Abstract
Mechanical stimulation is a known modulator of survival and proliferation for many cells, including endothelial cells, smooth muscle cells, and bone marrow-derived mesenchymal stem cells. In this study, we found that mechanical strain prevents apoptosis and increases the adhesive ability of dermal fibroblasts in vitro and thus confers the survival advantage in vivo after transplantation of fibroblasts into the full-thickness wound of diabetic mice. Cyclic stretch at a frequency of 0.5 Hz and maximum elongation of 20% stimulates cellular survival mediated by the activation of extracellular signal-regulated kinases (ERKs), c-Jun N-terminal kinases (JNKs), and the serine/threonine kinase Akt (AKT). Stretching of the fibroblasts increases the synthesis of extracellular matrix proteins and the formation of denser focal adhesion structures, both of which are required for fibroblast adhesion. The stretched fibroblasts also upregulate the expression of vascular endothelial growth factor (VEGF) and stromal cell-derived factor-1α (SDF-1α), which enhanced wound healing in vivo. Indeed, preconditioning with mechanical stretch allows better survival of the transplanted fibroblasts, when compared to unstretched control cells, in the wound environment of mice with streptozotocin-induced diabetes and thus accelerates the wound-healing process in these mice.
Introduction
Human cells receive mechanical stimuli throughout their life from the beginning of the early developmental stage. Mechanical stimuli affect various fundamental cellular processes, such as development, migration, determination of mitotic spindle position, proliferation, apoptosis, adhesive interaction, the extracellular matrix (ECM), paracrine factor production, and pathogenesis (6,10,14,16,17,20,24,31,48,53). Current studies on mechanical stimuli focus on the molecular mechanisms by which cells sense the mechanical stimulus, transduce the mechanical signal into the intracellular chemical signal, and respond to the mechanical stimulus in a physiological environment (41,50).
Many clinical trials on the transplantation of cells isolated from various tissues into wound sites have been performed (23). Survival and engraftment of the transplanted cells are very critical to successful cell therapy. However, the rate of survival/engraftment is remarkably low owing to the unfavorable wound environment caused by inflammation and low blood flow following poor angiogenic response. One typical example of an unfavorable wound environment is the diabetic wound (15). Poor skin wound healing in diabetes has been thought to result in part from impaired neovascularization (7,15,29). Several studies yielded preliminary results to support wound-healing enhancement by transplantation of various stem cells or precursors, such as circulating endothelial precursor cells (EPCs) and bone marrow-derived mesenchymal stem cells, into diabetic skin wounds (3,5,44,46). The transplanted cells are able to heal the diabetic wounds through differentiation into vascular and nonvascular cells and/or production of paracrine factors. For example, transplanted EPCs stimulate the migration of endothelial cells by releasing vascular endothelial growth factor (VEGF) and stromal derived factor-1 (SDF-1), both of which enhance angiogenesis (5,22,49). However, a major problem in cell therapy for diabetes is low survival rates of transplanted cells in the endogenous diabetic wound environment due to factors such as low oxygen level, high glucose level, and accumulated reactive oxygen species (ROS) (4,30,47). Therefore, several trials to promote the survival of the transplanted cells have been attempted (23).
A strategy to enhance the survival of transplanted cells might be to precondition the cells with mechanical strain prior to transplantation because mechanical strain affects cellular proliferation and apoptosis of various cells, including human mesenchymal stem cells (19,21), endothelial cells (27,37), and human skin keratinocytes (52). The mechanical strain is transduced inside the cells through various intracellular signals to affect apoptosis and proliferation (20). Mechanical strain has also been reported to induce proliferation of human dermal fibroblasts through the activation of extracellular signal-regulated kinase (ERK), p38 mitogen-activated protein kinase (MAPK), and V-akt murine thymoma viral oncogene homolog 1 (AKT) (36). However, whether mechanical strain increases the cellular survival of dermal fibroblasts remains unclear.
Adult skin wound healing is a complex process requiring the collaborative efforts of several different types of cells. During the wound-healing process, each of the various types of cells contributes to proliferation, migration, and contraction, as well as the synthesis of growth factors, matrix elements, and proteases at a wound site (34). In particular, dermal fibroblasts play essential roles in the repair of skin wounds through remodeling of the wound bed by synthesis of new ECM and growth factors and the formation of thick actin bundles (43). In this study, we examined the effects of cyclic equibiaxial stretch on the cellular proliferation, apoptosis, adhesive ability, and paracrine factor production of human dermal fibroblasts. Moreover, we evaluated whether preconditioning of fibroblasts with mechanical stretch provided an advantage in cell survival following transplantation and accelerated the wound-healing process in diabetic mice.
Materials and Methods
Primary Cell Culture
Human dermal fibroblasts (hDFs) isolated from circumcised foreskins were purchased from MCTT (Seoul, Korea), and mouse enhanced green fluorescent protein (EGFP)–dermal fibroblasts (mDFs) were isolated from 8- to 10-week-old FVB.Cg-Tg(ACTB-EGFP)B5Nagy/J strain [EGFP directed to widespread tissues by the cytomegalovirus-immediate early (CMV-IE) enhancer/chicken β-actin/rabbit β-globin hybrid promoter strain of Friend leukemia virus, strain B] mice (The Jackson Laboratories, Bar Harbor, ME, USA) through the treatment of type I collagenase (Washington Biochemical Corp., Freehold, NJ, USA). The fibroblasts were cultured in fibroblast growth media (FGM2) containing 2% fetal bovine serum, 5 mg/ml of insulin, 1 ng/ml of basic fibroblast growth factor (bFGF; all from Lonza, Walkersville, MD, USA) at 37°C in a humidified atmosphere of 5% CO2. The medium was exchanged every 2 days, and the cells were split at 80–90% confluent state by 0.05% trypsin/0.02% ethylenediaminetetraacetic acid (EDTA; Invitrogen, Carlsbad, CA, USA) treatment. The cells at passages between four and six were used for this study. The experiments were approved by the animal experiment ethics committee of Kyung Hee University [KHUASP(SU)-07-007].
Application of the Cyclic Equibiaxial Stretch to the Cells
hDFs and mouse EGFP–mDFs were plated at a density of 1.5 × 105 cells per well on the type I collagen-precoated silicon membrane of the six-well plate (Flexcell Inc., Hillsborough, NC, USA) and allowed to attach for 24 h in FGM2. If necessary, the cells were starved with the serum-free fibroblast basal media (FBM) (Lonza, San Diego, CA, USA) for 24 h before applying the strain. The cells on the flexible silicone membrane repetitively received the cyclic equibiaxial stretch for 24 h at 0.5 Hz frequency and 20% maximum elongation according to the manufacturer's manual for the Flexercell® FX-4000™ apparatus (Flexcell Inc.) as described in Figure 1A and B. With the Flexercell apparatus, the diameter of the circular silicone membrane is enlarged 1.2-fold by periodic vacuum loadings, which could result in a 1.44-fold increase of total cell culture surfaces (pr2). The cells at the center of the circle do not receive any stretch stimulus, but the cells at the periphery receive maximal stretch (Fig.1C, D). The stretch effects, such as apoptosis and bromodeoxyuridine (BrdU) incorporation, were observed at the two-thirds distant area from the center of the circle, and all of the fibroblasts on the silicon membrane were used both in in vitro replating experiments and in vivo experiments.
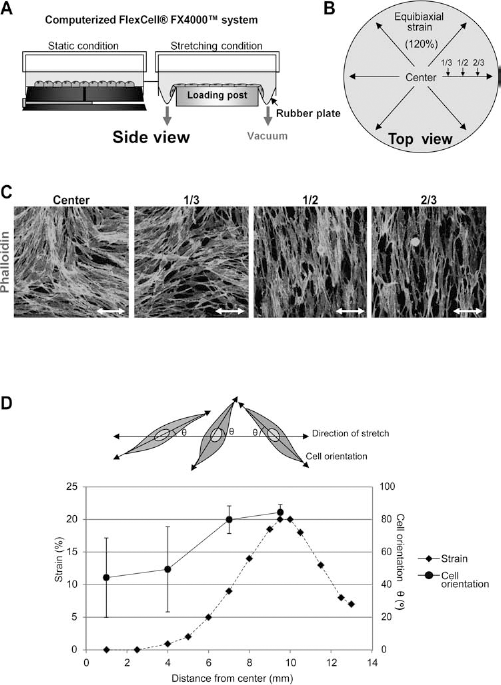
A device for in vitro application of the cyclic stretch and the analysis of radial distance-dependent orientation of dermal fibroblast applied with the stretch. The operating principle (A) and the top view (B) of the Flexcell® FX-4000™ Tension System are shown. (C) Cells and actin fibers were reorganized by the cyclic stretch. The actin fibers were parallel to the orientation of the cells applied with the cyclic stretch. Double-head arrows indicate the direction of the stretch. The cells were stained with tetramethyl-rhodamine isothiocyanate (TRITC)-conjugated phalloidin in order to show the actin fibers. (D) Orientation of cells was determined at several distances from the center of the silicone membrane after the application of stretch. More than 400 cells were analyzed to determine the orientation of cells at each distance, and the results are shown by mean ± SD. The broken line indicates the radial strain at each distance, while the stretch is applied.
Western Blot Analysis
For Western blot analysis, the cells were rinsed with ice-cold phosphate-buffered saline (PBS; WelGENE, Daegu, Korea) three times and disrupted by 100 μl of 2× lysis buffer (40 mM Tris-HCl pH 7.5, 300 mM NaCl, 2 mM Na2EDTA, 2 mM EGTA, 2% Triton-X 100, 5 mM sodium pyrophosphate, 2 mM β-glycerophosphate, 2 mM Na3VO4, and 2 μg/ml leupeptin, all from Cell Signaling Technology Inc., Beverly, MA, USA) per well of six-well plates. The cell lysates were incubated on ice for 20 min and centrifuged at 10,000 × g for 30 min at 4°C, and the supernatants were saved. Total protein amount was measured by Quick Start™ Bradford dye reagent (BioRad Lab., Inc., Hercules, CA, USA), and the same amount of proteins of each sample was denatured and resolved by 10% SDS polyacrylamide gel electrophoresis (SDS-PAGE; Sigma, St. Louis, MO, USA) and electrophoretically transferred to nitrocellulose membranes (Protran®, Whatman, GE Healthcare, Freiburg, Germany). Blots were blocked by filtered 5% nonfat dry milk (BD Pharmingen, San Jose, CA, USA) in 200 mM Tris-buffer saline pH 7.6 (TBS; Sigma), incubated with primary antibodies in TBS containing 0.1% Tween-20 (Sigma) overnight at 4°C, incubated with horseradish peroxidase (HRP)-conjugated secondary antibody (BioRad Lab., Inc.), and visualized by the ECL detection system (Santa Cruz Inc., St. Louis, MO, USA). Primary antibodies against pan-ERKs, phospho-ERKs, phospho-AKT, and phospho-c-Jun N-terminal kinases (JNKs) were purchased from Cell Signaling Technology, Inc., and an antibody against α-tubulin was purchased from Sigma. For Western blot analysis of fibronectin and type I collagen, samples were prepared by the use of the collected culture media and separated in 8% Tris-HCl SDS PAGE gels (Sigma). The number of the cells in each culture sample had been determined, and it had been used as the internal control to determine the volume of media that was used for Western blot analysis. Clone HFN7.1 hybridoma (American Type Culture Collection, Rockville, MD, USA) culture supernatant was used for Western blot of fibronectin, and antitype I collagen antibody was purchased from Biodesign Inc. (Kennebunk, Maine, Germany).
Terminal Deoxynucleotidyltransferase-Mediated dUTP-FITC Nick-End Labeling Assay (TUNEL Assay)
Apoptotic cells were detected by TUNEL assay with fluorescein system (Roche, Indianapolis, IN, USA). The cells were washed with PBS three times and fixed in 3.7% paraformaldehyde (Sigma) in PBS for 20 min at 4°C. After extensive washing with PBS, the cells were permeabilized with 0.2% Triton X-100 (Sigma) in PBS for 5 min, incubated at 37°C for 60 min with an equilibration buffer containing 2-deoxynucleotide 5′-triphosphate and terminal deoxynucleotidyltransferase (TdT) enzyme, according to the manufacturer's protocol. After the TUNEL assay, the cells were counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma), mounted with Vectashield mounting medium (Vector Lab., Burlingame, CA, USA), and immediately examined by fluorescence microscope (Leica Microsystems, Inc., Bannockburn, IL, USA). For the quantitative analysis of apoptotic cells, 12 fields per sample were analyzed (~400 cells/field). The cells showing green fluorescence in the nucleus were counted as apoptotic cells. Results were expressed as a percentage of apoptotic cells among DAPI-positive cells.
5-Bromo-2′-Deoxyuridine (BrdU) Incorporation Assay
The cellular proliferation was measured by DNA incorporation of BrdU. Briefly, the cells were labeled with 20 μM BrdU (Sigma) for 24 h of the mechanical stretch application. After fixing the cells as described above, the cells were incubated with 2 N HCl (Duksan Pure Chemical Company, Ansan, Korea) for 30 min, washed with PBS, incubated with mouse anti-BrdU antibody (Roche, Indianapolis, IN, USA), incubated with fluorescein-conjugated secondary antibody (Roche), counterstained with propidium iodide (PI; Sigma), and mounted with Vectashield mounting medium. The cells with green fluorescence in the nucleus were counted as proliferating cells. For the quantification of proliferating cells, 12 fields per sample were analyzed (~400 cells/field). Results were expressed as a percentage of BrdU-positive cells among PI-positive cells.
Inhibitor Study
PD98059 (Millipore Corporation, Billerica, MA, USA), a specific inhibitor of the ERK1/2 activation by mitogenactivated protein kinase kinase (MAPKK or MEK), LY294002 (Millipore), a selective inhibitor of phosphatidylinositol 3-kinase (PI3K), and SP600125 (Millipore), an anthrapyrazolone inhibitor of Jun N-terminal kinase (JNK), were dissolved in dimethyl sulfoxide (DMSO; Sigma). The cells were preincubated with PD98059 (100 μM), LY294002 (10 μM), and SP600125 (1 μM) for 1 h prior to the application of the cyclic stretch.
ELISA of VEGF and SDF-1α in Culture Supernatant of hDFs During Mechanical Stretch Application
VEGF or SDF-1α in the culture supernatants was measured by sandwich ELISA purchased from R&D Systems (Abingdon, UK). The human recombinant VEGF, SDF-1α, or test samples were added to the wells and then incubated for 2 h at room temperature. The plates were incubated with HRP-conjugated polyclonal antihuman VEGF or SDF-1 antibody at room temperature for 2 h. The color reaction was induced by the addition of substrate solution and was stopped 20 min later by the addition of 2 N of sulfuric acid. An automated microplate reader (BioRad Lab. Inc.) was used to measure the optical density (O.D.) at a wavelength of 450 nm. Between each of these steps, the plates were washed four times with PBS containing 0.1% Tween 20.
Immunocytochemistry
hDFs cultured on the type I collagen precoated silicone membrane were fixed with 3.7% formaldehyde for 20 min and permeabilized with 0.2% Triton X-100 in PBS for 5 min at room temperature. The silicone membrane of a well was subdivided into six pieces and stained using a general procedure for immunocytochemistry. Immunocytochemistry was performed by the use of the following primary antibodies: anti-α-smooth muscle actin (α-SMA; Dako, Burlingame, CA, USA), anti-α-tubulin (Sigma), or anti-vinculin (Sigma). In order to stain the actin fibers, cells were incubated with tetramethylrhodamine isothiocyanate (TRITC)-conjugated phalloidin (Sigma). Images were collected using a confocal microscope (Leica Microsystems, Inc.).
Induction of Experimental Diabetes
Streptozotocin (STZ, Sigma) at a dose of 50 mg/kg in 50 mM sodium citrate (Sigma), pH 4.5, was intraperitoneally injected daily for 5 days to wild-type male FVB mice at 8–12 weeks of age in order to induce the experimental diabetes. At 16 days after the final injection, serum glucose was measured from the blood of the tail vein using the glucose meter, Accucheck (Roche, Indianapolis, IN, USA). Mice were considered diabetic if they had hyperglycemia (>300 mg/dl), and it was checked weekly that the mice remained hyperglycemic for at least 5 weeks. At 3 weeks after the final injection of STZ, the full-thickness wound was made on the dorsal skin of the mice with hyperglycemia of 300~400 mg/dl to perform transplantation experiments.
Transplantation of EGFP–Dermal Fibroblasts in Wound
The full-thickness excision wound was made on the dorsal skin of the STZ-induced diabetic mouse using an 8-mm-diameter punch (Kai Europe GmbH, Solingen, Germany) biopsy while sparing the underlying panniculus carnosus muscle after depilation under anesthesia with ketamine (106.25 μg/kg; YuhanYanghang, Seoul, Korea). In order to transplant mouse EGFP–fibroblasts applied with the stretch or not, all of the fibroblasts on the silicon membrane were harvested with treatment of 0.3% type I collagenase. At a density of 6 × 105 cells/40 μl, the wound received a mixture of poloxamer F127 (Sigma) hydrogel (or fibrin glue; Greenplast®; Green Cross Corp., Yongin, Korea) and mouse EGFP–fibroblasts, which had been applied with the stretch or not. Wound sites were covered with Mepitel (Mölnlyke Health Care AB, Gothenburg, Sweden) and then Tegaderm (3M, London, ON, Canada), and the animals were kept in individual cages. At 2 or 7 days posttransplantation, the mice were euthanized by inhalation of CO2 gas. The wound tissues, including normal adjacent tissue and fascia, were dissected out. Then, the wounds were prefixed with 3.7% formaldehyde solution in PBS overnight and divided into two pieces, which were embedded in paraffin or OCT compound (Shandon Cryomatrix™, Thermo Fisher Scientific Inc., Waltham, MA, USA), respectively. A series of serial sections (10 μm) of the frozen tissue crossing the longest diameter of the wound was obtained. The entire wound site of the section was subdivided into four divisions, and EGFP-positive cells were counted under the fluorescence microscope (Leica Microsystems, Inc.).
Histological Scoring of Wound Healing
After fixation of the wound tissues in formalin, the samples were embedded in paraffin and sectioned (5 μm). For histological evaluation, sections were deparaffinized and rehydrated followed by Masson's trichrome staining (Sigma) or hematoxylin/eosin (H&E; Sigma) staining. In order to evaluate neovascularization, sections were subjected to immunohistochemical staining against Griffonia simplicifolia 1 (GS-1) lectin (Sigma). All slides were then evaluated under light microscopy by two independent observers, using a semiquantitative score to evaluate vascularity, granulation, and dermal/epidermal regeneration as previously described (1). We used four-point scales to evaluate vascularity (1, severely altered angiogenesis with one or two vessels per site, endothelial edema, thrombosis, and/or hemorrhage; 2, moderately altered angiogenesis with three to four vessels per site, moderate edema and hemorrhage, but absence of thrombosis; 3, mildly altered angiogenesis with five to six vessels per site, moderate edema, but absence of thrombosis and hemorrhage; and 4, normal angiogenesis with more than seven vessels per site with only mild edema but absence of thrombosis and hemorrhage) and granulation tissue formation (1, thin granulation layer; 2, moderate granulation layer; 3, thick granulation layer; and 4, very thick granulation layer) and a three-point scale to evaluate dermal and epidermal regeneration (1, little regeneration; 2, moderate regeneration; and 3, complete regeneration).
Statistical Analysis
Data were presented as means ± standard deviation. Statistical analysis was performed using t test and calculating one-tailed p values. If necessary, differences between experimental groups were compared by ANOVA followed by Bonferroni's test. Significance of in vivo transplantation experiments was determined with the Wilcoxon signed-rank test. Values of p < 0.05 were considered significant.
Results
Cyclic Stretch-Induced Reorientation of Cell and Actin Stress Fibers
To examine the effects of cyclic stretch on hDFs, we cultured the cells on collagen-coated silicone elastomere until they reached confluence. The cells were then exposed to an equibiaxial cyclic stretch (stretching condition) with a maximum elongation of 20% and a frequency of 0.5 Hz for 24 h by using the Flexercell® FX-4000™ system. As a control, cells were cultured under static conditions (Fig. 1A). Cells at different distances from the center received different degrees of stretch. The cells received stretch of approximately 1–2%, 10%, or 20% elongation at one third, one half, or two thirds of the diameter from the center of silicon elastomere, respectively (Fig. 1D). To estimate the stretch effects on cell orientation and cytoskeletal architecture, we observed actin stress fibers through TRITC-phalloidin staining (Fig. 1C, D). The orientation of most cells at the center and one-third diameter was random. However, the cells at one-half or two-thirds diameter, receiving more than 10% stretch, were significantly reoriented away from the direction of stretch (approximately 85°) (Fig. 1D). Interestingly, the actin stress fibers were aligned along the longest axis of reoriented cells subjected to stretch, compared to the control cells (Fig. 1C). Thus, approximately 75% of the cells (from one-half to two-thirds diameter) receiving more than 10% stretch at 0.5 Hz were reoriented away from the stretch direction, and actin stress fibers were reorganized along the reorientation direction of the cells.
Cyclic Stretch-Induced Early Transient Activation of ERKs, AKT, and JNKs
Mechanical stimuli are known to activate intracellular signaling pathways (28,37) in various types of cells. To examine whether the cyclic stretch affects the intracellular signaling pathways of the dermal fibroblasts, we harvested hDFs cultured with normal growth media (FGM2) at the indicated time after the application of the cyclic stretch. Activation of ERK1/2, AKT, and JNK1/2 was then determined by Western blot analysis (Fig. 2A). ERK1/2, AKT, and JNKs pathways were activated transiently. The phosphorylation level of ERK1/2 increased within 10 min after the cyclic stretch was sustained up to 1 h and thereafter decreased to basal levels. AKT and JNKs were also activated within 10 min after application of the cyclic stretch, but their phosphorylation started to rapidly decline after 30 min. The transient activation of ERK1/2, AKT, and JNKs mediated by cyclic stretch was also observed in the cells cultured in mitogen-free, serum-starved media (FBM) (Fig. 2B). These results suggest that cyclic stretch transiently activates ERK1/2, AKT, and JNKs in hDFs independently of various mitogens.

The cyclic stretch enhanced the survival of cells through the activation of ERKs, PI3K, and JNKs pathways. (A) Extracellular signal-regulated kinases (ERKs), AKT, and c-Jun N-terminal kinases (JNKs) pathways were activated in hDFs cultured with normal culture media (fibroblast growth media; FGM2) by the cyclic stretch. The cyclic stretch was applied to the cells for various incubation times (0 min~24 h) and Western blot analysis was performed with the cells to determine the level of phosphorylated ERKs, AKT, and JNKs. Pan-ERK or α-tubulin was used as the internal control. (B) The cyclic stretch activates ERKs, AKT, and JNK pathways in hDFs cultured with the serum-starved condition (fibroblast basal media; FBM). (C) The cyclic stretch decreased apoptosis of hDFs cultured with FGM2. A terminal deoxynucleotidyltransferase-mediated dUTP nick-end labeling (TUNEL) assay was performed to determine apoptosis. The number of apoptotic cells was significantly downregulated in the stretched cells (ST) compared to the control group (CON) (n = 4 for each group, *p < 0.002). (D) The cellular proliferation was enhanced by the cyclic stretch. Bromodeoxyuridine (BrdU) incorporation assay showed that the proliferation of cells increased in the stretch group (ST) (n = 4 for each group, *p < 0.012). (E) Apoptosis of the serum-starved cells was also decreased by the cyclic stretch (ST) compared to the static condition (CON) (n = 4 for each group, *p < 0.0017). (F) The mechanical stretch for short term (1 h) was enough to decrease apoptosis. Serum-starved hDF was cultured under the stretch condition for 24 h (24 h ST) or under the transient stretch for 1 h following culture under static culture condition (1 h ST/23 h CON). The cells were cultured under the full static condition for 24 h (24 h CON) as a control group (n = 5 for each group, *p < 0.016, **p < 0.022) (p < 0.002 by a one-way ANOVA and Bonferroni's test, CON; no stretch, ST; stretch). (G) The downregulation of apoptosis induced by the cyclic strain was mediated by the activation of ERKs, phosphatidylinositol 3-kinase (PI3K), and JNKs. PD98059 (PD, 100 mM), LY294002 (LY, 10 mM), or SP600125 (SP, 1 mM) was pretreated to the cell cultured under the condition of serum starvation (FBM) before the application of the cyclic stretch (ST) in order to inhibit the ERKs, AKT (a downstream of PI3K), or JNK pathway, respectively (n = 5 for each group, *p < 0.007, **p < 0.005, ***p < 0.00007, ****p < 0.000001, *****p < 0.0001 (*****determined by using a one-way ANOVA and Bonferroni's test). Dimethyl sulfoxide (DMSO) was also pretreated as a control of the treatment of the inhibitors. (C–G) More than three independent experiments were performed for analyses, and a representative result was shown as mean ± SD for each quantitative analysis.
Cyclic Stretch-Inhibited Apoptosis and Stimulated Cellular Proliferation
Studies using various cell types have shown that stretchmediated early activation of intracellular signaling affects cell apoptosis and/or cell proliferation (9,37). Therefore, we examined the effects of cyclic stretch on proliferation and apoptosis of hDFs. TUNEL assays and BrdU incorporation assays were performed using the cells that had been stretched for 24 h in FGM2 culture media (Fig. 2C, D). Approximately 35% of total cells underwent apoptosis in the confluent culture. However, the apoptosis rate of the cells that were subjected to stretch was reduced to ~9%, corresponding to an approximately fourfold lower number than the number of unstretched cells undergoing apoptosis (Fig. 2C). On the other hand, the cellular proliferation rate determined by BrdU incorporation assays increased from 58% to 68% by the application of cyclic stretch (Fig. 2D). Cyclic stretch also decreased apoptosis of the cells cultured under serum-starved conditions (FBM, Fig. 2E). The apoptosis rate was approximately 15% in the nonstretch condition but decreased to 3% upon the application of stretch. Under the starved conditions, the stretch effect on cell proliferation was marginal because the percentage of BrdU-incorporating cells among the nonstretched cells was less than 1% even though BrdU labeling was performed for 24 h (data not shown). Thus, the stretch reduced apoptosis under serum-starved conditions by approximately fivefold. It is likely that the cyclic stretch not only stimulated the proliferation of hDFs but also downregulated apoptosis, independent of mitogen-mediated regulation of proliferation or apoptosis.
ERK1/2, AKT, and JNK Pathways Mediated the Decrease in Apoptosis Induced by Cyclic Stretch
The stretch-stimulated transient activation of intracellular signaling molecules occurred early after the application of stretch. We asked whether the stretch-induced early/transient activation of the ERK1/2, AKT, or JNK pathway mediated the decreased apoptosis that was observed 24 h after the application of stretch. To examine this possibility, we subjected hDFs to stretch for 1 h and then incubated these cells under the static culture condition for 23 h (1 h ST/23h CON, Fig. 2F). The application of the stretch for 1 h dramatically decreased apoptosis, compared to the control group. Moreover, the level of antiapoptosis was very similar to the level induced when the stretch was applied for 24 h (24 h ST, Fig. 2F). Alternatively, we inhibited the signaling pathways to observe the effects of the inactivated signaling on apoptosis. The pathway-specific inhibitors were pretreated for 1 h before the application of the 24-h cyclic stretch to the cells that had been cultured under serum-starved conditions (FBM) (Fig. 2G). PD98059, LY294002, or SP600125 was used specifically to inhibit the ERK1/2 pathway, PI3K (a kinase of AKT), or JNK pathway, respectively. The application of the cyclic stretch significantly decreased apoptosis, compared to the control group. However, all of the inhibitors nullified the antiapoptotic effects of stretch on the cells (Fig. 2G). Therefore, the results suggest that the early/transient activation of ERK1/2, AKT, and JNKs mediated the antiapoptotic effects of the cyclic stretch.
Cyclic Stretch Enhanced the Secretion of ECM Proteins, VEGF, and SDF-1
Mechanical strain enhances the synthesis of ECM proteins (11,18), and the strain-mediated cellular reorientation requires the production of ECM protein, such as pericellular fibronectin (32). Therefore, we determined the secretion level of type I collagen or fibronectin with Western blot analysis using culture media from hDFs, which had been cultured under the cyclic stretch conditions or static conditions for 24 h or 48 h (Fig. 3A). The stretch increased the secretion of type I collagen 1.7-fold at 24 h or 2.5-fold at 48 h, compared to what was observed in the static culture. The secretion of fibronectin was also increased twofold at 24 h or fourfold at 48 h. Moreover, mechanical strain has been reported to regulate the expression of proinflammatory cytokines and angiogenic factors (8,25,35,51). To examine whether cyclic stretch would affect the expression of VEGF, we determined the levels of VEGF in culture media by ELISA assay at 24 h after the stretch application (Fig. 3B). The levels of secreted VEGF increased approximately fivefold at 24 h in the stretched conditions, compared to the nonstretched condition. We also examined whether mechanical stretch affects the expression of SDF-1α, which plays important roles in wound healing (2,40). Secreted SDF-1α also increased 2.5-fold at 24 h in the stretched condition. Therefore, cyclic stretch enhanced the expression of VEGF and SDF-1 as well as the ECM proteins type I collagen and fibronectin.

The cyclic stretch increased the expression of the extracellular matrix proteins and promoted the secretion of VEGF and SDF-1. (A) The cyclic stretch enhanced the expression of type I collagen and fibronectin in hDF. Western blot analysis was performed in order to determine the expression level of type I collagen and fibronectin in culture supernatant of the cells cultured under the static condition (CON) or stretch (ST) for 24 h or 48 h. The volume of the culture supernatant for the Western blot analysis was determined with the number of live cells in each experimental group. (B) The increased secretion of vascular endothelial growth factor (VEGF) or stromal cell-derived factor 1 (SDF-1) was induced by the cyclic stretch. Enzyme-linked immunosorbent assay (ELISA) analysis was performed with culture supernatant of hDFs cultured under the static condition (CON) or stretch (ST) for 24 h. The volume of the culture supernatant for ELISA analysis was also normalized with the number of live cells of each experimental group, n = 3 for each group, *p < 0.003, **p < 0.003. Two independent experiments had been performed for analyses, and a representative result was shown as mean ± SD for each quantitative analysis.
The Effects of Cyclic Stretch Were Maintained Under Static Conditions After the Cessation of the Stretch Stimulus
We asked whether the effects of cyclic stretch would continuously affect the cells after the cessation of the stimulus. Therefore, hDFs preconditioned with cyclic stretch for 24 h were replated onto coverslips and cultured under static conditions after their single-cell suspension was made through type I collagenase treatment. Attachment potential and cellular proliferation of the single cells were then examined (Fig. 4A, B). The number of the attached cells preconditioned with the stretch, compared to the control cells, increased (~2.6-fold) 1 h after replating (Fig. 4A). Moreover, proliferation of the cells preconditioned with the cyclic stretch increased after replating because the number of BrdU-positive cells was approximately fourfold higher among the cells preconditioned with the cyclic stretch than among the unstretched cells (Fig. 4B).

Characteristics obtained by the cyclic stretch preconditioning were maintained even after replating. (A) The number of the attached cells increased after reseeding the cells trained with the cyclic stretch. Human dermal fibroblasts trained with the mechanical stretch for 24 h were attached for 1 h on the cover glasses with the normal culture media (FGM2). The number of the attached cells was determined by counting cells per captured picture, and one picture was taken from one cover glass (n = 8 per each group, *p < 0.0001). Representative images are shown. Scale bar: 100 μm. (B) BrdU incorporation increased in the cells pretrained with the cyclic stretch. The cells trained with the cyclic stretch were incubated for 24 h with the normal culture containing BrdU media after reseeding (n = 8 per each group, *p < 0.0001). (C) The reorganization of actin fibers was maintained after reseeding of the cells trained with the cyclic stretch. At 1 h after reseeding of the strained cells, the cells were immunostained with antibody against α-tubulin (green) and TRITC-conjugated phalloidin (red). Scale bar: 10 μm. (D) Actin-related protein vinculin showed the ends of the reorganized actin fibers after reseeding of the pretrained cells. The pretrained cells were incubated for 20 h before immunocytochemistry against vinculin (green). Scale bar: 10 μm. (E) α-Smooth muscle actin (α-SMA) was not expressed either in the control cells (CON, a, c, e) or in the cyclic stretch applied cells (ST, b, d, f). Nuclei were stained in blue with 4′,6-diamidino-2-phenylindole (DAPI) (a, b, e, f), and α-SMA was immunostained in green (c–f). (A–E) CON, control; ST, stretch.
Cyclic stretch induced reorientation of hDFs and reorganization of actin fibers (Fig. 1C, D) and microtubules (data not shown). After replating and culturing of the stretched cells for 20 h under the static culture condition, reorganization of microtubules and the actin stress fibers were still maintained (Fig. 4C). Moreover, mechanical strain is known to induce reorganization of focal adhesion proteins (45), which are required for signal transduction and increased survival of cells mediated by strain (13). After replating, compared to the control cells, the preconditioned cells formed denser focal adhesion complexes that included vinculin (Fig. 4D). A previous study showed that mechanical stress induces the transdifferentiation of rat fibroblasts into myofibroblasts expressing α-smooth muscle actin (α-SMA), which is a marker protein of myofibroblasts (26). In our system, however, the preconditioned cells did not express α-SMA (Fig. 4E). Therefore, these results suggest that the stretch-mediated effects on cell adhesion, cell proliferation, and cytoskeletal reorientation were sustained for at least 24 h after the cessation of stretch stimulus.
Transplantation of the Stretched Dermal Fibroblasts Stimulated Wound Healing in Streptozotocin-Induced Diabetic Mice
This study showed that mechanical stretch stimulated cell survival, cell proliferation, expression of ECM proteins, VEGF expression, and SDF-1 expression in in vitro cultured hDFs. Moreover, preconditioning of the cells with stretch enhanced the survival and proliferation thereafter under an in vitro static culture system for 24 h. We hypothesized that the characteristics acquired by the stretched cells might enhance their survival after transplantation into in vivo wound sites and thereby potentially affect the wound-healing process. Therefore, we examined whether the preconditioned cells would show increased survival after transplantation into in vivo wound sites. Dermal fibroblasts isolated from the FVB–EGFP mouse were used in the transplantation experiments. First, we analyzed whether dermal fibroblasts isolated from FVB–EGFP mice are affected by the cyclic stretch (0.5 Hz frequency, at maximum 20% elongation, for 24 h) in the same way as hDFs. Mouse fibroblasts transiently activated the ERK1/2 pathway in response to the mechanical stretch (Fig. 5A). Moreover, mouse fibroblasts also showed the reduced apoptosis (0.56-fold) and increased in the proliferation (1.36-fold) compared to the control mouse fibroblasts (Fig. 5B). Furthermore, comparing to the control cells, mouse EGFP fibroblasts preconditioned with the stretch (ST-mDF) showed the increase both in attachment potential and cellular proliferation after replating on coverslips and culturing under the static condition. The number of the attached cells preconditioned with the stretch (ST-mDF) increased 1.36-fold, and the number of BrdU-positive cells increased 1.32-fold compared to the control cells (NT-mDF) (Fig.5C). The stretched cells or the control cells were then mixed with fibrin glue, and the mixture was then transplanted into the full-thickness wound inflicted on the back of a FVB mouse with STZ-induced diabetes (Fig. 5D). The transplantation of the stretched cells (ST-mDF) with fibrin glue resulted in a higher number of EGFP-positive cells in the wound sites 7 days after transplantation than the control cells (NT-mDF) expressing EGFP (Fig. 5E–H).
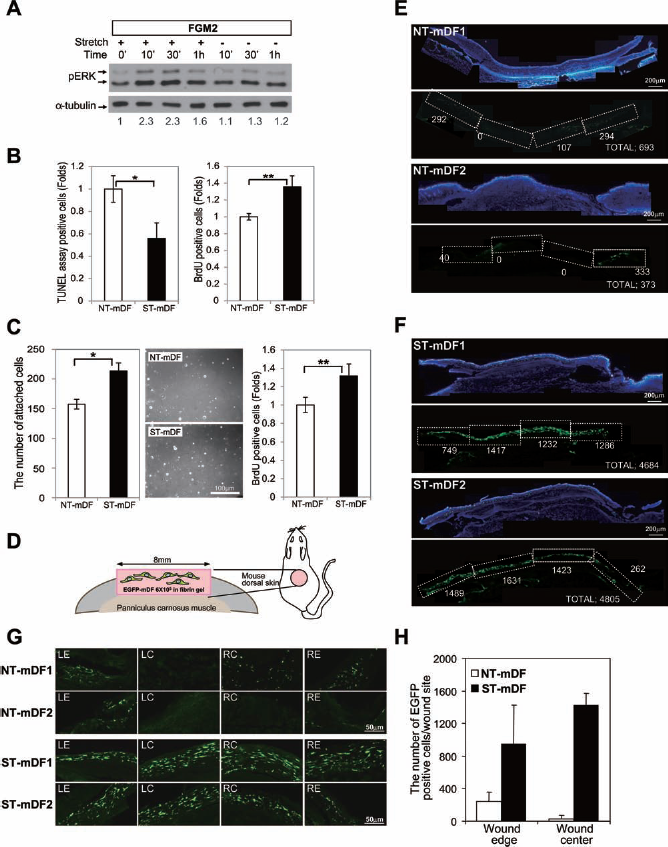
The cell preconditioning with the cyclic stretch resulted in the higher number of live transplanted cell within fibrin glue vehicle at the transplantation site. (A) The ERK pathway was activated by cyclic stretch in mouse fibroblasts after cyclic stretch application (20% elongation, 0.5 Hz frequency for 24 h). Dermal fibroblasts isolated from enhanced green fluorescent protein (EGFP) mouse were cultured with FGM2, and Western blot analysis was performed to determine the level of phosphorylated ERKs (pERK) after the application of cyclic stretch. The density of phosphorylated ERK bands were normalized with the density of α-tubulin for the quantification of pERK bands (relative fold increases of pERK are shown). (B) The cyclic stretch reduced apoptosis and enhanced cell proliferation of mouse fibroblasts. TUNEL assay and BrdU incorporation assay were performed to determine the number of apoptotic cells and proliferating cells after cyclic stretch application (20% elongation, 0.5 Hz frequency for 24 h) in FGM2, respectively. (The result is presented as mean ± SD, n = 5 for each group, *p < 0.011, **p < 0.0022.) (C) Attachment potential and cellular proliferation of mouse fibroblasts increased after replating mouse fibroblasts trained with cyclic strain. The number of attached cells at 1 h (n = 5 for each group, *p < 0.00018) and the number of BrdU-positive cells at 24 h (n = 6 for each group, **p < 0.002) were increased after replating of stretch-applied mouse fibroblast in FGM2. The result is presented as mean ± SD. Scale bar: 100 μm. (D) The experimental diagram shows that mDFs isolated from enhanced green fluorescent protein transgenic mice (EGFP–mDF) were transplanted onto full-thickness wounds of dorsal skin of diabetic mice. The stretched cells were premixed with fibrin gel before transplantation. More EGFP-positive cells were observed in cryosections of wound sites transplanted with the stretched dermal fibroblasts isolated from EGFP transgenic mice (ST-mDF, F) than with the control cells (not stretched mDFs, NT-mDF, E) at 7 days after transplantation. The white numbers indicate the total number of EGFP-positive cells found in the regions boxed with the white broken line of each section. The total number of the EGFP-positive cells in each section (total) is also shown. Scale bar: 200 μm. (G) The representative images of EGFP-positive cells in wound sites. LE, left wound edge; LC, left wound center; RE, right wound edge; RC, right wound center. Scale bar: 50 μm. (H) The number of EGFP-positive cells was counted in the wound edge or wound center of cryosections and is presented as mean ± SD (n = 2 mice, four cryosections). NT-mDF, not stretched mouse dermal fibroblasts; ST-mDF, stretched mouse dermal fibroblasts.
The stretched cells or the control cells were also transplanted into the full-thickness wound of the mouse with poloxamer hydrogel (Fig. 6A). The transplantation of cells enhanced the process of wound healing. Surprisingly, the stretched cells (ST-mDF), compared to control cells (NT-mDF), were found to accelerate wound healing in the diabetic mice at 7 days or 14 days (Fig. 6B). The tissue, including the wound, was harvested and fixed 2 or 7 days after transplantation, and EGFP-positive cells were counted in the serial sections representing the middle part of the wound (Fig. 6C, D). The number of cells expressing EGFP in the wound transplanted with the stretched cells was higher than the number of control cells expressing EGFP even 2 days after the transplantation (Fig. 6C). At 7 days, the number of EGFP-positive cells in the mice that received the stretched cells significantly differed from the number of EGFP-positive control cells (Fig. 6C). The number of cells expressing EGFP both at the wound edge and in the wound center of the dorsal wound transplanted with the stretched cells was significantly higher than the number of control cells expressing EGFP (Fig. 6D, *p < 0.001, **p < 0.0005 by Wilcoxon signed rank test). We also confirmed the result through digital image analysis done by TissueFAXS (TissueGnostics, Austria). A higher number of EGFP-positive cells were found both at the edge and in the center of the wound transplanted with the stretched cells than control EGFP-positive cells (data not shown). However, whether the increase in the number of the stretched cells in the wound sites is due to a better survival/graft rate, higher proliferation rate, or both, is uncertain.
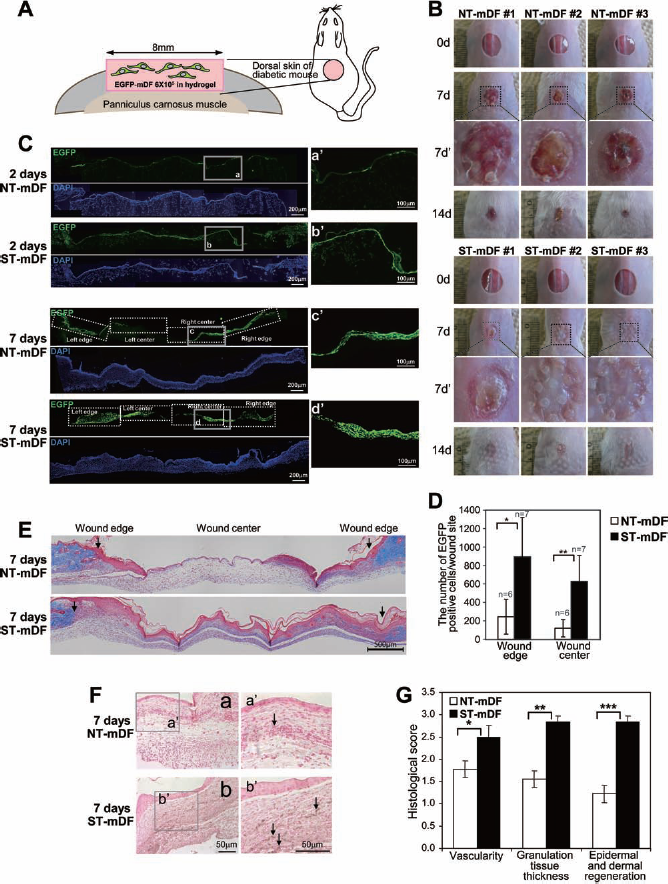
Wound healing was enhanced by the transplantation of mDFs applied with the cyclic stretch. (A) The experimental diagram shows that mDFs isolated from enhanced green fluorescent protein transgenic mice (EGFP-mDF) were transplanted onto full-thickness wounds of the dorsal skin of diabetic mice. The stretched cells were premixed with hydrogel before transplantation. (B) Wound healing at 0 days (0d), 7 days (7d), or 14 days (14d) after the transplantation of not stretched fibroblasts (NT-mDF) or the cyclic stretchapplied fibroblasts (ST-mDF). Broken black boxed regions in 7d are magnified in 7d′. (C) The number of EGFP-mDF applied with the mechanical stretch (ST-mDF) was higher in the wound sites after transplantation than the control EGFP-mDF (NT-mDF). Wound sites were fixed at 2 days or 7 days after transplantation, and representative fluorescent images of cryosectioned wound sites were shown. The number of EGFP-positive cells was counted both in left/right edge and left/right center of wound in each section (more than six sections were analyzed). The gray boxed regions (a, b, c, d) are magnified in a′, b′, c′, d′. Scale bar: 200 μm. The quantitative results for 7 days posttransplantation are shown as mean ± SD in (D) (*p < 0.001, **p < 0.0005, n = 6 mice for NT-mDF, n = 7 mice for ST-mDF). (E) The transplantation of the cyclic stretch-applied fibroblasts (ST-mDF) stimulated reepithelialization of the full-thickness wound of diabetic mice compared to the control fibroblasts (NT-mDF). Masson's trichrome and hematoxylin and eosin (H&E) staining were performed with paraffin sections of wound sites fixed at 7 days posttransplantation. Arrows indicate the boundary of wound sites. n = 3 mice per each group, Scale bar: 500 μm. (F) Griffonia simplicifolia-1 (GS-1) lectin-positive cells were more abundant in the wound sites transplanted with the fibroblasts applied with the cyclic stretch. a, the control group (NT-mDF); b, the mechanical stretch-applied group (ST-mDF). The boxed regions in a and b are magnified in a′ and b′. n = 4 mice per each group, Scale bar: 50 μm. (G) The semiquantitative evaluation of wound healing was performed using a semiquantitative scoring method to evaluate vascularity, granulation, and dermal/epidermal regeneration as previously described (1). The results are shown as mean ± SD. Eight or nine sections per mouse were analyzed for the scoring. n = 3 or 4 mice for not stretched (NT-mDF) or stretched groups (ST-mDF), respectively. *p < 0.012, **p < 0.0001, ***p < 0.0001. (B–G) NT-mDF, not stretched mouse dermal fibroblasts; ST-mDF, stretched mouse dermal fibroblasts. p values were determined by Wilcoxon signed-rank test.
Diabetic wounds in diabetic mice exhibit a delayed rate of healing as a consequence of the decrease in the formation of granulation tissue and neovascularization (Fig. 6E–G). We initially observed the formation of granulation tissue in the wounds of diabetic mice 7 days after the transplantation of the stretched cells or the control cells (Fig. 6E, G). Through Masson's trichrome and H&E staining, we observed that the transplantation of the stretched cells enhanced reepithelialization and the formation of collagen-enriched dermis in the full-thickness wound of the diabetic mice compared to the unstretched cells. Moreover, neovascularization was stimulated by the transplantation of the stretched cells (Fig. 6F, G). Vascular density in the central zone of wounds was evaluated through immunostaining of GS-1 lectin. The positive signal of GS-1 lectin increased in the wounds transplanted with the stretched cells compared to the unstretched cells (Fig. 6F). The scores of wound healing after transplantation of mDFs preconditioned with mechanical stretch were significantly higher than those obtained using the unstretched cells (Fig. 6G, *p < 0.012, **p < 0.0001, ***p < 0.0001 by Wilcoxon signed rank test). For the scoring, H&E-stained serial sections were analyzed with light microscopy by two independent observers to evaluate vascularity, granulation, and dermal and epidermal regeneration, as previously described (1). Taken together, these results suggest that the fibroblasts preconditioned with the cyclic stretch achieved better survival and/or proliferation rates and accelerated the wound-healing process in the diabetic mice.
Discussion
Exogenous mechanical stretch affects fundamental processes, such as proliferation, apoptosis, and fate selection of stem cells in various cell types, including hDFs (12,36,37,42). Our current study shows that dermal fibroblasts preconditioned with cyclic stretch accelerate the wound-healing process in the full-thickness wound of diabetic mice.
The cyclic stretch mediated the increase in cellular proliferation and the cell survival through ERK, JNK, and PI3K signaling, and a single inhibitor specific to each signaling decreased the effects of the stretch on cell survival. Indeed, each inhibitor increased apoptosis up to the level of the control group. Therefore, the activation of all three signaling pathways, ERK, JNK, and PI3K, is required for the downregulation of apoptosis. Individual signaling pathways might regulate different processes, but all of the processes may be required to enhance cell survival. For example, both reorganization of actin stress fibers and stimulated cell survival were inhibited by ERK- or JNK-specific inhibitors. LY294002, a PI3K-specific inhibitor, was able to inhibit increased cell survival but did not perturb the reorganization of actin stress fibers (data not shown). This finding suggests that AKT regulated by PI3K does not affect the reorganization of actin fibers but is required for the cell survival.
The application of mechanical strain stimulates cellular proliferation and cell survival in various cell types (12,37). Therefore, the application of strain could be an important strategy for promoting cell survival/proliferation in cell therapies that require good survival rates of the transplanted cells (23). Moreover, it would be more important that the stretched cells are able to maintain the advantages obtained from preconditioning with mechanical stretch when they return to static culture. Because the preconditioned cells would be transplanted into in vivo wound sites where the cyclic stretch no longer exits, the sustained effect of the cell preconditioning might be critical for engraftment, proliferation, and survival of the transplanted cells as well as for cell-mediated wound healing. Indeed, hDFs, which had been preconditioned with the stretch, showed higher adhesive potential and cellular proliferation even after they were replated on the static surface in vitro. In addition, vinculin-related focal adhesion complexes were more densely formed after replating of the cells preconditioned with the stretch than after replating of the control cells. Considering that interactions between adhesive cells and ECM, which are mediated by focal adhesion (33,38), are essential for proliferation, survival, and differentiation, the formation and mechanical stability of focal adhesions may be important for cell adhesion under mechanical stress. Therefore, the denser focal adhesions of the preconditioned cells after replating (shown in Fig. 4) would contribute to the enhanced engraftment, cell survival, and cellular proliferation under in vivo static conditions after in vivo transplantation as well as in vitro. It has been reported that mechanical stress induces the formation of focal adhesion complexes (39,42) and that protein tyrosine kinases then mediate reorganization of these focal adhesion complexes in response to rigid adhesion surfaces (39). In the future, it would be interesting to determine which signaling pathways mediate the formation of denser focal adhesions in the stretched hDFs.
The transplantation of the stretched mouse fibroblasts accelerates healing of the full-thickness wound in diabetic mice. Wound healing is impaired in the chronic wound of diabetic patients because of the reduced peripheral blood flow following impaired angiogenesis/VEGF expression and reduced mobilization of endothelial progenitor cells into sites of ischemia (7). Therefore, the expression of an EPC mobilizer, such as SDF-1, and a neovasculogenetic factor, such as VEGF, would be essential for the rescue of the defective wound healing in diabetic patients. Mechanical stretch induces increased expression of both VEGF and SDF-1 in fibroblasts, which would accelerate the wound-healing process in diabetic patients. Indeed, improved neovascularization was detected in the wound site transplanted with stretched fibroblasts. To express VEGF/SDF-1 in the chronic wound sites, the cells first need to survive under the hypoxic wound environment where neovasculogenesis is poor. On day 2 after the transplantation of the stretched cells isolated from EGFP transgenic mice, the number of EGFP-positive cells was threefold higher in the wound sites than the number of EGFP-positive unstretched cells. Moreover, 7 days after the transplantation of the stretched cells, the number of EGFP-positive cells was fourfold or fivefold higher at the wound edge or at center sites than the number of EGFP-positive control cells, respectively. Although a direct cause(s) of the increased number of transplanted cells is not clear, the stretched cells might have a higher initial survival rate than the control cells and higher proliferation rates later. The higher survival or lower apoptosis rates of the cells preconditioned with stretch might be contributed by the result of higher attachment ability in the wound environment. The cellular proliferation of the transplanted EGFP-positive cells might increase through both the direct effects of the stretch on the cells, such as the activation of ERK/JNK/AKT signaling, and the indirect effects of the stretch on the wound environment, such as increased neovasculogenesis following increased expression of VEGF or the mobilization of EPCs by SDF-1α. It would be very interesting in the future to observe whether EPCs would mobilize into the wound sites after the transplantation of the stretched cells and if the mobilization is mediated by SDF-1 expressed by the stretched cells.
Myofibroblasts in the wound may cause the formation of hypertrophic scars. If the stretch stimulates the differentiation of cells into myofibroblasts, then scar formation would be one of the unwanted effects of stretch preconditioning. The stretched hDFs did not express α-SMA, a marker used to characterize myofibroblasts among dermal fibroblasts both in vitro and in vivo (data not shown). Therefore, it is very likely that the stretched cells did not transdifferentiate into myofibroblasts. We transplanted the stretched cells by using two types of vehicle, poloxamer hydrogel and fibrin glue. We found that stretched cells accelerated the wound-healing process by the use of either hydrogel or fibrin glue, while the mobilization of the transplanted cells into the whole wound region was better in the hydrogel vehicle than in fibrin glue, regardless of the preconditioning of the cells.
In conclusion, dermal fibroblasts preconditioned with the cyclic equibiaxial stretch (0.5 Hz frequency, 20% maximum elongation, 24 h) accelerate the wound-healing process in diabetic mice. The transplantation of the stretched cells did not cause the formation of hypertrophic scars during the healing of the diabetic wounds. The acceleration of the wound-healing process in the diabetic wounds may have been due to the effects of stretch preconditioning on the cells, the enhanced cell survival or antiapoptosis effects and the stimulated cellular proliferation mediated by ERK/JNK/AKT signaling, the increase in the adhesive interaction between the stretched cells and the ECM, and the stimulated expression of VEGF and SDF-1.
Footnotes
Acknowledgments
This work was supported by the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (HI13C1479), the Future-Based Technology Development Program (NRF-2010-0020405), the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Ministry of Science, ICT & Future Planning (NRF-2012M3A9C6050485), and Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2012R1A1A2042265).
