Abstract
Stroke is the third leading cause of death worldwide and a huge perpetrator in adult disability. This pilot clinical study investigates the possible benefits of transplanting multiple cells in chronic stroke. A total of 10 consecutive stroke patients were treated by combination cell transplantation on the basis of an intraparenchymal approach from November 2003 to April 2011. There were six males and four females. Their age ranged from 42 to 87 years, and the course of disease varied from 6 months to 20 years. Six patients suffered cerebral infarction, and four patients suffered a brain hemorrhage. The olfactory ensheathing cells, neural progenitor cells, umbilical cord mesenchymal cells, and Schwann cells were injected through selected routes including intracranial parenchymal implantation, intrathecal implantation, and intravenous administration, respectively. The clinical neurological function was assessed carefully and independently before treatment and during a long-term follow-up using the Clinic Neurologic Impairment Scale and the Barthel index. All patients were followed up successfully from 6 months to 2 years after cell transplantation. Every subject achieved neurological function amelioration including improved speech, muscle strength, muscular tension, balance, pain, and breathing; most patients had an increased Barthel index score and Clinic Neurologic Impairment Scale score. These preliminary results demonstrate the novel strategy of combined multiple cell therapy based on intraparenchymal delivery: it appears to be relatively clinically safe and at least initially beneficial for chronic stroke patients. This manuscript is published as part of the International Association of Neurorestoratology (IANR) supplement issue of Cell Transplantation.
Introduction
Stroke is one of the leading causes of mortality and disability in the world and results in substantial burdens for patients, their families, and society. Functional recovery in the chronic stage mainly depends on intensive physical, occupational, and speech rehabilitation, which may enhance restorative processes through brain plasticity mechanisms (17,18). The literature shows that, in large case series, functional recovery from stroke reaches its maximum by 3–6 months after onset and that no further recovery occurs beyond this time. Nevertheless, about 80% of these patients reach their maximum function for activities of daily living within 6 weeks from onset (17). Moreover, these functional recoveries may occur in a small or a localized brain insult using rehabilitation measures. For large strokes, recovery may require more comprehensive neurorestorative processes like neurogenesis, angiogenesis, neural/synaptic plasticity, and immunomodulation to lead to functional improvement after stroke (4,18,19).
Obviously, the self-repair mechanism by endogenous neural stem cells is insufficient to treat stroke, causing extensive neuronal death, although regeneration of the brain after damage is still active long after a stroke occurs (13,43). In a lesion area, the core may not respond to any pharmacological or rehabilitative interventions. For these reasons, the prospect of repairing the nervous system using cell transplantation seems promising and may offer a unique approach for brain repair and restoration of functions. Indeed, cell-based therapy represents a third wave of therapeutics for stroke and one focused on restorative processes with a longer time window of opportunity than neuroprotective therapies (20). Since transplantation for stroke in the rat model was first reported in 1992, demonstrating graft survival and even evidence of a connection with the host brain, the role of cellular therapies to promote functional recovery of stroke has recently received considerable attention (22,56,65).
Previous studies have shown that a variety of cell types can provide some functional recovery in animal models of stroke and several clinical trials (5,6,28,41,44,73,77). Currently, new ideas abound, and many strategies have been suggested and tested, including local delivery of cells to the stroke area versus grafting systemic route, cell therapy for reconstitution of structure and function versus use of cell grafts to support intrinsic repair and recovery mechanisms, one administration of cells versus combination grafts, or cografting of several cell types or cells and other substances (24,54,64). To date, the brain repair for stroke is more likely to require some form of combination therapy designed to support structure, attract new blood supply, enhance intrinsic repair and plasticity mechanisms, as well as the replacement of lost cells for the maximum possible restoration (52,56). Therefore, focusing on the chronic stage of stroke patients, the aim of this pilot study is to explore the possible role of multiple cells though different implant routes in these selected subjects.
Materials and Methods
Patient Data
The study was approved by the research ethics committee of Beijing Rehabilitation Hospital Affiliated Capital Medical University. Ten consecutive patients with sequelae of stroke or in a chronic phase who remained neurologically unchanged over at least 4 weeks of observation were recruited for this clinical study. All subjects gave their informed consent to participate, according to the Declaration of Helsinki, from November 2003 to April 2011 at our department. All subjects provided written informed consent for their clinical data being used for research. Subjects underwent standard examinations, including medical history, medical and neurological examinations, routine laboratory tests, a functional scales evaluation, and a magnetic resonance imaging (MRI) and/or computerized tomography (CT) scan of the brain.
Subjects' mean age was 55.00 ± 14.38 years (42–87 years), and four (40%) were women. There were six patients who suffered cerebral infarction, while four had brain hemorrhage. Course of the disease ranged from 6 months to 20 years, with an average of 5.80 ± 5.71 years. Table 1 presents the demographic characteristics of the study sample.
Ten Stroke Patients' Data Before Cell Transplantation and After Long-Term Follow-up

OEC, olfactory ensheathing cell; NPC, neural progenitor cell; UCMSC, umbilical cord mesenchymal stromal cell.
Cell Preparation
Tissue was obtained from elective abortions in accordance with guidelines issued by the Chinese Ministry of Health (91-006) (53).
Olfactory ensheathing cells (OECs): OECs were isolated from aborted human fetal olfactory bulbs (gender unknown) with proper consent of the donors. The OECs were cultured in Dulbecco's modified Eagle's medium/Ham's F12 (DMEM/F12; Hyclone, Logan, UT, USA) with 10% fetal bovine serum (FBS; Hyclone) and propagated for 2–3 weeks. The cells were then characterized by immunostaining with antibodies against p75 (Sigma, St. Louis, MO, USA) (27,29,47,75). Cells taken from one fetus were used in one patient.
Schwann cells (SCs): SCs were isolated from human fetal sciatic nerve after the specimens were cut into slices, digested in trypsin (Invitrogen, Carlsbad, CA, USA), and made into a monoplast suspension. These cells were cultured in DMEM/F12 with 15% FBS and harvested using trypsin after 7 days. Double-label immunofluorescence staining of S100 (Sigma) and Hoechst (Sigma) were obtained to calculate the purified rate of SCs (70). The donors are the same as for OECs.
Neural progenitor cells (NPCs): Under aseptic conditions, tissue from the subependymal zone of the fetal brain was taken. A cell suspension was prepared after repeated mechanical percussion by pushing media through a suction tube, and then the cells were cultured in DMEM/F12 (Hyclone) with 20% FBS (Hyclone) and passaged by plating on poly-l-lysine (PLL)-coated dishes (Corning, Corning, NY, USA). A proportion of the cells were characterized by immunostaining for nestin (Chemicon, Temecula, CA, USA) (32). The donors were the same as for OECs.
Umbilical cord mesenchymal stromal cells (UCMSCs): Tissues of umbilical cord were harvested after birth (unknown sex), and tissue blocks were implanted into dishes (Corning) in DMEM/F12 with 10% FBS and incubated in a 37°C 5% CO2 incubator. After 10 days, the cells that migrated from the tissue block were digested by using 0.25% trypsin + EDTA (Invitrogen) and propagated for five to six passages (70). Cells originating from one cord were used for all 10 patients.
Surgical Procedure
Patients were taken to the operating room, and cell transplantation was performed using stereotactic surgery under local anesthesia. The cells were suspended in saline and administered as follows: OECs (dose 1.0 × 106 /50 μl media; n = 2) alone or OECs (dose 1.0–2.0 × 106) + NPCs (dose 2.0–4.0 × 106; n = 8) mixed in 50 μl media were implanted into the perilesional area following MRI or CT targeting. NPCs (dose 2.0–5.0 × 106/500 μl media; n = 4) alone or NPCs (dose 2.0–5.0 × 106) + SCs (dose 2.0 × 106) mixed in 500 μl media (n = 1) were transplanted intrathecally in 5 ml of the patient's own cerebrospinal fluid (CSF) through cerebellar cistern puncture. UCMSCs with a dosage of 1.0–2.3 × 107 in 100 ml saline were transplanted by an intravenous route (n = 2). Patients received one or more of the above treatments as shown in Table 1.
No pharmacological immunosuppressive therapy was performed.
Physical and mental exercises, including Active Movement–Target Enhancement–Neurorehabilitation Therapy (AMTENT) (30) for 4–6 h/day, were required to support further brain restoration following cell therapy.
Functional Assessment
The patients' pretreatment functional stage was used as the base line controls. Neurological function was evaluated by two independent neurologists both before cell treatment and after long-term follow-up. The quality of life was assessed using the Clinic Neurologic Impairment Scale and the Barthel index. The Barthel index is comprised of 10 questions, measuring disability and functional independence in a person's activities of daily living. Item scores are summed to generate a total score, where 0 indicates maximum dependence (i.e., severe disability) and 100 indicates maximum independence (no disability) (16).
Statistical Analysis
Results are expressed as mean ± SD. The data were analyzed with SPSS for Windows (version 10.0; SPSS, Inc., Chicago, IL, USA), for paired-sample t test. A significance level of 0.05 was used.
Results
Ten patients were followed from 6 months to 2 years (mean 1.45 ± 0.60). No adverse events were observed during or after the operative procedure or during the long-term follow-up. All patients achieved some degree of neurological function amelioration, including improvements in speech, muscle power, muscular tension, balance, pain relief, and respiratory ability (see Table 1). The Barthel index score increased from preoperative 59.50 ± 28.52 to postoperative 64.00 ± 29.70 (p = 0.041). The Clinic Neurologic Impairment Scale score decreased from preoperative 13.60 ± 8.44 to postoperative 12.30 ± 8.55 (p = 0.039).
Illustrative Case Report
Patient #2, a 43-year-old Italian woman, was admitted to our department on April 15, 2005 with dysarthria and motor disturbance on her left limbs due to cerebral infarction of 3 years. Initially, after she was given symptomatic treatment and rehabilitation, her right limbs improved. During the next 2 years, there was no improvement. Mental status was normal. She could speak several words with dysarthria but could not say whole sentences. She could limp with a brace. It was more difficult for her to move her right fingers. The Babinski sign was positive on the left side. Head CT revealed focal left-side basal ganglia and temporal lobe encephalomalacia (Fig. 1). The procedure of OEC transplantation was done on April 18, 2005 under local anesthesia. At discharge, her speech was much clearer, and her right limbs were more flexible than before treatment. During 2 years of follow-up, she could speak whole sentences and spoke much clearer than before. She could walk without her brace, and her gait became almost normal. Her score of Barthel index increased from 85 to 100 and included an improvement in walking from 10 to 15, transfers (bed to chair and back) from 10 to 15, and climbing stairs from 5 to 10.
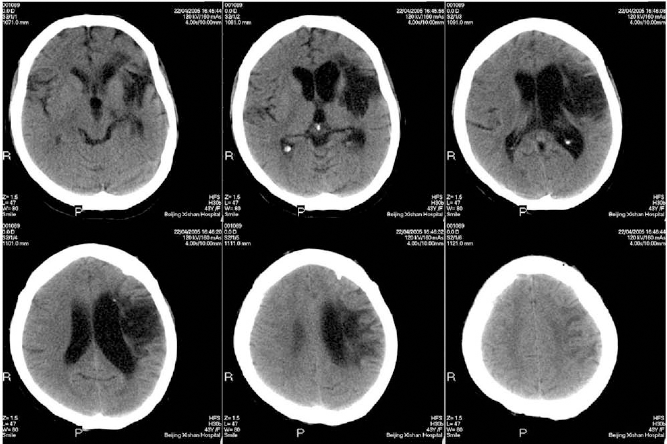
Findings of brain CT scan of patient #2 showing the cerebral infarction located at the left-side basal ganglia and temporal lobe.
Discussion
Cell transplantation has emerged as an experimental approach to restore brain functions. Our data support the hypothesis that multiple cell-based transplantation (OECs, Schwann cells, NPCs, and UCMSCs) might play a part in the functional recovery within the chronic phase of stroke patients, including patients that are 87 years old (patient 5) or with a clinical course of 20 years (patient 6). This is similar to a previous report that advanced age does not preclude a beneficial response to NPC transplantation following stroke (35).
Current Clinical Studies of Cell Therapy for Stroke
Our findings are supported by other studies, though so far only small pilot trials have tested the effects of cell therapy in stroke patients (2,36,63). Kondziolka et al. reported that there were no cell-related adverse effects during a maximum of 5 years follow-up after human neuronal cell transplantation into and around the small stroke volume of 26 patients with basal ganglia region motor stroke (40). In another clinical study, five patients with sequelae of stroke were given autologous bone marrow stromal cells (BMSCs) by stereotactical transplant into the perilesional area, and they showed some neurological function improvements and no serious adverse events caused by the surgery or cell implant during the 1-year follow-up (66). In an open-label, observer-blinded clinical trial, patients with severe middle cerebral artery territory infarct were randomly divided into two groups: the intravenous autologous marrow stromal cell (MSC) group (n = 16) or the control group (n = 36), and each subject was followed for a maximum of 5 years. No differences in the occurrence of seizures or recurrent vascular episodes were observed between the groups; however, MSC transplantation appeared to be safe and could improve the patients' function (42). Other ongoing clinical studies are now further exploring the safety and feasibility of cell-based therapies for stroke (3).
Possible Mechanisms of Different Cell Implants
Cell transplantation therapy offers an exciting multimodal strategy for brain repair in stroke patients (23). Stroke exploration with cell-based approaches aiming for trophic/growth actions, modulation of inflammation, neuroprotection, and neuronal replacement are ongoing (12,67). Cell therapy may provide significant benefit to stroke patients through upregulation of angiogenesis in the ischemic brain (22). The paracrine effects of secreted or released factors are believed to be responsible for most of the benefits observed after cell transplantation, rather than direct cell replacement (55,61). Although a comprehensive understanding of the mechanisms by which combined cell therapy may exert their therapeutic impact is currently lacking, increasing research data are adding knowledge of the mechanisms of each type of transplanted cells separately into the sphere of scientific evidence as described below.
OECs secrete numerous neurotrophic factors, including stromal cell-derived factor 1-α (SDF-1α) and brain-derived neurotrophic factor (BDNF), and promote axonal regeneration to potentiate neuroplasticity in neurodegenerative diseases (8). Our laboratory evaluated whether OECs are capable of eliciting repair of the white matter postgraft in middle cerebral artery occlusion (MCAO) rats (60). OEC transplantation into the periinfarct immediately after reperfusion was found to decrease infarct volume, promote survival, and improve neurological deficits, which immunohistochemical analysis suggested may have resulted from remyelination and axon regeneration (60).
Human OECs/olfactory nerve fibroblasts (hOECs/ONFs) have also been used in stroke models, and significant neurological improvement with increased glucose metabolic activity was observed along with enhanced neuroplasticity and the homing of endogenous stem cells in the rat and mouse ischemic brain (60).
Human umbilical cord tissue can be an ideal source of mesenchymal stromal cells due to its accessibility, vast abundance, and safety.
hUCMSCs implanted into the infarcted cortex of rats survived and secreted neuroprotective and growth-associated cytokines such as BDNF, platelet-derived growth factor-AA, basic fibroblast growth factor, angiopoietin-2, chemokine (C-X-C motif) ligand 16 (CXCL-16), neutrophilactivating protein-2, and vascular endothelial growth factor receptor-3 (46). Infarct volume and atrophy were reduced, while cortical metabolic activity and revascularization/ angiogenesis were enhanced (39,46). Since inflammatory signaling is involved in all stages of the ischemic cascade, from the early damaging events to the late repair processes (31), the beneficial effects of UCMSCs may include activation of endogenous CNS protective and reparative procedures as well as modulation of immunity and inflammation (68). Other possible explanations for the reduction in infarct size include diminished inflammation, inhibition of glial scar formation, enhanced subventricular zone neurogenesis, and survival of newly generated neuroblasts (51).
Enhancement of angiogenesis may be sufficient for an acute injection of hUCMSCs to be beneficial, but intracerebral injection in postacute ischemic stroke-treated rats causes rapid and transient changes in the cerebral microvasculature (50) and upregulation of β1-integrin expression. The latter was shown to be important for angiogenesis and recovery (15). Intracerebral grafts of UMSCs were equally effective in intracerebral hemorrhage (ICH) rats (45).
Intravenously transplanted human NPCs can enter the rat brain following an ICH and differentiate into neurons and astrocytes (32,57). NPCs transplanted into the cortex were observed to migrate long distances toward an ischemic lesion and, in most cases, adopted a neuronal phenotype (38), while in another study, they reduced the infarct size and adopted a neural phenotype (11). Furthermore, transplanted cells could differentiate and express electrophysiological properties expected of mature neurons (71).
It is becoming clear that cell transplantation of NPCs or NSCs may display their therapeutic effects by neuroprotection and immunomodulation rather than direct cell replacement. They can mediate efficient bystander myelin repair and axonal rescue (9,14,48), display anti-inflammatory actions, which can promote neuroprotection (59), reorganize white matter (33), and increase dendritic plasticity (49). Grafted Schwann cells can also contribute toward remyelination as well as promote nerve restoration in the rat model of cerebral hemorrhage (69).
Routes of Cell Implants
For all 10 patients, we first transplanted OECs alone or with NPCs by an intraparenchymal approach. The cell parenchymal implant is a key step in our protocol, which is supported by a prior study in which the therapeutic benefits of intracerebral and intravenous BMSC transplantation for cerebral infarct were compared (37). BMSCs were transplanted stereotactically (1 × 106 cells) or intravenously (3 × 106 cells) at 7 days after stroke. Results showed that intracerebral, but not intravenous, transplantation significantly enhanced functional recovery, and near-infrared (NIR) fluorescence imaging clearly visualized migration of intacerebral, but not intravenous, transplants toward the cerebral infarct 4 weeks following transplantation. This supports the idea that intracerebral administration may be more effective clinically (37), though this requires further study as this is an animal study, and not all animal studies agree with this observation (74).
Furthermore, intraparenchymal cell implants improved sensorimotor dysfunctions and motor deficits, but an intracerebroventricular injection of cells did not result in any improvement (64). Similarly, we strictly implanted cells into the peristroke tissue under the guidance of MRI and CT, rather than the lesion cyst.
For some patients, we combined the intracerebral transplant with intravenous and intracisterna magna delivery. Given the very limited sample, we did not compare the different outcomes for each subgroup, but our initial observations suggest that the procedure is safe as no side effects were apparent. Also, the Chopp group tested the effects of human umbilical cord tissue (hUTC) treatment in a rat model of stroke using different routes of administration including intravenous, intracisterna magna, or intracerebral injection at 24 h after stroke onset. All treatments led to significant functional improvements starting from day 14, but only the intravenous and intracerebral routes persisted to day 60, while the others had declined by day 49 (76).
Combination Cell Implants
Various cell types, including fetal cells, neural progenitor cells, glial cells, and stromal cells, are currently under investigation in experimental and clinical stroke trials, and the different graft sources possess unique advantages and limitations (58). Further studies integrating clinical trials and preclinical work with a focus on determining the mechanisms of action is needed to evaluate the true potential of cell therapy (20).
Enriched Environment and Cell Transplantation
Intensive training is strongly recommended to all of our patients. Environmental enrichment with rehabilitation has been shown to enhance the migration of transplanted cells to the injured area, promote functional recovery, and enhance subventricular zone neurogenesis after stroke (21).
Immunosuppression Application
No immunosuppression was performed because it is unclear whether it is required following cell transplantation. In addition, most stroke patients are aged subjects and so immunosuppressive therapy may be more harmful for them. However, CsA administration has been demonstrated to improve the survival of chromaffin cell xenografts (10); more experience is required before any conclusions can be drawn.
In summary, our findings must be interpreted with caution due to some limitations. In consideration of the small sample size, the methodological limitations, incomplete outcome data, and the absence of environmental enrichment or cell-only treatment groups, treatment with combination cells cannot be judged as completely safe and feasible: large, well-designed trials are needed. It is also necessary to gain a better understanding of the mechanisms of action of the cells after transplantation, the therapeutic time window for cell transplantation, the optimal route of cell delivery to the brain, the more suitable cell types and sources, the selection of patients likely to benefit from such a therapy, the development of imaging techniques to monitor cell therapy, and the best methods to control stem cell proliferation, survival, migration, and differentiation in the pathological environment (1,18,38). To ensure the best functional recovery, neurorestorative therapy is likely to require the application of a combination approach (7,25,26,30). Together, despite the above-mentioned limitations, our pilot study raises hope to provide a therapeutic option for patients with sequelae of acute or chronic stroke as it appears to be relatively safe and certainly initially beneficial.
Footnotes
Acknowledgments
We thank the patients who participated in this clinical study. The authors declare no conflict of interest.
