Abstract
In a previous study, we established a prenatal surgical approach and transplanted mesenchymal stem cells (MSCs) into the fetal rat spinal column to treat neural tube defects (NTDs). We found that the transplanted MSCs survived and differentiated into neural lineage cells. Various cytokines and extracellular signaling systems in the spinal cord niche play an important role in cell differentiation. In this study, we observed the differentiation of transplanted MSCs in different spinal cord niches and further observed the expression of neurotrophic factors and growth factors in the spinal cord at different developmental stages to explore the mechanism of MSC differentiation in different spinal cord niches. The results showed that transplanted MSCs expressed markers of neural precursor cells (nestin), neurogliocytes (GFAP), and neurons (β-tubulin). The percentages of GFP+/nestin+ double-positive cells in transplanted MSCs in E16, P1, and P21 rats were 18.31%, 12.18%, and 5.06%, respectively. The percentages of GFP+/GFAP+ double-positive cells in E16, P1, and P21 rats were 32.01%, 15.35%, and 12.56%, respectively. The percentages of GFP+/β-tubulin+ double-positive cells in E16, P1, and P21 were 11.76%, 7.62%, and 4.88%, respectively. The differentiation rates of MSCs in embryonic spinal cords were significantly higher than in postnatal spinal cords (p < 0.05). We found that the transplanted MSCs expressed synapsin-1 at different developmental stages. After MSC transplantation, we observed that neurotrophic factor-3 (NT-3), fibroblast growth factor-2 (FGF-2), FGF-8, transforming growth factor-α (TGF-α), vascular endothelial growth factor (VEGF), and platelet-derived growth factor (PDGF) significantly increased in the MSC transplantation group compared with the blank injection group. Furthermore, FGF-2 and VEGF expression were positively correlated with the number of surviving MSCs. In addition, we found that the expression of brain-derived neurotrophic factor (BDNF), NT-3, FGF-8, TGF-β, epidermal growth factor (EGF), and insulin-like growth factor (IGF) decreased with age, and the expression of FGF-2, FGF-10, FGF-20, TGF-α, and PDGF increased with age. Our data suggest that the embryonic spinal cord niche is more conducive to MSC differentiation after transplantation.
Introduction
Neural tube defects (NTDs) are complex congenital malformations that result from incomplete neurulation. With the development of prenatal diagnostic technology, NTDs have been found in the middle of pregnancy and even during the early stages of pregnancy, but the lack of effective treatments during pregnancy remains problematic. Advances in stem cell research have brought good prospects for the rehabilitation and reconstruction of nervous system damage. We have shown that sensory and motor neuron deficiency is a primary anomaly that coexists with spinal malformation (18,23,70), suggesting that neuronal replacement therapy based on neuron regeneration or cell replacement of the defective spinal cord is a promising approach to achieve a better functional outcome in spina bifida. Thus, in a previous study, we successfully established a new strategy for the potential treatment of spina bifida aperta using in utero transplantation of mesenchymal stem cells (MSCs). Our data indicated that MSCs survived, migrated, and differentiated into neurons in the spinal cord (30). Our results suggest that prenatal MSC transplantation can be used to treat spinal neuron deficiency in NTDs and might serve as a potential treatment option for other congenital anomalies. However, the mechanism and influence of the migration and differentiation of transplanted MSCs in the embryonic spinal cord niche have not yet been investigated.
MSCs are multipotent stem cells that can differentiate into different cell types, such as osteoblasts, adipocytes, chondroblasts, skeletal muscle cells, vascular cells, neuronlike cells, astrocyte-like cells, intestinal cells, and liver cells in response to different factors (25–27,36,44,63). Numerous studies indicated that MSCs have transdifferentiated into neurons both in vitro and in vivo (8,10,21,28,43,48,60). After implantation into sites of traumatic brain injury (TBI), MSCs may achieve long-term survival and differentiate into neural cells, including neurons and neurogliocytes (34). When exposed to an inflammatory environment or ischemic brain extracts in vitro, MSCs alter their growth factor expression profile (9,73), indicating that they are responsive to environmental cues. In some studies, fibroblast growth factor-2 (FGF-2) has been shown to improve the chondrogenic differentiation of MSCs (11,17,54,57). Furthermore, NT-3 stimulates MSCs to differentiate into neuronal phenotypes (72). Some authors have suggested that alterations in MSC culture conditions might have a marked impact on differentiation (2,55,64). Growth factors have become an important element in the effort to control the differentiation properties of MSCs. This research indicates that the differentiation of MSCs into nerve cells requires a suitable microenvironment.
Niches are specialized microenvironments that regulate stem cell activity. This niche is the sum of all factors, both cellular and molecular, that interact with and regulate stem cells. Stem cells reside in specialized microenvironments or “niches” that regulate their self-renewal and differentiation activity. Therefore, the niches where they reside regulate the behavior of stem cells. The concept of the stem cell niche was first introduced by Schofield (51). Mammalian niches are important regulatory structures that are complex. In the development of the nervous system, niches control stem cell maturation and the formation of the neuronal network. Horner et al. reported the proliferation and differentiation of progenitor cells throughout the intact adult rat spinal cord (22). The spinal cord contains cells, stroma, and other factors, and the environment induces stem cells to differentiate into a neuronal lineage. Neurogenesis has been associated with angiogenesis in the hippocampus, and endothelial cells release soluble factors that stimulate the self-renewal of stem cells, defining the angiogenic or vascular niche for neurogenesis (40,53). Recent studies of niches have begun to determine some of the functional components of individual niches. The niche provides support and maintenance for stem cells, regulates their proliferation, and may also be considered to direct downstream differentiation. Pertinent signals to the stem cell may include direct interactions with resident niche cells, extracellular matrix molecules, or diffusible autocrine, paracrine, and even endocrine signals (50), which are all integrated to provide the proper regulation and maintenance of the stem cell. However, the molecular and cellular mechanisms that underlie the interactions between stem cells and their niche need to be elucidated.
The spinal cord niche refers to its organization, cells, extracellular matrix, spinal fluid, and numerous factors. Various cytokines and extracellular signal transduction systems play an important role in MSC migration and differentiation in the spinal cord niche. Neurotrophic factors, neurotransmitters, and growth factors in the spinal cord niche can promote repair after spinal cord injury, and myelin-related lipoprotein, sulfide chondroitin protein, and polysaccharide inhibit the growth of axons. Therefore, the correlation between stem cells and the spinal cord niche has received increasing attention. Among the various cytokines, the family of neurotrophic factors, including nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), and neurotrophic factor 3 (NT-3), play an important role in maintaining neuron survival, promoting neuronal differentiation, inducing neurite outgrowth, regulating neural connections, and repairing injured nerves (7,67). Furthermore, fibroblast growth factors (FGFs) have been implicated in numerous cellular processes, including proliferation, migration, differentiation, and survival. Fibroblast growth factor family members also have broad effects on nervous system development, neural disease, and neural repair (19,75), and various FGF family members (e.g., FGF-2, FGF-8, FGF-20, etc.) have been used for the neural induction of MSCs in vitro. Transforming growth factor-β (TGF-β), the prototypical member of the TGF-β family, regulates a broad range of cellular responses, including cell proliferation, differentiation, adhesion, migration, and apoptosis. TGF-β has also been reported to regulate the differentiation, expansion, and migration of MSCs in vitro (24,59). TGF-α is an important mediator of wound healing and the injury response and has been shown to increase paracrine factor secretion of MSCs in vitro (66). Epidermal growth factor (EGF) is a general growth factor that exerts various actions, including cell migration and proliferation, in a wide variety of cells. MSC proliferation and migration significantly increased in response to EGF, and EGF significantly protected MSCs from apoptosis (16,58). Insulin-like growth factor (IGF) mediates several regenerative processes, including the modulation of inflammatory responses, apoptosis, and proliferation, and it was shown to stimulate the differentiation and migration of cultured MSCs (33). Vascular endothelial growth factor (VEGF) is involved in many central nervous system disorders. It is a key regulator of the paracrine effects of MSCs and was reported to significantly enhance the dopaminergic differentiation of human umbilical cord-derived MSCs (hUC-MSCs) in vivo in a rat model of Parkinson's disease (PD) (35,69). Platelet-derived growth factor (PDGF) can increase the differentiation of stem cells into a neuronal phenotype. Platelet-derived growth factor A (PDGF-A) affects cell proliferation, survival, migration, and differentiation through a paracrine or autocrine interaction, and the migratory activity of MSCs was greatly increased by PDGF-A treatment in vitro (3). Cizkova et al. (14,15) and Nandoe Tewarie et al. (39) showed that MSC transplantation improved functional recovery after spinal cord injury (SCI) in rats. Zhang et al. (71) demonstrated the implantation of bone marrow-derived MSCs (BM-MSCs) that were transfected with the neurotrophin-3 gene in a completely transected spinal cord. Multiple components, such as cells, the extracellular matrix, and various cytokines that are closely associated with stem cells, play important roles in stem cell survival, proliferation, and differentiation. However, to our knowledge, no studies have reported changes in the spinal cord niche during development. Research on the interaction between stem cells and the spinal cord niche could aid in the development of new therapies for fetal nerve tissue injury.
Therefore, we established MSC transplantation in embryonic and postnatal rats at different times. Studying MSC differentiation and exploring trends in the expression of neurotrophic factors and growth factors during neural development may provide theoretical and experimental evidence of new methods for the treatment of congenital spinal bifida aperta.
Materials and Methods
Experiment Animals
Outbred Wistar rats (female) of 10–12 weeks of age (250–300 g) and 4 weeks of age (approximately 100 g) were purchased from the animal center of China Medical University, Shenyang, China. The animals were maintained in an environment controlled for temperature (20–24°C) and humidity (50–70%) with a 12-h light/dark cycle. Solid laboratory chow and water were available ad libitum. Female rats were mated with male rats overnight. The appearance of vaginal plugs in female rats the morning after mating was considered embryonic day 0 (E0). All of the animal experiments were performed with approval from the ethics committee of China Medical University.
Isolation, Culture Expansion, and Transfection of Bone Marrow-Derived MSCs
Mesenchymal stem cells were isolated from a 4-week-old Wistar rat (female) following a previously published protocol (10). Rat MSCs were cultured in Dulbecco's modified Eagle medium/F12 (Life Technologies, Shanghai, China) supplemented with 10% fetal bovine serum (FBS; Hyclone, Shrewsbury, NJ, USA) and 100 IU/ml penicillin/100 μg/ml streptomycin (Life Technologies) on 25-cm2 tissue culture flasks (BD Biosciences, Franklin Lakes, NJ, USA). Primary isolated MSCs were defined as P0. When confluence was reached, the cells were passaged (1:2 dilutions) with fresh medium, and the MSCs were cultured until passage 15. Different passages of MSCs were used for transplantation. Twenty-four hours before transplantation, the MSCs were transfected with the enhanced green fluorescent protein (eGFP) expression adeno-5 vector (100 pfu/cell; SinoGenoMax Co., Ltd., Beijing, China) for the visualization of MSCs after transplantation into the rat spinal cord. Before transplantation, the cells were trypsinized, centrifuged, and resuspended in a small aliquot of fresh medium. The number of GFP-positive cells was counted, and the final concentration was adjusted to give a serial concentration that ranged from 7,000 to ~35,000 cells/μl.
Fetal Surgery and Microinjection of MSCs in E16 Embryonic Rats
We developed a new approach to transplant the cells into fetuses with the combined techniques of fetal surgery, microinjection, and microsurgery. The pregnant E16 rats were anesthetized with pentobarbital sodium (40 mg/kg body weight; Sinopharm Chemical Reagent Co., Ltd., Shanghai, China). An incision was made in the abdominal wall, and the uterine horn was exteriorized. To relieve uterine spasm, the uterus was covered with wet gauze immersed in warm physiological saline, and atropine (0.1 mg/kg body weight; Henan Runhong Pharmaceutical Co., Ltd., Henan, China) was administered intraperitoneally. Under the microscope, the position of the lumbosacral spine of the fetus was identified through the uterine wall. Afterward, a 7-0 nylon purse-string suture and small incision were made on the wall of the uterus. The amniotic sac was opened, and the spinal cord was exposed. A GFP-transfected MSC suspension (0.2 μl/injection site) was injected into the spinal cord with a micropipette (internal tip diameter 100 μm) connected to a Hamilton syringe. The micropipettes for injection were made from borosilicate glass capillaries (model GD-1; Narishige Scientific Instruments, Tokyo, Japan) using a micropipette puller (model PB-7; Narishige Scientific Instruments). After MSC injection, the fetuses were returned to the uterus, and the wound of the uterus was closed. On average, two to three fetuses could be injected in one dam without compromising the survival of the fetuses. The pregnant rats recovered from anesthesia within 1 h and were returned to their home cage.
Microinjection of MSCs in Postnatal Rats
MSCs were transplanted into Wistar rats (females) on postnatal day 1 (P1) and postnatal day 21 (P21). The rats were anesthetized with pentobarbital sodium (40 mg/kg body weight) and fixed on a rat board, with the spinous process as the center to make the incision. The skin of the dorsal side was incised, and the muscle was isolated. The spine was then exposed and cut, and spinal cord tissues were exposed. A GFP-transfected MSC suspension (0.2 μl/injection site) was then injected into the lumbar L1-L3 levels of the spinal cord with a micropipette (internal tip diameter 100 μm) connected to a Hamilton syringe, followed by layered sutures. The postnatal rats continued to feed after they awoke in the cage.
Sample Collection
Samples for Immunofluorescence
Four days after the transplantation of the MSCs, the pregnant rats and postnatal rats were anesthetized with an overdose of 10% chloral hydrate (Sinopharm Chemical Reagent Co., Ltd.). The fetuses and postnatal rats that received the transplants were transcardially perfused with 15 ml physiological saline followed by 25 ml of 4% paraformaldehyde (Sinopharm Chemical Reagent Co., Ltd.). The spinal column was dissected and postfixed in the same fixative for 24 h at 4°C, followed by cryopreservation in 20% sucrose for 24 h. The spinal column was then sectioned into 30-μm serial transverse sections, and all GFP-positive MSCs in the spinal column were observed and counted with a fluorescence microscope (Nikon, Tokyo, Japan). Images were acquired with a C1 confocal microscope (Nikon) or fluorescence microscope connected to a CCD camera (Nikon). Sections with GFP-positive MSCs were marked and stored at −80°C in the dark for further immunofluorescence analysis.
Samples for Real-Time PCR Analysis
The pregnant rats were sacrificed at E12, E14, E16, E18, and E20 by an overdose of 10% chloral hydrate injected into the abdominal cavity. The embryos were harvested, and the posterior spinal cord (from the inferior margin of the forelimb bud to the tail bud) was dissected. The postnatal rats were sacrificed at P1, P3, P5, P7, P11, P14, and P21 by an overdose of 10% chloral hydrate injected into the abdominal cavity, and the posterior spinal cords (L1–L3) were dissected. All of the samples were stored at −80°C for RNA extraction.
Immunofluorescence
To evaluate the differentiation of the MSCs before and after transplantation, the sections were analyzed by immunofluorescence using antibodies against nestin, β-tubulin, glial fibrillary acidic protein (GFAP), synapsin-1, and GFP as described previously (4). The primary antibodies included mouse anti-nestin (1:100; MAB353; Millipore, Billerica, MA, USA), mouse anti-GFAP (1:200; MAB3402; Millipore), rabbit anti-synapsin-1 (1:200; AB1543; Millipore), and mouse anti-β-tubulin (1:200; MAB1637; Millipore). After antigen retrieval, the sections were blocked with phosphate-buffered saline (PBS) that contained 10% fetal bovine serum (FBS) and 0.1% Triton X-100 (Sinopharm Chemical Reagent Co., Ltd.). The specimens were incubated with primary antibodies in combination with rabbit anti-GFP (1:200; AG279; Beyotime Institute of Biotechnology, Shanghai, China) or mouse anti-GFP (1:200; AG281; Beyotime Institute of Biotechnology) in 10% FBS-PBST overnight at 4°C. After primary incubation with the antibodies, the sections were washed three times with PBS, followed by incubation with Alexa Fluor 488-conjugated goat anti-rabbit immunoglobulin G (IgG) antibody (Invitrogen, Carlsbad, CA, USA)/Alexa Fluor 488-conjugated goat anti-mouse immunoglobulin G (IgG) antibody (Invitrogen) and TRITC-conjugated goat anti-mouse IgG (AP124R, Millipore)/TRITC-conjugated goat anti- rabbit IgG (AP 187 R, Millipore) in 10% FBS-PBST for 2 h at room temperature. After washing, the sections were stained with 4′,6-diamidino-2-phenylindole (DAPI) (C1002; Beyotime Institute of Biotechnology) and mounted with antifade mounting medium (P0126; Beyotime Institute of Biotechnology). To determine the percentage of MSCs that were differentiated, all GFP-immunopositive cells and all cells that were double-positive for GFP plus the aforementioned neuron/glia markers were counted in each section. The number of MSCs that expressed the specific neuron/glia markers was counted by observers who were blind to the experiments. The percentages of MSCs that expressed specific markers were determined as the number of double-positive cells/GFP-positive cells.
Real-Time PCR Analysis
Total RNA was quantified, and RNA with an A260 nm/A280 nm ratio less than 1.8 was discarded. Total RNA (2 μg) was reverse-transcribed at 37°C for 15 min, 85°C for 5 s, and 4°C with random 6-mer oligos (50 pmol), oligo dT primer (25 pmol), and the PrimeScript RT Reagent kit (Takara, Tokyo, Japan) in 20 μl of reaction solution. Real-time PCR (RT-PCR) products were diluted 1:10. Diluted RT-PCR products (2 μl) were subjected to quantitative real-time PCR using a SYBR® Premix Ex Taq™ II (Perfect Real Time) kit (Takara) in 20 μl of reaction solution that contained 2 μl cDNA templates, 0.4 μM of each primer, and 10 μl of 2× SYBR Green Master Mix (Takara) and brought to a final volume with RNase-free water. The reaction was performed in triplicate in a MicroAmp Optical 8-Tube Strip (4316567; ABI) with the 7500 Real-time PCR system (7500 Fast Real-time PCR System; Applied Biosystems, Foster City, CA, USA). PCR was performed as follows: predenaturation at 95°C for 30 s, 45 cycles of denaturation at 95°C for 5 s, and annealing at 58–63°C for 20 s. The negative control (i.e., without reverse transcriptase) gave no signal. The relative level of gene expression for each sample was calculated using the 2–ΔΔCt method and expressed as fold induction compared with the blank injection control from the same pregnant rats after β-actin normalization. GFP expression was analyzed with the Premix Ex Taq™ (Perfect Real Time) kit (Takara). Detailed information on the primers and probes used in this study are listed in Table 1.
Sequences of Primers and Probes Used in This Study

Statistical Analysis
All analyses were performed in a double-blinded manner. All values are expressed as mean ± SD. Student's t-test and one-way analysis of variance (ANOVA) followed by Bonferroni's post hoc analysis were used for single and multiple comparisons, respectively. Pearson correlations were calculated using SPSS 19.0 (IBM, Armonk, NY, USA). Values of p < 0.05 were considered statistically significant.
Results
Differentiation of MSCs Before and After Transplantation Into the Spinal Cord at Different Developmental Stages
A total of 102 fetuses received fetal surgery and microinjection of MSCs on E16, and 86 fetuses were harvested. A total of 82 postnatal rats received surgery and microinjection of MSCs on P1, and 68 were harvested. A total of 54 postnatal rats received surgery and microinjection of MSCs on P21, and 49 were harvested. Transplanted MSCs survived, and grew in the rat spinal column at different developmental stages (Fig. 1).
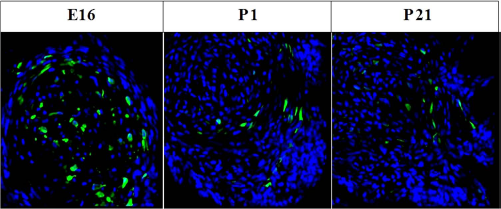
Survival of transplanted MSCs in rat spinal cord. Representative confocal images of transplanted MSCs (GFP+, green) that survived in the rat spinal column at different developmental stages. Scale bars: 50 μm.
We analyzed the markers for neural precursor cells (nestin), neurogliocytes (GFAP), and neurons (β-tubulin) in cultured undifferentiated MSCs at the same passages as those that were transplanted (Fig. 2). The results showed that the percentages of MSCs that expressed nestin, GFAP, and β-tubulin were 10.49%, 11.15%, and 1.67%, respectively (p < 0.05) (Table 2). The percentages were significantly lower in the cultured, undifferentiated MSCs compared with that of MSCs transplanted into spinal cord tissue.

The markers for neural precursor cells (nestin), neurogliocytes (GFAP), and neurons (β-tubulin) are in undifferentiated MSCs. Scale bars: 50 μm.
Effect of Different Developmental Stages on the Expression of Various Neural Markers in MSCs Before and After Transplantation

p < 0.05 compared with the MSCs transplantation in E16 and P1.
p < 0.05 compared with postnatal (P1 and P21) groups.
p < 0.05 compared with P21 groups.
To observe if transplanted MSCs generated different types of cells in the rat spinal cord at different developmental stages, we examined the expression of specific cell markers in transplanted MSCs on E16, P1, and P21. During the 4 days of rat development, transplanted MSCs expressed markers for neural precursor cells (nestin), neurogliocytes (GFAP), and neurons (β-tubulin and synapsin-1) (Figs. 3 and 4). The percentages of GFP+/nestin+ double-positive cells in transplanted MSCs at E16, P1, and P21 were 18.31%, 12.18%, and 5.06%, respectively. The percentages of GFP+/GFAP+ double-positive cells at E16, P1, and P21 were 32.01%, 15.35%, and 12.56%, respectively. The percentages of GFP+/β-tubulin+ double-positive cells at E16, P1, and P21 were 11.76%, 7.62%, and 4.88%, respectively. MSCs in the embryonic spinal column displayed significantly higher neuronal marker expression compared with postnatal rats (p < 0.05) (Table 2). The percentages of the above three kinds of double-positive cells in the embryonic spinal column were significantly higher than those in the P1 spinal column and the P21 spinal column (p < 0.05) (Table 2). However, only the percentage of GFP+/nestin+ double-positive cells and the percentage of GFP+/β-tubulin+ double-positive cells in the P1 spinal column were significantly higher compared with the P21 group (p < 0.05) (Table 2). To clarify whether transplanted MSCs differentiate into functional cells, we analyzed the expressions of synapsin-1 in transplanted MSCs on E16, P1, and P21 (Fig. 4). We found that the transplanted MSCs also expressed synapsin-1 at different developmental stages.

Differentiation of transplanted MSCs in the spinal cord at different developmental stages. Confocal images show the colocalization of GFP and cell-specific markers (red, arrows). (A) Nestin expression. (B) GFAP expression. (C) β-tubulin expression. Scale bars: 50 μm. (D) The percentages of GFP+/nestin+ double-positive cells, GFP+/GFAP+ double-positive cells, and GFP+/β-tubulin+ double-positive cells on E16, P1, and P21. The percentages were determined as the number of double-positive cells/total GFP+ cells. *p < 0.05 compared with postnatal day 1 and postnatal day 21 groups. #p < 0.05 compared with postnatal day 21 groups.
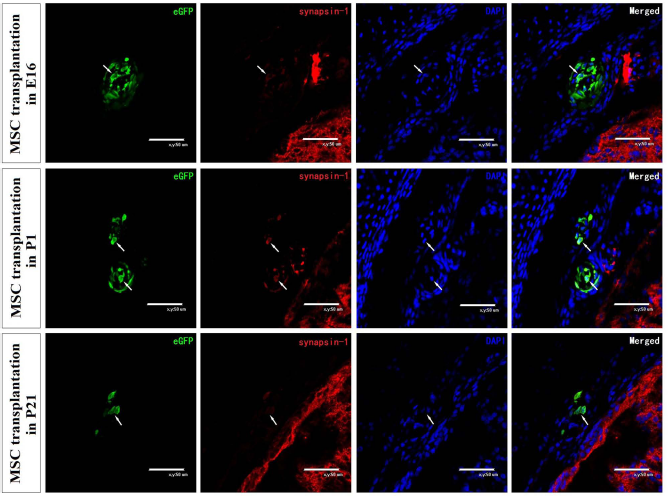
The expressions of synapsin-1 in transplanted MSCs on E16, P1, and P21. Confocal images show the colocalization of GFP and cell-specific markers (red, arrows). Scale bars: 50 μm.
All of the results described above indicate that MSC transplantation into fetal rat spinal cords favored differentiation to neural precursor cells, neurogliocytes, and neurons.
The Expression of Neurotrophic Factors and Growth Factors After MSC Transplantation Into the Spinal Cord
To clarify whether the expressions of neurotrophic factors and growth factors were affected after transplantation of MSCs, we analyzed the expressions of neurotrophic factors and growth factors in the rat spinal cords. After transplantation, NT-3, FGF-2, FGF-8, TGF-α, VEGF, and PDGF expression levels were significantly higher in the MSC transplanted groups than in the no injection group. The expressions of BDNF, NGF, FGF-10, FGF-20, TGF-β, EGF, and IGF were also elevated in the MSC transplantation group, although not significantly different from the no injection group (Fig. 5A). These results indicate that neurotrophic factors and growth factors were induced by MSCs transplantation. To determine whether the embryonic spinal cord niche is more favorable for MSC differentiation, we calculated the percentage of expression of neurotrophic factors and growth factors after MSC transplantation. The results showed that there were seven neurotrophic factors and growth factors, including PDGF, FGF20, BDNF, TGF-α, NGF, EGF, and FGF-10, with increased expression levels in the fetal spinal cord after MSC transplantation compared to those of the postnatal spinal cord and only six neurotrophic factors and growth factors, including VEGF, NT-3, FGF-2, FGF-8, TGF-β, and IGF, had decreased expression levels that were lower than those of the postnatal spinal cord. Meanwhile, the overall increased percentages of all tested neurotrophic factors and growth factors in the fetal spinal cord were obviously higher compared with those of the postnatal spinal cord (Fig. 5B). This could indicate that MSC transplantation into rat fetuses favored MSC survival and differentiation.

(A) Real-time PCR analysis of neurotrophic factor and growth factor expression in rat spinal cords transplanted with MSCs (MSC transplantation, n = 6), or injected with the same amount of medium (no injection, n = 6). The values are presented as fold expression differences compared with no injection. The p values are indicated by Bonferroni's test. (B) The percentage of expression of neurotrophic factors and growth factors in the embryonic group and postnatal group after MSC transplantation. The percentage of expression = (the expression with MSCs transplantation – the expression with no injection)/the expression with no injection × 100%.
To ascertain whether increased NT-3, FGF-2, FGF-8, TGF-α, VEGF, and PDGF expression levels were correlated with the number of surviving MSCs, we investigated correlations of GFP expression with NT-3, FGF-2, FGF-8, TGF-α, VEGF, and PDGF expression levels, as shown in Figure 6. FGF-2 and VEGF expression presented a significant correlation with GFP expression, and NT-3, FGF-8, TGF-α, and PDGF expression levels presented less of a correlation with GFP expression. The results indicate that FGF2 and VEGF were mainly expressed in transplanted MSCs themselves, and NT-3, FGF-8, TGF-α, and PDGF were expressed in both transplanted MSCs and host spinal cord tissue.
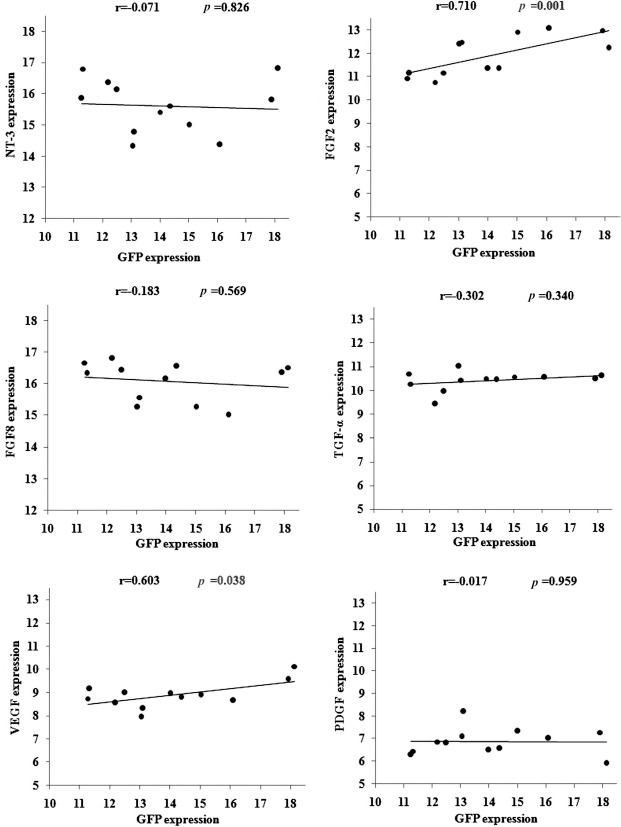
Correlation of GFP expression with NT-3, FGF-2, FGF-8, TGF-α, VEGF, and PDGF expression in rat spinal cord. Pearson's correlations between relative GFP expression with NT-3, FGF-2, FGF-8, TGF-α, VEGF, and PDGF expression levels were analyzed in spinal cords transplanted with MSCs (n = 12). The x-axis represents the relative GFP expression level. The y-axis represents the relative neurotrophic factor and growth factor expression levels.
The Expression of Neurotrophic Factors and Growth Factors in Normal Rat Spinal Cord at Different Developmental Stages
To clarify if the neurotrophic factors and growth factor expression were changed in the rat spinal cord niche at different development stages, we observed the expression of various neurotrophic factors and growth factors in the normal rat spinal cord without MSC transplantation at different time points. The results showed that the expression of BDNF, NT-3, FGF-8, TGF-β, EGF, and IGF tended to decrease during development, whereas the expression of FGF-2, FGF-10, FGF-20, TGF-α, and PDGF was increased. No obvious tendency in the expression of NGF and VEGF in either direction was observed (Fig. 7).

The expression of neurotrophic factors and growth factors during development. Neurotrophic factor and growth factor expressions were analyzed by real-time PCR analysis in normal rat spinal cords (n = 6).
Discussion
The migration and differentiation of MSCs are mainly determined by the spinal cord niche. Therefore, understanding changes in the molecular network related to MSC differentiation in the spinal cord niche is very important. After MSC transplantation, the early changes in the spinal cord niche mainly involved cell proliferation, differentiation, and migration. Subsequently, this niche maintains cell metabolism and phenotypic stability. In the present study, transplanted MSCs survived and expressed markers of neural stem cells (nestin), neurogliocytes (GFAP), and neurons (β-tubulin) in spinal cord tissues. The number of MSCs transplanted in the spinal cord at different developmental stages and the percentage of MSC differentiation were significantly higher in the embryonic spinal column than in the postnatal rat spinal columns. These results showed that the embryonic spinal cord niche was the more favorable niche for MSC differentiation. The process of cell migration and differentiation in vivo reflects an interaction between transplanted MSCs and various cytokines in the spinal cord niche. Various growth factors have been reported to be involved in stem cell survival, migration, and differentiation (1,42,61,65). These cytokines can be used as cell-mediated signals to regulate cell proliferation and differentiation, regulate the nutritional support of nerve cells, and promote functional recovery. To clarify if the neurotrophic factors and growth factors affect the fate of transplanted MSCs in the spinal cord, we evaluated the expression of various neurotrophic factors and growth factors. The results showed that neurotrophic factors and growth factors increased in the rat spinal cord niche after MSC transplantation. The expression of NT-3, FGF-2, FGF-8, TGF-α, VEGF, and PDGF significantly increased, which can promote axon extension, microvascular generation, myelin regeneration, and a reduction of apoptosis. Transplanted MSCs secrete neurotrophic factors that protect themselves and surrounding cells. In our previous study, the results showed that EGF was not expressed in cultured MSCs, but after transplantation EGF expression was observed in some of the transplanted MSCs in the spinal column, whereas FGF-2 was highly expressed in both cultured and transplanted MSCs (31). Meanwhile, we also found that transplanted MSCs not only expressed BDNF and NGF themselves, but also induced BDNF and NGF expression in the host spinal tissue (30). These data suggest that both transplanted MSCs and intrinsic responses of fetal spinal cords are responsible for increased growth factor expression. Meanwhile, the number of surviving MSCs could be affected by the status of the rodent immune system, which is still developing at E16. In the present study, we choose Wistar rats to perform the MSC transplantation. There is minimal rejection after organ transplantation from Wistar to Wistar, and it serves as a syngeneic control in many studies (56,68). Moreover, in the present study, the Wistar rat was transplanted with the same species of MSCs that possess immunosuppressive properties and may not induce an immune response. MSCs secrete soluble factors to create an immunosuppressive milieu. Studies on the interaction between MSCs and T cells support the potential use of allogeneic MSCs in regenerative medicine (47). It has been reported that MSC allografts can persist long term in immunocompetent hosts (52). Therefore, greater numbers of transplanted MSCs in the fetal spinal cord may be more related to the observed changes in growth factors than to the status of the rodent immune system.
The expression of various neurotrophic and growth factors has been investigated in other diseases (12,13,20, 29,41,46). However, neurotrophic and growth factor expression during the developmental stages of the spinal cord has not been reported previously. In the present study, the results showed that with increasing age expression of BDNF, NT-3, FGF-8, TGF-β, EGF, and IGF gradually declined in the spinal cord. The function of these factors is mainly neuronal survival and differentiation. BDNF is a neurotrophin with important functions in neuronal survival and differentiation. However, beyond its classic neurotrophic role, it is directly involved in the control of neuronal activity and synaptic plasticity as a neuromodulator (37,38,49). NT-3 supports sensory and motor neuron survival and promotes the proliferation and differentiation of neural crest cells. FGF-8 was reported to effectively predetermine bone marrow cells to differentiate into osteoblasts and increase bone formation in vitro (62). FGF-8 is an important factor in limb, ear, eye, and brain development and affects the development of the forebrain (45). TGF-β stimulates cell proliferation in the central nervous system (CNS) and inhibits neuronal apoptosis. EGF can promote mitosis and regeneration and cause neural stem cell proliferation and survival. In vitro, EGF can induce neural stem cells to differentiate into neurons and glial cells. IGF is a multifunctional cell proliferation regulation factor. It stimulates nerve cells to split between proliferation and survival and promotes the growth of axons. The expression of these factors in the embryonic stage is higher than in the postnatal stage, with the main effect being on neural development. This suggests that the factors in the embryonic spinal cord niche are conducive to MSC differentiation into nerve cells and are present at higher levels than in the postnatal period. However, the present study also found that the expression of FGF-2, FGF-10, FGF-20, TGF-α, and PDGF increased with age. This may have occurred because different cytokines play different roles during development. The primary functions of FGFs include the promotion of endothelial cell mitosis, enhancement of differentiation, chemotaxis, angiogenesis, and the promotion of neural ectoderm and mesoderm cells to survive and grow. Fibroblast growth factors are known to have effects on different tissues, depending on the developmental stage of the organ and concentration of growth factor present there. Furthermore, FGFs were found to have a significant effect on cell migration, proliferation, differentiation, and angiogenesis. The effects of FGF-2 on differentiation have also been closely studied, and it was found that FGF-2 inhibited the differentiation of oligodendrocyte progenitors (5). However, FGF-2 also has angiogenic properties, promotes proliferation and migration, and inhibits apoptosis in endothelial cells. FGF-10 is a presynaptic organizer that is involved in vesicle clustering and neurite branching (74). In many contexts, PDGFs are mitogenic during early developmental stages, driving the proliferation of undifferentiated mesenchymal cells and some progenitor cell populations (6). The development of vascular mural cells requires PDGF signaling (32). PDGF aids in the chemotaxis of neutrophils, smooth muscle cells, and fibroblasts. Altogether, the results suggest that various cytokines have different functions in the nervous system during development, and the embryonic spinal cord microenvironment is perhaps more conducive to the differentiation of MSCs after transplantation.
In conclusion, we found that transplanted MSCs survived, migrated, and expressed markers of neural precursor cells, neurons, and neurogliocytes in the rat spinal column. The differentiation rate of transplanted MSCs in the embryonic rat spinal cord was higher than in the postnatal rat spinal cord. After MSC transplantation, the expression of neurotrophic factors and growth factors significantly increased, indicating that transplanted MSCs can promote the expression of cytokines in the spinal cord niches, which is beneficial to the survival and differentiation of MSCs in the spinal cord. We also found that the expression of neurotrophic factors and growth factors changed with the development of the spinal cord. According to the number and abundant expression of neurotrophic factors and growth factors, the embryonic spinal cord niche was more favorable to neural differentiation. Because of the special microenvironment associated with embryonic development, in utero stem cell transplantation may have the best potential for the treatment for NTDs. However, the molecular and cellular mechanisms that underlie the interactions between stem cells and their environment remain to be elucidated at different developmental stages.
Footnotes
Acknowledgments
This study was supported by the National Natural Foundation of China (Grant Nos.: 81370717, 81171072, 81270755), National Basic Research Program of China (973 program, No. 2013CB945402), and Program for Liaoning Innovative Research Team in University (LT2013016). The authors declare no conflicts of interest.
