Abstract
Multiple sclerosis (MS) is a complex disease of neurological disability, affecting more than 300 out of every 1 million people in the world. The purpose of the study was to evaluate the therapeutic effects of human umbilical cord-derived mesenchymal stem cell (hUC-MSC) transplantation in MS patients. Twenty-three patients were enrolled in this study, and 13 of them were given hUC-MSC therapy at the same time as anti-inflammatory treatment, whereas the control patients received the anti-inflammatory treatment only. Treatment schedule included 1,000 mg/kg of methylprednisolone intravenously (IV) daily for 3 days and then 500 mg/kg for 2 days, followed by oral prednisone 1 mg/kg/day for 10 days. The dosage of prednisone was then reduced by 5 mg every 2 weeks until reaching a 5-mg/day maintenance dosage. Intravenous infusion of hUC-MSCs was applied three times in a 6-week period for each patient. The overall symptoms of the hUC-MSC-treated patients improved compared to patients in the control group. Both the EDSS scores and relapse occurrence were significantly lower than those of the control patients. Inflammatory cytokines were assessed, and the data demonstrated a shift from Th1 to Th2 immunity in hUC-MSC-treated patients. Our data demonstrated a high potential for hUC-MSC treatment of MS. This manuscript is published as part of the International Association of Neurorestoratology (IANR) special issue of Cell Transplantation.
Keywords
Introduction
Multiple sclerosis (MS) is an inflammatory disease of the central nervous system (CNS) that may result in severe disability and neurological defects (20). The onset of MS is usually between the ages of 20 and 40 and is twice as common in females than in males (12). It is characterized by damage of myelin sheaths, the protective material around nerve fibers in brain and spinal cord, and therefore results in disruption of normal conduction of nerve impulses. It is commonly believed that MS is an autoimmune disease; however, there is also evidence pointing to the loss of oligodendrocytes for myelin generation as an underlying mechanism for MS (7,12,19,20). Four subtypes of MS have been classified: relapsing–remitting MS (RRMS), secondary progressive MS (SPMS), primary progressive MS (PPMS), and progressive-relapsing MS (PRMS).
No therapeutic method that has been developed for MS is a cure. Nonetheless, anti-inflammatory or immunosuppressive agents are commonly used as treatment options for symptom alleviation. These medications often have severe adverse effects and fail to prevent disease progression in many circumstances (6,15). Emerging data suggested that stem cells have great potential in regenerative medicine, and such cell-based therapy may provide an alternative approach to the currently approved MS treatments (5,10,11). Mesenchymal stem cells (MSCs) are adult stem cells typically found in the bone marrow and in other tissues including umbilical cord (UC), fetal liver, and adipose tissue. MSCs are considered to be multipotent and have anti-inflammatory as well as regenerative properties (3,5,10). MSCs derived from the human umbilical cord (hUC-MSCs) are more primitive and possess multiple advantages including ethical agreeableness, a less-invasive procedure for isolation, low immunogenicity, high proliferation capacity, and multilineage differentiation capability (13,15). Several studies have used hUC-MSCs to modulate immune responses in autoimmune diseases including rheumatoid arthritis, type 1 diabetes, and encephalomyelitis (8,13,14). Therefore, it is reasonable to hypothesize that hUC-MSCs may have a significant therapeutic effect on MS.
We reported a study of 23 MS patients treated in our hospital from January 2010 to December 2012. Thirteen patients received anti-inflammatory and immunosuppressive reagents combined with hUC-MSC therapy, while the other 10 only received anti-inflammatory and immunosuppressive therapy. The efficacy and toxicity of hUC-MSC therapy in MS patients were evaluated, and its possible mechanisms were explored.
Materials and Methods
Subjects
The study was approved by the Ethics Committees of the 148th Hospital, and written informed consent was obtained from both hUC-MSC donors and MS patients. A randomized, two-armed study was designed to assess the effect of hUC-MSC therapy in addition to the conventional anti-inflammatory and immunosuppressive remedy. The inclusion criteria were set as 1) RRMS or SPMS with an average Expanded Disability Status Scale (EDSS) of 5.0 (range 4.0–8.0) at the time of enrollment and clinical deterioration at 1.0 point or higher on EDSS over the past 12 months; 2) males or nonpregnant females with an age range from 25 to 55; 3) had two or more attacks in the last 2 years with a disease course of more than 2 years; 4) characteristic abnormalities on magnetic resonance imaging (MRI) scan. Patients were excluded from the study if they 1) were treated with cytotoxic medications (i.e., cyclophosphamide, mitoxantrone, and azathioprine) in the past 3 months before the trial; 2) had significant cardiac, renal, or hepatic failure or any other severe diseases that may interfere with result interpretation; 3) had an active infection; 4) showed severe cognitive decline; and 5) were unable to understand and sign the informed consent. The patients did not pay for their inclusion or treatment in this study.
hUC-MSC Isolation, Expansion, and Characterization
hUC-MSCs were isolated from human UCs and cultured as previously described (13). Fresh, smooth, flat, relatively thin, hUCs of uniform thickness were obtained from healthy donors (both genders) in our hospital after normal deliveries and were processed immediately. The UCs were washed by phosphate-buffered saline (PBS; Zhongshan Golden Bridge Biotechnology Co., Ltd., Beijing, China), and blood vessels were removed. The remaining tissue was sectioned to approximately 2-mm pieces and transferred to a 25-cm2 tissue culture flask (Dongge Biotechnology Co., LTD, Beijing, China) containing Ham's F12 media (Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (Biochrom AG, Berlin, Germany). Tissue was incubated at 37°C with 5% CO2 in a humidified chamber. The cultured cells were harvested within two to three passages and assessed for lineage markers via flow cytometry. Briefly, cells were fixed in neutralized 2% paraformaldehyde (PFA; Dongge Biotechnology Co., Ltd.) solution for 30 min. Fixed cells were washed twice with PBS and incubated for 30 min with fluorescein isothiocyanate (FITC) or phycoerythrin (PE)-conjugated antibodies against cluster of differentiation 44 (CD44), CD29, CD105, CD31, CD45, and human leukocyte antigen (HLA)-DR (Abcam, Cambridge, UK; antibody concentration in accordance with the instructions) before being analyzed by flow cytometry (BD, Franklin Lakes, NJ, USA). Microorganisms or endotoxin in hUC-MSCs were tested in the clinical laboratory of our hospital. In order to detect whether there was bacterial contamination, the hUC-MSC medium was cultured overnight on nutrient agar plates (Haibo Biological Technology Co. Ltd., Shanghai, China). The chromogenic end-point tachypleus amebocyte lysate (CE-TAL) assay (Ruicheng Biotechnology Co., Ltd, Shanghai, China) was used to detect cytotoxins.
Treatment
Baseline data were collected from each patient prior to any treatment, including serological samples. At acute relapsing phase, all patients were given anti-inflammatory and immunomodulation reagent methylprednisolone (Jin-yao Pharmaceutical Co., Ltd, Tianjin, China) intravenously 1,000 mg/kg daily for 3 days, 500 mg/kg for 2 days, followed by oral prednisone (Xian Ju Pharmaceutical Co., Ltd, Zhejiang, China) 1 mg/kg/day for 10 days. Subsequent dosage of prednisone was reduced by 5 mg every 2 weeks until reaching a 5-mg/day maintenance dosage. Serum γ-globulin (KangBao Biological Products Co., Ltd, Shanxi, China) was simultaneously supplemented at 400 mg/kg/day for 5 days. Thirteen patients were randomly selected for hUC-MSC therapy (intravenous infusion, hUC-MSCs suspended in 100 ml normal saline) at a dosage of 4 × 106 cells/kg once every 2 weeks for three consecutive times. The control group did not receive a saline infusion. All patients continued the maintenance dosage of anti-inflammatory and immunosuppressive therapy during the follow-up stage. Safety surveillance included physical examination, adverse event documentation, and quarterly brain MRI (Siemens, Munich, Germany).
Endpoint Evaluation
The EDSS was used to assess the status of neurological function. The scores range from 0 to 10, representing normal to death, respectively (2). Patient scores ranged from 5 to 8 at enrollment, indicating impairment of daily activities and degrees of disability ranging from inability to walk longer than 200 m to restriction to a wheelchair. Scheduled clinical evaluations were performed before hUC-MSC treatment (baseline) and at 1, 2, 3, 6, 9, and 12 months after treatment. Two neurologists who had no knowledge of the study evaluated every case independently to minimize bias.
Detection of Cytokines with ELISA
Serum of every patient at enrollment and 1, 2, 3, 6, 9, and 12 months posttreatment was collected. Cytokines hepatocyte growth factor (HGF), interleukin-4 (IL-4), IL-10, interferon-γ (IFN-γ), IL-17, and tumor necrosis factor-α (TNF-α) were detected with ELISA kits (R&D System, Minneapolis, MN, USA) according to the manufacturer's instructions.
Statistical Analysis
Data analysis was performed by SPSS v19.0 software (IBM, Armonk, NY, USA). Normality of the data was confirmed by Kolmogorov–Smirnov and Shapiro–Wilk tests. In order to compare data at different time points, chi-square test and analysis of covariance were applied. Mann–Whitney and Wilcoxon tests were used when a nonnormal distribution of data was determined. Statistical significance in serum targets between the control and experimental group at every time point were assessed by Student's t test. The relapse occurrences of patients in both control and experimental group in a 1-year period were assessed by the Student's t test (and confirmed by Mann–Whitney test). The data of ELISA results and relapse occurrences were expressed as mean ± standard deviation (SD). It was considered to be statistically significant when the value of p < 0.05.
Results
Patient Characteristics
All patients were enrolled into the study from January 2010 to December 2012 in our hospital. The experimental group included 13 patients (four males and nine females), with an average age of 41.7 ± 5.6 years, and had either RRMS or SPMS with mean disease duration of 2.90 ± 0.9 years and EDSS score of 6.98 ± 1.2 (range 4.0–8.0). The control group included 10 patients (three males and seven females), with average age of 39.4 ± 3.8 years. The control patients had either RRMS or SPMS with mean disease duration of 2.57 ± 0.8 years and EDSS score of 6.02 ± 1.6 (range 4.0–8.0).
hUC-MSC Isolation
hUC-MSCs were cultured for two to three passages before harvesting (Fig 1A). The purity of the hUC-MSCs was examined by flow cytometry. They were positive for MSC marker CD105 (90.03%), integrin markers CD29 (94.20%) and CD44 (95.63%), but negative for endothelial cell marker CD31 (6.89%) and hematopoietic cell marker CD45 (5.07%) or lymphocyte surface markers HLA-DR (0.33%) (Fig 1B). After a stringent quality-control procedure, the hUC-MSCs used for infusion were prepared to a final concentration of 2 × 108/ml cells (Table 1).

Human umbilical cord-derived mesenchymal stem cells (hUC-MSCs) and evaluation of their membrane proteins by flow cytometry with CellQuest software. (A) Morphology of hUC-MSCs in culture. Scale bar: 100 μm. (B): Evaluation of hUC-MSC membrane proteins by flow cytometry with CellQuest software. Acquisitions are shown in histograms of flow cytometry; the number of acquired events was 20,000 in each acquisition. The hUC-MSCs were positive for CD29 (94.20 ± 1.56%), CD44 (95.63 ± 1.23%), and CD105 (90.03 ± 0.64%), and were negative for CD31 (6.89 ± 0.09%), CD45 (5.07 ± 0.068%), and HLA-DR (0.33 ± 0.04%). IgG1-FITC and IgG1-PE antibodies were utilized as isotype controls. FSC, forward scatter; SSC, side scatter.
Safety Tests of hUC-MSCs

Clinical Assessment of Neurological Function
First, the normality of EDSS values at each time point and age was analyzed by SPSS software, and the results showed that only the EDSS value before treatment for the control group was not normally distributed (Table 2). The three factors of gender, type, and incidences of relapse were analyzed by chi-square test in turn. As shown in Tables 3 and 4, the value of p (0.968) suggested that gender and type has no effect on the experimental group (patients treated with hUC-MSCs) and control group (the patients who only received anti-inflammatory and immunosuppression treatment). The recurrence frequency was significantly different between the two groups (p < 0.038) (Table 5). The patients treated with hUC-MSCs (experimental group) had a more steady disease course with fewer incidences of relapse than the patients who received anti-inflammatory and immunosuppression treatment only (control group) (Fig. 2).
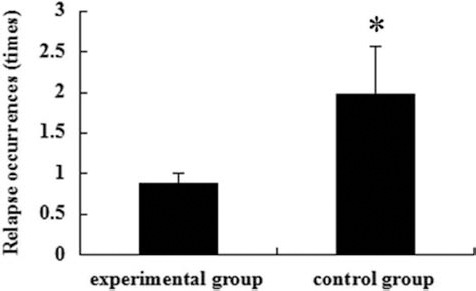
Average relapse incidence of MS patients. Patients in both control and experimental group were followed up in a 1-year period for the relapse occurrence. Data were expressed as mean value of each group. *p < 0.05.
Tests of Normality

Group 1, control (anti-inflammatory and immunosuppressive therapy only); Group 2, experimental (hUC-MSCs).
Lilliefors Significance Correction.
This is the lower boundary of the true significance.
Group × Gender Crosstab

Two cells (50.0%) have expected count less than 5. The minimum expected count is 3.04.
Computed only for a 2 × 2 table.
Group × Type Crosstab
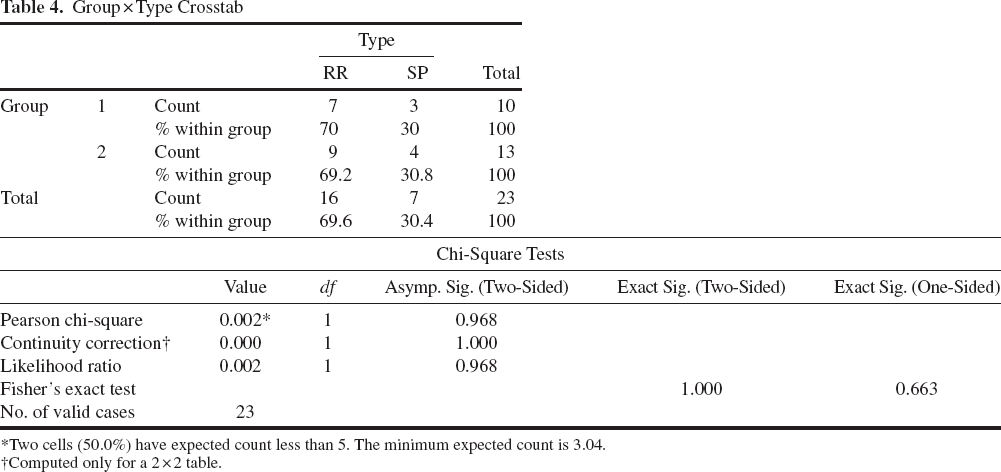
Two cells (50.0%) have expected count less than 5. The minimum expected count is 3.04.
Computed only for a 2 × 2 table.
Group × Relapse Incidence Crosstab

10 cells (100.0%) have expected count less than 5. The minimum expected count is 0.43.
Since EDSS values before treatment were nonparametrically distributed, they were also analyzed by Mann–Whitney test, as well as the relapse incidence. No significant difference was observed in the control and treated groups before treatment values (Table 6). In the present study, the collected sample size was very small, meaning that several of the statistical lattice values were 0, so it is not possible to test the impact of disease type on the EDSS at the same time. Therefore, the type and frequency of recurrence was not suitable for analysis of covariance. In this study, age, gender, and the EDSS in two groups can be carried out by analysis of covariance. The results showed that there were obvious differences between the two groups from the third point in time, that is to say EDSS values of the second month after the treatment (p < 0.05) (Table 7). Age and gender did not affect the EDSS values of the two groups (data not shown). Figure 3 confirms this, demonstrating that the EDSS values of the experimental group were lower than those of the control group over the last five time points. Both treatment groups show an initial improvement (decrease in EDSS values), but over time, the EDSS values increased again, but a significant difference between the control and treated groups remained. Therefore, we can believe that the effect of stem cells combined with conventional hormone therapy is significantly better than hormone therapy alone on MS treatment.

The profile plots of the univariate analysis of variance for the EDSS. In the profile (estimated marginal mean EDSS + SD), there was a clear difference between the two groups from the third time point (the second month after treatment; *p < 0.05). Key to time points: 1: before treatment, 2: first month after treatment, 3: second month after treatment, 4: third month after treatment, 5: sixth month after treatment, 6: ninth month after treatment, 7: 12th month after treatment.
Mann–Whitney Test

Not corrected for ties.
Grouping variable: Group.
The p Values of the Univariate Analysis of Variance Between the Control Group (Conventional) and the Experimental (hUC-MSC) Group

Data was not normally distributed, and so comparison was by Wilcoxon test. n.s., not significant (p > 0.05).
The Cytokine Expression
The serum HGF levels of patients who received hUC-MSC transfusion gradually increased in months 1, 2, and 3 posttreatment, and then slowly returned to the pretreatment levels, whereas the serum HGF levels in the control group did not significantly change throughout the monitoring period. The differences of serum HGF levels between the two groups were statistically significant at 1, 2, and 3 months after treatment (p < 0.05), but not at the later time points (Fig. 4). Similarly, we assessed the serum levels of T helper 1 (Th1)/Th2/Th17 cytokines, including IL-4, IL-10, IFN-γ, TNF-α, and IL-17 in patients of both groups (Fig. 4). The serum levels of the Th2 cytokines IL-4 and IL-10 increased significantly over the first 3 months after treatment in patients in the experimental group compared to those in the control group (p < 0.05). The serum levels of Th1/Th17 cytokines TNF-α and IL-17, however, significantly decreased in the first 3 months after treatment in the patients of the experimental group compared with those in the control group (*p < 0.05).

Serum levels of cytokines HGF, IL-4, IL-10, IFN-γ, IL-17, and TNF-a. Sera were collected before (0) and after (month 1, 2, 3, 6, 9, 12) stem cell transplantation, and cytokines were measured with enzyme-linked immunosorbent assay (ELISA). All assays were repeated three times, and the data were expressed as mean ± SD. Statistical significance in serum targets between experimental and control groups were assessed by the Student's t test. *p < 0.05 and **p < 0.01 when compared with the control. HGF, hepatocyte growth factor; IL, interleukin; IFN, interferon; TNF, tumor necrosis factor.
Interestingly, IFN-γ, an important Th1 cytokine, followed the Th2 cytokine trend in that its level increased in patients of the experimental group, but not the control group. All of the differences in cytokine levels in the early time of treatment had disappeared by 6 months after treatment.
Discussion
This investigation aimed at exploring the efficacy and safety of hUC-MSC therapy in MS patients. The study was designed to apply hUC-MSC therapy while patients were treated with traditional anti-inflammatory medicine. We did not set up a hUC-MSC treatment-only group because giving patients the best available treatment was our top priority, even during a clinical trial. For the same reason, we applied hUC-MSCs with traditional treatment. During a 1-year observation, no significant adverse effects were found, indicating the clinical safety could be well accepted. The significantly lower relapse occurrence and EDSS scores were found in the experimental group compared to the control group. Even though the EDSS scores at the end of the 1-year period were only slightly lower than that of the baseline in the experimental group, the ability of the treatment to apparently prevent, in the short term, the continuous deterioration of MS can be considered a relatively successful measurement.
Appropriate cell type, dose of cells, and the time of application of stem cell therapy are important for obtaining satisfactory outcomes. These factors might depend on the target mechanisms of the cell therapy (i.e., cell replacement vs. restorative effects via trophic support). We chose to use hUC-MSCs at a dose of 4 × 106 every 2 weeks for three doses based on our previous experiences as well as a search of the literature (4,25).
Efforts to develop cellular therapy in MS have long been directed toward implanting cells capable of replacing lost oligodendrocytes and regenerating myelin sheaths. With the increasing understanding of the complexity of MS, cellular therapy development has been increasingly focused on achieving far more than simply offering a source of replacement myelin-forming cells (22). Inflammation of the CNS is a prominent feature of MS (12,20). It is most notable in acute lesions that inflammation plays a role in the breakdown of the blood–brain barrier, migration of immunocytes into the CNS, production of proinflammatory cytokines, and neuronal damage (7). A previous study verified that hUC-MSCs can cross the blood–brain barrier via intravenous transplantation (16). They not only replaced diseased tissues, but also exerted several trophic, regenerative, and anti-inflammatory effects (3,5,10,17). Human MSCs significantly inhibited autoimmune progression (26). Growing evidence indicates that MSCs have immunomodulatory effects and enhance the shift from a Th1 to Th2 phenotype in vitro and in vivo (9,24). As a Th1-mediated immune system disorder, MS may benefit from such a shift (18,23).
To confirm that the shift from Th1 to Th2 plays a role in the stem cell treatment, we assessed the cytokines that represent either Th1 or Th2 immunity. Our data showed that anti-inflammatory Th2 cytokines (IL-4 and IL-10) had remarkable increases in the experimental group. These increases were accompanied by significant decreases of pro-inflammatory Th1 cytokines (IL-17 and TNF-α). The alterations of the cytokines concomitant with the change of EDSS scores indicated that the shift from Th1 toward Th2 phenotype had a positive correlation with disease amelioration. This shift was obviously not related to the traditional anti-inflammatory treatment, since it was not as significant in the control group. Through literature search, we found one contradictory result: that no Th1 to Th2 shift was observed in an earlier study (18). One of the possible reasons might be that a much higher dose of hUC-MSCs was adopted in this study, and the different administration route may also play a certain role.
Our data demonstrated that there was also an increase in HGF and IFN-γ, which may also have played a role in the ability of hUC-MSC therapy to improve MS. HGF is a multifunctional cytokine that acts primarily on cells of epithelial origin. Its ability in stimulating mitogenesis, cell motility, and matrix invasion makes it critical for tissue regeneration. A recent study suggested that HGF was also an important neurotrophic factor (3,21). IFN-γ, a major Th1 cytokine, was also elevated in the experimental group. It might be possible that the immune modulation effect of hUC-MSCs on the immune system may activate other immune cells, such as natural killer (NK) cells and macrophages, thus increasing secretion of IFN-γ. In short, the increase of both HGF and IFN-γ suggested a strong immunomodulation effect of hUC-MSCs in MS patients.
The therapeutic efficacy of hUC-MSCs on MS has been investigated in several studies (1,18,24), but none of these studies have presented convincing data and a protocol for future clinical application. Our study contributed, in part, to providing more evidence for the potential of hUC-MSCs as a therapy for MS.
Footnotes
Acknowledgments
This project was supported, in part, by the Chinese National Science Foundation (No. 30500208) and IAEA Research Project (No. CPR-13305). The authors declare no conflicts of interest.
