Abstract
Clinical islet transplantation is characterized by a progressive deterioration of islet graft function, which renders many patients once again dependent on exogenous insulin administration within a couple of years. In this study, we aimed to investigate possible engraftment factors limiting the survival and viability of experimentally transplanted human islets beyond the first day after their transplantation to the liver. Human islets were transplanted into the liver of nude mice and characterized 1 or 30 days after transplantation by immunohistochemistry. The factors assessed were endocrine mass, cellular death, hypoxia, vascular density and amyloid formation in the transplanted islets. One day posttransplantation, necrotic cells, as well as apoptotic cells, were commonly observed. In contrast to necrotic death, apoptosis rates remained high 1 month posttransplantation, and the total islet mass was reduced by more than 50% between 1 and 30 days posttransplantation. Islet mass at 30 days posttransplantation correlated negatively to apoptotic death. Vascular density within the transplanted islets remained less than 30% of that in native human islets up to 30 days posttransplantation and was associated with prevailing hypoxia. Amyloid formation was rarely observed in the 1-day-old transplants, but was commonly observed in the 30-day-old islet transplants. We conclude that substantial islet cell death occurs beyond the immediate posttransplantation phase, particularly through apoptotic events. Concomitant low vascularization with prevailing hypoxia and progressive amyloid development was observed in the human islet grafts. Strategies to improve engraftment at the intraportal site or change of implantation site in the clinical setting are needed.
Introduction
Clinical islet transplantation is a possible treatment for patients with type 1 diabetes mellitus, who experience frequent episodes of hypoglycemia due to severe glycemic lability. Islet transplantation can result in independence of exogenous insulin administration in up to 50% of cases 3–5 years posttransplantation, which is comparable with pancreas transplantation (3,4). However, despite the fact that in recent years immunotherapy strategies have improved, there is a progressive deterioration in islet graft function. Occurrence of an immediate blood-mediated inflammatory reaction when exposing islets to portal blood has been shown to damage the endocrine cells (20) and cause a loss of islet mass up to 50% within the first minutes posttransplantation (11). Whether the clinically observed progressive loss of islet function merely represents β-cell exhaustion in patients with a marginal islet mass, or whether other factors are involved, is not completely understood. Islet amyloid formation (31), gluco- and lipotoxicity (17), immunological events (1), and toxicity of the immunosuppressive drugs used after transplantation (26) have all been suggested as potential contributing factors to graft failure. Moreover, we have observed prevailing hypoxia (23) due to insufficient revascularization and blood perfusion (15) in syngeneic mouse islets implanted into the liver.
Also at extrahepatic implantation sites, such as beneath the renal capsule (9) or intramuscularly (13), substantial β-cell death occurs during the early posttransplantation period. Nevertheless, mouse islet grafts syngeneically transplanted into the renal subcapsular site of normoglycemic mice show no decrease in β-cell mass when investigated 1 month posttransplantation, indicating this to be a transient phenomenon if the blood glucose is properly controlled (5). At the clinically used intraportal site, there have been no studies investigating more than immediate changes in islet mass. Moreover, the development of intraislet blood vessels and amyloid during the first month, as well as the degree of hypoxia in intraportally transplanted human islets, has thus far not been characterized. In this study, we investigated the engraftment of human islets experimentally transplanted into the liver and observed that poor engraftment with associated apoptotic death contributed to an extensive loss of islet mass beyond the immediate posttransplantation phase.
Materials and Methods
Human Pancreatic Islets
The regional ethical board in Uppsala approved the use of human pancreatic tissue for this study. Pancreata from adult heart-beating brain-dead donors were provided by the Nordic Network for Clinical Islet Transplantation and used for isolation of human islets at the human islet isolation facility, Rudbeck Laboratory, Uppsala University Hospital. The average age of the organ donors was 55 ± 4 years (range 33–69), and they were all nondiabetic with an HbA1c of 38 ± 1 mmol/mol (5.6 ± 0.1% DCCT). Available information on islet donors, pancreata, and islet characteristics are presented in Table 1.
Islet Donor Information and Culture Period
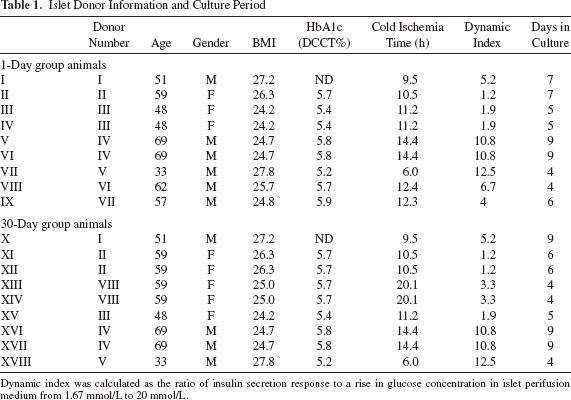
Dynamic index was calculated as the ratio of insulin secretion response to a rise in glucose concentration in islet perifusion medium from 1.67 mmol/L to 20 mmol/L.
Islet Culture
The islets were cultured at the human islet isolation center for the Nordic Countries, Rudbeck Laboratory, Uppsala, as per clinical standard in CMRL1066 medium (Gibco, Life Technologies, Carlsbad, CA, USA) supplemented with 8.7% human serum (Department of Transfusion Medicine, Uppsala University Hospital, Uppsala, Sweden), 139.5 mmol/L nicotinamide (APL Pharma Specials, Umeå, Sweden), 69.7 mmol/L sodium pyruvate (APL Pharma Specials), 10.5 mmol/L HEPES (Life Technologies), 2.1 mmol/L L-glutamine (Life Technologies), 17.4 μg/mL ciprofloxacin (Fresenius Kabi, Bad Homburg, Germany), and 43.6 μg/ml gentamycin (Life Technologies) at 37°C for the first 24 h and then at 25°C in 95% air and 5% CO2. After transfer to our experimental laboratory they were incubated in the same culture medium, but at 37°C, up to 24 h prior to their transplantation.
Animals
Adult male immune-deficient C57BL/6 (nu/nu) mice (n = 18) (M&B, Bomholtgaard, Denmark) were used as human islet recipients. The animals had free access to water and pelleted food throughout the course of the study. The animal housing and all experiments were approved by the local animal ethics committee for Uppsala University.
Transplantation
After 3–7 days of culture, 0.9 μl of human islet tissue (~300 human islets) was packed in a butterfly needle (25 G) in a volume of 0.1–0.2 ml of serum-free medium.
The mice were anesthetized with an intraperitoneal administration of Avertin (0.02 ml/g body weight) (Sigma-Aldrich, Saint Louis, MO, USA and BDH Merck, Poole, UK). A midline abdominal incision was made, and the islets were infused into the portal vein through the butterfly needle. Islets were transplanted selectively into the right liver lobe by temporarily clamping other portal vein tributaries during the islet infusion (15). This procedure enabled us to quantify surviving islet mass without consecutive sectioning of whole livers.
For experimental transplantation in mice, a 25-G needle was used rather than a 19-G needle that is commonly used clinically. In order to separately evaluate the influence of infusing the islets through a 25-G needle, five groups of 300 islets (from three different donors) were passed through a 25-G butterfly needle in vitro. Before and after this experiment, the islets were incubated at 37°C for 2 h in Krebs–Ringer bicarbonate HEPES (KRBH) medium [114.25 mmol/L NaCl (Merck, Kenilworth, NJ, USA), 4.74 mmol/L KCl (Merck), 1.18 mmol/L MgSO4 (Merck), 1.15 mmol/L KH2PO4 (Merck), 25.00 mmol/L NaHCO3 (Merck), 10 mmol/L HEPES (Sigma-Aldrich), 4.26 mmol/L NaOH (Merck), and 2.54 mmol/L CaCl2 (Merck)] at a glucose concentration of 5.6 mmol/L. An insulin ELISA (Mercodia, Uppsala, Sweden) measured the human insulin concentrations in medium. Passing the 25-G needle increased insulin concentrations in the medium from 47 to 2,757 pmol/L (range) in individual preparations to 294 to 8,151 pmol/L (range). This experiment indicates that the shear stress of the narrow needle indeed caused insulin leakage from some islets, but that the individual variations between preparations were as important for variations in medium insulin concentrations.
Retrieval of Organs and Immunohistochemistry
Human islet grafts transplanted into the liver were collected for analysis at either 1 or 30 days posttransplantation. Two hours before the animals were killed by cervical dislocation, the hypoxia marker pimonidazole (60 mg/kg; Hypoxyprobe, Burlington, MA, USA) was injected intravenously through the tail vein of the animals. The graft-bearing liver lobes were retrieved and fixed in 4% (v/v) paraformaldehyde (VWR International, Radnor, PA, USA), before they were embedded in paraffin. The liver lobes were sectioned to a thickness of 7 μm, and every fifth slide was stained for insulin. Adjacent slides were stained, respectively, for apoptosis (cleaved caspase 3), blood vessels (with lectin BS-1), hypoxia (pimonidazole) and amyloid (with Congo red). The staining protocols, outlined below, were performed according to the manufacturers' instructions, and all sections were counterstained in Mayer's hematoxylin (Histolab, Göteborg, Sweden).
Staining for insulin was performed as previously described (18). Briefly, hydrogen peroxide and normal swine serum (Vector Laboratories, Burlingame, CA, USA) were used as blocking solutions. A polyclonal guinea pig anti-insulin antibody (Fitzgerald Industries International, Concord, MA, USA) and a secondary swine anti-rabbit antibody (Dako, Glostrup, Denmark) were used as primary and secondary antibodies, respectively. Both antibodies were diluted in phosphate-buffered saline (PBS) to a concentration of 1:400 and 1:40, respectively. The slides were developed with a solution of 3,3′-diaminobenzidine (DAB) (Dako) for a maximum of 10 min.
Staining for cleaved (activated) caspase 3 was performed as previously described (23). The primary antibody used was a rabbit anti-caspase 3 antibody (Cell Signaling Technology, Danvers, MA, USA) diluted 1:100 in TNB (PerkinElmer, Waltham, MA, USA). The secondary antibody, a biotinylated goat anti-rabbit antibody (Southern Biotechnology, Birmingham, AL, USA), was diluted 1:300 in TNB. Streptavidin–horseradish peroxidase (SA-HRP) (PerkinElmer) diluted 1:100 in TNB and biotinyl tyramide (PerkinElmer) were used before dye development with DAB.
The microvascular endothelium was stained with the lectin BS-1 (Sigma-Aldrich), as previously described (18). Briefly, the sections were exposed to neuroaminidase type V from Clostridium perfringens (Sigma-Aldrich) for antigen retrieval. The slides were then blocked with goat serum (Dako) for 30 min. Biotinylated BS-1 (Sigma-Aldrich) was applied to the sections, which were incubated at 4°C overnight. The slides were incubated with TrekAvidin-AP label (Biocare Medical, Concord, CA, USA) for 20 min followed by developing with Vulcan Fast Red (Biocare Medical). Human pancreata (n = 5) were simultaneously stained and evaluated for comparison between native and transplanted islets.
Pimonidazole accumulates in cells, including islet endocrine cells, at a tissue oxygen tension < 10 mmHg (23,24) and is therefore commonly used as a marker for hypoxia. Liver tissue served as a positive control for the pimonidazole staining, since the liver is one of the least oxygenated organs in the body with a physiological oxygen tension of 10 mmHg, which becomes even lower closer to the central veins (2). Livers of mice that had not been injected with pimonidazole were used as negative controls. Mouse anti-pimonidazole (Bioscience Research Reagents, Temecula, CA, USA) diluted at 1:40 was used as the primary antibody. The secondary antibody was a biotinylated goat anti-mouse antibody (Southern Biotechnology) diluted at 1:250 and Vulcan Fast Red (Biocare Medical) was utilized for detection. Congo red (VWR International) was used for the staining of amyloid.
Data Analysis
Sections from all livers were systematically analyzed by staining for insulin, and the area was assessed with a morphometric computerized system (ImageJ 1.47v; National Institutes of Health, Bethesda, MD, USA). For every 105 μm of liver, a section was chosen and evaluated for total islet area.
Assessment of islet vascular density and evaluation for percentages of caspase-positive cells were made using a morphometric computerized system (ImageJ 1.47v). In order to determine the intraislet capillary density, only vessels surrounded by islet tissue were counted and compared to the same assessment of native human pancreata. The percentage of pimonidazole-positive islets in the grafts was evaluated under a light microscope. The amyloid content was evaluated by fluorescence microscopy and polarized microscopy; in the latter amyloid displays an apple-green birefringence (29).
The mean numbers of islets evaluated in the immunohistochemical analysis were as follows: for the total islet area, 79.0 ± 9.4 islets (n = 15 animals); for caspase 3, 21.2 ± 3.9 islets (n = 15 animals); for BS-1, 16.5 ± 2.2 islets (n = 10 animals); for pimonidazole, 32.3 ± 3.0 islets (n = 13 animals); for amyloid, 41.9 ± 4.6 islets (n = 15 animals). Five human pancreata were assessed for native vascular density, and the mean number of islets evaluated in each pancreas was 84.4 ± 6.0.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism® version 6.0 (GraphPad Software, La Jolla, CA, USA). All values are expressed as means ± SEM. When only two groups were compared, a Student's unpaired t-test was used. Pearson's product moment correlation was used for linear regression analysis. Multiple comparisons between data were performed using analysis of variance (ANOVA) followed by Bonferonni's post hoc test. A Kruskal–Wallis test was used for nonparametric data (the data was considered nonparametric based on Shapiro–Wilk normality test). For all comparisons, a value of p < 0.05 was considered statistically significant.
Results
Viability of Transplanted Islet Cells
Intraportally transplanted islets were identified in the liver based on immunohistochemical staining for insulin and further characterized by analysis of cellular death, vascular density, hypoxia, and the presence of amyloid (Fig. 1). In order to determine the viability of the transplanted islets, cellular apoptosis was evaluated. Prior to transplantation, 6.5% of the isolated islets were positive for cleaved caspase 3 (Fig. 2A). At 1 day posttransplantation, 21% were identified as positive for the same marker (Fig. 2A). Extensive necrosis (pyknotic nuclei, cellular debris) was observed in ~40% of transplanted islets and sometimes even in the surrounding liver parenchyma, at 1 day posttransplantation (Fig. 1A). The necrotic changes were rarely observed at 30 days posttransplantation, but instead the rate of apoptosis remained high (34%) (Fig. 2A). Noteworthy, the number of caspase 3-positive cells at 30 days was correlated with increased culture time prior to transplantation for the individual islet preparations (Fig. 2B). There was no correlation of posttransplant islet death with any of the donor characteristics, cold ischemia time of the pancreas, or insulin secretion capacity of the islets prior to their transplantation.

Micrographs of performed morphological stainings. Islet grafts were localized with an immunohistochemical staining visualizing the insulin-containing β cells (brown) in 1-day-old (A) and 30-day-old islet grafts (B). Necrotic islet areas and affected surrounding liver parenchyma (arrows) were commonly observed in the 1-day group (A). Apoptotic cells were detected by staining for active cleaved caspase 3 (brown, arrows; day 30; C). The lectin Bandeiraea simplicifolia-1, which binds to Gal-epitopes present selectively on endothelial cells, was used for the estimation of islet vascular density (day 30; D). Hypoxia was demonstrated by the usage of the biochemical marker pimonidazole, whose metabolites accumulate intracellularly in cells with an oxygen tension below 10 mmHg. Pimonidazole was visualized in pink through an immunohistochemical staining with the dye developed by Vulcan Fast Red (day 30; E). With Congo red staining, amyloid fluoresces in red (day 30; F) when using a red filter, while a green filter was used to exclude autofluorescence (G).

Apoptosis, islet area, vascular density, and hypoxia at 1 and 30 days posttransplantation. The frequency of caspase 3-positive cells was above 20% in the islet grafts after both 1 and 30 days posttransplantation, while the rate in isolated islets was in mean less than 7% (A). At 30 days posttransplantation, the number of caspase 3-positive cells correlated positively to culture time prior to transplantation for the individual islet preparations (B). The total islet area decreased by 52% between 1 and 30 days posttransplantation (C). A correlation was observed between a lower remaining islet area and a higher rate of apoptosis at 30 days posttransplantation (D). The vascular density increased between 1 day and 1 month after transplantation, but was still only 30% of that in native human islets in the 30-day-old islet grafts (E). The vascular density in the isolated islets prior to their transplantation was at the same level as in the 30-day-old grafts (E). The percentage low oxygenated islets (pimonidazole positive) did not differ between 1 and 30 days posttransplantation (F). All values are given as means ± SEM for six to eight animals. The evaluation of native and isolated pancreatic islets was performed on samples from five human individuals, respectively. *p < 0.05.
Total Islet Area
The total area of viable islet cells within each liver was estimated as a representation of islet mass. The mean area was reduced by 52% in the 30-day group compared to what was found 1 day after transplantation (Fig. 2C). A correlation was observed between a low islet mass and a high rate of apoptosis at 30 days posttransplantation (Fig. 2D).
The Vascular Density and Rate of Hypoxia
One day posttransplantation, the mean vascular density in the islets was 56 vessels/mm2, while the 30-day-old islet grafts contained twice as many blood vessels (115 vessels/mm2) (Fig. 2E). However, the blood vessel number at 1 month posttransplantation was still only approximately one third of that in native human islets (Fig. 2E). The vascular density in the isolated islets prior to their transplantation was at the same level as in the 30-day-old grafts (Fig. 2E). Both at 1 day and 30 days posttransplantation, approximately 45% of islets stained positive for the low oxygen marker pimonidazole (Fig. 2F).
Islet Amyloid
Islets containing amyloid were much more common 30 days after transplantation than 1 day after transplantation or newly isolated islets (Fig. 3A). Very few of the isolated islets contained amyloid (0.02%). At 1 day after transplantation, 1.7% of the islets contained amyloid, and 27% had amyloid deposits 1 month later. At this latter time point, amyloid-positive islets with substantial amounts of amyloid also contained a higher frequency of caspase 3 positive cells than amyloid-negative islets (Fig. 3B).
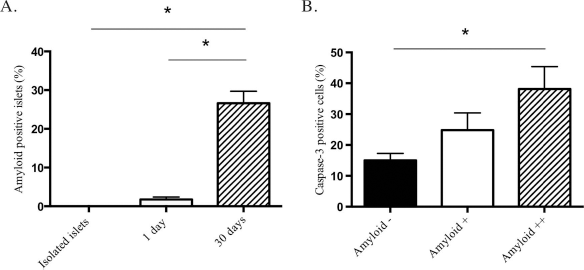
Amyloid in islets. Amyloid was rare in isolated islets prior to their transplantation, and in 1-day-old islet grafts, but was more commonly found in the 30-day-old islet grafts (A). At 30 days posttransplantation, the islets containing extensive amounts of amyloid had a higher frequency of caspase 3-positive cells. Individual islets were investigated for amyloid and caspase 3 in adjacent slides, and the islet amyloid was quantified either by none (-), moderate (+), or extensive (++) amyloid content (B). Islets with a larger amyloid content exhibited more caspase 3-positive cells. All values are given as means ± SEM for six to nine animals in each transplant group and for five human donors for the isolated islets. (A) *p < 0.0001 and (B) *p < 0.01.
Discussion
The main finding of the present study was that substantial islet cell death occurs beyond the immediate posttransplantation phase when islets are transplanted into the liver. In the present model of immune-deficient nude mice transplanted with islets, cell death could be attributed to engraftment factors rather than an adaptive immune response. This is supported by the fact that low vascularization with prevailing hypoxia and progressive amyloid development in the grafts were observed.
Cellular death may occur either by necrosis or by programmed cell death, known as apoptosis. Large necrotic areas in the liver could be observed 1 day after transplantation, which not only affected some of the transplanted islets but also in some cases the surrounding liver parenchyma. This finding likely reflects the embolization of portal blood vessels with islets at transplantation, which causes ischemia in some areas. Necrosis is regularly associated with acute toxic or hypoxic events and is therefore an expected event soon after transplantation, when islet cells depend solely on oxygen by diffusion for their survival. A noninvasive method of magnetic resonance imaging has previously been used for assessment of liver damage associated with clinical islet transplantation (25). Similar to the findings in our study, ischemic and necrotic areas were observed in the liver early after islet transplantation (2 days posttransplantation), but not 4 weeks later. A peak of insulin presence in peripheral blood was also observed early after transplantation in the clinical study, which likely reflected β-cell death with leakage of insulin (25). In our study, apoptosis occurred 1 day after transplantation of the human islets and to the same extent as we have previously reported for syngeneically transplanted murine islets at this site (23). However, we also observed an increased frequency of apoptotic human islet cells 1 month posttransplantation, similar to what was observed in murine islets (23), whereas necrotic islets were not discerned at this later time. The rate of apoptosis was even higher at 30 days than 1 day after transplantation of the human islet grafts. This indicates that a more regulated cell death persists in intraportally transplanted human islets for at least 1 month. Noteworthy, the human islet mass continued to decrease after the first day, and 1 month later, only slightly less than half of the graft area remained. A negative correlation was even found between apoptosis and islet mass at 30 days posttransplantation, indicating that these late events were important for islet loss. There was no correlation of cellular death posttransplantation with any of the donor characteristics, cold ischemia time of the pancreas, or insulin secretion capacity of the islets in vitro. However, an extended culture period had a negative influence on the posttransplant survival of islet cells, which suggests that the current restriction in culture time (0–48 h) applied in most clinical protocols is indeed important.
Islet endothelial cells were still present in cultured islets and 1 day after transplantation, but the density posttransplantation decreased to 20% of that in native islets. Endothelial cells present in the 1-day-old islet grafts likely reflect donor endothelial cells that survived the islet culture and transplantation procedure. However, when culturing rodent islets, their endogenous vessels are known to fully disappear within 2–3 days (21). This discrepancy is likely explained by the lower glucose concentrations used in the culture medium of human islets (12). Glucose has previously been reported to inhibit angiogenesis in human islets in vitro (10), and hyperglycemia has also been shown to induce apoptosis in pancreatic islet endothelial cells via reactive nitrogen species-mediated Jun N-terminal kinase activation (14). The residual endothelial cells are not connected to the recipient vascular system 1 day after islet transplantation and therefore do not contribute to blood perfusion at this stage. Indeed, 44% of islets stained positive for the hypoxia marker pimonidazole 1 day posttransplantation, which indicates poor oxygen delivery of the tissue. Moreover, since necrotic and apoptotic cells do not accumulate the marker (22), the degree of hypoxia in cells may even be an underestimation.
We have previously reported a decreased vascular density in human islets experimentally transplanted into the liver at 1 month posttransplantation (16), a finding which was confirmed in the present study. One month posttransplantation, the islet transplant vascular density had doubled, but was only approximately one third of that in native human islets. In the present study, we show that also the oxygen delivery to the human islet cells at this stage was inadequately low, since the percentage of hypoxic cells remained high 1 month posttransplantation. However, it should be noted that the absolute number of hypoxic islets had decreased, as only half of the tissue present at 1 day remained 1 month thereafter. Also for syngeneically transplanted mouse islets, low vascularization with increased numbers of pimonidazole positive islets was observed 1 month posttransplantation (23). In that study, a late revascularization seemed to occur, which increased islet vascular density and decreased the number of pimonidazole-positive islets 3 months posttransplantation to levels of native islets.
Some amyloid was observed in the human islets only 1 day after islet transplantation, which may reflect a tendency for amyloid formation during culture (31) or islet amyloid already being present in the islet donor. However, much more amyloid was observed at 30 days follow-up, which supports previous findings of a tendency for progressive amyloid formation in human islets posttransplantation at the renal subcapsular site (30). One possible reason for this may be inadequate drainage, since substantial formation of amyloid in an avascular setting has been observed in studies using microencapsulated human and recombinant human IAPP murine islets (6). Moreover, it has been reported that hypoxia plays a central role as a trigger for amyloid formation in Alzheimer's disease (7,32) and that this is in part mediated by the oxidative stress elicited by a hypoxic insult (27). Whether hypoxia triggers amyloid formation in islets is presently unknown. However, it has been postulated that oxidative stress may induce islet amyloid formation, leading to a positive feedback loop in which islet amyloid formation also induces oxidative stress (33). Notably, there was also a tendency toward higher frequency of apoptosis in islets containing an excessive amount of amyloid. In the end, progressive amyloid formation has the potential to cause graft failure due to triggering apoptotic events with concomitant loss of β-cell mass (28).
In the present study, we chose to use normoglycemic mice as recipients in order to avoid islet cell death due to hyperglycemia. We consider this a setting that better mimics the blood glucose control in patients, receiving islet transplants, who are either rendered normoglycemic by their grafts or subjected to intense supplemental insulin treatment regimes. We speculated that the chosen model would, if anything, underestimate the islet cell death posttransplantation. We have previously shown that the revascularization or blood perfusion of transplanted islets are not influenced by the blood glucose concentrations (8,19), but that oxygen tension levels may be decreased under hyperglycemic conditions due to the increased need for insulin secretion (8).
In conclusion, extensive loss of islets beyond the immediate posttransplantation phase occurs in islets transplanted into the liver. Novel strategies that support the engraftment of islets at this site or an alternative site of implantation need to be further explored in future studies.
Footnotes
Acknowledgments
The authors would like to thank Eva Törnelius, Anders Ahlander, Björn Åkerblom, and Marie Oskarsson for their support and technical assistance, as well as Professor Gunilla Westermark for expert advice on the islet amyloid evaluation. We would also like to thank the Nordic Network for Clinical Islet Transplantation (NNCIT) and the human islet isolation laboratory at Uppsala University Hospital for the generous supply with human islet tissue for the experiments. The study was generously supported by the Swedish Research Council, the Swedish Diabetes Association, the Swedish Juvenile Diabetes Foundation, Diabetes Wellness Sverige, and the Novo Nordisk Foundation. The authors declare no conflicts of interest.
