Abstract
Possible strategies for treating stroke include neuroprotection in the acute phase of cerebral ischemia and stem cell therapy in the chronic phase of cerebral ischemia. Previously, we have studied the temporal and spatial expression patterns of c-fos, hypoxia inducible factor-1α (HIF-1α), heat shock protein 70 (HSP70), and annexin V after 90 min of transient middle cerebral occlusion in rats and concluded that there is a time window for neuroprotection from 12 to 48 h after ischemia. In addition, we have estimated the neuroprotective effect of glial cell line-derived neurotrophic factor (GDNF) by injecting Sendai viral vector containing the GDNF gene into the postischemic brain. This Sendai virus-mediated gene transfer of GDNF showed a significant neuroprotective effect in the ischemic brain. Additionally, we have administered GDNF and hepatocyte growth factor (HGF) protein into the postischemic rat brain and estimated the infarct size and antiapoptotic and antiautophagic effects. GDNF and HGF significantly reduced infarct size, the number of microtubule-associated protein 1 light chain 3 (LC3)-positive cells, and the number of terminal deoxynucleotidyl transferase-mediated dUTP-biotin in situ nick-end labeling (TUNEL)-positive cells, indicating that GDNF and HGF were greatly associated with not only the antiapoptotic effect but also the antiautophagic effects. Finally, we have previously transplanted undifferentiated iPSCs into the ipsilateral striatum and cortex at 24 h after cerebral ischemia. Histological analysis was performed at 14 and 28 days after cell transplantation, and we found that iPSCs could supply a great number of doublecortin-positive neuroblasts but also formed tridermal teratoma in the ischemic brain. Our results suggest that iPSCs have a potential to provide neural cells after ischemic brain injury if tumorigenesis is properly controlled. In the future, we will combine these strategies to develop more effective therapies for the treatment of strokes.
Keywords
Introduction
Stroke is the second leading cause of death in the world and results in a drastic reduction in the quality of life (30). However, an effective therapy is not yet available. Both a better understanding of the mechanisms of ischemic brain damage and a new strategy for patients who have suffered a stroke are urgently required. Multiple molecular penumbra has been proposed, and this attracts the attention as the new concept for understanding the mechanisms of ischemic brain. Possible strategies for treating ischemic strokes are broadly categorized into two groups: (i) neuroprotection, which prevents damaged neurons from death in the acute phase of cerebral ischemia, and (ii) stem cell therapy, which can theoretically repair broken neuronal networks with newly born neurons/glias in the chronic phase of cerebral ischemia (Fig. 1). In this review article, we focus on our findings with multiple molecular penumbra, neurotrophic factors for neuroprotection, and the transplantation of induced pluripotent stem cells (iPSCs) for stem cell therapy.
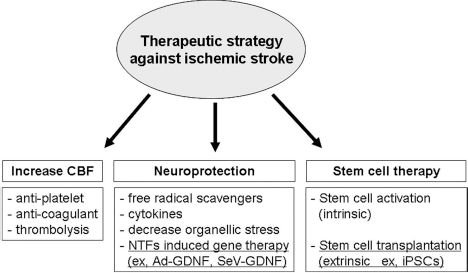
Therapeutic strategy for ischemic stroke. Possible therapeutic strategies for treating ischemic stroke include neuroprotection in the acute phase of cerebral ischemia, and stem cell therapy in the chronic phase of cerebral ischemia. CBF, cerebral blood flow; NTFs, neurotrophic factors; ad-GDNF, adenoviral vector containing glial cell line-derived neurotrophic factor; SeV-GDNF, Sendai virus vector containing glial cell line-derived neurotrophic factor.
Multiple Molecular Penumbra
The penumbra is regarded as an area of brain tissue that is damaged but not yet dead after focal ischemia. This concept was first proposed by Astrup et al. (4). They reported that measurement of electrical activity revealed regions that were dysfunctional but not yet dead after the induction of focal ischemia in a nonhuman primate brain (4). Additional definitions for the ischemic penumbra have been proposed from a metabolic viewpoint (10). More recently, the ischemic penumbra has been defined by characterizing its multiple gene/protein expression. Multiple molecular expression patterns have been reported to be involved with the mechanisms of ischemic brain damage (26); the concept of multiple molecular penumbra may therefore allow for the development of novel treatment strategies. The c-fos gene is an immediate early gene, and it is induced by cerebral ischemia (7). Hypoxia inducible factor-1α (HIF-1α) is a transcription factor induced by hypoxia. HIF-1α is known to play an important role in the promotion of anaerobic metabolism and angiogenesis (24). The heat shock protein 70 (HSP70) is a major molecular chaperone, which can be induced by hypoxia, cerebral ischemia, and heat shock. HSP70 can prevent intracellular aggregation of denatured proteins, supporting the recovery of brain cells (2). These proteins are known to be important components of molecular penumbra, but the temporal and spatial expression were largely unknown. We have previously demonstrated temporal and spatial changes in the molecular penumbra proteins that occur 1, 3, 12, 24, and 48 h after 90 min of transient middle cerebral artery occlusion (tMCAO) in rats. Immunohistochemistry and Western blot analysis showed that c-fos expressed quickly and widely within and outside of the infracted lesion (peak at 1 h). HIF-1α levels reached a maximum at 12 h, and HSP70 peaked at 24 h. On the other hand, an apoptotic marker, annexin V, was only slightly expressed at 3 h and gradually increased until 48 h. These results are consistent with the previous findings of other researchers (6,16), suggesting that there is a therapeutic time window for ischemic neural cells from 12 to 48 h after ischemic injury (34).
Neuroprotection with a Novel Viral Vector
Glial cell line-derived neurotrophic factor (GDNF) is a member of the transforming growth factor-β superfamily and has potent neuroprotective effects in ischemic brain injury (1). GDNF could be one of the promising candidates for therapy of acute cerebrovascular injury, but GDNF protein is difficult to administer in the clinical setting because of its inability to cross the blood–brain barrier and its short half-life of bioavailability. On those limitations, the gene therapy approach has great advantages in that an amount of protein could be produced continuously by a cell following insertion of a viral vector. We have reported that treatment with an adenoviral vector containing the GDNF gene could reduce the infarct volume in rat brain (13). However, the adenoviral vector has also been reported to induce inflammation and cytotoxicity (15); we therefore have also used a novel gene transfer vector derived from Sendai virus (SeV). SeV is classified as a type 1 parainfluenza virus belonging to the family paramyxoviridae with a negative-strand RNA genome (19). SeV has a great advantage in that it is nonpathogenic to humans because its genomic RNA is restricted to the cytoplasm without any risk of chromosomal interaction. In our previous studies, we have used the 90 min of tMCAO model in rats. The SeV-containing GDNF gene was directly injected into the cerebral cortex at 0 or 1 h after the reperfusion, and histological analysis was performed at 24 h after ischemia. We found that the SeV vector-mediated gene transfer of GDNF significantly reduced the number of terminal deoxynucleotidyl transferase-mediated dUTP-biotin in situ nick-end labeling (TUNEL)-positive cells as well as ischemic infarct volume. Another research group has also reported that administration of not only SeV-containing GDNF gene but also a SeV-containing NGF gene 30 min after the ischemic insult effectively prevented the delayed neuronal death of the hippocampal CA1 pyramidal neurons (27). These results indicated the high therapeutic potency of SeV/neurotrophic factors for cerebral ischemia (11).
Antiapoptotic and Antiautophagic Effect of Neurotrophic Factors
In recent years, new types of cell death have been described, and three types of cell death have currently been distinguished mainly by morphological criteria. Type I cell death is better known as apoptosis, and autophagy with the presence of autophagic vacuoles inside the dying cell are typical for type II cell death. Type III cell death is better known as necrosis (8). This autophagy is a regulated process of degradation and recycling of cellular constituents, participating in organelle turnover and in the bioenergetic management of starvation (14). Recent studies indicate the existence of autophagy in cerebral ischemia, and autophagy may be an important modulator of cell death and survival, interacting with necrosis and apoptosis (3). Hepatocyte growth factor (HGF) is a multi-functional growth factor identified as a potent mitogen for hepatocytes in primary culture (20,23). HGF binds and activates a tyrosine kinase receptor encoded by the c-Met proto-oncogene (5). HGF plays important roles in mitogenesis, motogenesis, morphogenesis, antiapoptosis, and angiogenesis (17,31). Both HGF and GDNF have previously been reported to be not only antiapoptotic but also antiautophagic in primary culture (22,33,35). However, the antiautophagic effect of HGF and GDNF in the ischemic brain was unknown, and so we have examined the antiapoptotic and antiautophagic effects of HGF and GDNF after 90 min of tMCAO in rats. Just after restoration of cerebral blood flow (CBF), HGF or GDNF was administered into Wister rats by placing a sponge containing HGF or GDNF protein in contact with the surface of the cerebral cortex, and the animals were sacrificed 24 h after the restoration of CBF. We found that GDNF and HGF evidently reduced infarct size (25). Western blot analysis of microtubule-associated protein 1 light chain 3 (LC3), which is a marker of autophagy, showed that cerebral ischemia increased LC3 expression, but GDNF and HGF significantly decreased it. We also performed double immunofluorescence analysis of LC3/TUNEL staining. The number of LC3/TUNEL double-positive cells significantly decreased in the HGF- and GDNF-treated groups. These results suggested that the neuroprotective effect of GDNF and HGF were associated with not only the antiapoptotic, but also the antiautophagic effect (25).
Stem Cell Therapy with iPSCs
Murine induced pluripotent stem cells (iPSCs) were first established by Takahashi and Yamanaka by introducing four transcriptional factors [c-Myc, octamer-binding transcription factor 3/4 (Oct3/4), sex-determining region Y box 2 (Sox2), and Krüppel-like factor 4 (Klf4)] into mouse fibroblasts (28). The iPSCs are regarded as a promising cell source for cell transplantation therapy to supply new neurons into the infarcted brain, because iPSCs retain a high replication competence and pluripotency to differentiate into various kinds of cells similar to embryonic stem cells (ESCs) (24). iPSCs can be produced from a patient's skin; therefore, iPSCs are not expected to have any immunoreactive or ethical problems like those found with ESCs. We have previously transplanted murine iPSCs into the ischemic mouse brain and investigated the survival and differentiation of transplanted iPSCs in the ischemic brain environment. Undifferentiated iPSCs (5 × 105) were transplanted into the ipsilateral striatum and cortex 24 h after cerebral ischemia, and histological analysis was performed 14 and 28 days after cell transplantation. We found that the iPSCs supplied a great number of doublecortin (DCX)-positive neuroblasts in the ischemic region (Fig. 2). However, the transplanted iPSCs expanded and formed tridermal teratomas in the ischemic brain (12). On the other hand, recent reports have shown that differentiated neuronal cells derived from human iPSCs could contribute functional recovery without tumor formation in stroke animal models (9,18,21,29) (Table 1). These results indicated that iPSCs have a promising potential to supply neuronal cells for postischemic brain, if tumorigenesis is properly controlled.

Induced pluripotent stem cells (iPSCs) have a potential to produce neuroblasts in postischemic brain. Anti-doublecortin (DCX), a neuroblast marker, staining of the infarcted lesion from the middle cerebral artery occlusion (MCAO) + PBS group (A, B), MCAO + iPSC group (C, D) at 14 days after cell transplantation. The boxed areas in (A) and (C) indicate the fields shown in (B) and (D), respectively. DCX-positive cells could be detected in the tumors of ischemic brain (C, D). Scale bars: 2 mm (A, C) and 50 μm (B, D).
Scientific Reports Showing Induced Pluripotent Stem Cell (iPSC) Transplantation Therapy in a Transient Middle Cerebral Artery Occlusion (tMCAO) Animal Model of Stroke

In this review, we briefly highlighted our recent progress in the development of distinct new strategies for the treatment of damaged brains following a stroke. To realize more effective therapies for patients suffering from stroke, it is important to combine these strategies in the acute or chronic phase following a stroke.
Footnotes
Acknowledgments
This work was partly supported by Grant-in-Aid for Scientific Research (B) 25293202, (C) 15K09316, and Challenging Research 15K15527 and Young Research 15K21181, and by Grants-in-Aid from the Research Committees (H. Mizusawa, K. Nakashima, M. Nishizawa, H. Sasaki, and M. Aoki) from the Ministry of Health, Labour and Welfare of Japan. The authors declare no conflicts of interest.
