Abstract
Pancreatic islets implanted heterotopically (i.e., into the kidney, spleen, or liver) become poorly revascularized following transplantation. We hypothesized that islets implanted into the pancreas would become better revascularized. Islets isolated from transgenic mice expressing enhanced yellow fluorescent protein (EYFP) in all somatic cells were cultured before they were implanted into the pancreas or beneath the renal capsule of athymic mice. Vascular density was evaluated in histological sections 1 month posttransplantation. EYFP was used as reporter for the transgene to identify the transplanted islets. Islet endothelial cells were visualized by staining with the lectin Bandeiraea simplicifolia (BS-1). Capillary numbers in intrapancreatically implanted islets were only slightly lower than those counted in endogenous islets, whereas islets implanted beneath the renal capsule had a markedly lower vascular density. In order to determine if this high graft vascular density at the intrapancreatic site reflected expansion of remnant donor endothelial cells or increased ingrowth of blood vessels from the host, also islets from Tie2-green fluorescent protein (GFP) mice (i.e., islets with fluorescent endothelial cells) were transplanted into the pancreas or beneath the renal capsule of athymic mice. These islet grafts revealed that the new vascular structures formed in the islet grafts contained very few GFP-positive cells, and thus mainly were of recipient origin. The reason(s) for the much better ingrowth of blood vessels at the intrapancreatic site merits further studies, because this may help us form strategies to overcome the barrier for ingrowth of host vessels also into islets in heterotopic implantation sites.
Introduction
Pancreatic islets postnatally develop an abundant glomerular-like microcirculation system, which ensures that no portion of an islet is more than one cell away from arterial blood (4). This arrangement optimizes the condition for supply of oxygen and nutrients to the islet cells, their metabolic sensing, and the distribution of secreted hormones to target organs in order to maintain glucose homeostasis. When pancreatic islets are isolated for transplantation, the vascular connections become interrupted. Transplanted islets are therefore initially dependent on diffusion of oxygen and nutrients from the surrounding tissue for their survival and function. A new islet vascular system is formed within 7–14 days (13,23,26), but irrespectively whether the islets are implanted into the kidney, spleen, or liver, the acquired vascular network is significantly less dense than the original one, and moreover transports insufficient amounts of oxygen to the tissue (6,7,20). For murine islets implanted to the kidney, spleen, or liver, the revascularization is similarly low and the islet vascular network constitutes approximately one third of that in native islets (20). Also for human islets experimentally transplanted to nude mice the intraislet vascular density is similarly low in islet grafts implanted intraportally to the liver or at the renal subcapsular site (16a), and decreased compared to native islets (7). Attempts to improve islet revascularization are clearly warranted, as this may significantly decrease the number of transplanted β-cells needed to obtain insulin independence. However, the molecular events within the islet tissue permissive for a better revascularization are largely unknown, although recruitment of angioblasts, as well as vascular endothelial growth factor (VEGF) overexpression in the donor tissue, have been shown to improve islet graft revascularization and function (9,15,39).
Clinically, islet transplantation into the liver through the portal vein is almost exclusively applied. Orthotopic implantation of islets (i.e., implantation into the pancreas) has been described in experimental settings (12,31,32), but heterotopic implantation sites, predominantly kidney, spleen, or liver, are much more commonly chosen. We hypothesized that islets implanted into appropriate surroundings (i.e., into the pancreas) would become better revascularized than islets implanted heterotopically. To test this, we used islets isolated from transgenic YC-3.0 mice, characterized by expression of enhanced yellow fluorescent protein (EYFP) in all pancreatic islet cells. EYFP was used as a reporter for the transgene to identify the transplanted islets. In order to investigate the origin of endothelial cells in the acquired new vasculature in transplanted islets, we used islets isolated from Tie2-green fluorescent protein (GFP) transgenic mice with specific expression of GFP in the endothelial cells as donor islets.
Materials and Methods
Animals
YC-3.0 transgenic mice were kindly donated by Professor R. Y. Tsien at the University of California and kept in a local colony at Karolinska Institutet (Stockholm, Sweden). The genetic background of the YC-3.0 transgenic mouse is 129J × C57B1/6, backcrossed once with 129J before propagated by brother–sister mating and identification of homozygous animals. The generation and characterization of the YC-3.0 mice has been described previously (27,33). Briefly, transgenic YC-3.0 mice are characterized by the expression of the yellow chameleon protein-3.0 (YC-3.0) under the regulation of the β-actin and cytomegalovirus promoters (24), resulting in the expression of EYFP, one part of the hybrid YC-3.0 protein, in most tissues including all pancreatic islet cells. YC-3.0 mice have been characterized in detail with regard to glucose homeostasis, islet function, islet mass, islet vascularity, and islet blood perfusion, and no differences were found compared to control C57B1/6 mice (27). Tie2-GFP transgenic mice (i.e., animals that express GFP behind the Tie2 promoter) were purchased from The Jackson Laboratories [STOCK Tg(Tie2GFP) 287Sato/J] and kept in a local colony at Karolinska Institutet. Male recipient C57B1/6 (nu/nu) mice weighing approximately 30 g were purchased from M&B Research and Breeding Center (Ry, Denmark). The animals had free access to tap water and pelleted food throughout the course of the study. All experiments were approved by the animal ethics committee of Uppsala University.
Islet Isolation and Transplantation
Pancreatic islets from YC-3.0 and Tie2-GFP mice were prepared by collagenase digestion and cultured free-floating for 4–6 days in RPMI-1640 medium supplemented with L-glutamine, benzylpenicillin (100 U/ml; Roche Diagnostics Scandinavia, Bromma, Sweden), streptomycin (0.1 mg/ml), and 10% (v/v) fetal calf serum. The culture medium was changed every second day. After culture, groups of 200 islets were packed in a braking pipette and implanted beneath the capsule of the left kidney, or packed and injected through a butterfly needle (25 gauge) into the pancreas in C57B1/6 (nu/nu) mice anesthetized with avertin [a 2.5% (v/v) solution of 10 g 97% (v/v) 2,2,2-tribromoethanol (Sigma, St Louis, MO, USA) in 10 ml 2-methyl-2-butanol (Kemila, Stockholm, Sweden)].
Histological Staining Procedures
The graft-bearing organs, or pancreata from age-matched control YC-3.0 and C57B1/6 (nu/nu) mice, were removed 2 days, 1 month, or 6 months posttransplantation, fixed in 10% (v/v) formaldehyde, and embedded in paraffin. Consecutive sections (4–5 μm thick) of the islet grafts or control pancreata were mounted on glass slides, deparaffinized in xylene, and dehydrated in graded alcohols. For antigen retrieval, slides were immersed in Target Retrieval solution® pH 6.0 (DAKO, Carpinteria, CA, USA), and boiled for 25 min in a microwave oven. Incubation of slides with Peroxidase blocking reagent® (DAKO) for 10 min to block endogenous peroxidase was followed by washing in Wash buffer® (DAKO). The slides were then incubated with DAKO protein block for 30 min. A green fluorescent protein (GFP) antibody known to detect not only GFP but also EYFP (Invitrogen, Carlsbad, CA, USA) was diluted in antibody diluent (1:1200 for YC-3.0 islets and 1:200 for Tie2-GFP islets, respectively) with background reducing components® (DAKO). The slides were incubated with this for 30–60 min at room temperature (RT) followed by washing in Wash buffer®. The ChemMate EnVision detection kit® (DAKO) was used on the sections according to the manufacturer's instructions. The sections were developed with diaminobenzidine before counterstained with hematoxylin. Finally, the sections were dehydrated and mounted in Pertex mounting medium (HistoLab, Göteborg, Sweden). Negative control slides were processed identically except that the primary antibody was omitted. The immunohistochemical staining was performed using an automated immunostaining instrument, Autostainer plus® (DAKO).
Adjacent sections of the grafts were stained with hematoxylin and eosine to visualize any infiltrating inflammatory cells. Moreover, adjacent sections of the 1-month-old grafts were stained with the lectin Bandeiraea simplicifolia (BS-1) to visualize the islet blood vessels, as described in detail previously (20), before counterstaining with hematoxylin.
Evaluation of Vascular Density
In each animal, 10 or more tissue sections stained with BS-1 from the islet transplants or all parts of the control pancreas were randomly chosen and evaluated. The numbers of stained blood vessels in endogenous and transplanted islets were quantified under a light microscope. Connective tissue surrounded the individual islets, especially in the renal subcapsular grafts. The numbers of blood vessels in the endocrine and connective tissues were here counted separately. The respective fractions of islets and connective tissue in the islet grafts were determined by a direct point counting technique (37). For this purpose, the intersections overlapping the stroma and endocrine cells within the islet grafts were counted. The areas of the investigated endogenous islets and grafted islets were determined using a computerized system for morphometry (Easy Image 3000, Tekno Optik, Huddinge, Sweden). Vascular density—the number of blood vessels found per measured islet or graft area (mm2)—was then calculated.
Statistical Analysis
Values are expressed as the means ± SEM. When only two groups were compared, Student's unpaired two-tailed t-test was applied. Multiple comparisons between data were performed using analysis of variance (ANOVA) and Bonferroni's post hoc test. For all comparisons, values of p < 0.05 were considered statistically significant after correction.
Results
The YC-3.0 islets retained their EYFP expression and remained at least 6 months after intrapancreatic transplantation (two out of two animals). Following immunohistochemical staining they could easily be distinguished from endogenous islets of the recipient pancreas (Fig. 1). Islets in both the intrapancreatic and renal subcapsular islet grafts showed a gross normal morphology with no infiltration of inflammatory cells within the endocrine tissue. However, intrapancreatic islet grafts 2 days after transplantation showed inflammatory cells in the vicinity of islets in three out of three animals (Fig. 1B). In both implantation organs the transplanted islets were grouped in clusters, but vivid connective tissue interspersing the individual islets in the grafts was only found in the renal subcapsular grafts [15.7 ± 2.5% (n = 6) vs. 2.7 ± 0.7% (n = 6) for renal subcapsular and intrapancreatic islet grafts, respectively; p < 0.05].

Micrographs of intrapancreatically implanted YC-3.0 islets staining positive for enhanced yellow fluorescent protein (EYFP) (brown) 2 days, 1 month, or 6 months posttransplantation (A, C, and D, respectively). Neighboring endogenous C57B1/6 (nu/nu) islets are not stained (C, upper right corner). Inflammatory cells were detected in the vicinity (arrow), but not within islets implanted into the pancreas 2 days after transplantation (B). Scale bar: 100 μm (A, B), 300 μm (C), and 200 μm (D).
The capillary numbers in the endocrine tissue of the intrapancreatic islet grafts were slightly lower than those counted in endogenous islets (Fig. 2). Islets implanted into the renal subcapsular site had a markedly lower vascular density than both endogenous islets and intrapancreatic islet grafts. Connective tissue surrounding the islets implanted at the renal subcapsular site was, on the other hand, richly vascularized. Intrapancreatic islet grafts had a sparse connective tissue stroma, which made the evaluation of the connective tissue vascular density impossible. The implantation of islets into the pancreas did not affect the vascular density of the endogenous islets, because islets in control pancreata had a similar vascular density (Fig. 2D). Investigation 1 month posttransplantation of Tie2-GFP islets implanted into the pancreas or into the renal subcapsular site revealed that the new islet vascular structures formed in the islet grafts were almost exclusively negative for GFP [<5% of endothelial cells in all transplants at both the intrapancreatic site (n = 5 animals) and the renal subcapsular site (n = 3 animals)], and thus mainly of recipient origin (Fig. 3).

Micrographs of endogenous YC-3.0 pancreatic islet (A) and YC-3.0 islet grafts 1 month after implantation into the pancreas (B) or beneath the left renal capsule (C) of athymic C57B1/6 mice. Islet endothelial cells were visualized by staining with the lectin Bandeiraea simplicifolia. Scale bar: 50 μm (A, C), 100 μm (B). (D) Vascular density in endogenous islets of nontransplanted transgenic YC-3.0 mice (control islets), in endogenous islets of recipient athymic C57B1/6 (endogenous islets), and in the endocrine (closed bar) and connective tissue (hatched bar) of islet grafts implanted intrapancreatically or beneath the renal capsule. The intrapancreatic islet grafts had very sparse connective stroma, which made the evaluation of its vascular density impossible. All values are given as means ± SEM for 6–8 experiments. *p < 0.05 when compared to control islets; †p < 0.05 when compared to the intrapancreatically implanted islets. All comparisons were made using ANOVA.
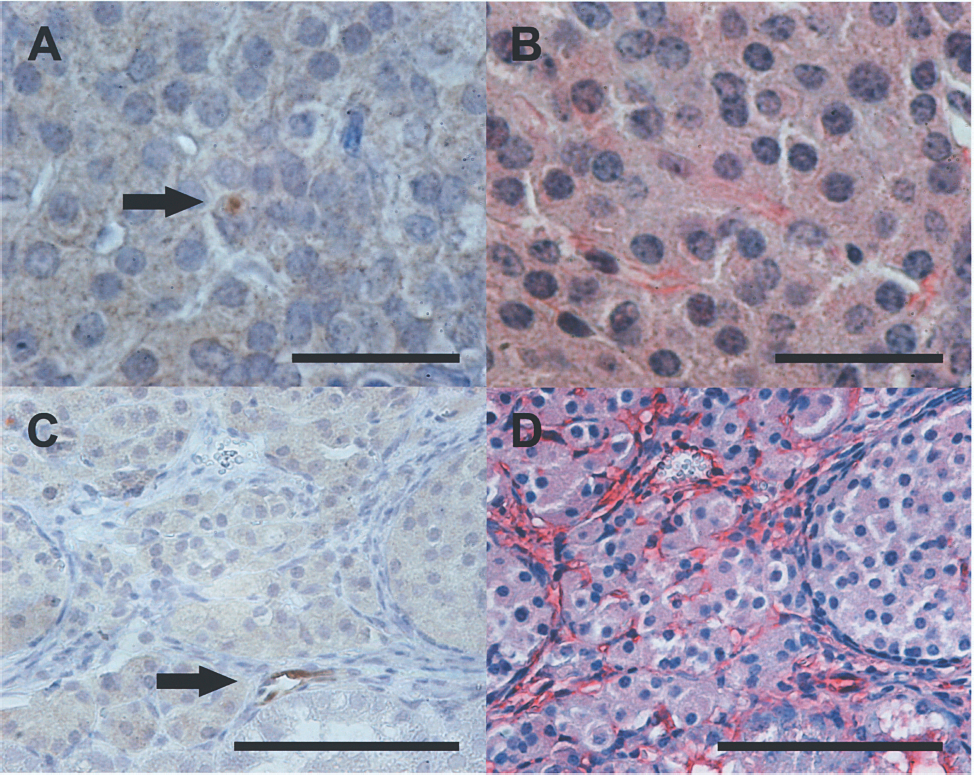
Micrographs of Tie2-GFP donor islets implanted into the pancreas (A–B) or beneath the renal capsule (C–D). Very few GFP-positive endothelial cells (dark brown indicated with arrows) were detected in the intrapancreatic (A) and renal subcapsular islet grafts (C) 1 month posttransplantation. Adjacent tissue sections of the islet grafts were stained for islet endothelium with Bandeiraea simplicifolia (red) to visualize the acquired new vasculature in transplanted islets (B, D). Scale bar: 30 μm (A, B), 100 μm (C, D).
Discussion
Currently, islets from at least two donor pancreata are needed to reverse diabetes in clinical islet transplantation. In contrast, only 10–20% of the native islet mass seems to be needed to maintain normoglycemia in a healthy individual. Continued focus to advance immunological therapies is essential. However, more knowledge on the influence of the implantation organ, as well as means to improve engraftment and revascularization could dramatically improve islet survival and function after transplantation (25). Currently, extensive islet cell death occurs in the immediate posttransplantation period (2,3,10,11), as well as a functional impairment in the surviving endocrine cells (16,21). Our previous studies have shown that heterotopically implanted islets become poorly revascularized. Similar results were obtained irrespective of whether the islets were implanted beneath the renal capsule, intraportally to the liver as in the clinical situation, or into the spleen (20). In the present study we tested the influence of a foreign microenvironment on islet revascularization by comparison to intrapancreatic islet transplantation and found that there is a clearly better revascularization of islets grafted into the pancreas than into a heterotopic site such as the renal subcapsular site. Indeed, the islet vascular density was almost restored to that of the endogenous islets.
Recent studies, primarily on transplanted freshly isolated islets, have highlighted that not only host blood vessels, but also remnant donor islet endothelial cells, participate in the formation of a new islet vascular network (5,17,26). In this study, we chose to culture islets before transplantation, which has been shown to disrupt and dedifferentiate, or degenerate, the endogenous islet vasculature (26,28). Thereby, the graft vascular network will mainly arise from blood vessels of recipient origin (7,34). This was confirmed by the near absence of GFP expression in the formed islet graft vasculature. The higher vascular density found in the orthotopically implanted islets, therefore, seems to relate to a better revascularization from the host blood vessels, rather than expansion of the remnant donor islet endothelial cell pool.
In the immediate vicinity of islets implanted heterotopically to the kidney, spleen, or liver, richly vascularized connective tissue is formed. However, recipient blood vessels attracted to the islets rarely enter the endocrine tissue in this setting (19,21). This suggests location of matrix or tissue-bound angiostatic factors within the implanted islets. The difference in capacity to vascular ingrowth of pancreatic endothelial cells and of endothelial cells of the other organs may reflect differences in angiogenetic response (18,29,30). Interestingly, however, the pattern bears a clear resemblance to the findings in heterotopically implanted embryonic lung, where host vessels were unable to enter the graft (35). Accumulating data indicate the importance of a great number of guidance proteins, such as ephrins, semaphorins, slit, and the respective receptors, not only for directing the ingrowth of nerve cells but also for regulating selective ingrowth of endothelial cells into different tissues forming the tissue-specific vascular network during organogenesis (1,8,36). Several of these guidance proteins continue to be expressed in the adult organism including their pancreatic islets (19) and have also been shown important for islet function (14). Their receptors have been shown to be expressed on islet endothelial cells (38). It is possible that the observed findings of selective poor revascularization of heterotopic implants reflect the inhibitory influences of such guidance proteins to foreign-organ endothelial cells.
Although being the most physiological environment for islets, the pancreas has rarely been considered as a potential implantation organ. Surgical interventions and injections in the pancreas are difficult and there is a high risk of complications due to leakage of enzymes from the exocrine cells that causes tissue damage and inflammation. The pancreas will therefore probably never become a choice of implantation organ in clinical islet transplantation. However, in our experiments, none of the animals seemed to develop acute pancreatitis posttransplantation. Inflammatory cells in limited numbers could in some cases be observed in the vicinity of the implanted islets, but no inflammation in the endocrine tissue could be discerned 2 days, 1 month, or 6 months posttransplantation. It is possible that the early inflammation around the intrapancreatically transplanted islets stimulated the angiogenetic response to the islets, but in the absence of vivid intraislet inflammation it seems unlikely to contribute to islet core revascularization. In previous studies, experimental intrapancreatic islet transplantation in rodents and canines have proven possible to provide cure to diabetic animals and even to be superior to renal subcapsular islet transplantation (12,31,32). We decided not to try to repeat these findings, because due to the blind injection of the islets into the pancreas we sometimes (~10%) partially or totally lost the graft, most likely due to leakage of islets at injection. In order to avoid variable blood glucose levels in the recipients we instead used normoglycemic recipients. Remaining native pancreatic islets do not seem to interfere with the formation of new blood vessels in islet graft; the vascular density in the grafts is similar to that obtained in islets implanted to cure diabetic recipients (22). As an alternative strategy to test intrapancreatic islet graft function, we have developed a technique to reisolate transplanted islets from the pancreas and liver and found that intrapancreatically transplanted islets, in contrast to intrahepatically transplanted islets, retain a lot of function, which also correlates to fewer changes in gene expression for key enzymes in β-cell metabolism (16).
The reasons for the much better development of an islet vascular network at the intrapancreatic site clearly merit further studies. In order to restore the islet vascular system also at heterotopic implantation sites, either stimulated expansion of the remnant endothelial cell pool or strategies to overcome the barrier for ingrowth of host vessels into the endocrine tissue are clearly warranted.
Footnotes
Acknowledgments
The skilled technical assistance of Astrid Nordin and Eva Törnelius is gratefully acknowledged. This work was supported by the Swedish Research Council (55X-15043), the Juvenile Diabetes Research Foundation, EFSD/JDRF/Novo, the Swedish Diabetes Association, the Family Erling-Persson Foundation, the Swedish Juvenile Diabetes Fund, the Novo Nordic Fund, the Anér Foundation, Lundströms Memorial Fund, the Åke Wiberg Foundation, the Magnus Bergvall Foundation, and the Family Ernfors Fund.
