Abstract
Stress urinary incontinence (SUI) is characterized by the involuntary leakage of urine from the urethra due to increased abdominal pressure. The complex pathophysiological mechanisms underlying SUI have driven the development of diverse therapeutic strategies. Current treatment options encompass both conservative and surgical interventions, with surgical approaches generally often regarded as the most effective option approach for severe cases. However, many surgical techniques carry significant risks of complications. In this context, urethral injection therapy, primarily based on stem cell–mediated regenerative approaches, has emerged as a minimally invasive alternative. Stem cell therapies leverage their multipotent differentiation capacity and paracrine signaling pathways to directly target the pathophysiological contributors to SUI, including urethral sphincter dysfunction, neuromuscular junction degeneration, and imbalances in elastin and collagen homeostasis. This narrative review provides a critical evaluation of current stem cell–mediated regenerative strategies for SUI, focusing on cellular mechanisms and the therapeutic effects driven by paracrine signaling. Recent clinical advances, unresolved scientific controversies, and innovative combinatorial delivery systems incorporating targeted therapeutic approaches are analyzed. Despite challenges remain, such as determining the optimal stem cell dosage and improving in vivo survival rates, ongoing research offers valuable insights into the development of cell-free bioactive derivatives, advanced combination delivery systems, and precise molecularly targeted therapies.
Keywords
Introduction
The International Continence Society (ICS) defines stress urinary incontinence (SUI) as involuntary leakage of urine in the absence of detrusor muscle contraction, particularly during increases in abdominal pressure caused by activities such as sneezing, coughing, or laughing 1 . A research report from Peking Union Medical College Hospital identifies SUI as the most prevalent form of urinary incontinence among women, with a reported prevalence rate of 18.9% in Chinese female populations. This prevalence escalates significantly with age, reaching 28% among women aged 60 years and older. Established risk factors for SUI include obesity, smoking, and aging 2 (Fig. 1). Given its profound impact on quality of life and overall health, timely intervention is critical to mitigate disease progression.

Pathophysiological mechanisms of SUI. Due to obesity, smoking, childbirth, aging, and other factors, the pelvic floor muscle tissue and the related microenvironment are damaged, resulting in stress urinary incontinence.
A pelvic floor–related pathophysiological framework provides insight into SUI, encompassing both structural and functional dimensions. Structurally, the urethral sphincter consists of inner smooth muscle and outer striated muscle layers. Urine leakage occurs when these muscles are compromised, resulting in bladder pressure exceeding urethral closing pressure. Neural regulation forms a second core component of urinary control. Damage to nerves innervating the pelvic floor and external urethral sphincter can independently precipitate urinary leakage. Functionally, pelvic floor fascia and ligaments comprise dense connective tissues containing fibroblasts and extracellular matrix (ECM) secreted by these cells. Fibroblasts regulate ECM remodeling through cytokine and matrix protein secretion, and their dysfunction directly compromises connective tissue integrity3,4. Key molecular and cellular pathways implicated in SUI pathogenesis include myocyte apoptosis, collagen degradation, fibrotic muscle replacement, and vascular or neurological impairments 5 . Preclinical SUI models frequently employ nerve transection, vaginal balloon dilation, and ovariectomy to replicate pathophysiological conditions. In recent years, leakage point pressure (LPP), defined as the minimum bladder pressure required to induce urethral leakage, has been adopted as a standardized metric to evaluate model reliability and therapeutic efficacy 6 .
Based on the described pathogenesis, multiple clinical interventions for SUI have been developed and demonstrated therapeutic efficacy. Current treatment strategies are categorized into conservative and surgical approaches. Conservative management includes pelvic floor muscle training, bioelectric stimulation, pharmacotherapy, and lifestyle modifications such as weight reduction and smoking cessation. These interventions enhance urethral sphincter strength, improve pelvic floor muscle coordination, and modulate neuromuscular function 7 . Surgical intervention is typically reserved for cases refractory to conservative therapy, aiming to augment bladder or urethral support, increase urethral resistance, and restore urinary continence. The midurethral sling procedures are widely adopted, as they maintain an effective functional urethral length during episodes of elevated abdominal pressure 3 . However, critical considerations include complications related to suspension materials and the risk of recurrent urinary incontinence following subsequent pregnancies 8 .
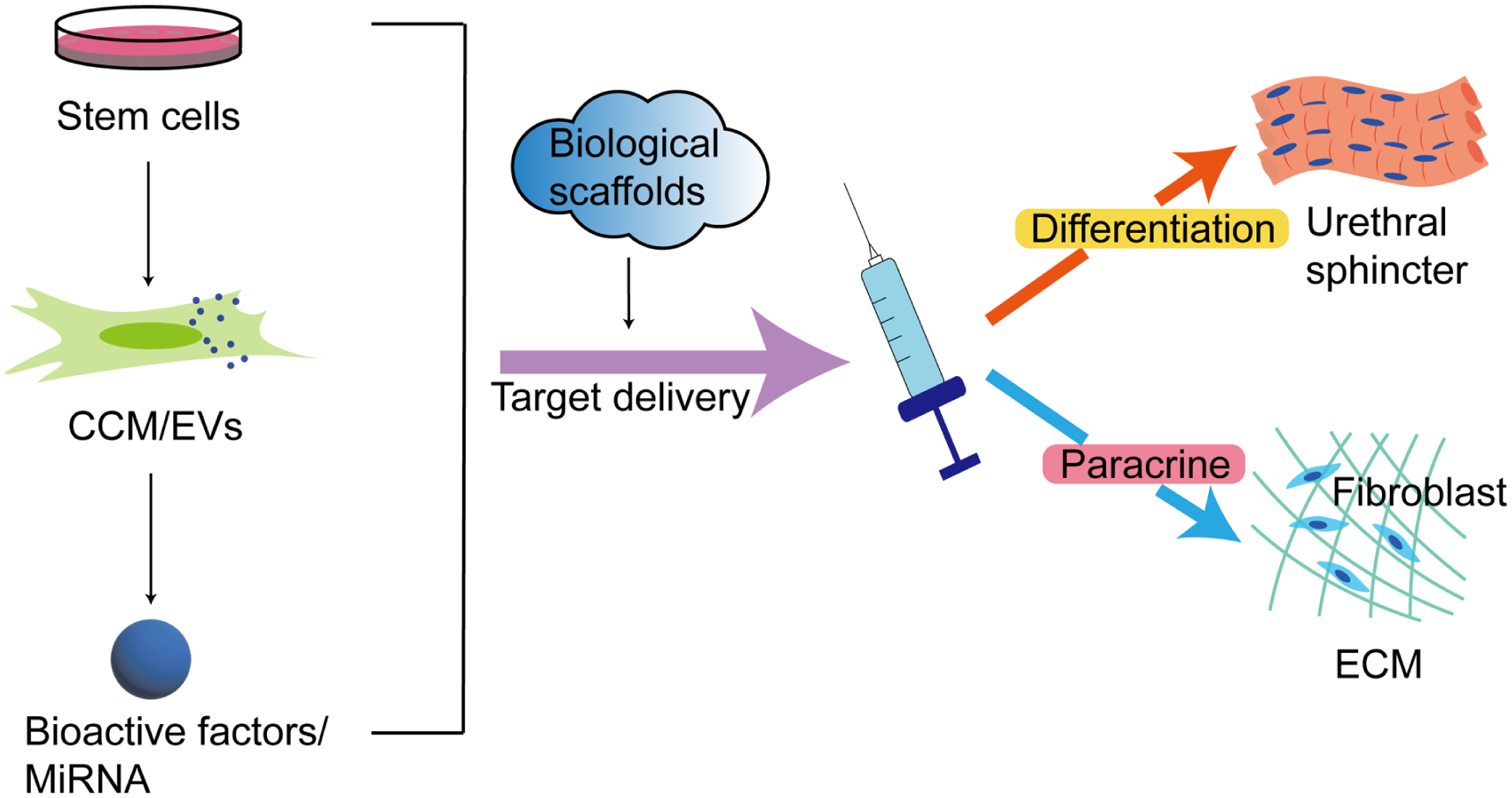
Various injectable materials have been investigated for SUI treatment. Urethral fillers aim to augment mucosal coaptation and restore urethral resistance. However, inert substances may fail to replicate physiological sphincter function in injured or atrophic tissues, potentially causing hypersensitivity reactions, abscess formation, or urinary retention9,10. Recent advancements have prioritized minimally invasive regenerative therapies, such as stem cell injections, which reduce surgical invasiveness and recovery time compared with traditional methods 11 . Stem cells exert therapeutic effects through paracrine signaling, releasing bioactive mediators, including chemokines, cytokines, angiogenic factors, and secreted proteomes that regulate cell proliferation, differentiation, and tissue repair 12 . Preclinical studies demonstrate that stem cell–derived exosomes (Exos) enhance myocyte proliferation and differentiation, accelerate collagen synthesis, and inhibit fibrosis via signaling pathways such as Ras-ERK, PI3K/AKT, and Sirt7/TGF-β113–15. In addition, combinatorial delivery systems incorporating stem cells with biomaterial scaffolds may further amplify regenerative outcomes in urethral sphincter repair. This less invasive approach, coupled with its capacity for tissue regeneration, positions stem cell–based therapies as a promising strategy for SUI (Fig. 2).

Stem cell–based regeneration therapy for the treatment of SUI. Injection of stem cells and their paracrine products to optimize delivery conditions and studying the therapeutic effect on urethral sphincter function. MDSCs: muscle-derived stem cells; ADSCs: adipose tissue–derived stem cells; BMSCs: bone marrow mesenchymal stem cells; UCMSCs: umbilical cord–derived mesenchymal stem cells; CCM: concentrated conditioned medium; EVs: extracellular vesicles.
This review commences with an in-depth analysis of stem cell–mediated regenerative therapies for SUI, focusing on molecular mechanisms underlying decellularization approaches and targeted interventions. Subsequently, strategies to optimize delivery systems are proposed to improve therapeutic efficacy and clinical outcomes.
Diversification and selection of cells
Recent advances in cellular regenerative medicine have enabled minimally invasive harvesting and scalable expansion of autologous cells, thereby reducing treatment costs. In addition to autologous transplantation, allogeneic stem cell transplantation is increasingly viable due to the immunoprivileged properties of these cells 16 . Stem cell therapy for SUI aims to regenerate damaged urethral sphincter complex tissues through targeted delivery or guided migration of stem cells to injury sites, with the ultimate goal of regenerating muscles and nerves to restore normal function. Preclinical studies have systematically elucidated the mechanisms underlying stem cell–based interventions, providing critical insights into optimizing SUI management (Table 1).
Preclinical studies of therapy on injection of stem cells.

ADSCs: adipose tissue–derived stem cells; BMSCs: bone marrow–derived stem cells; EBO: excision of bilateral ovaries; HE: hematoxylin/eosin staining; IV: intravenous; LPP: leak point pressure; MDSCs: muscle-derived stem cells; PNT: pudendal nerve transection; SNT: sciatic nerve transection; VBD: intravaginal balloon dilatation.
Muscle-derived cells
Skeletal muscle, constituting a significant proportion of body mass, represents a readily accessible source of cells with robust regenerative potential. Research indicates that both pelvic floor muscles and the urethral sphincter, which are critical for urinary continence, rely on muscle stem cells and myosatellite cells for tissue repair. Myosatellite cells originate in the mesodermal layer of skeletal muscles and remain quiescent under normal conditions. Upon activation, these cells differentiate into myoblasts, monopotent progenitors capable of mediating muscle regeneration, reinforcing urethral wall integrity, and enhancing urethral sphincter and detrusor muscle contractility 29 . Preclinical studies demonstrate that allogeneic myoblast injections integrate into the smooth muscle layer of the lower urinary tract, forming myotubes and sustaining protein expression. These findings suggest that myoblasts may augment urethral support and improve sphincter and detrusor function 17 . However, challenges persist, including the low survival rate of transplanted myoblasts. Muscle-derived stem cells (MDSCs) are increasingly recognized as pluripotent progenitors with superior regenerative capabilities compared with myoblasts. Characterized by self-renewal capacity, prolonged proliferation, broad differentiation potential, and low immunogenicity, MDSCs offer distinct advantages. Compared with satellite cells and fibroblasts, MDSC injections exhibit prolonged efficacy without inducing immunogenic reactions18,19. High-dose MDSCs do not cause excessive proliferation or migration into adjacent tissues, nor do they lead to urethral obstruction. In experimental models, urethral segments treated with MDSCs showed a 27% greater maximum contraction force than fibroblast-treated controls, accompanied by significant restoration of sphincter morphology and function 20 . This outcome contrasts with fibroblast proliferation, which promotes urethral fibrosis and reduces tissue compliance. Notably, TGF-β1-induced spontaneous fibroblast differentiation in MDSCs disrupts normal muscle fiber formation, exacerbating fibrosis and limiting regenerative efficacy 30 . Smad proteins, as downstream mediators of TGF-β1 signaling, present a therapeutic target: Smad3 inhibition has been shown to suppress fibroblast differentiation and enhance urethral sphincter recovery 31 . These findings address a critical barrier to MDSCs-based therapies by mitigating fibrotic complications. Clinically, autologous MDSCs derived from skeletal muscles are the most extensively studied stem cell population for treating female SUI. Initial clinical validation began in 2008 with a trial involving eight SUI patients who received MDSCs injections and were monitored for 12 months. Six participants exhibited symptom improvement, including one case of complete resolution 32 . Subsequent dose-escalation studies demonstrated a positive correlation between MDSCs dosage and patient response rates during 12-month follow-ups33,34. However, a 24-month follow-up revealed SUI recurrence in some individuals 35 . Collectively, these data indicate that intraurethral MDSCs injection is a minimally invasive and safe intervention with short-term efficacy. However, the decline in therapeutic effects over time suggests limitations in MDSCs proliferation capacity, necessitating higher dosages or adjunctive strategies 36 .
Adipose tissue–derived stem cells
Adipose tissue–derived stem cells (ADSCs) offer distinct advantages over MDSCs, including readily accessible isolation protocols and sustained proliferative potential. These cells exhibit pluripotent differentiation capacity, enabling their commitment to myogenic lineages under appropriate induction, which positions them as promising candidates for regenerative therapies targeting tissue repair. Wu et al. pioneered the adaptation of human urodynamic assessment methods to evaluate urethral sphincter function in rat models. Their findings demonstrated that ADSCs transplantation significantly improved sphincter morphology and functional parameters, achieving durable therapeutic outcomes in SUI 21 . Notably, 3 months post-injection, ADSC-induced myoblasts accelerated the recovery of damaged urethral sphincter muscles. Effective sphincter regeneration requires not only enhanced myotube formation but also concurrent restoration of vascular networks and ECM integrity, both of which are indispensable for normal urethral function22,37. Further investigations highlight the paracrine mechanisms of ADSCs in promoting tissue repair. Upon migration to injury sites, ADSCs secrete bioactive factors with anti-inflammatory and antiapoptotic properties, which aid in muscle and peripheral nerve regeneration while stimulating angiogenesis 38 . CXCL12 (stromal-derived factor-1), a critical chemokine for stem cell homing, plays a pivotal role in directing ADSCs migration to injured urethral tissues. Experimental models confirm that ADSCs administered via intravenous or urethral routes successfully localize to the submucosal layer of the urethral sphincter, where CXCL12 guide their integration into damaged regions, thereby facilitating structural repair 23 . In addition, elevated vascular endothelial growth factor\ levels following ADSCs treatment enhance vascularization and nutrient supply, further supporting sphincter regeneration24,25. Recent studies also reveal ADSCs’ capacity to differentiate into neurally induced adipose-derived stem cells, which promote Schwann cell proliferation and sustain neural regeneration 26 . Clinical applications of autologous ADSCs have demonstrated promising safety profiles and therapeutic efficacy in small-scale trials. A pilot study involving periurethral ADSCs transplantation in SUI patients reported significant symptom improvement in 10 participants during a six-month follow-up period. Phase I and II clinical data corroborate these findings39,40. However, current evidence remains limited by small sample sizes, short follow-up durations, and undefined optimal dosing regimens.
Other types of mesenchymal stem cells
Bone marrow–derived mesenchymal stem cells (BMSCs), among the earliest identified and most extensively studied mesenchymal stem cells (MSCs) populations, exhibit unique therapeutic potential for SUI due to their exceptional multipotency. Preclinical studies demonstrate that BMSCs transplantation in denervated SUI rat models significantly improves LPP while promoting structural and functional recovery of the damaged external urethral sphincter. These effects are attributed to dual mechanisms: direct myogenic differentiation and paracrine activation of tissue regeneration pathways 27 . Notably, repeated BMSCs administration prolongs cellular proliferation, reduces fibrosis, and enhances repair of urethral connective tissue and neuromuscular junctions 28 . BMSCs represent compelling candidates for regenerative therapy owing to their relative ease of isolation and robust in vitro expansion capacity. However, clinical translation faces critical barriers. Conventional bone marrow aspiration requires general or spinal anesthesia, posing inherent invasiveness. More importantly, ectopic ossification has been observed in vivo following BMSCs injection into myocardial tissue, independent of dose or tissue microenvironment, raising concerns about uncontrolled differentiation potential 41 . These findings underscore the necessity for preclinical investigations to validate directional differentiation control post-transplantation.
In contrast, umbilical cord–derived mesenchymal stem cells (UCMSCs) demonstrate superior translational advantages. Clinical studies report that more than half of SUI patients treated with UCMSCs exhibited symptomatic improvement during 12-month follow-ups 42 . Experimental evidence reveals that UCMSCs enhance fibroblast proliferation and viability through anti-inflammatory cytokine secretion while suppressing apoptosis. In addition, these cells modulate the PI3K/AKT/PTEN pathway to inhibit collagen hypermetabolism in anterior vaginal wall fibroblasts, thereby restoring pelvic floor structural support 13 . UCMSCs harvesting is noninvasive and ethically uncomplicated, with fetal-origin cells offering greater differentiation plasticity and reduced immunogenicity compared with ADSCs 43 . These attributes position UCMSCs as a scalable, immunoprivileged resource for clinical-grade cellular therapies.
Despite the therapeutic promise of BMSCs, MDSCs, ADSCs, and UCMSCs, significant translational challenges persist. The invasive nature of BMSCs harvesting hinders their widespread clinical application. Similarly, the preparation processes for MDSCs, ADSCs, and UCMSCs are complex, requiring the establishment of standardized protocols for collection. Furthermore, clarifying the differentiation pathways and primary mechanisms of action of these cells post-injection is critical, as is evaluating potential adverse effects through long-term clinical follow-up36,44,45. Future research priorities should focus on optimizing delivery systems, including biomaterial scaffolds and targeted homing strategies, and defining standardized dose-response relationships. Expanded phase II randomized controlled trials are essential to mitigate clinical translation risks and accelerate evidence-based implementation.
Decellularized injection therapy and related mechanism
Stem cell injection therapy has demonstrated therapeutic efficacy in improving SUI; however, it carries risks such as infection, high procedural costs, cellular senescence, and tumorigenic potential 46 . To address these limitations, decellularization-based approaches combined with stem cell–derived products have gained prominence. These strategies focus on concentrated conditioned medium (CCM), extracellular vesicles (EVs), and Exos, which are proposed for urethral sphincter regeneration. Compared with cell-based interventions, these components offer a more stable and controllable therapeutic effect 47 . CCM contains diverse bioactive molecules secreted by stem cells, while EVs, nanoscale membrane vesicles present in CCM, carry proteins, mRNA, and miRNA capable of mediating intercellular communication and modulating the local microenvironment. Their advantages include ease of isolation and storage, minimal immunogenicity, and promising potential for enhancing urethral sphincter function 48 (Fig. 3).

The mechanism of action of EVs in fibroblasts and myosatellite cells. EVs are taken up by fibroblasts and deliver miRNA, promoting increased expression of collagen and elastin and ECM remodeling. EVs are taken up by myosatellite cells, which phosphorylate ERK or activate the NF-κB pathway to upregulate expression of the myogenic differentiation factors Myf5, Pax7, MyoD and promote differentiation of myosatellite cells. Upward green arrows indicate positive modulation and downward red arrows indicate negative modulation, respectively. ADSCs: adipose tissue–derived stem cells; BMSCs: bone marrow mesenchymal stem cells; USCs: urinary-derived stem cells; TGF-β: transforming growth factor beta; F3: coagulation factor III; EVs: extracellular vesicles; ERK: extracellular regulated protein kinases; PEP: platelet-derived extracellular vesicle product.
The ECM provides critical structural support to the urethra. Pathogenic alterations in ECM composition within the urethral sphincter, such as reduced elastin and collagen fiber density and disorganized elastin fiber architecture, are key contributors to SUI. Experimental studies demonstrate that BMSCs-CCM treatment reorients and intensifies elastin fibers near the urethral sphincter, indicating that CCM components effectively facilitate elastin repair. Given the short half-life of many bioactive factors in CCM, repeated administrations yield greater tissue restoration than single-dose interventions49,50. Stem cells exert their therapeutic effects on the urethral sphincter by secreting multiple bioactive molecules. Brain-derived neurotrophic factor (BDNF), a neurotrophic family member critical for neuronal plasticity, post-injury repair, and neuroprotection, has been identified as a pivotal component of CCM. Studies show that regenerative electrical stimulation upregulates BDNF expression, thereby enhancing β(II)-microtubule protein synthesis and nerve regeneration responses in bilateral nerve and muscle injury models51,52. These findings highlight BDNF’s essential role in nerve repair mediated by CCM, which likely contains additional unidentified bioactive factors. EVs exhibit superior biocompatibility and low immunogenicity compared with CCM, positioning them as a more precise therapeutic option 53 .
Subsequent investigations by Zhang et al. isolated EVs from CCM and elucidated their mechanistic roles in urethral repair. Their findings demonstrated that BMSC-derived EVs activate the Sirt7/TGF-β1 signaling pathway in primary fibroblasts, upregulating TGF-β1 expression and facilitating ECM remodeling in injured urethral sphincters 15 . Sirt7, a member of the mammalian sirtuin family, antagonizes TGF-β1 and Smad3 signaling to promote fibroblast activation and enhance elastin and collagen synthesis. Exos, a subset of EVs with diameters ranging from 30 to 150 nm, have also shown therapeutic potential. Studies reveal that ADSC-derived Exos increase collagen production in vaginal fibroblasts from SUI patients by enhancing synthesis and suppressing degradation pathways 54 . Beyond ECM remodeling, EVs from diverse stem cell sources, including ADSCs, BMSCs, and urinary-derived stem cells (USCs), play critical roles in sphincter muscle regeneration. For instance, ADSC-derived EVs promote skeletal muscle and Schwann cell growth in a dose-dependent manner. Compared with ADSCs transplantation alone, EV administration in rat models resulted in superior urethral functional recovery, accompanied by increased muscle and nerve fiber density 55 . Wang et al. further identified that ADSC-Exos carrying miR-93 target coagulation factor III (F3) in fibroblasts and muscle satellite cells. By downregulating F3, these Exos reverse its inhibitory effects on ECM remodeling in SUI fibroblasts and upregulate myokine expression in satellite cells, thereby activating primary satellite cells to drive regeneration 56 . Additional studies highlight the therapeutic efficacy of genetically modified EVs. Li et al. administered BMSC-derived EVs overexpressing SIRT1/EVs to a rat SUI model, and observed significant improvements in LPP and maximum bladder capacity (MBV). SIRT1, an NAD+-dependent deacetylase, mitigates cellular senescence and promotes muscle regeneration through extracellular regulated protein kinases (ERK) pathway activation, enhancing satellite cell proliferation and differentiation 57 . Similarly, USC-derived Exos induce ERK phosphorylation, stimulate myosatellite cell activity, and elevate LPP while reducing fibrosis during muscle repair 58 . Non–stem cell derived EVs also exhibit regenerative potential. Human platelet–derived EVs expressing CD41a and CD9 have demonstrated functional recovery of the external urethral sphincter in porcine SUI models by delivering collagen I and driving skeletal muscle satellite cell proliferation and differentiation via NF-κB signaling. These EVs preserve their lipid bilayer to stabilize collagen delivery, though further validation in chronic SUI models is required 59 .
Despite promising preclinical evidence, this secretome-based approach has not yet been evaluated in clinical settings. Subsequent research should conduct a comprehensive insights of secretory components and their activating signaling pathways, systematically comparing the therapeutic effects of different bioactive factors. Emphasis should be placed on regulating the ECM metabolic balance, improving fibroblast function, promoting collagen and elastin synthesis while inhibiting their degradation, and enhancing the elasticity and structural support of periurethral tissues. These efforts aim to refine regenerative strategies for SUI and achieve precise targeted therapy.
Optimization of delivery system
Current limitations of monotherapy using stem cells include substantial peripheral organ sequestration, diminished tissue retention rates, and transient cellular viability, which collectively hinder comprehensive urethral tissue reconstruction in SUI management. To address these challenges, critical emphasis must be placed on enhancing functional durability and precision differentiation of transplanted cells (Table 2). Recent investigations have advanced two principal optimization approaches: selection of delivery materials and targeted therapeutic strategies.
Preclinical studies of combination therapy based on stem cells.

PNT: pudendal nerve transection; VBD: intravaginal balloon dilatation.
Synergistic Combination Therapeutic Strategies
Biological scaffolds are employed to deliver cells for repairing damaged urethral sphincters. These scaffolds display excellent biocompatibility, promote cellular proliferation and regeneration, and undergo natural degradation to facilitate tissue repair and functional restoration 70 . Fibrin has demonstrated efficacy in wound healing by enhancing myoblast survival and differentiation. Furthermore, fibrin gel exhibits complete biodegradability, which optimizes cell delivery while minimizing foreign body implantation 71 . Combining MDSCs with fibrin glue (FG) yields superior outcomes compared with MDSCs alone. The MDSCs + FG group exhibits increased muscle mass thickness, improved muscle-to-collagen ratio, and elevated neovessel density, indicating that FG supports MDSCs differentiation toward myogenic lineages. In addition, fibrin-stabilizing factor XIII and the arginine-glycine-aspartate motif within FG enhance cell adhesion and proliferation 60 . However, FG exhibits rapid degradation kinetics, resulting in the dissipation of therapeutic effects within 4 weeks. Calcium alginate composite gels represent an alternative cellular carrier. These materials demonstrate prolonged in vivo retention relative to FG, alongside superior biocompatibility and hydrophilic properties, establishing a favorable microenvironment for tissue regeneration. When BMSC-induced myofibroblasts are administered with calcium alginate gel, they differentiate into muscle cells within the SUI microenvironment. Urethral resistance correlates positively with gel volume, suggesting that observed improvements in LPP may primarily reflect the physical bulking effect of the gel rather than myogenic regeneration 61 . Silk fibroin scaffolds derived from silkworms exhibit in vivo retention exceeding 8 weeks, coupled with favorable mechanical properties, biodegradability, and low immunogenicity. These characteristics make them effective carriers for reinforcing urethral junction integrity. When combined with ADSCs, this system restores urethral pressure to baseline levels within 12 weeks while promoting nerve and muscle fiber formation, offering durable therapeutic benefits 62 . ADSCs, as the most extensively utilized cell source, have demonstrated clinical safety when administered with collagen matrices. However, clinical observations indicate that collagen matrices exhibit progressive liquefaction during extended follow-up periods, potentially reducing their volumetric retention capacity 72 .
Bioactive growth factors, as soluble proteins secreted by cells, frequently result in inadequate targeting specificity due to their pleiotropic activation of extracellular and intracellular signaling pathways. By identifying a single, well-defined factor, therapies can achieve greater precision in regulating physiological processes
73
. For instance, neural growth factor (NGF) modulates both neuronal and non-neuronal cells, enhancing stem cell differentiation. Experimental data demonstrate that combining NGF with MDSCs significantly elevates LPP compared with MDSCs alone
63
. In this study, MDSCs and NGF-overexpressing genes were co-delivered via adenoviral vectors, forming a “cell-factor-vector” triple therapy system. However, adenoviral vectors exhibit immunogenic potential, necessitating further investigation into vector-associated inflammatory responses. Alternative delivery systems employ nanospheres as carriers. Insulin-like growth factor-1 (IGF-1), a critical regulator of cell proliferation, skeletal muscle metabolism, and angiogenesis, possesses a small molecular weight that predisposes it to rapid systemic clearance. To address this limitation, researchers developed a controlled release system using sodium alginate-poly-
Targeted Therapeutic Strategies
The improvement of target specificity in stem cell–based therapies is critical to addressing heterogeneous clinical outcomes across diseases. MSCs derived from distinct tissues may exhibit lineage-specific differentiation biases, for instance, BMSCs are prone to ectopic ossification 41 . Furthermore, stem cell therapies employ disease-specific repair mechanisms, and modulating key pathways can enhance therapeutic efficacy. Therefore, advancements in targeting strategies aim to resolve challenges related to inconsistent efficacy, safety risks, and the capacity to adapt to diverse disease mechanisms, thereby advancing precision regenerative medicine.
MicroRNAs (miRNAs) function as central regulators of stem cell differentiation, regeneration, and developmental processes, which has garnered significant research attention due to their therapeutic potential in disease management. MiRNA-mediated targeting strategies, either upregulating or inhibiting specific miRNA expression profiles, can modulate stem cell–derived therapeutic outcomes 75 . For example, MiR-124-3p enhances the survival and myogenic differentiation of ADSCs in vivo by targeting Caveolin-1. This leads to amplified regenerative efficacy in sphincter repair, increased elastin and collagen expression, and promotion of pelvic floor muscle ECM remodeling 67 . Similarly, MiR-181a-5p targets MFN1 to stimulate BMSCs differentiation into fibroblasts, enhance fibroblast-related gene expression, and upregulate collagen production, thereby accelerating structural restoration in injured tissues 68 . However, the effects of miRNAs on non-target tissues and the risk of ectopic differentiation of BMSCs grafts remain incompletely understood. In addition, the efficiency and stability of miRNA delivery in vivo require further validation. Current studies focus on optimizing delivery systems for miRNA mimics and inhibitors through chemical modifications to improve biostability and pharmacokinetic properties. These efforts aim to facilitate the clinical translation of miRNA-based therapeutics for tissue regeneration.
Chemokine signaling plays a pivotal role in MSCs implantation. Chemokine (C-C motif) ligand 7 (CCL7) enhances stem cell homing, implantation, and growth factor secretion, which correlates with improved outcomes in SUI models 76 . At the receptor level, chemokine (C-C motif) receptor 1 (CCR1) improves BMSCs retention and sphincter function when co-administered with CCL7 into the urethra. While these findings suggest a novel therapeutic approach, the molecular mechanisms underlying chemokine–stem cell interactions require further elucidation. Optimizing chemokine activity to enhance stem cell homing presents a promising avenue for improving SUI therapies 69 . CXCL12 has been demonstrated to direct ADSCs migration and colonization in damaged urethral tissues 23 . Subsequent studies reveal that CXCL12 not only improves cell survival at injury sites, resulting in increased LPP and MBV compared with skeletal muscle precursor cell injections, but also promotes sphincter vascularization and motor endplate formation. Research indicates that CXCL12 maintains stable expression in SUI models for 2 weeks post-injection, elevating vascular density and enhancing sphincter morphology 77 . A chronic sphincter injury model developed in non-human primates exhibits pathophysiological features closely resembling those in humans, offering a robust platform for translational research. However, whether CXCL12 acts directly or mediates regeneration via cellular homing mechanisms remains unclear 12 . Phase I and II clinical trials have confirmed the safety of CXCL12, and preclinical investigations utilizing human plasmids expressing CXCL12 in chronic injury models represent a promising direction for future studies 78 . Further research should delineate the specific pathways involved and identify optimal carriers to enable clinical application.
Clinical translational challenges and future directions
SUI is a chronic condition with rising prevalence. Its pathophysiology involves complex etiological factors, and restoring urethral sphincter function necessitates coordinated muscle regeneration, vascular and neural reconstruction, and ECM remodeling. Stem cell therapy demonstrates significant therapeutic potential in treating chronic diseases through mechanisms including targeted migration to injured tissues, stimulation of neuroangiogenesis, immunomodulation, and antifibrotic effects. Regenerative therapies serve as micro-innovative alternatives for diverse pathologies 79 . While preclinical studies have proven the capacity of stem cell–based interventions to improve sphincter function, persistent risks associated with abnormal cell population dynamics remain. In regenerative medicine, transplanted stem cells frequently fail to engraft or differentiate at injury sites, which may result in scar tissue formation or tumorigenesis80,81. As previously discussed, MDSCs may undergo spontaneous fibrosis 20 , and excessive administration of stem cells can induce myocardial fibrosis via paracrine activation of the TGF-β signaling pathway 82 . Therefore, a critical challenge lies in mitigating lineage-specific biases of precursor cell lines and guiding them toward appropriate differentiation pathways. Large-scale clinical trials are essential to establish standardized protocols for safe and reproducible outcomes.
As molecular-level investigations advance, targeted regulation of stem cells and multi-target interventions may become more feasible. Elucidating stem cell differentiation mechanisms and paracrine signaling pathways has enabled the identification of bioactive molecules with reparative properties for clinical application. However, stem cells and their cell-free products derived from stem cells require complex extraction processes, and improving yield remains a primary technical hurdle. Optimizing culture conditions is equally vital to ensure the consistent production of EVs and the stable expression of bioactive factors for injectable therapies. In addition, the mechanism of action of EVs has not been fully elucidated. For instance, the specific signaling pathways involved in USC-Exo-mediated repair of urethral sphincter or connective tissue in SUI, resulting in a lack of targeted therapeutic approaches. And the precise roles of miR-93 and Sirt7 in ECM degradation remain unpredictable. These limitations may hinder their optimized application in SUI treatment15,56,58. Protein stents and nanospheres can serve as delivery systems to extend the therapeutic effects of these interventions while being naturally degradable, thereby minimizing complications such as urinary retention. This offers innovative solutions for optimizing treatment outcomes. Moreover, Targeted therapy can further assist in directing bioactive factors to the site of injury, maximizing their reparative potential. Once validated through clinical trials, such approaches could greatly improve the convenience and accessibility of treatments. Variability in clinical outcomes has been observed, which may be influenced by individual patient differences and the complex pathological factors underlying SUI. Future research could explore the combination of regenerative therapies with conservative treatments, such as electrical stimulation, to achieve synergistic effects52,83. These advancements will further the implementation of personalized therapeutic strategies in clinical practice.
Conclusion
Although cell-free therapies and combination therapies demonstrate promising outcomes, the limited availability of clinical data necessitates further investigation prior to widespread clinical adoption. Standardizing the extraction processes of stem cells and their secretory components is crucial to ensure the sustained efficacy and safety of these treatments. Future research must prioritize elucidating mechanistic pathways, grounding findings in robust preclinical experiments, and rigorously assessing the safety profile and long-term effectiveness of reparative agents. In parallel, the development of humanized urethral sphincter models using organoid technology, will make it possible to enable a deeper understanding of disease progression mechanisms and contribute to advancing precision medicine for SUI. By integrating advancements in biomaterials, stem cell biology, and tissue engineering, multidisciplinary strategies can be developed to achieve significant clinical breakthroughs in the management of SUI.
Footnotes
Acknowledgements
No assistance in the preparation of this article is to be declared.
Ethical Considerations
Not applicable.
Author Contributions
Yao Chen: Writing—original draft, Visualization, Methodology, Formal analysis, Data curation, Conceptualization. Bao Li: Writing—original draft, Supervision, Conceptualization. Xiang Zhao: Writing—original draft, Data curation. Xinyu He: Writing—original draft, Data curation. Wei Gao: Writing—review & editing, Supervision. Qian Li: Writing—review & editing, Supervision, Funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 81802054); the support program for Youth Innovation Technology in Colleges and Universities of Shandong Province of China (grant number 2021KJ106); the Tai-Shan Scholar Program from Shandong Province (grant number tsqn202103116); the Shandong Provincial Natural Science Foundation (grant number ZR2022MH061); the Innovation Entrepreneurship Training Program for College Students of Shandong Province (grant number S202410438028); the Shandong Province Higher Education Youth Innovation Team Development Program; the Science and Technology Development Program, Weifang, China (grant number 2024GX011); and the Graduate Student Research Grant from Shandong Second Medical University (grant number 2024YJSCX020).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and material
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
