of approximately 10,000–12,000/kg. It has been general knowledge and practice in islet transplant centers that preservation of the entire islet structure is ideal for islet functionality posttransplant (1,17,21,29,33,40,55). There is evidence that pancreatic islets cannot be considered nonspecific aggregates of cells. Each islet should rather be considered as a microorganism with a complex and specific organization, and only intact islet architecture allows normal endocrine function (1,17,40). Several studies have put emphasis on the importance of the interactions among the diverse cell types (21,55) and cells of the same type (21,29) for optimal regulation of hormonal secretion and metabolic control. This is mostly due to the fact that pancreatic islets are clusters of five known different cell types that need to preserve proportion and interaction for a physiologic metabolic response (17,21,29,53,55).
Islet size has been a neglected factor and never considered as incidental on success of pancreatic ITX. Only recently, size has generated some interest as a potential condition for improving engraftment and/or functionality (24,26,35,52). The first observation was reported by Lehmann et al. in 2007 (24) following a retrospective analysis of clinical cases. This clinical observation demonstrating superiority of smaller islets by C-peptide production in seven clinical islet transplant cases was followed by animal studies confirming that smaller islets are preferable (26,35,52). More recently, Henriksnas and colleagues have shown a difference in blood perfusion related to size when islets are transplanted (14). However, the functional and observational data available so far does not seem to be sufficient to optimize and standardize protocols improving success and clinical applicability of pancreatic ITX. Nevertheless, no strategies have been investigated so far to obtain a larger number of smaller islets.
The primary aim of this study was to characterize the impact of size on engraftment and functionality of transplanted islets in a marginal mass mouse model. The study was based on the hypothesis that islet size may be a key factor that affects islet engraftment and/or functionality. Furthermore, we investigated potential strategies to manipulate the islet isolation technique to achieve optimal size and improve outcome.
Materials and Methods
Animals
C57/BL10 male mice (9–12 weeks old) were used as pancreatic islet donors and recipients (syngeneic islet transplant model). Animals were purchased from Harlan (Indianapolis, IN, USA), housed in a standard animal facility, and provided ad libitum with rodent chow and tap water. All animals were cared for according to the International Guidelines on Animal Care. The study protocol was approved by the University of Texas Medical Branch's Institutional Animal Care and Use Committee (IACUC) and performed under standard regulatory guidelines for research involving animals.
Diabetes Induction
Recipient animals were rendered diabetic 4–5 days prior to ITX by a single streptozotocin injection [intraperitoneal (IP), 220 mg/kg; Sigma, St. Louis, MO, USA]. Animals with a blood glucose level (BGL) of >300 mg/dl for 3 consecutive days became transplant candidates.
Islet Isolation
Donor animals were anesthetized by isoflurane (Terrell” Isoflurane, USP, Novaplus, Piramal Healthcare, Andhra Pradesh, India), and pancreas was harvested as previously described (47). In brief, a midline abdominal incision was performed. After clamping of the duodenal ampulla, the pancreatic duct was cannulated, and collagenase solution (0.8 mg/ml; Sigma) was injected. Following dequate distention, the pancreas was harvested and stored on ice.
Pancreata were digested, and islets were purified using Dextrane (Sigma) density gradients (1.092, 1.1, 1.083, 1.040). After purification, islets were separated by size in three different groups using stainless steel mesh (Bellco Glass, Inc., Tissue Sieve, Vineland, NJ, USA) filtrations by gravity (150-μm and 300-μm pore size, respectively) (Fig. 1). Following filtration, islet size was confirmed through observation at light microscopy (Fig. 2). Aliquots from each islet size batch were harvested and stained with dithizone (Sigma) for sizing and counting. Isolated islets were divided for ITX in three groups: small (<150 μm), medium (150–300 μm), and large (>300 μm) and transplanted as 600 (full mass), 400 (suboptimal mass), and 200 IEQ (marginal mass). In each islet-size group, the number of islets transplanted was adjusted to match the same islet mass. From each isolation, islet functionality was confirmed in vivo by diabetes reversal in control animals receiving 600, 400, or 200 IEQ of all sizes. Among all the isolations performed, only those isolations that led to reversal of diabetes in control (all sizes) animals who received 600 IEQ were considered successful and were included in the study and in the analysis.
Example of stainless steel filtration mesh. (A) Stainless steel filtration mesh used to separate islets according to their size. (B) A 150-μm pore size to separate small islets. (C) A 300-μm pore size to separate medium from large islets. Isolated islets divided by size using stainless steel mesh filtration. (A) Small islets (50–150 μm). (B) Medium islets (150–300 μm). (C) Large islets (>300 μm). Aliquots stained with dithizone and observed at light microscopy (4×).

Overdigestion
In view of our hypothesis that smaller pancreatic islets could have a better outcome in reversing diabetes, we performed isolations prolonging the digestion phase of 10 min to obtain islets of smaller size. Aliquots were stained with dithizone, counted by size, and islet size distribution was compared to control isolations.
Fragmentation of Medium and Large Islets
In view of our hypothesis that smaller pancreatic islets could have a better outcome in reversing diabetes, medium and large islets were separated from small islets and fragmented by vigorous shaking of the tissue for 15 min in a water bath at 37°C or by overexposing to collagenase for an additional 10 min (14). Islets were then filtered using a 150-μm mesh, and islet size and number were assessed by averaging, following dithizone staining of three aliquots.
Islet Transplantation
Animals were anesthetized by isoflurane. A midline incision was performed, and the right kidney was exposed. Following a puncture at the inferior pole with a 25 gauge Surflo® winged infusion set (Terumo Medical Corporation, Tokyo, Japan) connected to a 1-cc syringe, isolated islets were transplanted fresh under the kidney capsule (superior pole) using a transparenchymal approach.
Animals that experienced technical failures (anesthesia-related issues, bleeding, and death within 24 h postsurgery) were removed from the study (n = 4).
Engraftment and Functionality Assessment
Animals were monitored for BGL and body weight daily for the first 2 weeks and twice weekly thereafter. Blood glucose concentrations were determined using a blood glucose meter and strips (Accu-Chek; Roche Diagnostics, Indianapolis, UN, USA) after tail vein puncture. Transplanted islets were considered to be engrafted when a BGL of <150 mg/dl was attained and maintained. Recipients that experienced reversal of diabetes (i.e., normal BGL) within 7 days posttransplant were considered successful.
In animals that had reversed diabetes, islet functionality was assessed by multiple IP glucose tolerance tests (IPGTTs). IPGTTs were performed at short- (days 10–14) and long-term (day 60–120) follow-ups. IPGTT from naive (nontransplanted) animals (n = 2) were included as controls. Briefly, animals were fasted, and following the detection of baseline BGL, 2 mg/kg body weight of 50% dextrose (Hospira, Inc., Lake Forest, IL, USA) in 0.5 ml was injected IP. BGL was then measured at 15, 30, 45, 90, and 120 min after injection.
In all animals that reversed diabetes within 7 days and concluded the study, the graft-bearing kidney was removed to observe the diabetes reoccur and to perform histological examination. In brief, following isoflurane anesthesia, a midline incision was performed, and the right kidney was exposed. The renal peduncle was isolated and ligated using 4–0 nonabsorbable suture (Perma-Hand silk, Ethicon, Johnson & Johnson, New Brunswick, NJ, USA). The peduncle was cut on the kidney side, the graft-bearing kidney was removed, and the animal was sutured (Perma-Hand silk) and allowed to recover. When diabetes reoccurred, animals were humanely sacrificed, and the pancreas was harvested for histological analysis.
Groups
A total of 112 transplanted mice were entered in the study investigating the islet size. A total of 31 animals received all sizes of islets and functioned as control groups (n = 16, full mass and isolation control; n = 4, suboptimal mass; and n = 11, marginal mass). A total of 29 mice received small islet size (<150 μm; 9 full, 9 suboptimal, and 12 marginal). A total of 25 mice received medium islet size (150–300 μm; 8 full, 9 suboptimal, and 7 marginal). A total of 27 mice received large islet size (>300 μm; 7 full, 10 suboptimal, and 10 marginal). Blood glucose levels and body weight were recorded (data not shown) for up to 100 days posttransplant for all animals.
An additional 28 animals were transplanted with marginal masses of either overdigested islets (n = 14) or fragmented islets (n = 14). To determine the impact of possible manipulation on islet size, overdigested and fragmented islets were compared to marginal mass of control (all sizes, n = 11) and small naive islets (n = 12).
Statistical Analysis
The hypotheses of interest were whether islet size affects reversal of diabetes, and among animals that reversed diabetes posttransplantation whether islet functionality, tested with IPGTT, was affected by islet size. Owing to the small sample size, the hypotheses for islet size were tested with Fisher's exact test (FET). If the association was significant, significant subtables were identified using decomposition of chi-square based on the likelihood ratio (LR) chi-square. The log-rank test was used to compare diabetes reversibility (Kaplan–Meier plots). The hypothesis regarding how interventions on islet size could affect diabetes reversal posttransplantation was tested with the Freeman–Halton test [FET extended to general R × C tables by Freeman and Halton (12)]. The hypothesis concerning islet functionality was tested using a two-factor ANOVA with the Tukey–Kramer post hoc test to assess differences among groups. Onefactor ANOVA and Tukey–Kramer post hoc test was used to evaluate differences between groups at a certain time (120 min), and p < 0.05 was considered significant. All computations were performed with SAS 9.2 (Cary, NC, USA).
Results
Isolation Outcome by Islet Size, Overdigestion, and Fragmentation
Islet Percentage by Size per Donor

Islet size is reported as μm.
When the entire preparation was overdigested by increasing the digestion time, we observed a shift toward the left (smaller islets) compared to the control, indicating that we were actually able to reduce medium and larger islets to a smaller size (Table 1). When medium and large islets (150–350 μm) were fragmented by either prolonged shaking or extra collagenase exposure for 10 min, smaller islets could be obtained ranging from 50 to 150 μm in diameter (Table 1).
Engraftment by Islet Size
As expected, in the control group (all sizes islets), we observed a 100%, 75%, and 36% diabetes reversal in the full, suboptimal, and marginal islet mass, respectively (Table 2, Fig. 3). Within the full mass, only animals who received small islets were comparable to control and reversed diabetes 100% of the time compared to medium (75%) and large (71%) islets. In all masses, we observed a trend with increased success in engraftment from large to small islets (Table 2, Fig. 3). When marginal mass groups were compared, small islets performed significantly better than medium, large, and control size groups (75% vs. 37.5%, 10%, and 36%, respectively). The same significant difference was observed when the overall reversal of diabetes was achieved regardless of islet mass, with 86% reversal in the small group compared to 56%, 33%, and 77% in the medium, large, and control groups, respectively (Table 2, Fig. 3).
Reversal of diabetes. (A) Overall reversal by size. In the small islet group, 86% of the animals reversed diabetes (25/29) compared to medium islets 56% (14/25), large islets 33% (9/27), and control animals that reversed in 77% of the cases (23/31). Statistically significant difference in engraftment was observed with reduced engraftment as the islet size increased (FET, *p = 0.0033; †p = 0.0003 for groups marked with the respective symbols). (B) When the data were analyzed by size and mass, the same trend was observed with a confirmed increase in engraftment as the islet size decreased. This trend was highly represented in the marginal mass group where small islets reversed in 75% of the cases (9/12) compared to 37.5% (3/8), 10% (1/10), and 36% (4/11) in medium, large, and control groups, respectively (FET, *p = 0.015; †p = 0.008 for groups marked with the respective symbols). Reversal of Diabetes by Islet Mass and Size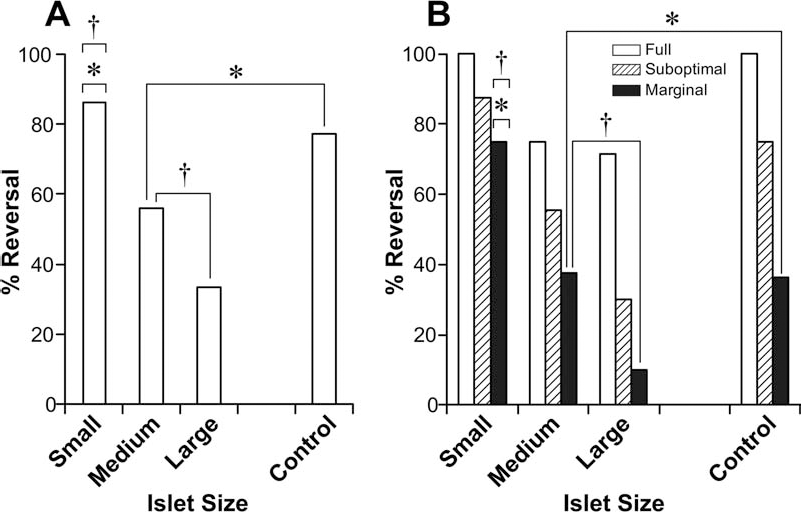

There was a statistically significant association between islet size and reversal of diabetes (FET, p = 0.0003; LR df = 3, value = 19.7896, p = 0.0002). Using the decomposition of chi-square, the association between small size versus all others was significant (FET, p = 0.015; LR df = 1, value = 9.1020, p = 0.0026).
Reversal of diabetes was uniformly distributed among each study group as shown in Figure 4 (Kaplan–Meier) and superiority of small islets was confirmed in the marginal group.
Engraftment–Reversal of Diabetes After ITX, Overdigested, and Fragmented Islet Study
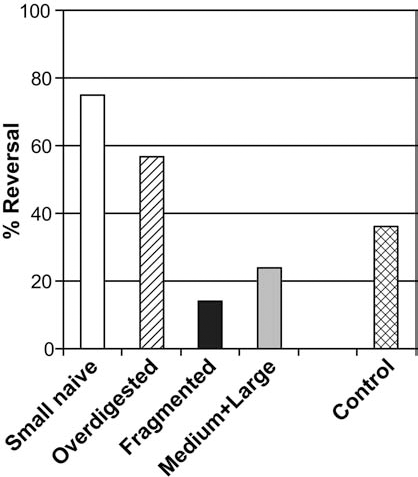
When mouse pancreata were overdigested with a shift toward smaller islets as already reported (Table 1), 57% of the animals reversed diabetes compared to 36% in the control group and 75% in the small naive group (Fig. 5). When medium and larger islets were treated and reduced to smaller islets (50–150 μm), only 14% of the animals reversed diabetes compared to 36% in the control, 75% in the small naive, and 57% in the overdigested group. Fragmentation of medium and large islets to a smaller size resulted in loss of functionality even when compared to naive medium and large islets (14% vs. 24%, respectively).
Difference in functionality was statistically significant among groups (Freeman–Halton test, p = 0.0067) (Fig. 5).
Short- and Long-term Graft Functionality
Animals that reversed diabetes were included in the functionality study. The short-term IPGTT curves were calculated as average per group, and they are reported in Figure 6. No significant difference was observed in IPGTT results from all animals that reversed diabetes. The two-factor ANOVA test did not show a difference in IPGTT when small, medium, large, control, and naive (nontransplanted) groups were compared among marginal (p = 0.1063), suboptimal (p = 0.4152), and full islet mass (p = 0.1242) (Fig. 6). No differences in short-term IPGTT were observed when naive (nontransplanted) animals and marginal mass recipients of fragmented, overdigested, small, and control islets were compared (p = 0.1469) (Fig. 7). At 120 min, we observed a statistically significant higher value of BGL in the fragmented group compared to all the other groups (p < 0.0001). Long-term IPGTT curves for each group did not differ from short-term curves (data not shown).
IPGTT curves. BGL value at different time points was calculated as average among each study group. (A) Full mass group included 16 control, 5 large, 6 medium, and 9 small islet animal recipients. (B) Suboptimal mass group included 3 control, 3 large, 5 medium, and 7 small islet animal recipients. (C) Marginal mass group included 4 control, 1 large, 3 medium, and 9 small animal recipients. IPGTT from naive (nontransplanted) animals were included for comparison (n = 4). The two-factor ANOVA test did not show a difference in IPGTT when small, medium, large, control, and naive (nontransplanted) groups were compared among marginal (p = 0.1063), suboptimal (p = 0.4152), and full islet mass (p = 0.1242). IPGTT curves: comparison in marginal mass. BGL value at different time points was calculated as average among each study group. Groups included are control (n = 4), fragmented (n = 4), overdigested (n = 8), and small (n = 9). IPGTT from naive (nontransplanted) animals were included for comparison (n = 4). The two-factor ANOVA test did not show a difference in IPGTT when naive (nontransplanted) animals and marginal mass recipients of fragmented, overdigested, small, and control islets were compared (p = 0.1469); The one-factor ANOVA at 120 min showed a statistically significant higher value of BGL in the fragmented group compared to all the other groups (*p < 0.0001).

Histological Examination
When the islet-bearing kidney was removed in anesthetized animals, diabetes reoccurred in 100% of the enrolled animals within 3 days. Animals were kept diabetic for 48 h and then sacrificed. Graft-bearing kidneys were stained for insulin, confirming pancreatic islet presence in the kidney. At sacrifice, pancreata were harvested and stained for insulin to confirm β-cell destruction (data not shown).
Time of diabetes reversal for each study group (Kaplan–Meier). (A) Full mass group. Log-rank test 0.1629. (B) Suboptimal mass group. Log-rank test 0.1204. (C) Marginal mass group. Log-rank test 0.0085. Comparison of reversal of diabetes in marginal mass. When pancreata were overdigested (n = 14), islets engrafted better than control (n = 11) (57% vs. 36%); when medium and large islets were fragmented (n = 14), islets performed worse than naive medium and large (n = 18) (14% vs. 24%). Small naive islets (n = 12) remain the most successful group with 75% of animals reversing diabetes. Freeman–Halton test, p = 0.0067.
Discussion
In the current study, we investigated the impact of size in pancreatic ITX. Pancreatic islets were transplanted under the kidney capsule of syngeneic mice, and engraftment and functionality were investigated in relation to three different islet sizes (small: <150 μm, medium: 150–300 μm, and large: >300 μm). The study was based on a marginal mass model compared to suboptimal and full to extrapolate any potential linear correlation between islet sizes and masses. All groups were compared to control animals receiving the three different masses of all islet sizes (0–>300 μm). The superiority of small islets was confirmed by significant improvement in reversal of diabetes within 7 days, regardless of the mass. When only marginal mass groups were analyzed, small islet superiority was even more evident. Based on our marginal mass model, we concluded that small islets are superior in engraftment following transplantation with a clear trend of decreased success as the islet size increases. Animals were then monitored for up to 120 days posttransplantation, and no significant differences were observed in functionality over time. While engraftment was clearly affected by size, functionality during follow-up was not, even when challenged by the glucose stimulation test (IPGTT) in the short- and long-term follow-up. Based on the evidence that smaller islets are superior in engraftment, we proceeded to investigate two approaches aimed to reduce larger islets to a smaller size. When medium and larger islets were reduced in size by fragmentation (14), islet structure resulted disrupted, and functionality was significantly impaired compared to control as well as small, medium, and large islets. When fragmented islets appeared to have engrafted, a distorted functionality was observed when challenged by glucose stimulation. On the contrary, when pancreata were slightly overdigested, the moderate shift toward smaller islets was associated to improved functionality, and although it was not as good as small islets, it was improved when compared to controls. To our knowledge, this is the most comprehensive study demonstrating, in an animal model, how different islet sizes have an impact on engraftment and long-term functionality. In addition, in the attempt to standardize the islet product to improve clinical transplant outcome, we are the first group investigating functionality of fragmented and overdigested islets in vivo.
Pancreatic ITX is a promising approach for the treatment of diabetes and can be considered as an alternative therapy to whole-pancreas transplantation (3,27,28,37,38). The establishment of ITX has gone through the development of new protocols and techniques. Recent improvements have been associated mostly to innovative immunosuppressive protocols focused on minimizing β-cell toxicity. With the experience reported by the Edmonton group (50), we learned that standardization plays a major role in the outcome. In the attempt to standardize the procedure, the field has further evolved with the establishment of the Collaborative Islet Transplant Registry (CITR) (9), a database collecting information from islet transplant centers that are participating. As the field improves, it is evident that still some limitations exist and that the procedure is missing optimal consistency. Quantification of the transplanted islets has been supported by the establishment of the count as IEQ and number of islets with an average diameter of 150 μm. IEQ was again an attempt to standardize the islet count and identify the islet mass required to reverse diabetes regardless of islet size. Based on this, it was established that an ideal transplant should aim to infuse approximately 10,000 IEQ/kg of body weight of the recipient. As reported by the CITR in 2008 and 2009, in islet-alone cases, approximately 6,500 IEQ (SD = 2,500) have been infused per single infusion (up to three infusions/patient) (9). From the same reports, it appears that higher IEQs (as cumulative IEQs infused) predict greater likelihood of achieving insulin independence (p < 0.001). Although this is an attempt to define a successful islet product, it does not take in consideration islet characteristics and viability. Most likely, these factors play a role in the overall outcome of clinical pancreatic ITX and may be responsible for the large number of islets that are required to reverse diabetes. The number of islets required in an islet transplant is, in fact, higher than the number of islets physiologically present in a pancreas. In addition, it is known that clinical signs of diabetes can be observed when approximately 90% of the β-cells are destroyed (57), implying that normal glucose control can be maintained with less than half of the islet mass usually present in a pancreas. Furthermore, partial removal of the pancreas is often associated with preserved glucose homeostasis, confirming again that some islet mass can be removed without having a major effect on glucose metabolism. Insulin resistance, resulting from longstanding diabetes, immunosuppression, and autoimmunity are all factors that are believed to affect graft functionality in islet transplantation. Most likely, these factors play a role in failure of engraftment and/or requirement of a larger mass to effectively control blood glucose. Although studies support these theories, pancreas transplant does not seem to be affected by the same limiting factors, and one organ is enough in fully reestablishing glucose homeostasis in diabetic patients (49,54). When islet autotransplantation is performed in patients undergoing a pancreatectomy, the number of IEQ/kg seems to be lower when compared to allotransplantation. Still, the success rate of autologous procedures is similar to the allotransplant, standing at approximately 55% as reported by one of the most active groups in this field (5). Even in this case, success appears to be correlated to the number of IEQ/kg transplanted with significant improved outcome when IEQ/kg is over 5,000. Based on the evidences just reported, other factors besides autoimmunity, rejection, and immunosuppression play a role and affect islet engraftment.
As a standard procedure, it was established that pancreatic islets should be maintained intact to preserve functionality because of the complex organization and cellular interaction that takes place in these multicellular structures (1,17,40). This concept was integrated on islet preparation up to the point that ideal isolations were including the so-called “mantled” islets: islets surrounded by a thin rim of exocrine tissue (9,44,45), as a sign of insular integrity and protection of the islets during the initial phase posttransplant (4).
While it is absolutely instrumental to assess islet composition and purity to characterize the product that is transplanted in diabetic patients, there are still limits to the possibility of having results before the transplant is performed (41). Even more helpful would be the possibility to perform isolations targeting a certain product known to provide a better outcome when transplanted.
Lehmann et al. (24) have shown that small islets are superior to large islets when compared for in vitro insulin secretion. In the same study, they observed a higher survival rate of smaller islets during normoxic or hypoxic culture compared to large islets. Islet cell death was strongly dependent on islet size and oxygen tension, doubling on islets over 100 μm in diameter compared to less than 50 μm. Of clinical relevance is what they retrospectively observed in seven clinical islet transplant cases. When they looked at prediction of functionality by C-peptide levels, size (using the isolation index: IEQ/islet number) seemed to be the best predictor for islet functionality. Although the number of patients used for the multiple regression analysis was small, they have shown that a higher islet volume with large islets was required to achieve the same insulin secretion provided by smaller islets. Superiority of small islets was previously shown in animal studies by static and dynamic perifusion of small (<125 μm) and large (>150 μm) islets in vitro (35) and by reversing diabetes in diabetic rats (26) or mice. In our study, we have moved forward through this investigation with the intent to determine the relationship between size and engraftment in a marginal mass model, to reveal any potential correlation as the size changes and to investigate potential interventions to reduce islet size while improving functionality.
Several mechanisms may be responsible for the improved functionality that we can observe in smaller islets. Smaller islets have a reduced diffusion barrier with the capacity to respond promptly and efficiently to glucose concentration. In a study, Papas et al. (39) demonstrated that small islets have superior glucose-dependent insulin secretion and higher oxygen consumption. Nam and colleagues (35) explain why small islets have better glucose-stimulated insulin secretion than large islets discussing several mechanisms. From physiology studies, we know that isolated islets need vascular support and innervations after isolation because vascular-dependent and neural-regulated signals are necessary for synchronization between β-cells in order to secrete insulin (17). Passive glucose diffusion and β-cell interaction through the gap junction are the mechanisms that allow the glucose-controlled insulin secretion in the isolated islets; therefore, a smaller diffusion barrier allows for a more efficient response to glucose change concentrations. Using a new computational model based on cellular-level glucose–insulin dynamics, the intraislet spatial distributions of insulin, glucose, and oxygen levels were described supporting the theory that smaller islets perform better when transplanted and/or encapsulated (7). Hence, smaller islets are able to function better than larger ones. In addition, it is known that diffusion may be the main mechanism by which islets get oxygen during the early period after transplantation. Small islets have less diffusion barriers to oxygen and nutrients and have a higher chance than large islets for survival in a hypoxic environment (26). All of this information leans toward a superiority of small islets in terms of opportunity to survive initially and preserve functionality posttransplant leading to a more successful ITX.
Our group and others (11,33) have previously shown that pancreatic islets undergo structural and ultrastructural changes, with complete loss of the microvascular network. This condition makes the early period following transplantation critical for cell survival, until the islets, within 10–14 days (30,56), become vascularized by a newly formed microvascular network arising from host vessels. Peripheral cells seem to be able to survive because of their proximity to vessels, while necrosis takes place in the core of the cell cluster of larger islets probably due to hypoxia (10,11). While vascularization takes place, larger islets seem to lose cells and possibly die off (34). In a very elegant study by Henriksnas and colleagues (14), a new method to detect islet graft blood perfusion is described. By using this method, the authors could detect the vascular density in the revascularization of islets of different sizes, shapes, and locations. Of major relevance is their discovery that blood perfusion of transplanted islets was found to be only 5% of that in native islets with great heterogeneity in blood flow between individual islets. In addition, they described the presence of a few dysfunctional blood vessels. Even more interesting is the findings that islets with disrupted integrity had a better vascularization compared to apparently intact islets where they could not even detect blood perfusion. This brought the authors to conclude that, although transplantation of islets can restore normoglycemia in rodents, this is mainly done through distorted islets. Their data were not supported by in vivo functionality (animals were not rendered diabetic, and they did not compare reversal of diabetes by intact vs. fragmented islets). In addition, as a potential experimental bias, this method (that we have adopted in our study as well) might have affected islet functionality beyond the reduction of islet size. Our study focused on functionality, and the kidney capsule was selected as a site of implantation to assure that glucose metabolism was attained and maintained by the graft. Only animals where diabetes reappeared following removal of the graft-bearing kidney were included in the analysis. Based on our study, we can conclude that smaller islets have better engraftment capabilities, and this could be due to a faster and complete vascularization. When we fragmented larger islets into smaller islets by either mechanical or enzymatic digestion, as described by Henriknas and his group (14), and we evaluated their engraftment, this was significantly decreased. Fragmented islets can be well vascularized, but we have shown that the loss of integrity does not allow them to fulfill their physiological duty. On the contrary, we can speculate that a slight overdigestion of the pancreas allows the downsizing of islets with improved vascularization while functionality is preserved.
In summary, a linear correlation between size and engraftment was demonstrated by investigating three different islet sizes and observing improved engraftment as the size decreases. Fragmentation of larger islets to the optimal islet size we have identified is not a successful strategy. Overdigestion of the pancreas can be safely performed with improved posttransplantation outcome. In the attempt to reconstitute pancreatic islets through disaggregation–expansion–reaggregation of β-cells, the size of 50–150 μm should be the targeted dimension. Strategies to improve vascularization should also be considered to preserve larger islets.
Footnotes
Acknowledgments
This work was supported in part by NIH T32 grant (DK007639–18) and the Gillson-Longenbaugh Foundation. The authors would like to thank Boyd Carr, MS, for his assistance with the laboratory work, and Steve Schuenke and Eileen Figueroa for their editorial support. The authors declare no conflicts of interest.
