Abstract
Our previous series of studies have proven that olfactory ensheathing cell (OEC) transplantation appears to be able to slow the rate of clinical progression after OEC transplantation in the first 4 months and cell intracranial (key points for neural network restoration, KPNNR) and/or intraspinal (impaired segments) implants provide benefit for patients (including both the bulbar onset and limb onset subtypes) with amyotrophic lateral sclerosis (ALS). Here we report the results of cell therapy in patients with ALS on the basis of long-term observation following multiple transplants. From March of 2003 to January of 2010, 507 ALS patients received our cellular treatment. Among them, 42 patients underwent further OEC therapy by the route of KPNNR for two or more times (two times in 35 patients, three times in 5 patients, four times in 1 patient, and five times in 1 patient). The time intervals are 13.1 (6–60) months between the first therapy and the second one, 15.2 (8–24) months between the second therapy and the third one, 16 (6–26) months between the third therapy and the fourth one, and 9 months between the fourth therapy and the fifth time. All of the patients exhibited partial neurological functional recovery after each cell-based administration. Firstly, the scores of the ALS Functional Rating Scale (ALS-FRS) and ALS Norris Scale increased by 2.6 + 2.4 (0–8) and 4.9 + 5.2 (0–20) after the first treatment, 1.1 + 1.3 (0–5) and 2.3 + 2.9 (0–13) after the second treatment, 1.1 + 1.5 (0–4), and 3.4 + 6.9 (0–19) after the third treatment, 0.0 + 0.0 (0–0), and 2.5 + 3.5 (0–5) after the fourth treatment, and 1 point after the fifth cellular therapy, which were evaluated by independent neurologists. Secondly, the majority of patients have achieved improvement in electromyogram (EMG) assessments after the first, second, third, and fourth cell transplantation. After the first treatment, among the 42 patients, 36 (85.7%) patients' EMG test results improved, the remaining 6 (14.3%) patients' EMG results showed no remarkable change. After the second treatment, of the 42 patients, 30 (71.4%) patients' EMG results improved, 11 (26.2%) patients showed no remarkable change, and 1 (2.4%) patient became worse. After the third treatment, out of the 7 patients, 4 (57.1%) patients improved, while the remaining 3 (42.9%) patients showed no change. Thirdly, the patients have partially recovered their breathing ability as demonstrated by pulmonary functional tests. After the first treatment, 20 (47.6%) patients' pulmonary function ameliorated. After the second treatment, 18 (42.9%) patients' pulmonary function improved. After the third treatment, 2 (28.6%) patients recovered some pulmonary function. After the fourth and fifth treatment, patients' pulmonary function did not reveal significant change. The results show that multiple doses of cellular therapy definitely serve as a positive role in the treatment of ALS. This repeated and periodic cell-based therapy is strongly recommended for the patients, for better controlling this progressive deterioration disorder.
Keywords
Introduction
Amyotrophic lateral sclerosis (ALS) is a progressive, fatal, neurodegenerative disease caused by the degeneration of motor neurons, the nerve cells in the central nervous system that control voluntary muscle movement. Death due to respiratory failure occurs typically 2–5 years after disease onset (84). Although the etiology of ALS remains largely unknown, the pathological findings are commonly the degeneration of the corticospinal tracts associated with loss of upper and/or lower motor neurons and demyelinated axons. The corticospinal tract degeneration is most evident in the lower parts of the spinal cord, but it can be traced up through the brain stem to the posterior limb of the internal capsule and corona radiata by means of fat stains, which show the macrophages that accumulate in response to myelin degeneration (74). The food and drug administration (FDA) of the US has approved only one drug treatment for the disease, Riluzole, which is believed to reduce damage to motor neurons by decreasing the release of glutamate via activation of glutamate transporters. Currently, no treatments can cure ALS. Cell-based therapy had been introduced clinically as a promising attempt to treat ALS and prolong the life span during the last 20 years through the potential neuroprotective effect, neurorestoration, and neuroplasticity, etc. (2,10,13,14,19,22, 33–35,37,61–63). Olfactory ensheathing cells (OECs) are a novel glia that displays Schwann cells together with astrocyte-like properties. They are a source of growth factors and adhesion molecules that have a very important role as neuronal support enhancing cellular survival (48,67).
Our previous studies have implied that OEC intracranial (key points for neural network restoration, KPNNR) and/or intraspinal transplantation are indeed beneficial for patients with ALS (12,34,35). Our further study has suggested that OEC transplantation is capable of slowing the rate of clinical progression within the initial 4 months after transplantation (34). The majority of the patients had neurological functional improvements and/or delayed the deterioration following the first cell therapy; however, they usually need to receive further cell medicine for maintaining the improved outcome. In this article, we report the effects of multiple cell therapies on neurological function, electromyogram, and pulmonary function in patients suffered with ALS on basis of the long-term observation.
Materials and Methods
Patients
Patients with a “probable” or “definite” ALS diagnosis (according to the revised El Escorial criteria) (9) were eligible for inclusion in the study. Between March of 2003 and January of 2010, a total of 507 ALS patients underwent our cellular treatment. Of them, 42 patients (31 males and 11 females) underwent further OEC therapy by the route of KPNNR for two or more times (two times in 35 patients, three times in 5 patients, four times in 1 patient, and five times in 1 patient). Their ages ranged from 20 to 73 with an average of 49.9 ± 10.7 years. The duration before the first cell transplantation varied from 0.5 to 8.0 (mean 2.9 ± 2.0) years. In this study, upper limb onset occurred in 22, lower limb onset in 11, bulb onset in 7, upper and lower limb onset in 1, and neck onset in 1. The time intervals were 6–60 (mean 12.1 ± 8.9) months between the first therapy and the second one, 8–24 (mean 15.0 ± 5.7) months between the second therapy and the third one, 6–26 (mean 16.0 ± 14.1) months between the third therapy and the fourth one, and 9 months between the fourth therapy and the fifth one. The study was conducted at Beijing Hongtianji Neuroscience Academy in conjunction with Beijing Rehabilitation Center in accordance with guidelines issued by the Chinese Ministry of Health (91–006) (69). All eligible patients and/or families were fully informed about the nature of the study, and provided written informed consent to participate in the study.
OEC Harvest
OECs were isolated and cultured in GMP standard laboratory as described previously (55). In brief, OECs were isolated from aborted human fetal olfactory bulb with donor consent. The OECs were cultured and propagated for 2–3 weeks and characterized by immunostaining with antibodies against p75 (a neurotrophin receptor that is specific for OECs, Sigma) (55). OECs from one to two fetuses, which represent 2 × 106 cells/100 μl, were transplanted for each patient. In accordance with the Chinese Pharmacopoeia procedure, the bacteria, viruses, fungi, mycoplasma, and endotoxin testing were done. Cell quality indicators: cell activity ≥85%. Human leukocyte antigen-DR (HLA-DR)-matching tests were carried out before transplantation to ensure the histocompability between the donors and the recipients.
OEC Surgical Transplantation
Initially, 1 × 106 cells/50 μl were transplanted into the involved spinal cord under general anesthesia. Then the cell transplantation procedure consisted of the injection of 2 × 106 cells/100 μl into KPNNR within the corona radiata of the frontal lobes bilaterally using stereotactic techniques under local anesthesia (11) as T2 weighted and, especially, fluid attenuated inversion recovery (FLAIR) MR images show signal pathological changes that reflect Wallerian changes in the corticospinal tracts (Fig. 1).

Fluid attenuated inversion recovery magnetic resonance imaging (FLAIR MRI) films show the degenerative findings though the corticospinal tract and/or corticobulbar tract at the level of the medulla oblongata (A), pons (B), midbrain (C), subinternal capsule (D), and the corona radiate (E) in amyotrophic lateral sclerosis (ALS). The two brightened dots show the targets of cell transplant: two injection sites of olfactory ensheathing cells (OECs) in the corona radiate of both-sided frontal lobes (key point for neural network restoration, KPNNR) (E).
Clinical Evaluation (Including Neurological Function, MRI, EMG, and Pulmonary Function)
The neurological function of patients was evaluated by three neurologists independently before and at 2–4 weeks after cell intracranial transplant. The ALS Functional Rating Scale (ALS-FRS) and the Modified Norris bulbar and Limb scale (23), together with a detailed video record of each patient were taken.
A magnetic resonance imaging (MRI) scan was done before each cellular surgery. Electromyogram (EMG) and pulmonary function test were performed pre- and post-cell transplantation (2–4 weeks after operation).
We used Keypoint-4 electromyograph of evoked potential instrument from Denmark. In EMG, 8–12 muscles were tested, which included proximal and distal part of four limbs, muscles of tongue, and sternocleidomastoideus muscle. The amount of motor unit potentials (MUPs), wave amplitude, and persistent discharge ability were recorded during maximal voluntary contraction of tested muscle. The results described as follows mean EMG improvements: spontaneous potentials diminished, recruitment pattern of the maximal voluntary contraction of involved muscles became more intensive and partially together with an increased number of MUPs, and MUPs recorded successfully which failed to occur preoperatively.
We performed pulmonary function by using pulmonary function analyzer (CHEST HI-801, Japan). We tested vital capacity (VC), forced vital capacity (FVC), forced expiratory volume (FEV1), FEV1/FVC, maximum minute ventilation (MVV), maximal expiratory pressure (MEP), and maximum inspiratory pressure (MIP) pre- and post-cell transplantation (2–4 weeks after operation).
Statistical Analysis
The data were expressed as means ± SD. The data were analyzed with SPSS for Windows (version 13.0; SPSS, Inc., Chicago, IL), for either two-tailed paired samples t-test, or one-way ANOVA test (Bonferroni values for equal variances, but Tamhane's T2 values for no homogeneity of variances). A significance level of 0.05 was used.
Results
Neurological Function
All of the patients had neurological functional recovery after multiple cell-based administrations. After the first treatment, the scores of ALS-FRS increased from 24.5 ± 7.1 to 27.1 ± 7.1 (t = −7.084, p < 0.001) and ALS Norris Scale increased from 55.9 ± 20.9 to 60.8 ± 22.1 (t = −6.148, p < 0.001); after the second treatment, ALS-FRS score increased from 20.1 ± 7.3 to 21.1 ± 7.4 (t = −5.200, p < 0.001) and Norris score increased from 43.0 ± 21.6 to 45.6 ± 21.8 (t = −5.003, p < 0.001); after the third treatment, ALS-FRS score increased from 18.3 ± 7.7 to 19.4 ± 8.4 (t = −2.066, p = 0.084) and Norris score increased from 36.1 ± 20.9 to 39.6 ± 23.1 (t = −1.310, p = 0.238); after the fourth treatment, ALS-FRS score had no change with 20.5 ± 4.9 and Norris score increased from 37.5 ± 24.7 to 40.0 ± 28.3 (t = −1.000, p = 0.500). The scores of ALS-FRS and ALS Norris Scale increased by 2.6 ± 2.4 (0–8) and 4.9 ± 5.2 (0–20) after the first treatment, 1.1 ± 1.3 (0–5) and 2.3 ± 2.9 (0–13) after the second treatment, 1.1 ± 1.5 (0–4) and 3.4 ± 6.9 (0–19) after the third treatment, 0.0 ± 0.0 (0–0) and 2.5 ± 3.5 (0–5) after the fourth treatment, and 1 point after the fifth cellular therapy (Table 1, Fig. 2).

Electromyogram (EMG) finding in one patient (A, B). At 2 weeks (C, D) and 6 months (E, F) after the first cell treatment, EMG showed the recruitment of the maximal voluntary contraction of both sides of first dorsal interosseous (Int) muscles was changed, and its pattern was improved and more intensive. Following the third operation, recruitment of maximal voluntary contraction increased in the vastus medialis (Med) muscles bilaterally, and the pattern was more intensive (G, H). After the fourth cell transplantation, the motor unit potentials were recorded in the left vastus med muscle. The recruitment of maximal voluntary contraction was improved and more intensive in the right vastus med muscle (I, J). This patient's medical status keeps very well during the 5 years after four cell transplantations (K, red arrows show the cell transplantation and blue arrow shows the attack of lacunar infarction).
Amyotrophic Lateral Sclerosis Functional Rating Scale (ALS-FRS) and Norris Scale Score and Increased Score After Four Treatments

There was a statistically significant difference between pretreatment ALS-FRS and Norris scale score and posttreatment score after the first and second doses of cell therapy (p < 0.01). Increased scores of ALS-FRS in first group was significantly more than the other three groups (p < 0.05) and no statistic differences were shown between second and third, third and fourth groups (p > 0.05), but it is difference between second and fourth groups (p < 0.05). There was statistic difference on increased score of Norris scale between first and second cell transplant (p = 0.019).
No patients in this study experienced side effects or complications following the operation. Re-MRI studies did not show any development of tumors, hemorrhages, edema, cyst formations, infections, disruption of neural structures, and other pathological findings in the transplanted sites.
Electromyogram (EMG) Study
After the first treatment, among the total of 42 patients, 36 (85.7%) patients' EMG results become better and the remaining 6 (14.3%) patients showed no remarkable change. Improvements of EMG include: a) spontaneous potentials diminished in 10 patients (23.8%); b) recruitment pattern of the maximal voluntary contraction of involved muscles improved, which became more intensive and partially together with increased number of motor unit potential in 33 patients (78.6%); c) motor unit potential recorded successfully, which failed to occur before surgery in 3 patients (7.1%).
After the second treatment, out of the 42 patients, 30 (71.4%) had improvement, 11 (26.2%) showed no remarkable change, and 1 (2.4%) worsen. Improvements of EMG include: a) spontaneous potentials diminished in 8 patients (19.0%); b) recruitment pattern of the maximal voluntary contraction of involved muscles improved, which became more intensive and partially together with increased number of motor unit potential in 28 patients (66.7%); c) motor unit potential recorded successfully, which failed to occur before surgery in 2 patients (4.8%).
After the third treatment, among the 7 patients, 4 (57.1%) patients' EMG results improved, 3 (42.9%) patients no change. Improvements of electromyogram includes: a) spontaneous potentials diminished in 1 patient (14.3%); b) recruitment pattern of the maximal voluntary contraction of involved muscles improved, which became more intensive and partially together with increased number of motor unit potential in 2 patients (28.6%) (Fig. 2); c) motor unit potential recorded successfully, which failed to occur before surgery in 1 patient (14.3%).
Two patients received the fourth treatment, and both EMG results ameliorated. The improvements were both in the recruitment pattern of the maximal voluntary contraction of involved muscles (Fig. 3).
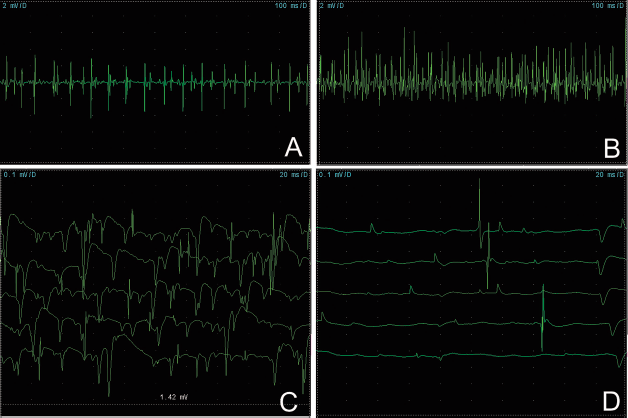
EMG finding in one patient. At 17 days after the first cell treatment, EMG showed the recruitment of the maximal voluntary contraction of left-sided vastus medialis improved. Following the third operation, recruitment of maximal voluntary contraction increased in vastus med muscles bilaterally, and the pattern was more intensive (A, B). At 20 days after the second cell transplantation, spontaneous potentials diminished in right tibialis anterior (C, D).
One patient received the fifth treatment and the EMG results improved. The improvement was successful recording of motor unit potential.
Pulmonary Function
The First Treatment
Twenty-one patients' pulmonary function was normal. So we analyzed the other 21 patients' pulmonary function. The details of the results were shown in Table 2. There was no significant difference between pretreatment and posttreatment.
The Results of Pulmonary Function After the First Treatment

VC, vital capacity; FVC, forced vital capacity; FEV1, forced expiratory volume; MVV, maximum minute ventilation; MEP, maximal expiratory pressure; MIP, maximum inspiratory pressure.
The Second Treatment
Fifteen patients' pulmonary function was normal. So we analyzed the other 27 patients' pulmonary function. The details of the results were shown in Table 3. No significant difference between pretreatment and posttreatment was found.
The Results of Pulmonary Function After the Second Treatment

The Third Treatment
We analyzed these 7 patients' pulmonary function. The details of the results were shown in Table 4. There was no significant difference between pretreatment and posttreatment.
The Results of Pulmonary Function After the Third Treatment
The Fourth and Fifth Treatments
Two patients received the fourth treatment and 1 patient received the fifth treatment. After the fourth treatment, 1 patient became better and the other patient revealed no change. After the fifth treatment, the patient's pulmonary function kept stable.
Discussion
The pivotal practical therapeutic goal in neurodegenerative diseases is currently to diminish neural dysfunction using varied strategies (1,32,41,64,77). There have been accumulating basic and clinical researches on Neurorestoratology (3,6,7,28,42,59,80,88,93,97). Numerous studies on treating ALS with cells have been accomplished. Cell-based restorative treatment has become a new trend and rapidly increasing data worldwide have proven that it has a crucial therapeutic value in ALS (Tables 5 and 6).
Selected Preclinical Literature of Cell-Based Therapy for ALS (Data From PubMed)

Literature on Cell-Based Therapy in ALS in Humans (Data From PubMed)

The community of clinical physicians, however, still believe that there are no methods that can control or stabilize the progressive deterioration for ALS patients; therefore, self-comparison design was used in this study as an effective tool to answer whether the treatment of multiple cell therapies have a positive role on deteriorated ALS, like the cell transplant research for corneal lesions, although the randomizing double blind control study including placebo or sham-surgery is an important design to assess the effect in clinical medicine trials (38,70).
Herein, we provided the evidence of the feasibility and efficacy of multiple OEC neurorestorotherapy in ALS patients and these new findings reinforced our previous findings (9,34,35). Our results clearly demonstrate that ALS patients recovered their neurological functions and received benefit from multiple grafted OEC transplantation, which did not cause significant side effects. The patients appear to have a similar level of positive reaction after the first, second, third, fourth, or even fifth cell therapy, although there were no statistical differences in several measures, which were most likely due to the obvious different sample size and homogeneity of variances, etc. Some of our patients obtained long-term control of the progression of ALS and improved their daily quality of life.
Given the potential risks following the traditional spinal cord surgery under general anesthesia in ALS patients, Huang et al. first proposed the key point for neural network restoration (KPNNR) in brain based on a set of successful cases in clinical practice (8,35). The core target where the cells should be transplanted into is located anterior 1/4–1/3 of the lateral ventricle and 23–27 mm away from the midline. This site is where the frontal corona radiata pyramidal tract passes through, and represents a point at which numerous projection fibers, association fibers, and commissural fibers converge. After being transplanted into this important “point” in the brain, the cells will initiate an extensive bidirectional remodeling in the entire neural network including the cerebrum, cerebellum, and spinal cord. Evidence shows that the procedure of KPNNR cell transplantation under local anesthesia is safer and better tolerated, especially in ALS patients with impairment of breathing.
The olfactory system is unique in that it supports continuous growth of axons from the olfactory epithelium into the CNS throughout the lifetime of the individual. The olfactory system's capacity for axonal extension and target-specific synaptic interaction has been attributed to the growth-promoting properties of OECs (40,80). Transplantation of these cells has been shown to have a neuroprotective effect, support axonal regeneration, remyelinate demyelinated axons, neuroplasticity, neuromodulation or neural signal repair, neurogenesis, angiogenesis, anti-inflammatory response, reducing scar and cavity formation, and/or strong phagocytic activity (23,49,51,56,58,60,62,68,71,72,78,79,83,85–87,89,91,92, 94,96).
A preclinical study by Martin and Liu (60) shows that adult olfactory bulb multipotent neural precursor cell (NPC) grafts provide temporary protection from motor neuron degeneration, improve motor function, and extend survival in ALS mice. They delayed disease onset to approximately 117 days, compared with control onset at approximately 90 days. The life span of NPC recipient mice was extended (approximately 170 days) compared with the life span of controls (approximately 140 days). In our lab, we found that OECs injected into the dorsal funiculus of thoracic spinal cord of SOD1G93A rats is feasible and safe; OECs prolong the survival of transgenic SOD1G93A rats compared with sham controls. The mechanism tests at 4 weeks after OEC transplantation reveal that the transplanted OECs not only can migrate at least 4.2 mm away from the injecting site, but also can protect motor neurons from death and remyelination of the nerve fibers (53). Furthermore, a report of autopsy cases has shown that the transplanted cells can survival for 12–24 months (28).
Therefore, the results demonstrate that the cell-based therapy definitely has the capability of playing an essential role for the treatment of catastrophic ALS. The regime of periodic OEC intracerebral transplantation is strongly recommended to patients for better controlling the nature and progressive deterioration of this disastrous CNS disorder, although the cure has not yet been available so far.
Footnotes
Acknowledgments
Financial support from Capital Medical Development Scientific Research Fund of Beijing Municipal Health Bureau of China (2009-3229) is gratefully acknowledged. We thank Ms. Wei Wang for preparing the clinical materials. The authors declare no conflicts of interest.
