Abstract
Transplantation of human hepatocytes is gaining recognition as a bridge or an alternative to orthotopic liver transplantation for patients with acute liver failure and genetic defects. Since most patients require multiple cell infusions over an extended period of time, we investigated hepatic functions in cells maintained in University of Wisconsin solution at 4°C up to 72 h. Eleven different assessments of hepatic viability and function were investigated both pre- and posthypothermic storage, including plating efficiency, caspase-3/7 activity, ammonia metabolism, and drug-metabolizing capacity of isolated hepatocytes. Long-term function, basal, and induced cytochrome P450 activities were measured after exposure to prototypical inducing agents. Cells from 47 different human liver specimens were analyzed. Viability significantly decreased in cells cold stored in UW solution, while apoptosis level and plating efficiency were not significantly different from fresh cells. Luminescent and fluorescent methods assessed phases I and II activities both pre- and post-24-72 h of cold preservation. A robust induction (up to 200-fold) of phase I enzymes was observed in cultured cells. Phase II and ammonia metabolism remained stable during hypothermic storage, although the inductive effect of culture on each metabolic activity was eventually lost. Using techniques that characterize 11 measurements of hepatic viability and function from plating efficiency, to ammonia metabolism, to phases I and II drug metabolism, it was determined that while viability decreased, the remaining viable cells in cold-stored suspensions retained critical hepatic functions for up to 48 h at levels not significantly different from those observed in freshly isolated cells.
Introduction
Hepatocyte transplantation (HTx) is gaining acceptance as an alternative therapy, in particular for patients with inherited metabolic liver diseases. These diseases are characterized by lack of one particular enzyme or protein, giving rise to hepatic and/or extrahepatic disease. As the liver has a high redundancy in function, replacement of 5-20% of the liver with healthy donor hepatocytes could correct a wide range of inherited metabolic liver diseases (8). However, due to the risk of portal thrombosis, only 5% of the liver mass can be infused at one transplant session (9). In current clinical protocols, hepatocytes are transplanted by intraportal infusion over a period that may last up to 48 h (9,15,19). As a result, hepatocytes need to be cold stored between repeated infusions (9). The deterioration of the integrity and function of hepatocytes in suspension after a few hours of incubation at 37°C is well recognized, while the beneficial effect of the hypothermic preservation of hepatocytes in culture media (i.e., Leibowitz or Williams E solutions) (7,20,26,30) or University of Wisconsin (UW), histidine—tryptophan—ketoglutarate (HTK), and Celsior solutions has only been partially investigated (11,14,18,23,30).
Although hypothermia has a protective effect due to lowering metabolic functions, cold ischemic damage is evident upon rewarming (3,14). Belzer's or UW solution was originally developed for organ/tissue preservation and is currently the preferred solution for cold storage of isolated hepatocytes (4) and has been used for longdistance shipments of hepatocytes between the isolation center and transplantation sites (9). This solution has been adapted to the intracellular ion composition with a high buffering capacity and contains impermeable agents to help maintain osmotic gradients through cold storage (4). Because of these properties UW is the solution included for cold storage of hepatocytes in the clinical application submitted from our institutions to the regulatory authorities. It is known that the time span for preservation for whole organs is limited to several hours. The time limit for cold preservation of isolated hepatocytes is not known.
There are a number of investigations of the effect of cold ischemia—warm reperfusion injury and the underlying mechanisms involved in hepatocyte cell death (16,22,28) but mainly for experimental purposes. How the duration of cold storage of hepatocytes affects the quality of cells for clinical cell infusion has not been systematically investigated.
In order to satisfy regulatory requirements for clinical transplants, one has to demonstrate that a specific step in the process such as cold storage of cells prior to transplantation still results in cells with adequate function to be useful for transplantation. In this study, we provide data on hepatic function from short-term and longer term experiments from cells cold stored in UW solution for up to 72 h, from up to 47 human hepatocyte isolations. The viability and function of cold-stored hepatocytes were analyzed using the same procedures employed at our center to assess the suitability of hepatocytes for clinical transplants. The results indicate that critical hepatic functions are well maintained in hepatocytes cold stored in UW solution for up to 48 h. The data obtained from these studies satisfy the regulatory requirement to demonstrate function in the cold-stored cells and provides a scientific basis to support the use of hepatocytes for clinical transplants for up to 48 h postisolation.
Materials and Methods
Hepatocyte Isolation
All tissues were collected with informed consent (IRB protocol 0411142, University of Pittsburgh, PA, USA). Hepatocytes were isolated from 47 different donors (Table 1): 19 samples were obtained from organ donors rejected for orthotopic liver transplantation (OLT) for reasons including prolonged warm/cold ischemia time, drug overdose, anoxia, steatosis, and fibrosis; and 28 specimens were derived from patients undergoing scheduled liver resection procedures performed for a number of different forms of neoplastic disease or undergoing OLT for genetic inborn errors. Whole donor organs not used for OLT were flushed in situ with cold preservation solution and maintained on wet ice and received for cell isolation 6-36 h post-cross-clamp. Residual liver tissue not needed for diagnostic purposes was transported to the laboratory from the operating rooms in ice cold UW (Viaspan; Bristol-Myers Squibb, New York, NY, USA) or Eagle's minimum essential medium (Lonza, Walkersville, MD, USA), respectively and perfused within 90 min of removal.
Characteristics of the Liver Tissues Investigated
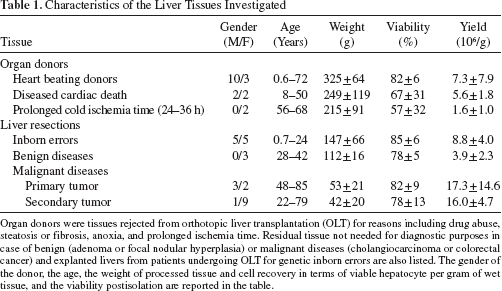
Organ donors were tissues rejected from orthotopic liver transplantation (OLT) for reasons including drug abuse, steatosis or fibrosis, anoxia, and prolonged ischemia time. Residual tissue not needed for diagnostic purposes in case of benign (adenoma or focal nodular hyperplasia) or malignant diseases (cholangiocarcinoma or colorectal cancer) and explanted livers from patients undergoing OLT for genetic inborn errors are also listed. The gender of the donor, the age, the weight of processed tissue and cell recovery in terms of viable hepatocyte per gram of wet tissue, and the viability postisolation are reported in the table.
The tissue dissociation and subsequent hepatocyte isolation procedures were performed as previously described (12). Cell viability, assessed by trypan blue (Sigma-Aldrich, St. Louis, MO, USA) exclusion method, and plating efficiency were performed as previously described (12).
For the purpose of this study, cells were analyzed immediately after isolation and after 24, 48, and 72 h in UW solution (concentration of 2-4 × 106 viable cells/ml) at 4oC. Every 24 h, an aliquot of cells was diluted 1:3 with cold Eagle's minimum essential medium and centrifuged at 80 × g for 5 min and resuspended in plating medium, as previously described (12).
Caspase-Glo™ Assays
Caspase-3 and caspase-7 were measured with a luminescent assay (Caspase-Glo® 3/7; Promega Corporation, Madison, WI, USA) according to the manufacturer's instructions with some minor changes as described here. Briefly, 40 ml of the cell suspension was seeded in a 96-well white plate (Thermo Scientific, Waltham, MA, USA) and allowed to equilibrate for 30 min at 37°C. An equal volume of Caspase-Glo™ reagent was then added directly to the well. After 30-min incubation at room temperature, luminescence was read directly on a white plate in a luminometer (Synergy HT, BioTek Instruments, Winooski, VT, USA) with an integration time of 1 s/well. Luminescence produced by the assay is proportional to the amount of caspase activity present in the cell sample. Results are expressed as luminescence counting units (LCU)/min and normalized to 1 million viable cells.
Drug Metabolism Studies
Cytochrome P450 (CYP) activities were measured immediately after washing, while the cells were in suspension and after plating and attachment. Methods to assess ammonia, testosterone, and 7-ethoxyresorufin metabolism [cytochrome p450 family 3 subfamily A polypeptide 4 (CYP3A4) and 1A1/2], resorufin conjugation, media, and culture conditions were as previously described including high-performance liquid chromatography on a Beckman System Gold HPLC using a Mono-Q anion exchange column (GE Healthcare, Piscataway, NJ, USA) to measure CYP3A4 activity (12). Additional cell-based assays for specific CYPs (2C9, 3A7, 3A4) were used according to manufacturer's instructions with minor modification. Briefly, 3 × 104 hepatocytes were added to a 96-well white plate and incubated for 30 min at 37°C with proluminogenic compounds for specific CYPs (CYP-Glo™, Promega Corporation). The luminogenic product was detected with a luciferin detection reagent, and after incubation for 20 min at room temperature, luminescence was read directly in a white plate in a luminometer (Synergy HT) with an integration time of 1 s/well. Attached cells were analyzed after 5 days in culture by exposing the cells in 96-well clear culture plates to different Glo™ substrates and transferring the suspension after lysing to a 96-well white plate and read with the same protocol. For the last 3 days in culture, specific CYP inducers were added to the media every 24 h: 25 mM β-naphthoflavone (BNF; Sigma-Aldrich) for cytochrome P450 family 1 subfamily A polypeptide 1/2 (CYP1A1/2), 10 mM rifampicin (rif; Sigma-Aldrich) for CYP 3A4, or 0.1% dimethyl sulfoxide (DMSO; Sigma-Aldrich) as vehicle control. The luminescence produced is proportional to CYP activity. Results are expressed as LCU/min and normalized to 1 million viable cells or to the double-stranded DNA (dsDNA) content.
dsDNA Amount Quantification
Double-stranded DNA (dsDNA) quantification was performed with a Quant-iT™ PicoGreen® dsDNA ultrasensitive fluorescent nucleic acid staining kit according to the manufacturer's instructions (Molecular Probes, Invitrogen Corporation, Camarillo, CA, USA). After the Glo™ assays were complete, each well was incubated with 100 μl Quant-iT™ PicoGreen® in Trisethylenediaminetetraacetic acid (EDTA) buffer, and the fluorescence intensity was read on a fluorescent spectrometer (Synergy HT) at an excitation wavelength of 488/15 nm and an emission wavelength of 528/20 nm. dsDNA concentration was quantified by interpolating the A528 values for the unknowns from a standard curve of λ DNA using the equation [dsDNA (mg/ml) = 0.1057 * A528 - 61.322; R2 = 0.9941].
Statistical Analysis
Experiments were performed in triplicates. Results are expressed as mean ± standard error of the mean (SEM). Unpaired t tests on pair group and analysis of variance (ANOVA) with Newman—Keuls multiple comparison tests have been used to determine statistical differences. A value of p < 0.05 was chosen as the minimum level of significance. Results are presented as histograms showing mean ± SEM. All data have been analyzed by GraphPad Prism software (version 5.03; San Diego, CA, USA).
Results
During the study period, 47 liver tissues were collected, and successful hepatocyte isolations were performed on all specimens. The study group was composed by patients undergoing liver resection due to primary or secondary malignancy (n = 18), liver transplantation due to metabolic liver diseases (n = 10), or organ donors rejected from organ transplant for different reasons (n = 19) (Table 1). The donors were equally distributed between females (47%) and males (53%); age ranged from 0.6 to 85 years (39 ± 4 years).
After isolation, hepatocytes were analyzed directly (fresh) or analyzed after 24, 48, and 72 h of cold storage in UW solution. Freshly isolated hepatocytes showed a mean viability of 81 ± 1%. A significant decrease in terms of viability was measured every 24 h of cold storage: 61 ± 2% at 24 h, 52 ± 3% at 48 h, and 48 ± 3% at 72 h (p < 0.0001) (Fig. 1A). The cell recovery after cold storage ranged from 50% to 75% of the total viable cell number initially added to aliquots.

Human hepatocyte viability, apoptosis, and plating efficiency. Human hepatocyte viability (A), caspase activity (B), and plating efficiency (C) for freshly isolated hepatocytes and cells in University of Wisconsin (UW) solution after 24 h, 48 h, and 72 h at 4°C. Bars show mean and standard error. *p < 0.01; **p < 0.001; ***p < 0.0001. Representative phase contrast photographs for freshly isolated hepatocytes (D) and cells stored in UW solution for 24 h (E), 48 h (F), and 72 h (G). Photos taken on day 3 of culture. Scale bar: 50 mm. LCU, luminescence counting units.
The level of apoptosis, quantified by caspase-3 and -7 activities, was evaluated in 23 of the preparations. A trend toward increased caspase activity was observed with longer cold storage, but differences did not reach statistical significance (Fig. 1B).
The ability of hepatocytes to attach to a cell culture plate coated with extracellular matrix requires that viable cells express functional cell adhesion molecules and maintain sufficient adenosine triphosphate (ATP) levels to maintain adherence (29). All hepatocyte suspensions showed high plating efficiencies on collagen substrate post-cold storage, and again, there was no statistical difference in plating efficiency between the cold-stored and freshly isolated cells (Fig. 1C). Once attached to the substrate, all cellular preparations showed typical primary human hepatocyte morphological (polygonal shape, one or more nuclei, granular cytoplasm with small vesicular inclusions and bright sharp cell membranes surrounded, and canalicular structures) without any noticeable differences between pre- and posthypothermic storage (Fig. 1D). After 2 days of culture, all the cell preparations formed a confluent or semiconfluent monolayer on collagen-coated plates.
Cytochrome P450 Activities
Since transplanted cells would be expected to perform metabolic functions immediately posttransplantation, activities mediated by CYP 1A1/2, 2C9, 3A4, and 3A7 were analyzed. CYP1A1/2 showed stable activity during the first 48 h of cold preservation and a slight, but not significant, increase in activity after 72 h of cold preservation (p = 0.42) (Fig. 2A). Since the toxic effects of long-term cold storage might not be obvious from analysis conducted immediately after recovery from cold storage, we also investigated long-term hepatocyte viability and function by plating the cells on collagen and analyzing basal and induced levels of CYP activities. Basal ethoxyresorufin-O-deethylase (EROD) activity and the induction in response to exposure to the prototypical inducer (BNF) were investigated (Fig. 2B). Exposure to BNF for 3 days induced up to a 24-fold increase in EROD activity over control (p < 0.0001).

Cytochrome P450 activity of human hepatocytes. Cytochrome p450 family 1 subfamily A polypeptide 1/2 (CYP1A1/2) measured by the conversion of 7-ethoxyresorufin to resorufin (by ethoxyresorufin-O-deethylase; EROD) in cells in suspension immediately after isolation and after cold storage for 24 h, 48 h, and 72 h in UW solution (A) and after plating and 5 days in culture, including the final 3 days with exposure to β-naphthoflavone (BNF) or vehicle control [dimethyl sulfoxide (DMSO) only] (B). CYP3A4 activity on cells immediately after isolation or recovered from cold storage and measured in suspension (C) and after culture and a 3-day exposure to rifampicin (Rif) measured by luciferin-isopropyl acetal (IPA) (D). Measurement of CYP3A7 (fetal form) in cells in suspension or immediately postrecovery from cold storage by luciferin-6-pentafluoro-benzyl ether (PFBE) (E). CYP2C9 activity measured on cells in suspension by luciferin-H (6′-deoxyluciferin) (F). Bars show mean and standard error. *p < 0.01; ***p < 0.0001.
Activities mediated by CYP3A were maintained through cold preservation. When CYP3A4-mediated metabolism was assessed immediately after isolation, an activity equal to 16,739 ± 2,644 LCU/min/million of hepatocytes was present (Fig. 2C). After 24 h in UW, a slight but not significant increase in activity in the cells was measured (23,894 ± 8,045 LCU/min/million; p = 0.54). Activity remained stable and was not statistically different from freshly isolated cells in hepatocytes recovered after up to 48 h or 72 h of cold storage (Fig. 2D). A 3-day prior exposure to rifampicin induced significant increases in CYP3A activities in fresh, 24 h, and 48 h cold-stored cells. Cells stored for 72 h showed an increase in activity, although the data did not achieve statistical significance. The results were the same as those with the luminescent assay when 3A4 activity was measured with the traditional high-performance liquid chromatography (HPLC) method to detect the 6-β-hydroxy metabolite of testosterone (data not shown).
Activities mediated by CYP3A7 showed a profile similar to that observed with CYP3A4 (Fig. 2E). As expected, in cells obtained from patients younger than 3 years of age (n = 7), activity of the fetal 3A, 3A7, was prominent.
Luciferin-H (6′-deoxyluciferin) metabolism is specifically mediated by 2C9 in human hepatocytes. Although a trend toward decreasing 2C9 activity was noted, the results in cold-stored samples were not significantly different from fresh cells (Fig. 2F).
Phase II Activity
Conjugation of resorufin is a measure of phase II metabolic activity and was evaluated immediately after recovery in cold-stored cells and also after attachment with 5 days in culture. There was no statistical difference in resorufin conjugation between fresh and cold-preserved cells (Fig. 3A). Conjugation activities are generally higher in plated cells (hatched bars) than those measured in suspension (open bars). This inductive effect on conjugation activity was observed in cells from nearly all human cases. A significant increase in conjugation activity in cultured cells was observed maintained out to 48 h of cold storage (Fig. 3A). At 72 h of cold storage, there was a slight, but nonsignificant, increase in conjugation activity in plated cells.

Phase II activity and ammonia metabolism. Resorufin conjugation (A), as a measurement for phase II activity, and ammonia metabolism (B) were performed with hepatocytes in suspension (open bars) and with hepatocytes maintained in culture for 5 days (hatched bars). Bars represent mean and standard error. *p < 0.01; **p < 0.001; ***p < 0.0001.
Ammonia Metabolism
Like conjugation capacity, ammonia metabolism is generally greater in fresh cells or hepatocytes maintained in culture for several days compared to those in suspension. There was no difference in ammonia metabolism between fresh and cold-stored hepatocytes when measurements were made immediately after recovery (Fig. 3B, open bars). A progressive and significant decline in ammonia removal was observed in cells kept at 4°C past 24 h when they were plated and analyzed 5 days postplating (Fig. 3B, hatched bars).
Discussion
There are a large number of patients awaiting liver transplantation, since the availability of donor organs is limited. Hepatocyte transplantation is a promising therapy for a variety of indications (5,8,21,24). A typical protocol will require infusion of billions of hepatocytes. The total number of hepatocytes injected in a HTx event is calculated to be in the range of 1-2 × 108 per kg body weight (6,10). Owing to the risk of portal thrombosis (2) from a single infusion of a large number of cells, the target cell number is frequently split into three to five separate infusions over 24-36 h (9,10). As a result, isolated hepatocytes need to be stored from several hours to several days prior to transplantation (9,27). During that time, isolated hepatocytes are usually cold preserved (2-8°C) until needed. Moreover, currently revised clinical protocols now include pretreatment of the recipient with local radiation (10) or partial liver resection (21). Since preconditioning of the recipient is not begun until it is clear that sufficient numbers of isolated hepatocytes will be available to complete the transplant, preconditioning can add several more hours of cold time postisolation. To date, there is no general consensus on an optimal protocol to maintain cells postisolation and prior to transplantation. In this study, UW was chosen as the preservation solution, since it has been previously used for storage of human hepatocytes for transport between isolation and transplant centers (9) and for its documented superior properties in preserving human hepatocytes compared to other organ preservation solutions (1). Because of these properties UW was the only solution included in the protocol submitted from our institution to the Food and Drug Administration for the cold storage of hepatocytes prior to transplant (10). The purpose of the study was to define the time limits of cold storage that still provided functional hepatocytes for transplantation. Since UW was the only preservation solution approved in our clinical transplant protocol, and the large numbers of cells required performing the assays included here, other storage solutions could not be included in the present study. We chose instead to provide the data on cold storage in UW on a large number of hepatocyte isolations (up to 47) to get a full picture of the data one might expect from future cell isolations. We utilized hepatocytes isolated both from organ donors (at the moment, the only clinical source of human hepatocytes for cell-based therapy) as well as tissues unsuitable for clinical applications, including tissue from liver resections for neoplasia and explanted tissues from inborn error patients (Table 1). In a recently published study, we demonstrated that cells isolated from patients with some metabolic liver diseases provide highly viable hepatocytes that engraft and repopulate liver when transplanted (13). The present results with cells isolated from these organs from metabolic disease patients indicate that these cells weather the stress of hypothermic storage as well as or better than cells isolated from organs rejected for transplantation. In the tissues received from liver resections from cancer patients, we have not observed any statistical difference in viability, plating efficiency, or drug metabolism between the patients that received chemotherapy and those that did not.
It is well known that isolated hepatocytes undergo progressive damage and subsequent cell death during cold storage (1,19,25). Although hypothermia slows cell metabolism and prevents a rapid loss in viability, extended low-temperature storage may cause cellular damage. Viability, as measured by trypan blue exclusion, was significantly reduced in cold-stored compared to fresh cells. A form of apoptosis due to lack of cell anchorage, called anoikis, may be involved in loss of viable hepatocytes maintained in suspension (25). However, in this study of cold-preserved human hepatocytes, only a trend toward increased caspase-3/7 activities was observed, which did not reach statistical significance even at 72 h. The plating efficiency of the cold-stored cells did not change significantly from that observed with freshly isolated hepatocytes. These results indicate that although the viability decreased with extended cold storage, the ability to attach to a culture substrate is well maintained in the remaining viable cells through 72 h of cold storage.
Biotransformation of drugs or chemical reagents as a measure of hepatic function has been proposed as a criteria for selection of hepatocyte cases for clinical transplant (9,17). Results presented here for the CYP 1A, 3A, and 2C families support the hypothesis that these endpoints provide data useful for the analysis of hepatic function prior to transplant.
While most studies focus on CYP450-mediated metabolism, relatively little attention has been paid to either conjugation activity or ammonia metabolism. In addition to phase I metabolism, long-term phase II and ammonia metabolism are highly relevant endpoints to measure, particularly since urea cycle disorders and Crigler—Najjar syndrome have been the most common indications for HTx.
However, for assessing the potentially toxic effects of extended cold storage, the way the assays are performed becomes quite critical. Except for viability as measured by trypan blue exclusion, all of the functional assays measured immediately after recovery of the cells from cold storage show no significant difference between freshly isolated and those stored up to 72 h. However, the data presented for CYP3A4 induction and ammonia metabolism in long-term plated cells suggests that when the previously cold-stored cells are allowed to attach to culture plates and are maintained for an additional 5 days in culture, subtle, previously latent, adverse effects of cold storage on hepatic functions are revealed. A significant induction of CYP3A4 metabolic activity following exposure of cultured cells to rifampicin is only observed in cells maintained out to 48 h of cold storage. Likewise, the normal increase in ammonia metabolism observed in cultured hepatocytes is only statistically significant for freshly isolated cells and those stored for 24 h.
Measurements of CYP1A induction and the conjugation of resorufin were identical to fresh cells out to 72 h of cold storage. The longer term assays on cultured cells require that the cells maintain high viability and function over an extended time in culture. Cells not sufficiently vital to withstand that level of stress are presumed to be lost to the analysis. Since these activities were found to decline in long-term cultured cells, measurements of CYP3A4 induction by rifampicin and the increase in ammonia metabolism may be the most sensitive indicators of cold storage damage to human hepatocytes.
In summary, isolated human hepatocytes were exposed to prolonged storage at 4°C and examined for viability and function with a battery of 11 tests that are used in the investigation of hepatocytes for possible clinical transplant at our centers. This study supports the hypothesis that isolated hepatocytes can be maintained in UW solution for a prolonged time without significant loss of most hepatic functions. Long-term culture of hepatocytes and assays of CYP induction and ammonia metabolism declined with prolonged cold storage greater than 48 h and may therefore be more sensitive indicators of cold storage damage than assays conducted immediately post-cell recovery. While viability decreased in cold-stored cells in the range of 50-60%, most hepatic functions were well maintained in the remaining viable cells out to at least 48 h postisolation.
Footnotes
Acknowledgments
Supported by NIH. N01-DK-7-0004/HHSN26700700004C and RC1DK086135 (S.C.S.), National PKU Alliance (R.G. and K.J.S.), COPEV Associazione per la Prevenzione e Cura dell'Epatite Virale “Beatrice Vitiello” ONLUS (R.G.). The authors declare no conflicts of interest.
