Abstract
We have previously shown that interspecies incompatibility of CD47 plays an important role in triggering rejection of xenogeneic hematopoietic cells by macrophages. However, whether CD47 incompatibility also induces rejection of nonhematopoietic cellular xenografts remains unknown. Herein, we have addressed this question in a mouse model of hepatocyte transplantation in which CD47-/- hepatocytes were used to resemble xenografts for CD47 incompatibility. We show that intrasplenic transplantation of CD47-/-, but not wild-type (WT) hepatocytes, into partially hepatectomized syngeneic WT mice resulted in a rapid increase in Mac-1+ cells with an activation phenotype (i.e., Mac-1+CD14+ and Mac-1+CD16/32high), compared to nontransplant controls. In addition, CD47-/- hepatocytes were more severely damaged than WT hepatocytes as indicated by the greater AST and ALT serum levels in these mice. Furthermore, long-term donor hepatocyte survival and liver repopulation were observed in mice receiving WT hepatocytes, whereas CD47-/- hepatocytes were completely rejected within 2 weeks. These results suggest that CD47 on donor hepatocytes prevents recipient myeloid innate immune cell activation, hence aiding in graft survival after hepatocyte transplantation. Thus, CD47 incompatibility is likely to present an additional barrier to hepatocyte xenotransplantation.
Keywords
Introduction
Hepatocyte transplantation (HTx) has been proposed as an alternative to organ transplantation for patients with liver disease or as a “bridge” to liver regeneration or transplantation (8,34). Unfortunately, the insufficient supply of accessible livers limits this therapy, but the use of hepatocytes from other species (i.e., xenotransplantation) could potentially overcome this problem (29). Successful xenotransplantation faces considerable hurdles, such as the immune response of the recipient against the graft (5). There is increasing evidence that cell components of the innate immune system, such as macrophages, neutrophils, and monocytes, play an important role in xenograft rejection (12,40). Macrophages mediate strong rejection of porcine hematopoietic cells even in the absence of adaptive immunity (1,18). Thus, improved understanding of the mechanisms involved in the innate immune xenoresponses may help in developing effective approaches to overcome xenograft rejection.
Innate immune cell activation is regulated by the balance between stimulatory and inhibitory signals. Previous studies have shown that one underlying mechanism relies on the interaction between inhibitory receptor, signal reg ulatory protein-α (SIRPα), expressed on myeloid cells, and its ligand cluster of differentiation 47 (CD47), a member of the Ig superfamily ubiquitously expressed in all tissues (4). CD47 interacts with SIRPα, leading to tyrosine phosphorylation of immunoreceptor tyrosine-based inhibition motifs (ITIMs), therefore inhibiting phagocytosis. The CD47–SIRPα interaction provides a “don't eat me” signal to myeloid cells, which is required for preventing phagocytosis of normal self-hematopoietic cells (31). Earlier studies have demonstrated the inability of porcine CD47 to functionally interact with mouse or human SIRPα, resulting in phagocytosis of porcine hematopoietic cells by mouse and human macrophages, respectively (19,38). However, it remains poorly understood whether lack of cross-species reactivity between CD47 and SIRPα also triggers nonhematopoietic xenograft rejection by myeloid-derived cells. Herein, we have explored the role of CD47 in regulation of innate immune cell activation and graft rejection in a mouse model of hepatocyte transplantation in which hepatocytes from CD47-/- mice were used to mimic xenogeneic hepatocytes that express a CD47 incapable of interacting with the recipient SIRPα (38). We found that transplantation of CD47-/-, but not CD47+/+, hepatocytes led to rapid activation and expansion of recipient macrophage 1 antigen-positive (Mac-1+) cells, a condition that was associated with poor graft survival in CD47+/+ recipient mice. These results indicate that the lack of cross-species interaction in the CD47–SIRPα pathway is likely to elicit innate immune cell activation and graft rejection in hepatocyte xenotransplantation.
Materials and Methods
Animals
Four- to 6-week-old male wild-type (WT) C57BL/6 (B6) and green fluorescent protein (GFP)-expressing (WT or CD47-/-) mice were used as the hepatocyte transplantation recipients and donors, respectively. WT B6 and GFP-transgenic B6 [C57BL/6-transgenic (UBC-GFP) 30Scha/J] mice were purchased from the Jackson Laboratory (Bar Harbor, ME, USA). GFP-transgenic CD47-/- (CD47-/- GFP) B6 mice were generated by crossing CD47-/- B6 mice (kindly provided by P-A Oldenborg, Umeå University, Umeå, Sweden) with WT GFP-transgenic (WT-GFP) B6 mice. All procedures performed on the animals were approved by the Massachusetts General Hospital Subcommittee on Research Animal Care.
Hepatocyte Isolation and Transplantation
Primary hepatocytes were isolated from WT and CD47-/- GFP mice by a two-step collagenase digestion method as previously reported (23). In brief, the liver was perfused in situ for 10 min via a portal vein with 50 ml of perfusion buffer (Earl's balanced salt solution; Gibco, Gaithersburg, MD, USA). After the initial flush out, a buffer supplemented with collagenase (0.5 g/L; Dako, Carpinteria, CA, USA) and CaCl2 (Sigma-Aldrich, St. Louis, MO, USA) 0.55g/L was perfused at a flow rate of 4–5 ml/min for 10 min. The liver was passed through a filter mesh (BD, Franklin Lakes, NJ, USA), and the hepatocytes were obtained by 40% Percoll (Amersham, Uppsala, Sweden) gradient centrifugation. Hepatocyte viability evaluated by trypan blue (Gibco) staining was routinely >90%. To facilitate hepatocyte engraftment and provide growth advantage over the host hepatocytes, all recipients were treated by two intraperitoneal injections of 30 mg/kg of retrorsine, a pyrrolizidine alkaloid from Senecioretrorsus (4 mg/ml in distilled water; Sigma-Aldrich) that inhibits hepatocyte proliferation in mice (11), with a 2-week interval followed by 70% hepatectomy 4 weeks after the last injection of retrorsine. The hepatectomy was performed by removing the left lateral and median hepatic lobes as previously described (16). WT-GFP and CD47-/- GFP mouse hepatocytes (1.5 × 106 per mouse; in 150 ml PBS; Sigma) were injected into the lower pole of the recipient spleen immediately following hepatectomy. The splenic artery was clamped for about 2 min to avoid immediate passage of hepatocytes into the portal vein; the infusion site was ligated, and the abdomen was sutured. Blood was collected from the tail 1 day before and every other day after 70% hepatectomy and transplantation for the first 7 days and every week thereafter for measuring aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels and innate immune cell activation. Serum AST and ALT levels were measured using a VetTest Chemistry Analyzer (IDEXX Laboratories, Westbrook, ME, USA). Mice were followed up to 5 weeks and then sacrificed for histological evaluation.
Flow Cytometric (FCM) Analysis
The following antibodies were used in FCM analysis of recipient myeloid cell activation in the blood [i.e., white blood cells (WBCs)]: anti-mouse Mac-1-PE (phycoerythrin; clone M1-70; R&D, Minneapolis, MN, USA), anti-mouse CD14-APC (allophycocyanine; clone Sa2-8; eBioscience, San Diego, CA, USA), anti-mouse CD16/32-PE [fragment, crystallizable γ receptor II/III (FcγR II/III); clone 2.4G2; BD Pharmingen, San Diego, CA, USA], and rat immunoglobulin G2a (IgG2a; isotype control; BD Pharmingen). Labeled cells were analyzed in a FACScalibur (BD Biosciences, Mountain View, CA, USA). CD47 expression on hepatocytes was determined by staining with rat anti-mouse CD47 (clone miap301; Biolegend, San Diego, CA, USA).
Detection of Donor Hepatocytes in the Spleen and Liver
Autopsies were performed at days 3, 7, and 14, and at the time of death or sacrifice, and the liver and spleen were fixed with 4% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA, USA), embedded in Tissue-Tek (Sakura Finetek U.S.A, Inc., Torrance, CA, USA), and stored at −80°C. Frozen sections (6-μm thick) were cut, nuclear counterstained with Vectashield mounting medium with 4′,6-diamidino-2-phenylindole (DAPI; Vector Labs, Burlingame, CA, USA), and observed under the fluorescence microscope for the presence of GFP+ cells. Ten randomly selected areas per sample were analyzed with the image J software (NIH, Bethesda, MD, USA) under 200× magnification, and the data are presented as the percentage of GFP-positive areas.
Statistical Analyses
Student's t test or one-way ANOVA with Tukey's posttest when appropriate was used to calculate statistical difference in mean values between groups. A value of p < 0.05 was considered statistically significant.
Results
CD47-/- Hepatocyte Transplantation Induces Rapid Expansion of Recipient Myeloid Cells
To investigate the role of CD47 expression on donor hepatocytes in regulating innate immune cell activation in the absence of immune activation by other mechanisms, a syngeneic mouse hepatocyte transplantation model was used. WT B6 mice were conditioned with retrorsine and 70% hepatectomy (as detailed in the Materials and Methods section) and received intrasplenic injection of syngeneic WT or CD47-/- hepatocytes or a sham operation (nontransplant controls). FCM analysis confirmed CD47 expression on isolated WT hepatocytes (Fig. 1). We first followed the kinetic changes in peripheral blood Mac-1+ cells in these mice. Mac-1 (CD11b/CD18) is a β2-integrin expressed on myeloid-derived cells (2). FCM analysis revealed that transplantation of CD47-/- hepatocytes, but not WT, led to a significant increase in Mac-1+ cells compared to nontransplant controls. Although an increase in Mac1+ cells was detected on day 3 in mice from all three groups compared to the pretransplant levels (approximately 50% measured 1 day prior to hepatectomy/hepatocyte transplantation), mice receiving CD47-/- hepatocytes had significantly higher levels of Mac-1+ cells at days 3 and 5 compared to WT hepatocyte recipients and nontransplant controls (Fig. 2A–D). In addition, a significant upregulation of Mac-1 expression was also observed in CD47-/- hepatocyte-transplanted mice at days 3 and 5 after transplantation when compared with those receiving WT hepatocytes or nontransplant controls (Fig. 2B–D). The similar levels of Mac-1+ cells between WT hepatocyte-transplanted mice and nontransplant controls throughout the experiments indicate that the observed Mac-1+ cell expansion in the recipients of WT hepatocytes was caused by hepatectomy and not WT hepatocyte transplantation (Fig. 2B–D).
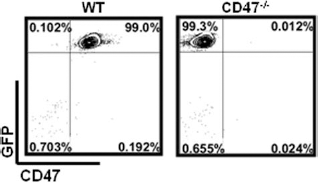
CD47 and GFP expression on hepatocytes. Representative flow cytometry (FCM) plot of green fluorescent protein-wild type (GFP-WT) and GFP-cluster of differentiation 47 knockout (CD47-/-) mouse hepatocytes stained with anti-CD47.

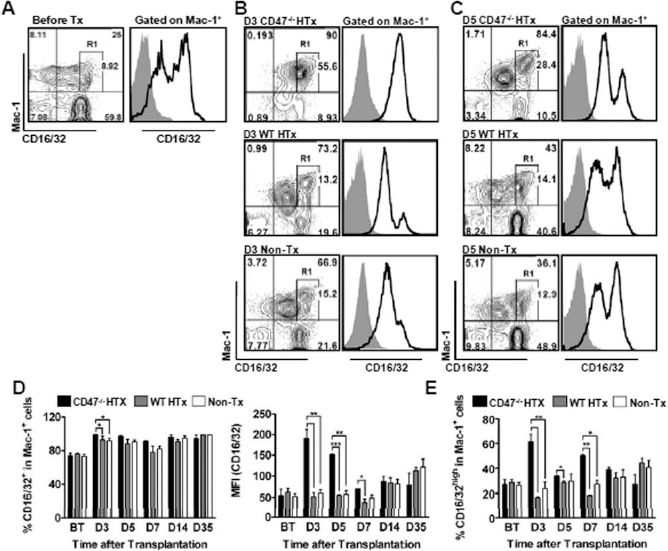
Early recruitment and activation of Mac-1+ and CD14+ after CD47-/- hepatocyte transplantation. White blood cells (WBCs) were prepared 1 day before transplantation (BT) and days 3, 5, 7, 14, and 35 posttransplantation from mice receiving CD47-/- (CD47-/-HTx) or WT (WT HTx) hepatocytes or nontransplanted controls (non-Tx) and analyzed for macrophage 1 antigen (Mac-1) and CD14 expression. (A–C) Representative FCM plots of WBCs stained with anti-Mac-1 and anti-CD14 and histograms depicting levels of Mac-1 and CD14 expression on gated Mac-1+ cells analyzed BT (mice treated with retrorsine alone (A) and at days 3 (B) and 5 (C) after transplantation. (D) Percentages of Mac-1+ cells in WBCs and levels of Mac-1 expression (shown as mean fluorescence intensity; MFI) at the indicated times. (E) Percentages of CD14+ cells in Mac-1+ cells and levels of CD14 expression. Data in (D) and (E) are expressed as mean ± SEM (n = 6/group). *p < 0.05; **p < 0.005; ***p < 0.0005; ****p < 0.0001.
The Lack of CD47 Expression on Donor Hepatocytes Induces a Strong Activation of Recipient Mac-1+ Cells
In order to evaluate whether the lack of CD47 on donor hepatocytes can induce activation of myeloid-derived Mac-1+ cells, we measured CD14 and FcγR expression on these cells in peripheral blood. CD14 is the receptor for the lipopolysaccharide (LPS)–binding protein complex (LBP) and is expressed on myeloid cells upon activation (10,24,25,39). The percentage of Mac-1+ cells coexpressing CD14 was low (approximately 4.2 ± 1.3%) in all groups prior to hepatectomy/hepatocyte transplantation (Fig. 2A). A significant increase in Mac-1+CD14+ cells compared to the pretransplant level was seen at day 3 posttransplantation in mice receiving WT hepatocytes (p < 0.005) and nontransplant controls (p < 0.05), which likely resulted from hepatectomy as the percentages of Mac-1+CD14+ were comparable between the two groups throughout the study period (Fig. 2A–E). Compared to WT hepatocyte recipients and nontransplant controls, a significantly greater increase in Mac-1+CD14+ cells was observed in mice receiving CD47-/- hepatocytes on days 3 and 5 (Fig. 2A–C, E). Percentages of Mac-1+ cells expressing CD14 in the recipients of CD47-/- hepatocytes at day 3 were approximately fivefold higher than those observed for the WT hepatocyte-transplanted and nontransplant mice, respectively (Fig. 2B, E). CD47-/- hepatocyte transplantation also led to a significant upregulation in the level of CD14 expression in Mac-1+ cells (Fig. 2B, C, E).
It has been shown that CD16/32 (FcγRIII/II) activates Mac-1, and acting synergistically with Mac-1 promotes phagocytosis and adherence of phagocytes at sites of infection (20), and myeloid cells show rapid upregulation of CD16/32 expression upon activation (17,33). In order to further investigate the activation status of Mac-1+ cells after transplantation of CD47-/- hepatocytes, we measured the expression of CD16/32 on peripheral blood Mac-1+ cells. Although significant increases in percentages of Mac-1+CD16/32+ cells and levels of CD16/32 expression were detected in all groups compared to pretransplant levels, the degrees of these increases were significantly greater in mice receiving CD47-/- hepatocytes compared to WT hepatocyte-transplanted and non-transplant controls (Fig. 3A–D). Noticeably, within the Mac-1+CD16/32+ cells, there was a population expressing high levels of CD16/32 (i.e., Mac-1+CD16/32hi cells in R1) (Fig. 3A–C), which increased even more pro foundly after CD47-/- hepatocyte transplantation. As shown in Figure 3E, the percentages of Mac-1+CD16/32hi in mice transplanted with CD47-/- hepatocytes were more than fourfold at day 3 and threefold at day 7, higher than those in the other two groups. Since no significant difference was seen between WT hepatocyte recipients and nontransplant controls throughout the observation period, the changes observed in both groups were likely caused by hepatectomy. A significant increase in the number of Mac-1+ cells (Fig. 4A, C) and the level of Mac-1 expression (Fig. 4B, D) was also seen in the spleen (Fig. 4A, B) and liver (Fig. 4C, D) in mice transplanted with CD47-/-hepatocytes compared to those receiving no hepatocytes or WT hepatocyte transplantation. Together, these results suggest that CD47 expression on donor hepatocytes plays an important role in controlling recipient innate immune cell activation.
Rapid increase in Mac-1+CD16/32hi cells in mice receiving CD47-/- hepatocyte transplantation. WBCs were prepared at the indicated time points and analyzed for CD16/32 expression on Mac-1+ cells. (A–C) Representative FCM plot of WBCs stained with anti-Mac-1 and anti-CD16/32 and histogram depicting CD16/32 expression levels on gated Mac1+ cells analyzed BT (A) and 3 (B) and 5 (C) days after transplantation. (D) Percentages of Mac-1+CD16/32+ cells and levels of CD16/32 expression on Mac-1+ cells. (E) Percentages of Mac-1+CD16/32hi cells (i.e., cells in R1 in Fig. 3A–C). Results in (D) and (E) are presented as mean ± SEM (n = 6 per group). *p < 0.05; **p < 0.005; ***p < 0.0005.

Rapid increase in Mac-1+ cells in the spleen and liver of mice transplanted with CD47-/- hepatocytes. Spleen (A–B) and liver cells (C–D) were prepared at days 3 and 7 posttransplantation from mice receiving CD47-/- (CD47-/- HTx) or WT (WT HTx) hepatocytes or no hepatocyte transplantation (Non-Tx) and analyzed for the numbers of Mac-1+ cells (A, C) and levels of Mac-1 expression (B, D) at the indicated time points. *p < 0.05; **p < 0.005; ***p < 0.001.
Evaluation of Hepatocyte Damage After Transplantation
Following intrasplenic hepatocyte transplantation, serum levels of ALT and AST were evaluated as a measure of hepatocyte destruction. Mice receiving CD47-/-hepatocyte transplantation showed a significant increase in AST levels compared to mice receiving WT hepatocytes and nontransplanted controls at day 1 (Fig. 5A) and a significant increase in ALT levels at days 1 and 3 compared to WT hepatocyte recipients (Fig. 5B), indicating that CD47-/- hepatocytes were severely destroyed. Notably, mice receiving WT hepatocytes had similar levels of AST and significantly reduced levels of ALT when compared to the nontransplanted controls (Fig. 5A, B), suggesting that transplanted WT hepatocytes may mediate protection against endogenous liver injury as previously reported (13,32).

Increased serum levels of AST and ALT in mice receiving CD47-/- hepatocyte transplantation. Shown are serum levels (mean ± SEM) of aspartate aminotransferase (AST) (A) and alanine aminotransferase (ALT) (B) in mice receiving WT (WT HTx; n = 8) or CD47-/- (CD47-/- HTx; n = 10) hepatocytes and in nontransplant controls (Non-HTx; n = 6). *p < 0.05; ***p < 0.001.
Failure of CD47-/- Donor Hepatocytes to Survive After Intrasplenic Injection Into Syngeneic WT Mice
We next compared the survival of WT versus CD47-/-hepatocytes in the spleen and liver of WT mouse recipients. Spleen and liver samples were prepared 5 weeks after GFP+ hepatocyte transplantation, and donor hepatocyte survival was examined by measuring GFP+ cells under a fluorescent microscope. GFP+ hepatocytes were found arranged in cordlike structures around the red pulp of the spleen in mice receiving WT hepatocytes, while no surviving GFP+ cells were observed in animals transplanted with CD47-/- hepatocytes (Fig. 6A, B). Furthermore, WT donor hepatocytes were found to engraft and repopulate the recipient liver (Fig. 6C, D) as previously reported (21). However, GFP+ hepatocytes were barely detectable in mice receiving CD47-/- hepatocytes (Fig. 6C, D). In a separate experiment, we assessed donor hepatocyte survival at early time points after transplantation. Compared to mice receiving WT hepatocytes, mice transplanted with CD47-/- hepatocytes showed a marked reduction in GFP+donor hepatocytes in the spleen at days 3 and 7 posttransplantation (Fig. 7), indicating a rapid destruction of CD47-/- donor cells in these mice (Fig. 7). Furthermore, no surviving CD47-/- hepatocytes were detected in the spleen at day 14 posttransplantation, whereas WT donor hepatocytes were detected in the recipient mice at all time points examined (Fig. 7).
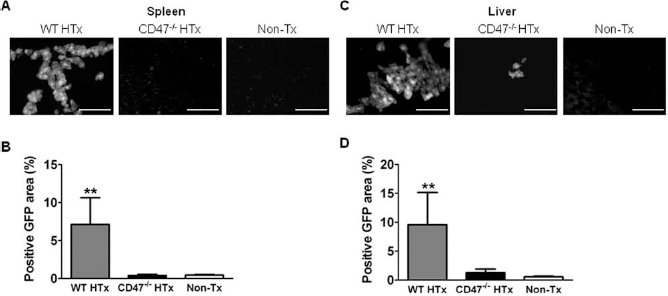
Lack of long-term surviving CD47-/- hepatocytes in WT mouse recipients. Mice subjected to retrorsine and 70% hepatectomy underwent intrasplenic hepatocyte transplantation from GFP-WT or GFP-CD47-/- donors. Spleen (A–B) and liver (C–D) tissue sections were prepared 5 weeks after transplantation and examined for surviving GFP+ donor hepatocytes. Shown are representative spleen (A) and liver (C) images of WT hepatocyte transplanted mice (WT HTx), CD47-/- hepatocyte transplanted mice (CD47-/- HTx) and nontransplanted controls (Non-Tx). Percentages (mean ± SEM) of GFP+ areas in the spleen (B) and liver (D) from the indicated groups (n = 8–10 per group). **p < 0.005.

Rapid destruction of CD47-/- hepatocytes after intrasplenic transplantation in WT mice. Mice subjected to retrorsine and 70% hepatectomy underwent intrasplenic hepatocyte transplantation from GFP-WT or GFP-CD47-/- donors. Spleen tissue sections were prepared at days 3, 7, and 14 posttransplantation and examined for surviving GFP+ donor hepatocytes. Shown are representative images of the spleens from mice receiving WT (WT HTx; left panel) or CD47-/- (CD47-/- HTx; right panel) hepatocytes at the indicated time points after transplantation. Scale bar: 100 mm.
Discussion
We investigated in this study the role of CD47 expression on donor hepatocytes in the regulation of recipient innate immune cell activation and graft rejection after hepatocyte transplantation. We used a mouse model of syngeneic hepatocyte transplantation to avoid interference from immune activation by other mechanisms. We found that transplantation of CD47-/-, but not WT, hepatocytes in syngeneic WT mice induced significant expansion and activation of recipient Mac-1+ cells. Furthermore, CD47-/- hepatocytes were barely detectable in recipient spleen or liver by day 7, while long-term survival of donor hepatocytes was observed for all animals receiving WT hepatocytes. These data, to our knowledge, provide the first direct evidence that CD47 expression on donor hepatocytes is critical for restraining innate immune cell activation and promoting graft survival after hepatocyte transplantation.
Mac-1 is constitutively expressed at varying degrees on monocytes, macrophages, and granulocytes and mediates binding to endothelial cells and extracellular matrix (2,35). Previous studies have shown that myeloid cells upregulate Mac-1 expression upon activation (26,36). Thus, the observed significant increase in Mac-1+ cells in mice receiving CD47-/- hepatocytes compared to those transplanted with WT hepatocytes indicates that the lack of CD47 expression on donor hepatocytes can induce rapid activation and expansion of recipient myeloid innate immune cells. This observation was further confirmed by significantly increased expression of two other activation markers, CD14 and FcγRs, on Mac-1+ cells from CD47-/- hepatocyte-transplanted mice compared to those receiving WT hepatocytes. In humans, CD14 has been considered a myelomonocytic differentiation antigen with expression by myeloid cell lineages (9,14,15). In mice, however, CD14 expression is either very low or undetectable in resting macrophages and granulocytes (6,7,28) but rapidly upregulated upon cell activation (27,28). Both human and mouse myeloid innate immune cells have been shown to upregulate CD16/32 (FcγRIII/II) expression upon activation (3,17,30,33). All data considered, the present study provides strong evidence that CD47 expression on donor hepatocytes can inhibit recipient myeloid cell activation. Studies are in progress to identify the Mac-1+ myeloid cell lineages that show rapid activation after CD47-/- hepatocyte transplantation.
A significant early increase in myeloid cell expansion and activation was also observed in mice following WT hepatocyte transplantation, although to a lesser extent than that observed in mice receiving CD47-/- hepatocytes. However, this effect following WT hepatocyte transplantation was likely due to hepatectomy and not hepatocyte transplantation as comparable changes were also seen in nontransplant controls. Full restoration of liver mass occurs in normal mice after 70% hepatectomy (32), but the remnant liver cannot undergo regeneration in retrorsine-treated mice following hepatectomy (11,32). In accordance, we observed high serum levels of AST and ALT in nontransplanted controls. The transient, but significantly lower, AST and ALT levels at day 1 posttransplantation in mice receiving WT hepatocytes compared to that measured in nontransplant controls suggests that the transplanted hepatocytes may provide extra liver mass to aid in liver function and alleviate the excess stress imposed to the native liver. However, mice receiving CD47-/- hepatocytes had paradoxically higher levels of AST and ALT, indicating that, in addition to the stress and injury from the native liver, the transplanted hepatocytes were also being destroyed.
Multiple barriers may restrict engraftment and proliferation of transplanted hepatocytes (21,22). Although 70–80% of transplanted cells were found to be cleared from the liver within 1 or 2 days after syngeneic cell transplantation (22), the remaining hepatocytes could survive and repopulate the liver rapidly if appropriate conditions are provided (11). In this study herein, longterm donor hepatocyte survival and liver repopulation were detected in mice receiving WT hepatocytes but not in those receiving CD47-/- hepatocytes in which donor hepatocytes were rapidly destroyed within 2 weeks (most cells were destroyed by 7 days) after transplantation (Figs. 6 and 7). Although the loss of CD47-/- hepatocyte grafts may possibly result from immune rejection and/or disadvantage in survival, the rapid expansion/activation of Mac-1+ cells and increased AST and ALT levels in these mice indicate that rejection by activated innate immune cells is likely to be a major cause of graft loss. In support of this possibility, preliminary observations (data not shown) revealed that long-term graft survival could be achieved when CD47-/- hepatocytes were transplanted into syngeneic CD47-/- mice, which are tolerant of CD47-/- cells (31,37).
In summary, using a syngeneic mouse hepatocyte transplantation model, the present study demonstrates that missing CD47 on donor cells alone can cause recipient myeloid cell activation and graft loss. We have previously shown that CD47 has no cross-reaction between species, and CD47 incompatibility contributes significantly to rejection of xenogeneic hematopoietic cells by macrophages (19,38). The data presented in this study indicate that CD47 incompatibility will likely also pose a barrier to nonhematopoietic cellular xenotransplantation, at least hepatocyte xenotransplantation.
Footnotes
Acknowledgments
The authors thank Dr. Hui Wang for the critical review of this manuscript. This work was supported by research grants from NIH (R01 AI064569, 2P01AI045897-11A). N. Navarro-Alvarez is partially supported by a Basic Science Fellowship from the American Society of Transplantation. The authors declare no conflict of interest.
