Abstract
Tissue engineering with stem cells is a fascinating approach for treating anterior cruciate ligament (ACL) injuries. In our previous study, stem cells isolated from the human anterior cruciate ligament were shown to possess extensive proliferation and differentiation capabilities when treated with specific growth factors. However, optimal culture conditions and the usefulness of fetal bovine serum (FBS) as a growth factor in in vitro culture systems are yet to be determined. In this study, we compared the effects of different culture media containing combinations of various concentrations of FBS and the growth factors basic fibroblastic growth factor (bFGF) and transforming growth factor-β1 (TGF-β1) on the proliferation and differentiation of ligament-derived stem cells (LSCs) and bone marrow mesenchymal stem cells (BMSCs). We found that α-MEM plus 10% FBS and bFGF was able to maintain both LSCs and BMSCs in a relatively undifferentiated state but with lower major extracellular matrix (ECM) component gene expression and protein production, which is beneficial for stem cell expansion. However, the differentiation and proliferation potentials of LSCs and BMSCs were increased when cultured in MesenPRO, a commercially available stem cell medium containing 2% FBS. MesenPRO in conjunction with TGF-β1 had the greatest ability to induce the differentiation of BMSCs and LSCs to ligament fibroblasts, which was evidenced by the highest ligamentous ECM gene expression and protein production. These results indicate that culture media and growth factors play a very important role in the success of tissue engineering. With α-MEM plus 10% FBS and bFGF, rapid proliferation of stem cells can be achieved. In this study, MesenPRO was able to promote differentiation of both LSCs and BMSCs to ligament fibroblasts. Differentiation was further increased by TGF-β1. With increasing understanding of the effects of different culture media and growth factors, manipulation of stem cells in the desired direction for ligament tissue engineering can be achieved.
Keywords
Introduction
Rupture of the anterior cruciate ligament (ACL) is one of the most common injuries encountered in orthopedic practice (4,6). Most ACL injuries do not heal spontaneously and cause residual instability and subsequent disabilities (25,32,45). Currently, ACL reconstruction requires harvesting of autograft material from the patella or hamstring tendon, which may cause donor site morbidity and hinder prompt recovery. In addition, long-term recovery after reconstruction is unsatisfactory because of residual instability and secondary osteoarthritic changes in the knee (1,5,7,12,18,23,24,29,30,33). With the rapid development of functional tissue engineering, there is renewed interest in different approaches to the treatment of ACL rupture. Tissue engineering with stem cells, growth factors, and scaffolds to repair or reconstruct a ruptured ACL instead of traditional autograft reconstruction has been widely investigated. Mesenchymal stem cells (MSCs) and several other types of cells have been used in tissue engineering and show tremendous potential (3,13,15,16,22,26,34,42,43). Combinations of different growth factors with basal media have been widely used in stem cell culture to expand or induce the differentiation of stem cells for tissue engineering (14,19,20,31,40). Basic fibroblast growth factor (bFGF) and transforming growth factor-β1 (TGF-β1) have been shown to increase the proliferation and differentiation of stem cells. Both of these growth factors also seem to be involved in the expression of genes related to the synthesis and degradation of the extracellular matrix (ECM) (8,9).
Our laboratory recently reported that stem cells isolated from the human ACL have higher proliferation and differentiation capabilities when treated with growth factors, a property that is beneficial for ligament tissue engineering (8–10). Although the effects of culture media on human bone marrow mesenchymal stem cells (BMSCs) have been investigated previously, the influence of different culture media and growth factors on the proliferation and differentiation of ligament-derived stem cells (LSCs) has not been addressed to date (8–10,19,20,37,38). Recently, commercially produced media, such as MesenPRO, have become available for the culture of human stem cells. These media might serve as a promising standard against which to compare the results of stem cell research in different laboratories. MesenPRO and TGF-β1 have been found to be the most potent inducers of smooth muscle cell differentiation in stem cell culture (17). However, the effects of MesenPRO on stem cells derived from ACL and bone marrow have not yet been reported. The aim of this study was to assess two different culture media in combination with growth factors, regarding their support of stem cell expansion and ligament differentiation from cultures of human LSCs and BMSCs.
Materials and Methods
Isolation of BMSCs and LSCs
Approval from the National Yang-Ming University's Institutional Review Board was obtained before starting the study. ACL tissue and bone marrow were collected from five donors (three males and two females, 21–39 years of age) aseptically through arthroscopic procedures after informed consent was obtained. The diagnosis was ACL rupture in all patients. Human LSCs and BMSCs were harvested from the same donor, as described previously (8–10,37,38). In brief, for LSC isolation, ACL ligament tissue samples were washed repeatedly in phosphate-buffered saline (PBS; Gibco, Grand Island, NY, USA). The tissue was minced into small pieces and digested with 1% collagenase in α-modified minimum essential medium (α-MEM; Gibco) containing 10% heat-inactivated FBS (Sigma-Aldrich, St Louis, MO, USA). Following overnight incubation at 37°C, cells were collected by centrifugation and washed repeatedly in PBS. The cells were plated at 5,000 cells/cm2 in T75 flasks (Becton Dickinson, Franklin Lakes, NJ, USA) with a growth medium (α-MEM supplemented with 10% FBS) as passage 0. To isolate BMSCs, bone marrow was aspirated from the distal femur when drilling the femoral tunnel for ACL reconstruction. Nucleated cells were isolated with a density gradient medium (Ficoll-Paque; Pharmacia, Uppsala, Sweden) and resuspended in a complete culture medium (α-MEM supplemented with 20% FBS). After 24 h, nonadherent cells were discarded, and adherent cells were washed with PBS. These adherent cells were defined as passage 0 cells. Once adherent passage 0 cells from ACL and bone marrow reached semiconfluence, cells were detached with 0.25% trypsin-ethylenediaminetetraacetic acid (EDTA; Gibco), washed twice with PBS, centrifuged at 1,000 rpm (200 × g) for 4 min, and replated as passage 1 at 50 cells/cm2 in each T75 flask with MesenPRO (MesenPRO RS™ with 2% FBS; Invitrogen, Carlsbad, CA, USA) or α-MEM plus 10% FBS. After an additional 14 days of growth, cells were harvested by washing aliquots of the trypsin-detached cells twice with PBS and cryopreserved in liquid nitrogen in FBS with 10% di methyl sulfoxide (DMSO; Sigma-Aldrich) (passage 1). The immunophenotypes and multilineage differentiation capabilities of these cells were confirmed, as reported previously (8,9). In brief, for immunophenotyping, stem cells from passages 4–6 were stained with fluorescein-or phycoerythrin-coupled antibodies and analyzed with a FACS Caliber (Becton Dickinson). Antibodies against human cluster of differentiation 14 (CD14), CD29, CD34, CD44, CD45, CD73, CD90, CD105, CD166, human leukocyte antigen (HLA)-ABC, and HLA-DR were all purchased from Becton Dickinson. For multilineage differentiation, stem cells from passages 4–6 were cultured in six-well plates (Becton Dickinson) at a density of 3,000 cells/cm2 for osteogenic differentiation and a density of 5,000 cells/cm2 for adipogenic differentiation. For chondrogenic differentiation, 5 × 105 of these cells in a 15-ml propylene tube (Becton Dickinson) was centrifuged at 500 rpm (100 × g) to form a pellet. These cells were cultured in osteogenic, adipogenic, and chondrogenic media for 3 weeks. Immunophenotypical analysis revealed that these cells were positive for CD29, CD44, CD73, CD90, CD105, CD166, and HLA-ABC, but negative for CD14, CD34, CD45, and HLA-DR. In osteogenic differentiation experiments, the cells were positively stained with Alk-p (Sigma-Aldrich) and alizarin red-s (Sigma-Aldrich), and the mRNA expression of osteocalcin and type I collagen were significantly increased when compared with the control group. In adipogenic differentiation experiments, mRNA expression of peroxisome proliferator-activated receptor γ (PPAR-γ) and fatty acid-binding protein (FABP) were significantly increased when compared with control group, and positive staining with oil red O (Sigma-Aldrich) was detected. In chondrogenic differentiation experiments, mRNA expression of type II collagen and cartilage oligomeric matrix protein (COMP) was significantly increased when compared with control group, and the pellets had a positive staining with alcian blue (Sigma-Aldrich). Primers used for confirming differentiation by RT-PCR are shown in Table 1. After their immunophenotyping and multilineage differentiation capabilities were confirmed, the cells were defined as LSCs or BMSCs. To expand the cells, a vial of cryopreserved cells was thawed and plated at a concentration of 5,000 cells/cm2 in a T75 flask with MesenPRO or α-MEM plus 10% FBS. Serial passages of the cells were performed using the same culture media.
The Primers Used to Confirm Differentiation Abilities of the Cells

PPAR-γ, peroxisome proliferator-activated receptor γ; FABP, fatty acid binding protein 4; COL1A1, collagen type I, α 1; COL2A1, collagen type II, α 1; COMP, cartilage oligomeric matrix protein.
Colony-Forming Assay
Passage 1 LSCs and BMSCs were diluted and replated onto 100-mm culture plates (Becton Dickinson) at a concentration of 200 cells/per plate in MesenPRO or α-MEM plus 10% FBS. The experiment was performed in triplicate for each donor. After 14 days, colonies that had formed from single cells were visualized by light microscopy. The colonies were fixed with 4% formaldehyde (Sigma-Aldrich) and then stained with 0.5% crystal violet (Sigma-Aldrich) in methanol (Avantor Performance Materials, Center Valley, PA, USA) for 5 min. The plates were washed twice with distilled water, and the number of colonies per dish was determined. Colonies of <2 mm in diameter or those that were faintly stained were ignored (8,9,38).
Population Doublings (PD) of Cells
To examine the proliferation and PD capacity of LSCs and BMSCs, passage 1 cells from the five donors were replated at 5,000 cells/cm2 in six-well plates in MesenPRO or α-MEM plus 10% FBS and serially passaged. Media were changed twice a week. The cells were counted and replated at the same density when confluence was reached. We calculated PD value for each passage using the formula PD = log2(NC/N0), where N0 is the inoculum cell population and NC the number of cells at confluence, and summed the values from each passage to obtain the final PD value. PD times (PDT) of all five donors were calculated and averaged.
Effects of Growth Factors on Cell Proliferation
The effects of growth factors on the proliferation of LSCs and BMSCs were determined using a commercially available 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt (MTS) kit (CellTiter 96 AQ; Promega Corp., Madison, WI, USA). In brief, 10,000 cells were placed in each well of a 96-well plate (Becton Dickinson). After stimulation with the growth media (MesenPRO or α-MEM plus 10% FBS) only or supplemented with either 10 ng/ml bFGF (Sigma-Aldrich) or 10 ng/ml TGF-β1 (eBioscience, San Diego, CA, USA) for 72 h, the relative cell number was estimated by measuring absorbance at 490 nm on a microplate reader (U3300 Pro; Amersham Biosciences, Freiburg, Germany).
Effects of Growth Factors on Cell Density
In each well of six-well plates, either 4,000 LSCs or BMSCs from passages 4–6 were placed and cultured with growth media (MesenPRO or α-MEM plus 10% FBS) with or without additional growth factors. The experimental treatment consisted of the growth media supplemented with 10 ng/ml bFGF or 10 ng/ml TGF-β1. The control groups were growth media without additional growth factors. Culture media were changed twice a week. The number of cells was determined on days 7 and 14 by trypan blue (Sigma-Aldrich) exclusion in a Neubauer counting chamber (Paul Marienfeld, Lauda-Königshofen, Baden-Württemberg, Germany). Three independent experiments were performed at each time point.
Quantification of Collagen and Total ECM Proteins
Collagen and proteins were quantified by colorimetric analyses, as described previously (21,27,41). The method used the selective binding of Sirius Red F3BA (Sigma-Aldrich) to collagen and Fast Green FCF (Sigma) to noncollagen protein when both are dissolved in aqueous saturated picric acid (Sigma-Aldrich). In brief, 4,000 LSCs and BMSCs were placed in each well of six-well plates and cultured with growth media (MesenPRO or α-MEM plus 10% FBS) with or without growth factors, similar to the cell density study. After 1 or 2 weeks of culture, cells in the six-well culture plates were incubated with 1 ml of saturated picric acid solution that contained 0.1% Sirius Red F3BA and 0.1% Fast Green FCF. The plates were incubated at room temperature for 30 min in a rotary shaker. The fluid was then carefully withdrawn, and the plates were washed repeatedly with distilled water until the fluid was colorless. After washing with water, 1 ml of 1:1 (v/v) 0.1% NaOH (Mallinckrodt Pharmaceuticals, St. Louis, MO, USA) and absolute methanol was added to the plates to elute the color. The eluted color was read immediately using a spectrophotometer (BIO-RAD, Philadelphia, PA, USA) at 540 and 605 nm (U3300 Pro). The cell numbers calculated in the cell density study were used to normalize and calculate collagen and noncollagen synthesis, and the data were presented as collagen and noncollagen protein synthesis per cell.
Real-Time Reverse Transcription-Polymerase Chain Reaction (RT-PCR) Analysis
In each well of the six-well plates, 4,000 LSCs and BMSCs were placed and cultured with media condition similar to the cell density study. Culture media were changed twice per week. Cells were harvested at days 7 and 14. Total RNA from the cells in each group was extracted using a commercially available kit (S.N.A.P.; Invitrogen, Groningen, The Netherlands), according to the manufacturer's instructions. Complementary DNAs (cDNAs) were obtained by RT of 1 μg total RNA using Advantage RT-for-PCR (Clontech, Palo Alto, CA, USA), according to the manufacturer's instructions. Quantitative PCR was performed on a Roche LightCycler® 480 (Roche Diagnostics, Laval, Quebec, Canada) real-time PCR system. The primer sequences were designed using the Universal ProbeLibrary for Humans (Roche Applied Science, Laval, Quebec, Canada) using nucleotide sequences deposited in the GenBank database. Primer sequences and PCR conditions are described in Table 2. Calculations for the determination of the relative level of gene expression were made using the cycle threshold (Ct) method. The mean Ct value from duplicate measurements was used to calculate the expression of the target gene, after normalization to a housekeeping control [glyceraldehyde 3-phosphate dehydrogenase (GAPDH)]. This value was expressed as a fold change compared to that of samples without additional growth factors. Negative control experiments without an RNA template and without addition of the reverse transcriptase enzyme were also performed.
Primers Used for Real-Time Reverse-Transcription Polymerase Chain Reaction (RT-PCR) Analysis

COL1A1, collagen type I, α 1; COL3A1, collagen type III, α 1; α-SMA, α-smooth muscle actin; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; Tm, melting temperature (°C).
Statistical Analysis
Results are shown as mean ± standard deviation. Statistically significant differences between groups were analyzed using the independent-sample t test and multivariate analysis of variance (ANOVA), followed by univariate ANOVA and Tukey's post hoc test (SPSS version 11.5; SPSS, Chicago, IL, USA). A value of p < 0.05 was considered statistically significant.
Results
Morphology and Proliferation of Cells
LSCs and BMSCs were extensively subcultured for about 20–22 passages in α-MEM plus 10% FBS or MesenPRO. Both LSCs and BMSCs were adherent to plastic and were spindle shaped, although a more extended cell shape with cell aggregation was noted in the culture with MesenPRO. LSCs and BMSCs cultured with growth media plus bFGF had higher cell densities. LSCs and BMSCs treated with growth media plus bFGF developed smaller cells with short cell processes, whereas those cultured in growth media plus TGF-β1 had a more flattened cell shape with longer cell processes; the latter group of cells tended to aggregate to form linear or circular multiple cell complexes with TGF-β1 treatment (Fig. 1A, B).

Morphology of LSCs and BMSCs. In each well of a six-well plate, 4,000 ligament-derived stem cells (LSCs) and bone marrow mesenchymal stem cells (BMSCs) from passages 4–6 were placed and cultured with growth media [a-modified minimum essential medium (α-MEM; a–c) plus 10% fetal bovine serum (FBS) or MesenPRO (d–f)] with or without growth factors (a, d). The experimental treatment was the growth media supplemented with 10 ng/ml basic fibroblast growth factor (bFGF; b, e) or 10 ng/ml transforming growth factor (TGF)-b1 (c, f). The control treatment lacked growth factors. Culture media was changed twice a week. The morphology of clonally expanded cells derived from anterior cruciate ligament (ACL; A) and bone marrow (B) were photographed. The cells were spindle shaped, similar to fibroblasts, and were adherent to plastic. LSCs and BMSCs at the first week were photographed in both the control and growth factor groups. Cell density and morphology differed, according to the growth factor used for treatment. Scale bars: 200 μm.
MesenPRO and bFGF Increased Cell Proliferation
Compared with the control group (α-MEM plus 10% FBS), the group treated with MesenPRO (with or without growth factors) showed significantly increased numbers of LSCs and BMSCs in the MTS assay. Cell numbers were also increased by treatment with α-MEM plus 10% FBS and bFGF after 3 days. The addition of TGF-β1 to α-MEM plus 10% FBS had no significant effect on either LSC or BMSC proliferation after 72 h of culture (Fig. 2A). At the end of the first week, the highest numbers of LSCs and BMSCs were found in MesenPRO plus bFGF, followed by α-MEM plus 10% FBS and bFGF. MesenPRO plus TGF-β1 increased LSC proliferation compared with α-MEM plus 10% FBS and TGF-β1, but there was no significant difference in BMSC proliferation (Fig. 2B). After 2 weeks of culture, LSC numbers were significantly higher in the MesenPRO plus bFGF group, MesenPRO plus TGF-β1 group, and the α-MEM plus 10% FBS and bFGF group. However, the MesenPRO plus bFGF group was the only group that had significantly higher BMSC numbers than the control group. In the first and second weeks, the treatment of MesenPRO plus TGF-β1 significantly increased the LSC cell number than in the control group, but no significant differences in BMSC numbers were found between the MesenPRO plus TGF-β1 and control groups (Fig. 2B, C).

Proliferation of LSCs and BMSCs under bFGF and TGF-β1 treatment. (A) In each well of a 96-well plate, 10,000 LSCs and BMSCs from passages 4-6 from all five donors were placed, deinduced, and then cultured in growth media with or without 10 ng/ml of bFGF or TGF-β1 for 72 h (the first three bars in each figure indicate α-MEM with or without growth factors; the next three bars indicate MesenPRO with or without growth factors). Relative cell numbers were calculated by measuring the absorbance of each well at 490 nm. Five independent experiments were performed for each donor. Results are expressed as mean ± standard deviation. The control treatment was α-MEM plus 10% FBS without growth factors **(p < 0.01, *p < 0.05). (B, C) Cumulative cell density at 1 and 2 weeks, respectively. Each experiment was performed in triplicate for each donor. Each bar indicates the average cell number for five donors with the same treatment. The control treatments were α-MEM plus 10% FBS without additional growth factors (**p < 0.01, *p < 0.05).
Higher Clonogenic Efficiency of α-MEM with FBS Compared with MesenPRO
The average colony-forming efficiency (mean ± standard deviation for five donors in each group) for LSCs was 39.5 ± 9.23 colonies/200 cells in α-MEM plus 10% FBS while only 16.8 ± 7.34 colonies/200 cells in MesenPRO. The average colony-forming efficiency for BMSC was 28.1 ± 5.17 in α-MEM plus 10% FBS, while only 10.8 ± 5.65 colonies/200 cells in MesenPRO. LSCs cultured in α-MEM plus 10% FBS had significantly greater colony-forming ability than LSCs cultured in MesenPRO (p = 0.02) and BMSCs cultured in MesenPRO (p = 0.002). Average colony size did not differ significantly between the groups (Fig. 3A).
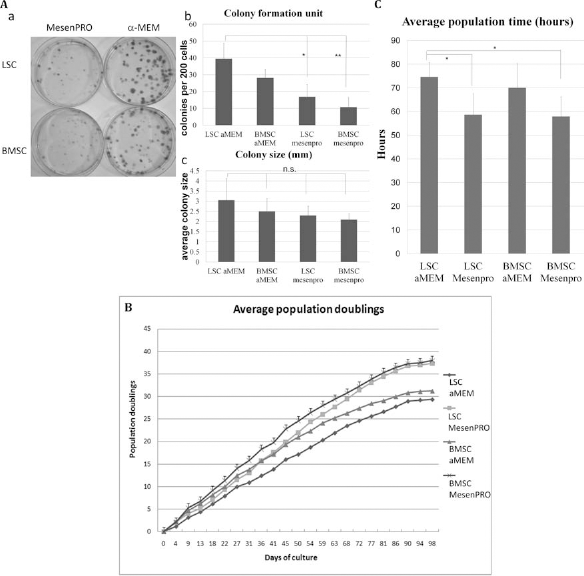
Colony-forming ability of LSCs and BMSCs with α-MEM plus 10% FBS or MesenPRO. (A) These plates indicate colonies formed by single LSCs and single BMSCs treated with MesenPRO or α-MEM plus 10% FBS (a). Average colony-forming efficiency was determined by crystal violet staining after 2 weeks of culture (b). Results are mean ± standard deviation for five donors in each group. Average sizes of colonies formed by LSCs and BMSCs were also compared (c). Passage 1 stem cells were used in this experiment. (B, C) Serial passage and population doublings (PDs) of LSCs and BMSCs. Both LSCs and BMSCs could be serially passaged in vitro with α-MEM (plus 10% FBS) or MesenPRO from passage 1. PD times were estimated for BMSCs and LSCs with α-MEM plus 10% FBS and MesenPRO. LSCs cultured in α-MEM plus 10% FBS showed significantly longer PD times than BMSCs cultured in MesenPRO (p = 0.026) as well as LSCs cultured in MesenPRO (p = 0.036) (C).
Shorter PDTs with MesenPRO
Both LSCs and BMSCs could be serially passaged in vitro for more than 90 days with more than 20 PDs with α-MEM plus 10% FBS or MesenPRO. PDT was estimated to be 70.1 ± 10.3 and 57.9 ± 8.3 h for BMSCs with α-MEM plus 10% FBS and MesenPRO, respectively, and 74.6 ± 6.1 and 58.7 ± 8.9 h for LSCs with α-MEM plus 10% FBS and MesenPRO, respectively. PDTs were significantly shorter in LSCs and BMSCs cultured in MesenPRO than in LSCs cultured in α-MEM plus 10% FBS (Fig. 3B, C).
MesenPRO with TGF-β1 Increased Collagen and Total ECM Protein Production
Phase contrast microscopic examination showed strong positive staining in the TGF-β1 group, followed by the bFGF and control groups (Fig. 4A). After 2 weeks of treatment, spectrophotometry showed that the MesenPRO plus TGF-β1 groups of LSCs and BMSCs showed a significantly higher noncollagen ECM protein production per cell than the control groups. LSCs in the α-MEM plus 10% FBS and TGF-β1 group had significantly higher noncollagen protein production than the control group. For BMSCs, significantly higher collagen production was noted in the MesenPRO plus TGF-β1 group, whereas the α-MEM plus 10% FBS and bFGF group showed significantly lower collagen production than the control groups. There was no significant difference in collagen production for LSCs between different treatments. The highest levels of noncollagen protein and collagen production were noted in LSCs treated with MesenPRO plus TGF-β1 (34.4 ± 4.6 and 2.1 ± 0.3 ng/per cell, respectively). The lowest levels of noncollagen protein and collagen production were noted in BMSCs treated with α-MEM plus 10% FBS and bFGF (12.2 ± 3.7 and 0.6 ± 0.2 ng/per cell, respectively) (Fig. 4B).

Collagen and noncollagen protein production. (A) In each well of a 12-well plate, 2,000 LSCs and BMSCs were placed and cultured in α-MEM plus 10% FBS (a–c, g–i) or MesenPRO (d–f, j–l) with or without additional growth factors [10 ng/ml bFGF (b, e, h, k) or 10 ng/ml TGF-β1 (c, f, i, l)] for 2 weeks. The control treatments were growth media without additional growth factors (a, g). LSCs and BMSCs from passages 4–6 were used in this experiment. Collagen and noncollagen protein production was determined by staining with Sirius Red and Fast Green at day 14 of culture. Micrographs were taken after staining. Scale bars: 100 μm. (B) After staining and photographing, the stains of the culture plates were eluted, and absorbance was determined using a spectrophotometer at 540 and 605 nm. Relative concentrations of noncollagen and collagen protein with different growth factors after culture for 2 weeks were compared. The control group was treated with α-MEM plus 10% FBS without additional growth factors (**p < 0.01, *p < 0.05).
MesenPRO Plus TGF-β1 Promotes Stem Cell Differentiation to Ligament Fibroblasts
Relative gene expression levels of collagen type I, collagen type III, fibronectin, α-smooth muscle actin (α-SMA), and tenascin-c in the control and growth factor treatment groups from 1 to 2 weeks are shown in Figure 5A–E, respectively. All the above-mentioned molecules are major ECM constituents in human ligaments. The mRNA expression of type I collagen was significantly increased in LSCs and BMSCs by treatment with TGF-β1 plus MesenPRO in the first and second weeks. α-MEM plus 10% FBS and bFGF had a less pronounced effect than MesenPRO on the increase in type I collagen expression in both LSCs and BMSCs. For both LSCs and BMSCs, the lowest type I collagen expression was noted in the α-MEM plus 10% FBS and bFGF groups followed by MesenPRO plus bFGF groups, but the differences in the expression between the groups and control was not statistically significant (Fig. 5A). For LSCs, compared with the control groups, type III collagen mRNA expression was significantly increased by treatment with MesenPRO and that with MesenPRO plus TGF-β1 in the first and second weeks. Type III collagen expression was increased in BMSCs after treatment with MesenPRO plus TGF-β1 in the first and second weeks and that with MesenPRO alone in the second week. In the second week, the α-MEM plus 10% FBS and bFGF group for LSCs showed significantly lower type III collagen expression than the control group; type III collagen expression was also decreased in both LSCs and BMSCs by treatment with MesenPRO plus bFGF, but the differences in expression between the groups and control was not statistically significant (Fig. 5B). Fibronectin mRNA expression was significantly increased above the control level only in LSCs treated with TGF-β1 plus MesenPRO in the second week. There were no significant differences in fibronectin mRNA expression among the different treatment groups for BMSCs in the first and second weeks or for LSCs in the first week when compared with control group (Fig. 5C). Significantly higher levels of α-SMA mRNA expression was observed in the MesenPRO plus TGF-β1 group for LSCs in the second week as well as in the MesenPRO plus TGF-β1 group for BMSCs in the first and second weeks compared with the control groups (Fig. 5D). Tenascin-c mRNA expression in LSCs was increased by treatment with MesenPRO plus bFGF in the first and second weeks and by MesenPRO plus TGF-β1 in the second week. In BMSCs, tenascin-c mRNA expression was significantly higher in the MesenPRO plus TGF-β1 group for the first and second weeks as well as in the MesenPRO plus bFGF group in the first week (Fig. 5E).

Relative transcript levels of genes with different treatments. In each well of a six-well plate, 4,000 LSCs and BMSCs from passages 4-6 were placed and cultured in α-MEM plus 10% FBS or MesenPRO with or without additional growth factors (10 ng/ml bFGF or 10 ng/ml TGF-β1) for 1 and 2 weeks. The control treatments were growth media without additional growth factors. LSCs and BMSCs from passages 4-6 were used in this experiment. Cells from each donor were harvested weekly, and the relative levels of collagen type I (A), collagen type III (B), fibronectin (C), α smooth muscle actin (α-SMA; D), and tenascin-c (E) were evaluated. All transcript levels were normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) transcript production. The control group was treated with α-MEM plus 10% FBS without additional growth factors (**p < 0.01, *p < 0.05).
Discussion
The ability of stem cells to undergo robust in vitro proliferation and differentiation upon various manipulations is a prerequisite in tissue engineering. Large-scale stem cell expansion with high capacity for differentiation and abundant secretion of ECM protein is also indispensable for successful tissue engineering. Various basal media with combinations of FBS with different growth factors have been evaluated for their ability to promote the in vitro proliferation and differentiation of stem cells. However, the results of different studies are difficult to compare because of differences in experimental conditions. In previous reports and our experiments, α-MEM plus 10% FBS was used for the in vitro culture of LSCs, synovial tissue stem cells, and BMSCs (8–10,37,38). MesenPRO, a commercially available medium with 2% FBS, has been used in the laboratory for stem cell culture and related experiments (17). However, the effects of MesenPRO on stem cell proliferation and differentiation in comparison with other culture media are not well understood. In this study, we compared MesenPRO with our homemade medium, α-MEM plus 10% FBS, alone or in combination with bFGF or TGF-β1, to elucidate whether MesenPRO is able to replace current media in stem cell research after eliminating the difficulties in interpretation of different results resulting from the use of different media.
Growth factors are cell-secreted peptide hormones that control cell function in an autocrine or paracrine manner. Several growth factors, including bFGF and TGF-β1, have been shown to be important in regulating stem cell proliferation and differentiation as well as the formation of ECM (8,9,39,44). bFGF is a strong mitogen for stem cells and has been used in stem cell culture studies to increase cell proliferation and maintain stem cell multipotency (35,40). TGF-β1 has been widely used in the induction of proliferation and differentiation of mesenchymal progenitor cells and ACL fibroblasts in vitro. It has been demonstrated to be able to increase the production of ECM protein in connective tissues (2,28,36).
In the in vitro stem cell cultures described in the present work, MesenPRO increased the proliferation of both LSCs and BMSCs in comparison with α-MEM plus FBS. PDTs were also decreased by treatment with MesenPRO. However, the colony-forming abilities of both LSCs and BMSCs were significantly decreased by MesenPRO. In addition, for both LSCs and BMSCs, MesenPRO plus bFGF was a more potent stimulator of proliferation than α-MEM plus bFGF. With a combined treatment of TGF-β1 plus MesenPRO, no clear increase in proliferation was noted in BMSCs compared with treatment with MesenPRO alone. However, LSC proliferation was increased by treatment with MesenPRO plus TGF-β1 compared to that with MesenPRO alone. Thus, MesenPRO is able to increase the proliferation ability of LSCs and BMSCs as well as decrease the PDTs of both LSCs and BMSCs. Nevertheless, the colony-forming ability of both LSCs and BMSCs was decreased by treatment with MesenPRO, which might indicate a decrease in pluripotency.
The production of collagen and noncollagen protein per cell was increased and was the highest with MesenPRO plus TGF-β1 treatment for both LSCs and BMSCs. Treatment with bFGF combined with either α-MEM or MesenPRO decreased collagen and noncollagen protein production in BMSCs and LSCs. Fibroblast differentiation was strongly enhanced both in LSCs and BMSCs with the addition of TGF-β1 to MesenPRO, but not to bFGF or α-MEM plus FBS. In addition, both LSCs and BMSCs had relatively low ECM gene expression and ECM production after treatment with bFGF or α-MEM plus FBS. Addition of bFGF significantly increased the proliferation and cell density of LSCs and BMSCs without increasing the gene expression levels of type I collagen, type III collagen, fibronectin, and α-SMA in BMSCs and LSCs.
Gong et al. reported that MesenPRO and TGF-β1 were the most potent inducers of human BMSCs for differentiation into smooth muscle cells, whereas their homemade medium (10% FBS with Dulbecco's modified Eagle's medium) could maintain human BMSCs in an undifferentiated state (17). However, only semiquantitative immunofluorescence staining for SMA and calponin and Western blotting for smooth muscle-related proteins (SMA, SMA22α, and calponin) were evaluated in their study. In our study, the results were confirmed by quantitative data in the form of significant increases in collagen and noncollagen production protein per cell and type I collagen, type III collagen, α-SMA, and tenascin-c in real-time RT-PCR after treatment with MesenPRO plus TGF-β1.
A number of experiments have been performed to determine the optimal conditions and materials for ligament tissue engineering (3,9–13,42,44). In this study, we compared human LSCs and BMSCs from the same donors to evaluate the proliferation, ECM protein production per cell, and gene expression after treatment with different growth factors and culture media, aiming to determine the optimal culture conditions for the different stages of ACL tissue engineering. We found that a combination of α-MEM plus FBS and bFGF was able to maintain both LSCs and BMSCs in a relatively undifferentiated state with low major ECM gene expression and protein production, which is beneficial for stem cell expansion with limited differentiation. On the other hand, MesenPRO plus TGF-β1 had the greatest ability to trans-differentiate BMSCs and LSCs to ligament fibroblasts, as evidenced by the highest ECM protein production and gene expression.
Three-dimensional (3D) growth and differentiation of stem cells are important for successful ligament tissue engineering. Fan et al. set up a 3D culture system that was constructed from a silk cable-reinforced gelatin/silk fibroin scaffold. MSCs were seeded in the 3D scaffold and then were either cocultured or not cocultured with ligament fibroblasts for 2 weeks. Cocultured MSCs showed faster proliferation and more collagen production compared with MSCs that were not cocultured. The ligament ECM-specific genes, including type I collagen, type III collagen, and tenascin-c, were expressed in MSCs cocultured with ligament fibroblast but not in non-cocultured MSCs. The behavior of MSCs cocultured with ligament fibroblasts in this 3D system was similar to that of our stem cells cultured in MesenPROplus TGF-β1. Further evaluation of the effect of different culture media and growth factors in the 3D coculture system would be a useful step in the pursuit of successful ligament tissue engineering and will be one of the main foci in our following study (15).
In summary, culture media and growth factors play very important roles in successful tissue engineering. With careful selection, conventional media such as α-MEM plus FBS can keep stem cells undifferentiated more successfully than commercial media. The addition of certain growth factors such as bFGF can further increase proliferation of stem cells without promotion of stem cell differentiation. However, certain commercially available media such as MesenPRO may be able to promote stem cell proliferation and differentiation, and their effect maybe further increased by the addition of TGF-β1. Increasing understanding of the effects of different growth media and growth factors may improve our ability to manipulate stem cells toward clinical applications.
Footnotes
Acknowledgments
This work was supported in part by the UST-UCSD International Center of Excellence in Advanced Bio-engineering sponsored by the Taiwan National Science Council I-RiCE Program under Grant Number NSC100-2911-I-009-101. The authors also acknowledge financial support from the Taipei Veterans General Hospital (VGH101E1-012, VGH101C-015, and VN101-07), the National Science Council, Taiwan [NSC101-2120-M-010-002, NSC100-2314-B-010-030-MY3, NSC101-2321-B-010-009, NSC 101-2911-I-010-503, Taoyuan General Hospital (PTH10129), and NSC 99-3114-B-002-005], the Ministry of Economic Affairs, Taiwan(101-EC-17-A-17-S1-203), and a Grant from National Research Program for Biopharmaceuticals (DOH101-TD-PB-111-NSC014), as well as the National Yang-Ming University/Cheng Hsin General Hospital Grant (101F195CY07). This study was also supported by the Aim for the Top University Plan from the Ministry of Education. The authors declare no conflicts of interest.
