Abstract
Normal wound healing is a complex, yet critical process. Unfortunately, delayed or impaired wound healing presents a significant burden for numerous patients worldwide. Recent research has demonstrated that exosomes, a subtype of extracellular vesicles, may play a pivotal role in mediating cell-cell communication during wound healing. This narrative review aims to explore the use of exosomes in wound healing, with a focus on in vivo application of exosomes in animal wound models. A total of 73 studies are outlined in this narrative review, exploring either excisional, diabetic, burn, ischemic, or pressure wounds. Overall, the most frequent source of exosomes was adipose-derived mesenchymal stem cells, and the most common application method was local or subcutaneous injection. While these studies demonstrated great promise for the use of exosomes for therapeutic use in wound healing, this review also discusses the challenges and obstacles that will need to be overcome, including standardizing methods, further characterizing exosomes and identifying the optimal sources and doses in the development of exosome therapies.
Keywords
Introduction
Wound healing
The skin, the body’s largest organ, is the first barrier against injury and pathogens. As such, wound healing is a critical, yet complex process that is of great importance for all patients. Indeed, in 2019, 10.5 million Medicare beneficiaries in the United States alone suffered from wounds, with chronic-wound related costs reaching US$22.5 billion 1 . Consequently, improving and accelerating wound healing remains a critical aspect of patient care, and requires a thorough understanding of the underlying pathophysiological mechanisms. Differentiating the type of wound also determines the best management of care. Primarily, one must differentiate between acute and chronic wounds, the latter of which is generally defined to be a wound lasting longer than 3 months 2 . In chronic wounds, accurate diagnosis of underlying systemic and local pathologies is essential to ensure optimal care. For example, within chronic ulcers alone, differentiating arterial from venous ulcers, or determining whether it is a mix of both is crucial as treatment differs greatly. While there are numerous types of wounds based upon etiology, some notable classifications include traumatic wounds, be it pressure, thermal, chemical or radiation wounds; metabolic wounds, such as with diabetic ulcers; inflammatory and autoimmune-induced wounds, such as with pyoderma gangrenosum; infectious wounds, or even neoplastic wounds, which includes melanomas, squamous or basal cell carcinomas 3 .
While the importance of understanding the underlying etiology of individual wounds cannot be understated, one must also grasp the mechanism of wound healing. Understanding the phases and process of wound healing allows for better management of wounds4–6.
Wound healing usually occurs in three main phases: inflammation, proliferation and regeneration. Prior to these main phases, hemostasis and coagulation starts immediately after injury. This requires immediate contraction of vascular smooth muscles to allow for vessel constriction to decrease bleeding. This primarily relies on the coagulation cascade, with activation of the extrinsic and intrinsic pathways, as well as platelet activation and aggregation. Ultimately, this leads to the formation of a thrombus, which is of great importance for further phases.
The second stage is the inflammatory phase, with the recruitment of immune cells to the site of injury, particularly neutrophils and macrophages. This is achieved by the secretion of several cytokines following the above-mentioned platelet activation, including tumor necrosis factor-alpha (TNF-α), transforming growth factor-beta (TGF-β), interleukin-1 (IL-1), platelet-derived growth factor (PDGF), and interferon-gamma (IFN-γ)4,7–9. Neutrophils are of great importance in the early phase of inflammation, and undergo a process of rolling and adhering to the surface of the endothelium in blood vessels, with subsequent migration out of the vessels toward the site of injury, at which point they use reactive oxygen species and proteases to phagocytose foreign material and microbes5,6,10. Macrophages continue this phagocytosis in the later phases of the inflammatory phase, as well as secreting several pro-inflammatory, fibrogenic, and angiogenic factors such as TGF-β, fibroblast growth factor (FGF), PDGF, and vascular endothelial growth factor (VEGF). This not only attracts further immune cells, including neutrophils, macrophages and finally, lymphocytes, as well as fibroblasts and endothelial cells5,6,10,11. Chronic wounds, in particular, tend to remain in the inflammatory phase and cannot proceed into the next phase, the proliferation phase.
Normal wound healing then proceeds into the proliferation phase that starts around day 3 following injury, and lasts 2 to 3 weeks4,5. This stage is critical to allow for initial closure of the lesions. Fibroblasts are especially important in this phase, and they initially migrate to the wound via factors such as TGF-β and PDGF. Once at the site, there is intense proliferation, as well as production of collagen, particularly type III, and other elements of the extracellular matrix. This leads to the formation of granulation tissue 12 . Following this step, fibroblasts may change their phenotype to become myofibroblasts to allow for wound retraction 13 . In terms of angiogenesis, while pro-angiogenic factors are already secreted during the inflammatory phase, during the proliferation phase, further endothelial cells undergo proliferation and migration to ensure the formation of new vessels. Macrophages, in particular, play an important role in regulating the degree of vascularization, which will be critical for successful wound healing 14 . Initial re-epithelization also occurs during this phase, with cells moving from the periphery and edges of the wound, with gradual proliferation and restoration of basement membrane 15 .
Finally, wound healing ends with the remodeling phase, which also represents the longest stage, lasting up to 1 to 2 years 5 . This stage requires the replacement of granulation tissue produced during the proliferative stage with the final scar. Following lesion closure with new keratinocytes and eventual epidermis, type III collagen is replaced by type I collagen. This leads to greater tensile strength, and up to 70% of its pre-injury strength may be recovered 15 . Metalloproteinases in particular actively take part in this remodeling, and further extracellular matrix remodeling. The new matrix eventually organizes and further contracts 5 . Finally, apoptosis plays a critical role in this phase, and allows for the final acellular, avascular mature scar 16 .
Extracellular vesicles and exosomes
Given the importance of proper wound healing and all its steps, the improvement of this process continues to be a crucial area of study. Exosomes are a subtype of extracellular vesicles (EVs), and are membrane bound vesicles that are secreted into the extracellular space. EVs have a broad range of sizes ranging from 30 to 2000 nm, and are composed of not only exosomes (30–150 nm), but also microvesicles (100–1000 nm) and apoptotic bodies (500–2000 nm). Exosomes are of endosomal origin via intraluminal vesicles that are secreted when multivesicular bodies fuse with the plasma membrane. Exosomes therefore contain proteins, lipids mRNA, miRNAs, lncRNAs, and DNA fragments that reflect their cell of origin17–19. Markers that are unique to exosomes include CD9, CD63, CD81, Alix, and TSG101. Because of the cargo delivered by exosomes, they have become a new and promising tool to not only study the processes of wound healing, but ameliorate them. It is important to emphasize that the wound environment undergoes significant changes throughout the different phases of healing. Consequently, exosome secretion and content also vary with each phase, suggesting that a phase-specific administration of exosomes may be necessary to support optimal wound healing. While the exact functions of exosomes has not yet been fully elucidated, exosomes have been shown to be involved in several processes, including immune regulation and cell-cell communication 20 . Indeed, several studies have demonstrated that exosomes encourage phenotypic macrophage change (from pro-inflammatory to pro-resolution), increase growth factor expression, fibroblast proliferation and migration; as well as improving angiogenesis via the release of bioactive and pro-angiogenic factors21–26.
When considering exosomes for their therapeutic use, particularly in terms of wound healing, one must consider the source of exosomes used. First, one must differentiate between human, or other animal sources-most commonly mice or rats 27 . The cell types used to extract exosomes also varies from study to study. Exosomes derived from adipose-derived stem cells (ADSCs) are most common in human and rodent studies. Umbilical cord mesenchymal stem cells are second most frequent in human studies; however, bone marrow–derived mesenchymal stem cells (BMSCs) are most common for rodent studies. All of the above-mentioned cells used for wound healing are considered members of mesenchymal stem cells (MSCs). These pluripotent stem cells have been particularly useful for studies, as they can originate from a large variety of different tissues, which makes them a particularly interesting source of exosomes. They have also been found to produce a large amount of exosomes compared with other cell types, as well as having immunomodulatory properties 28 .
Methodology of study selection
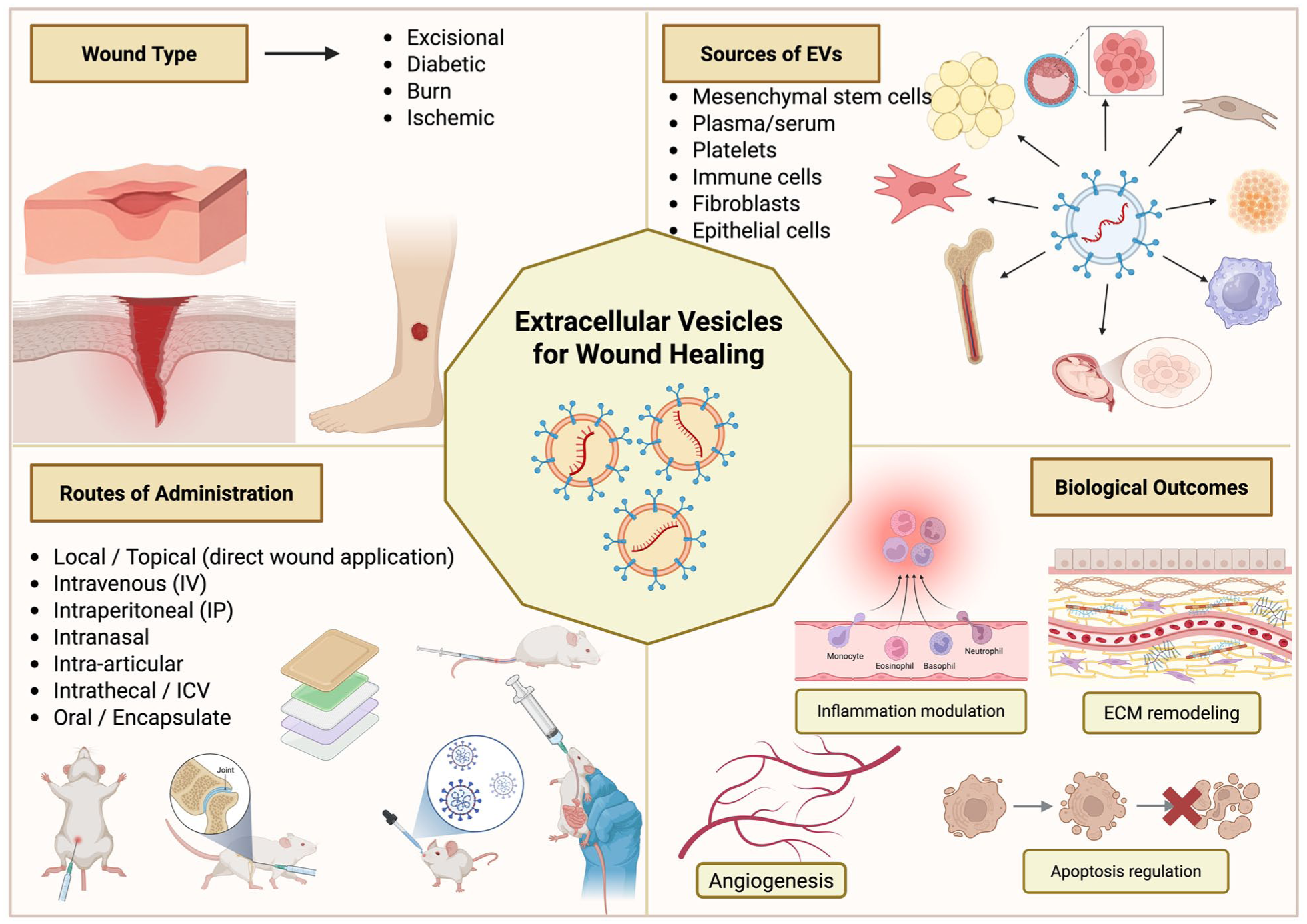
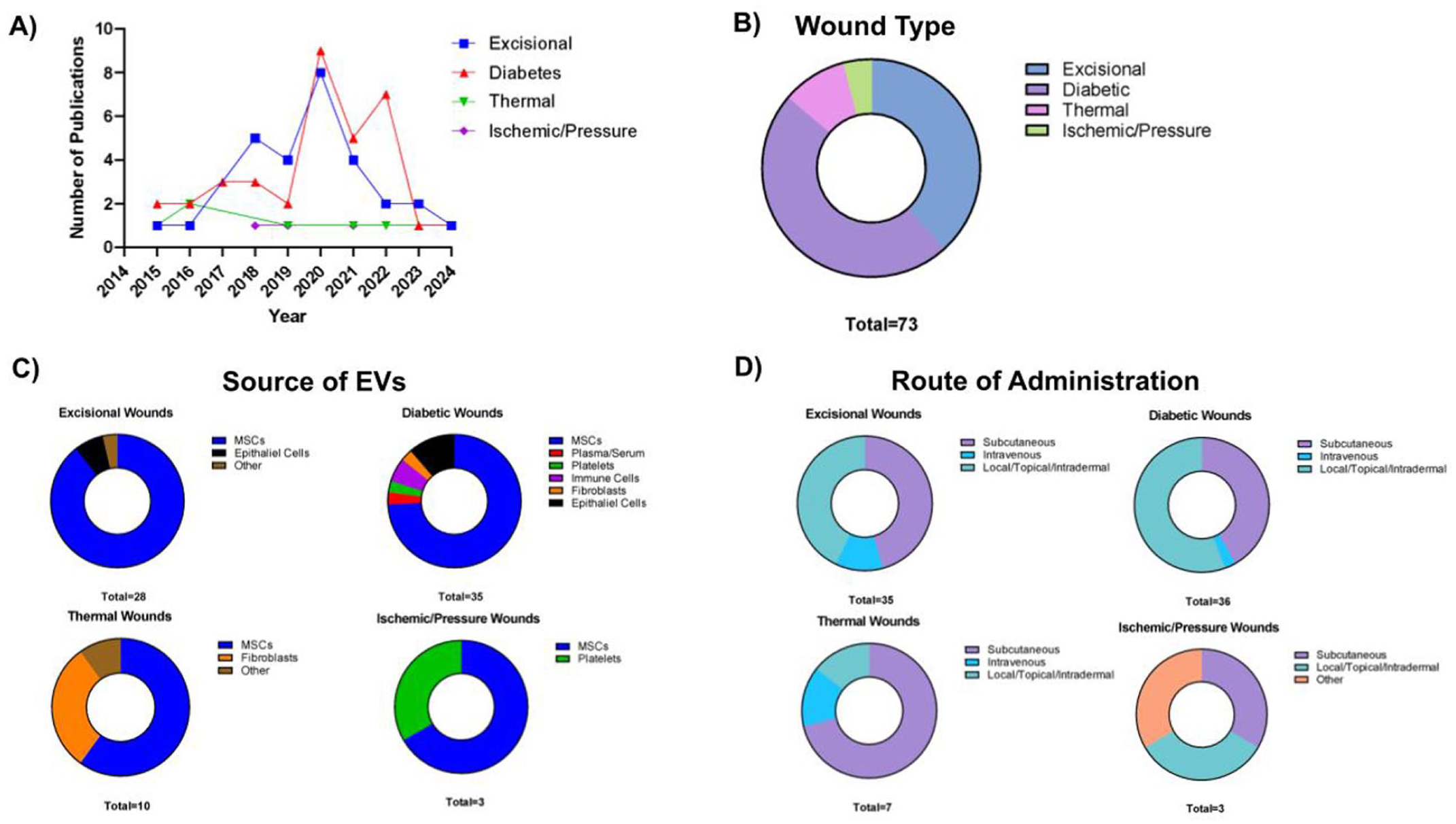
Given the results from studies mentioned in the introduction, it is unsurprising that many new investigations have been made regarding the therapeutic use of exosomes for wound healing. While several other reviews exist, the aim of this review in particular was to comprehensively outline how extracellular vesicles (EVs) and exosomes have been used in a variety of wound models. Recent reviews have summarized the potential of EVs in regenerative medicine; however, most have focused predominantly on in vitro studies or generalized EV biology without dissecting in vivo therapeutic applications for wound healing. This review provides a comprehensive synthesis of 73 studies, offering a comparative analysis across different wound types (excisional, diabetic, thermal, ischemic), EV sources, and delivery strategies; addressing translational gaps and comparing regenerative and immunomodulatory profiles of various EV sources, which has not been done in prior reviews. A summary of results based upon wound type can be found after each corresponding section.
This narrative review was conducted using PubMed until December 2024, using the keywords for initial search included “exosomes” OR “exosome,” AND “wound” OR “wound healing” OR “diabetic ulcer” OR “burn” OR “radiation wound” OR “pressure wound.” Studies were included if they met the following criteria: (1) involved in vivo animal models; (2) explored therapeutic applications of exosomes or extracellular vesicles (EVs) in wound healing; (3) provided measurable outcomes such as wound closure rate, re-epithelialization, angiogenesis, or scar formation. In particular, studies frequently cited across multiple reviews were prioritized for inclusion. Exclusion criteria included non-English manuscripts, studies without accessible full texts and any studies not supported by original data. For that reason, only original articles, and not review articles will be discussed in this review. While this is a narrative review, efforts were made to include representative studies across various EV sources and wound types, rather than relying on citation frequency or availability alone.
Exosome use in wound healing
As previously mentioned, not all wounds are created equally. Wound healing mechanisms may differ greatly depending on whether it is a simple excisional wound, or a chronic diabetic ulcer. As such, this review will be separated by wound type to better understand how exosomes may help for each wound model investigated. Summary of all study results seen in Figure 1.
Excisional wounds
There have been a large number of studies investigating the use of exosomes and their effect on wound healing in excisional or punch wounds. Most studies mentioned below did initial testing on in vitro scratch assays, then moved on to further investigate on animal wound models. A total of 28 studies regarding excisional wound healing will be discussed below.
Notably, most studies used subcutaneous or local wound injections (21/28), and the most frequent source of exosomes were adipose-derived mesenchymal stem cells (10/28). See Table 1 for summary of results for excisional wounds. Figure 2 shows a chronological summary of studies using excisional wounds.
In vivo use of exosomes in excisional wounds.

Note. Results are grouped by wound type, whether an in vitro or in vivo animal model was used, the source of exosomes, mode of delivery and a brief overview of the results. Studies that used the same source of exosomes are grouped together. Only statistically significant results from in vivo models were discussed.

Summary of studies examining excisional wounds.
Adipose-derived mesenchymal stem cells
First, the studies using adipose-derived mesenchymal stem cells will be discussed. Some studies investigated several delivery methods at once. Hu et al., for example, examined both subcutaneous and intravenous delivery of exosomes for wound healing. Initial in vitro testing was promising, demonstrating increased fibroblast proliferation and migration, as well as collagen synthesis with exosome exposure. Subsequent exosome injection in mice showed that both intravenous and subcutaneous application of exosomes led to statistically significantly increased wound closures on days 7, 14, and 21. Interestingly, intravenous injection showed improved results compared with subcutaneous application. Furthermore, collagen I and III expression showed increased peak levels on day 5 with intravenous injection, while both subcutaneous and intravenous injection showed overall increased collagen maturity on days 14 and 21 29 .
Zhou et al. similarly used both subcutaneous and intravenous exosome delivery; however, they also examined topical smearing application of exosomes. Initial results demonstrated that subcutaneous and topical exosomes significantly improved wound closure rates, although topical smearing application showed significantly improved wound closure velocity compared with other groups, with morphologically better epithelial tissue and reorganized collagen fibers. Notably, three times a day smearing treatment led to improved closure rates, re-epithelialization compared with twice or once daily. Increased smearing frequency also led to greater collagen maturity, with lower collagen I to collagen III ratios, a sign of scarless healing. Finally, similar to smearing, they found that systemic intravenous exosome application significantly accelerated wound closure, improved wound healing and decreased scarring 30 . Finally, Zhang et al. used both subcutaneous and intradermal exosomal treatment simultaneously in their study. They similarly found that exosomal treatment led to accelerated wound closure rate, enhanced re-epithelialization, decreased scar areas, increased collagen maturity and increased blood vessel density, a sign of improved angiogenesis 31 .
Several other studies investigated the use of ADSCs using only one method of delivery. Exosomal injection of wound edges showed promising results, with Yang et al. 32 demonstrating increased wound closure velocity and decreased scar areas on the final day. Zhu and Quan 33 similarly found increased wound closure rates, improved collagen deposition and blood vessel and granulation formation. Two further studies investigated subcutaneous exosome injection in particular, and found improved rates of wound closure34–36. Interestingly, two studies suggested that ADSC exosomes may act via microRNA 19b to improve wound healing, and Cao et al. 35 in particular showed that inhibition of microRNA 19b led to decreased rates of wound closure 34 .
Intravenous injection alone also showed promising results for wound healing, with significantly improved morphological features following exosome treatment, including improved epithelialization, keratinization and restoration of hair follicles compared with the non-exosome-treated group. Collagen deposition, which has been shown to be important for scar formation and overall wound size, was shown to be decreased in the exosome-treated group, and dermal thickness was also significantly decreased compared with the wound group, but much closer to normal values in the control, non-wounded group 36 .
Finally, ADSC exosomes were also applied topically. Zhou et al. investigated topical application of exosomes, using both exosomes in PBS and exosomes encapsulated in Pluronic F-127 hydrogel 37 . PF-127 hydrogel has been used as a scaffold to improve drug delivery for wound treatment 102 . Exosomal treatment, be it in Phosphate-buffered saline (PBS) or in the hydrogel, led to improved wound healing, with shorter wound length, increased markers of vascularization and proliferation, decreased inflammation, and decreased collagen I/III ratios, a sign of scarless healing. Interestingly, using the hydrogel did lead to some improved morphological characteristics by day 13, with new hair follicles and ordered collagen deposition. Notably, it also allowed for decreased application, and was only topically applied once every 3 days, compared with the thrice a day application of exosomes alone 37 .
Lee et al. similarly used topical exosome application with hyaluronic acid; however, they used a pig model for their study. This is of importance, as porcine skin is considered to be one of the most closely related to human skin103,104. Their results similarly showed that exosomal treatment led to improved re-epithelialization, wound closure and collagen III deposition 38 .
Bone marrow–derived mesenchymal stem cells
Bone marrow–derived stem cells were another common source of exosomes. Subcutaneous injection was also used, with a study by Jiang et al. investigating subcutaneous exosome injection compared with PBS control or intravenous BMSC injection. Interestingly, while intravenous BMSC did improve wound healing, subcutaneous exosomal treatment alone seemed to be more effective, with increased wound closure, increased cutaneous appendages, and increased markers of angiogenesis (VEGF). Decreased expression of TGF-β1, Smad2, 3, and 4 in the exosomal group suggests that inhibition of this pathway may be a possible mechanism 39 .
The type of cells from which exosomes are derived is of particular importance, which Kim et al. demonstrated when using bone marrow–derived macrophages as a source of exosomes. Notably, they used exosomes from either activated type 1 macrophages (pro-inflammatory), or reprogrammed type 2 macrophages (anti-inflammatory). The latter demonstrated significantly improved wound closure, as well as increased collagen, dermal thickness and granulation tissue. Exosome treatment with exosomes from type 1 macrophages did not show such effects. Understanding the source of isolated exosomes is therefore of great importance 40 .
Qiu et al. also investigated the importance of exosome source, whereby they intradermally injected excisional wounds with exosomes from neonatal or adult mice. By days 7 and 14, there was significantly increased healing with exosomes from neonatal mice, although exosomal treatment overall improved wound healing rate compared with control PBS treatment. This pattern was also shown in morphological differences, with increased integration of epithelia and appendages, more organized collagen deposition. Markers of angiogenesis and re-epithelialization were also increased, while markers of excessive scar formation were decreased 41 . Intravenous injection also accelerated wound healing, increased collagen formation, and increased vascular endothelial and proliferative markers. Notably, in this study, results between exosome treatment and BMSC were largely not significantly different 42 .
Finally, Su et al. used topical application of BMSC exosomes compared with subcutaneous injection on excisional wounds. Interestingly, topical exosome application had significantly increased wound closure, and improved morphological characteristics compared with controls, scaffold alone, or subcutaneous injection 43 .
Umbilical cord-derived stem cells
Umbilical cord-derived stem cells were another frequent source of exosomes. Studies included here mostly used subcutaneous or topical application. Subcutaneous injection of umbilical cord MSC-derived exosomes demonstrated increased wound closure rates, improved morphological characteristics, increased collagen and increased blood vessel formation44,45. Interestingly, Zhao et al. investigated the difference in effect when human umbilical cord MSCs (hucMSCs), hucMSC-conditioned medium, hucMSC exosomes, hucMSC exosome-deprived medium, or PBS and sham controls were injected. hucMSC, hucMSC-conditioned medium and hucMSC exosomes all showed different degrees of efficacy, although overall, the exosome group did tend to improve wound healing marginally. Notably, exosome-deprived medium showed no significant difference in results compared with the PBS-treated group, suggesting that the presence of exosomes is critical for improved wound healing 46 .
Topical application has also been investigated using umbilical cord-derived stem cells, with both leading to improved wound healing rates and morphological characteristics47,48. Zhang et al. chose to use a liquid chitosan band-aid for exosome delivery, as it can also form film once in place. Interestingly, the chitosan liquid band-aid alone also seemed to have a bacteriostatic effect on both staphylococcus aureus and Escherichia Coli. The inhibition zone was significantly increased compared with the control group, with an even greater increase in bacterial inhibition with the addition of exosomes 48 . Given that bacterial load can also present an obstacle for wound healing, this additional effect may be of interest for further studies.
Amnion or amniotic fluid-derived
There were also a few studies that investigated the injection of exosomes derived from amnion or amniotic fluid. Subcutaneous injection of exosomes from human amniotic fluid stem cells increased healing rate, improved morphological characteristics, and showed increased markers of proliferation, angiogenesis and nerve regeneration. Notably, exosomal injection was not significantly different from injection of human amniotic fluid stem cells alone 49 . Another study investigating the injection of exosomes derived from human amniotic epithelial cells found that exosomal treatment improved wound healing, as expected. However, they also found that exosomes treated with RNase or proteinases led to differing results. Rnase-treated exosomes led to decreased wound healing, while proteinase pre-treatment of exosomes did not abolish wound healing effects of exosomes 50 . The importance of RNA was further examined by Gao et al. They used exosomal treatment, as well as exosomes with either miR-135a overexpression or knockdown, to investigate wound healing. While exosomal treatment significantly improved wound healing compared with controls, miR-135a knockdown significantly decreased wound healing compared with exosomal treatment, while the fastest wound healing rate was found with miR-135a overexpression 51 . These studies in particular help elucidate the components in exosomes that may be critical for their beneficial effect on wound healing.
Other sources of exosomes
Finally, there were some rarer sources of exosomes that were investigated, including exosomes derived from iPSCs, fetal dermal stem cells, mucosal epithelia, saliva, epidermal stem cells and SK-MEL5 melanoma cells. Most of these were applied subcutaneously. Subcutaneous injection of exosomes from human iPSC cells also led to improved wound healing rates, with improved morphological characteristics (scar width, follicle and gland formation, vascularization) 52 . Interestingly, one study also used exosomes derived from host tissue, such as epidermal stem cells, with similar success, showing improved wound closure, decreased markers of excessive scarring, and increased markers of nerve and vessel regeneration 53 . Further studies with exosomes from fetal dermal mesenchymal cells and saliva-derived exosomes similarly demonstrated improved wound healing, angiogenesis and improved morphological characteristics54,55.
Moreoever, topical application of exosomes have been examined using mucosal epithelia. The investigators used exosomes from non-conditioned and conditioned media. The exosomes from conditioned media represented exosomes derived from the media in which the mucosal epithelial cells were cultured, while exosomes from non-conditioned media were not exposed to cell cultures. As expected, only exosomes from conditioned media led to a significant decrease in wound width. Furthermore, they found that more frequent application (compared with one larger dose) led to decreased wound width 56 .
The above-mentioned studies have shown that the use of exosomes, even from less common sources such as saliva, can be used for the treatment of excisional wounds. Further work characterizing and investigating the components in exosomes may be beneficial to better understand the mechanisms of action for improved wound healing with exosome treatment.
Diabetic wounds
Diabetic wounds have also been of great interest in the field of wound healing. Diabetic wounds are often much more difficult to heal, and multifactorial. Hyperglycemic states, together with increased inflammation, peripheral neuropathy, vasculopathy with significantly decreased perfusion, and oxidative stress make diabetic wounds all the more complex105,106. As such, these wounds are much likelier to turn chronic. Furthermore, diabetic wounds are more prone to infection, with a greater colonization of both Staphylococcus aureus and epidermidis 105 . Notably, the global prevalence of diabetic foot wounds is estimated to be around 6.3%, and up to 13% in North America 107 . Given the complexity in the pathophysiological mechanisms of diabetic wounds, as well as the significant global disease burden, finding viable treatment options is therefore all the more critical.
A total of 35 studies on the use of exosomes in diabetic wounds will be discussed below. As with excisional wounds, the most frequent source of exosomes were adipose-derived stem cells (10/35), and the most frequent application method was local or subcutaneous injection (23/35). See Table 2 for summary of results for diabetic wounds. Figure 3 shows a chronological summary of studies using diabetic wounds.
In vivo use of exosomes in diabetic wounds.
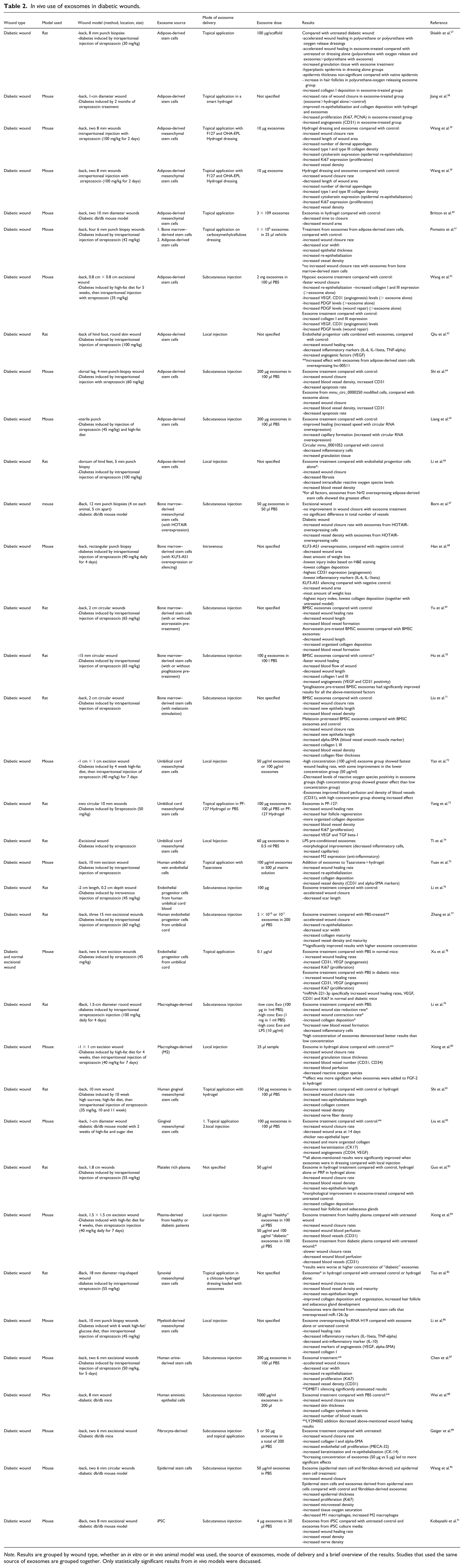
Note. Results are grouped by wound type, whether an in vitro or in vivo animal model was used, the source of exosomes, mode of delivery and a brief overview of the results. Studies that used the same source of exosomes are grouped together. Only statistically significant results from in vivo models were discussed.

Summary of studies examining diabetic wounds.
Adipose-derived stem cells
Adipose-derived stem cells were once again the most frequent source of exosomes for the treatment of diabetic wounds. Overall, either topical or local injection of exosomes were used as methods of application.
In terms of topical application, several different types of dressings were used. Shiekh et al. examined exosome delivery using either an antioxidant polyurethane dressing or an antioxidant polyurethane-based oxygen releasing scaffold, the latter of which used calcium peroxide to allow for oxygen release over 10 days. While the dressings alone already improved wound healing, adding exosomes further accelerated wound closure, with exosomes in the polyurethane-oxygen releasing scaffold performing the best. This trend was also true with highest granulation tissue thickness being achieved in the polyurethane-oxygen releasing exosome-treated groups. Increased collagen I deposition, collagen organization and maturity were also seen in exosome-treated groups. Finally, while epidermal thickness was greatly increased in the dressing alone groups, suggesting some hyperplastic effect on wound healing, the exosome-treated groups did not show this same trend 57 . Three further studies used different types of hydrogels to allow for exosome encapsulation delivery. Jiang et al. 58 demonstrated that their polyethylene glycol metalloproteinase degradable hydrogel alone already improved overall wound healing; however, adding exosomes further improved wound healing rate, angiogenesis, and cell proliferation significantly. Wang et al. 59 similarly saw overall improved results with their Pluronic F-127/oxidative hyaluronic acid hydrogel dressing alone, as well as exosomes alone; however, combining the two significantly increased wound closure rate, wound area length, number of dermal appendages, collagen remodeling, re-epithelialization, and angiogenesis. Britton et al. used a protein-based hydrogel, Q5 to allow for exosome delivery. Wounds treated with exosomes in hydrogel showed significantly decreased time to closure and decreased wound area. Notably, there was not a significant difference in results compared with local injection of exosomes in the wound 60 .
Finally, Pomatto et al. used a carboxymethylcellulose dressing to deliver exosomes from both ADSCs and BMSCs. Notably, they found that exosomes from BMSCs were not as effective, while exosomes from ADSCs had increased wound closure rates, decreased scar width, increased re-epithelialization and blood vessel density 61 .
Local injection of exosomes from ADSCs has also shown great potential. Wang et al. 62 found that exosomes derived from ADSCs under hypoxic conditions had particularly improved wound healing rates, re-epithelialization, angiogenesis, and collagen remodeling, even when compared with exosomes derived from non-hypoxic conditions. Changing the conditions or characteristics of ADSCs was further explored in other studies. Several studies investigated RNA, and what effect over or under expressing specific RNA may have on diabetic wound healing. Qiu et al. evaluated the effect of injecting endothelial progenitor cells, combined with exosomes or with exosomes from ADSCs overexpressing lnc-00511. While endothelial progenitor cells alone improved wound healing, adding exosomes, and particularly exosomes from lnc-00511 overexpressing cells, led to significantly increased wound healing, decreased expression of inflammatory factors and increased expression of angiogenic factors 63 . Two further studies investigated exosomes derived from circular RNA overexpressing ADSCs. Liang et al. used mmu_circ_0001052 overexpression, while Shi et al. used mmu_circ_0000250 overexpressing ADSCs. Both studies found that, while exosome treatment alone was beneficial, when treated with exosomes from ADSCs with the above-mentioned overexpression, wound healing overall improved64,65.
Finally, Li et al. examined how Nrf2 overexpression in ADSCs could improve wound healing, as this factor alone has been shown to improve wound healing 108 . They used exosomes to complement injection of endothelial progenitor cells. While endothelial progenitor cells already decreased ulceration size, adding exosomes promoted this effect, with the largest effect being seen with exosomes from Nrf2 overexpressing ADSCs. This overall trend was also true in terms of increased blood vessel density, decreased fibrosis and decreased oxidative stress 66 .
The studies mentioned above therefore not only demonstrate the use of ADSC exosomes on diabetic wound healing, but how manipulating the source may further improve outcomes.
Bone marrow-derived stem cells
Although adipose-derived stem cells have been the most frequent source of exosomes for the treatment of diabetic wounds, bone marrow-derived stem cells were also frequently used. As previously mentioned, Pomatto et al. 61 did not find any success with exosomes from bone marrow–derived stem cells. Notably, many of the below mentioned studies pre-treated or modified bone marrow-derived stem cells prior to exosome collection.
Born et al., for example, used extracellular vesicles (positive for exosomal markers) from BMSCs that were transfected to overexpress long non-coding RNA HOX transcript antisense RNA (HOTAIR). They hypothesized that HOTAIR overexpression would increase vascularization. Interestingly, they used both a simple excisional, and a diabetic wound model, and found that wound closure was only significantly increased in the diabetic wound model. In the diabetic model, only wounds treated with extracellular vesicles from HOTAIR-overexpressing cells showed significantly increased wound closure and vessel density 67 . Han et al. also investigated long non-coding RNA, specifically, KLF3-AS1 overexpression in BMSC and its effect on exosomal wound healing. This was the only case of intravenous injection, whereas the other application methods using BMSC derived exosomes involved local injection. They found that KLF3-AS1 overexpression led to improved wound healing, blood vessel formation and decreased inflammation and collagen deposition. Silencing, however, demonstrated very similar results to the untreated diabetic wound model, suggesting that this is indeed an important factor for wound healing 68 .
Further studies used other methods of BMSC pre-treatment prior to exosome collection, including atorvastatin, pioglitazone and melatonin69–71. Atorvastatin, commonly used to decrease lipid levels, was chosen based on findings demonstrating increased tissue repair in acute lesions in rats 109 . While BMSC derived exosomes did improve wound healing rate and blood vessel formation, the effect was significantly enhanced when using exosomes from atorvastatin pre-treated BMSCs 69 . This trend was also seen when Hu et al. 110 used pioglitazone, an anti-diabetic that has also been shown to decrease oxidative stress 111 . Wound healing rate, collagen deposition and angiogenesis were increased with BMSC exosomes; however, the effect was more distinct with pioglitazone pre-treatment 70 . Finally, Liu et al. 113 used melatonin treatment on BMSCs, as they particularly focused on its anti-inflammatory effect 112 . As before, melatonin treatment of BMSCs enhanced the positive effect of exosomes when used in an in vivo diabetic wound model, with increased rates of wound healing, re-epithelization, neovascularization and collagen synthesis 71 .
Umbilical cord origin
Several studies also used exosomes derived from the umbilical cord, or from endothelial cells derived from the umbilical cord or umbilical vein. In terms of exosomes derived from umbilical cord mesenchymal stem cells, three studies in particular will be outlined. The first study investigated how low or high exosome concentration affects wound healing, using either 50 or 100 µg/ml exosomes. For all variables, including wound closure rate, blood perfusion, angiogenesis and levels of reactive oxygen species, higher exosome concentration led to increased effect, although both groups had significant effects compared with the control 72 . Yang et al. similarly used umbilical cord mesenchymal stem cells as a source of exosomes; however, they used a topical application. They compared exosome treatment in PBS compared with in a hydrogel, and found that exosomes applied in a hydrogel led to significantly improved wound healing rate, angiogenesis and proliferation compared with exosomes in PBS, control, PBS or hydrogel alone 73 . One study also investigated how preconditioning could affect wound healing, and used lipopolysaccharide pre-conditioned umbilical cord mesenchymal stem cells as a source of exosomes. They found that compared with exosomes from unconditioned cells and untreated wounds, preconditioning led to improved morphological characteristics and wound healing 74 . Finally, Yuan et al. had promising results using exosomes derived from human umbilical vein endothelial cells. They used a hydrogel loaded with tazarotene, a retinoic acid. They found that addition of exosomes significantly improved wound healing rates, re-epithelialization, collagen deposition and neovascularization 75 .
All further studies discussed used endothelial progenitor cells from the umbilical cord as a source of exosomes. Two of the studies found that these exosomes did significantly improve wound healing, although Zhang et al. 77 did demonstrate that an increase in exosome concentration led to significantly better results with wound closure rates, re-epithelialization, scar width and blood vessel formation 76 , 77 . Xu et al. further showed that exosomes derived from these endothelial progenitor cells improved wound healing in both normal and diabetic mice, and specifically demonstrated that miRNA-221-3p alone, one of the top three miRNA expressed in the exosomes, could significantly improve the factors mentioned 78 . This may help further understanding of the specific factors that may be involved in enhancing wound healing.
Other: macrophage-derived, gingival mesenchymal stem cells, plasma, amniotic, synovial, IPSC, fibrocyte, epidermal stem cells, urine
Many other, less frequent sources of exosomes have also been used to treat diabetic wound models. Of the less frequent sources, two studies investigated macrophage-derived exosomes. Li et al. found that macrophage-derived exosomes improved wound healing rates, particularly at a higher concentration. Furthermore, direct addition of lipopolysaccharides to exosomes led to increased inflammatory markers, and attenuated exosomal wound healing effects 79 . Xiong et al. similarly used macrophage-derived exosomes; however, they examined its use together with fibroblast growth factor-2 (FGF-2) in a hydrogel. Exosomes in the hydrogel alone led to significantly improved wound closure rates, angiogenesis and decreased reactive oxygen species levels, with even further improvement when exosomes were combined with FGF-2 in the hydrogel 80 .
Two further studies examined the use of exosomes from gingival mesenchymal stem cells. While not as commonly used as bone marrow, adipose or umbilical cord counterparts, these mesenchymal stem cells are also highly proliferative, and easily accessible 114 . Shi et al. used gingival mesenchymal stem cell–derived exosomes in a topical application manner, and incorporated them in a chitosan/silk hydrogel. As expected, wound healing rate, re-epithelialization, collagen deposition, blood vessel and nerve fiber density were all increased in the group treated with exosomes 81 . Liu et al., while also using a topical application, compared this to local injection of exosomes. While local injection did show improvement in wound healing results, topical application in microspheres was significantly more effective 82 .
Plasma has also been studied as a source of exosomes. Results from the study by Guo et al. demonstrated that exosomes from platelet-rich plasma could improve wound healing and angiogenesis, even when compared with platelet-rich plasma alone 83 . Xiong et al. then further examined how exosomes derived from plasma from healthy or diabetic patients could affect wound healing. They showed particularly interesting results, with the expected improvement in wound healing with exosomes derived from healthy patients, while treatment with exosomes from diabetic patients led to significantly decreased wound closure rates, perfusion and angiogenesis. This was even more significant at higher concentrations of exosomes derived from diabetic patients. These results in particular demonstrate that the organism from which exosomes are sourced must be considered as a whole, and adds another layer of complexity when considering the use of exosomes in wound healing 84 .
Several studies also investigated how modifying less commonly used cell sources for exosomes could improve wound healing. Tao et al. used exosomes derived from synovial mesenchymal stem cells in a topical chitosan hydrogel dressing. They used stem cells overexpressing miR-126-3p for increased angiogenic activity, and found that wound healing, angiogenesis and re-epithelialization did in fact improve significantly when treated with these exosomes 85 . Another study investigated the use of exosomes overexpressing long non-coding RNA H19 derived from myeloid-derived mesenchymal stem cells. Interestingly, they did not find an increase in wound closure with exosome treatment alone, while H19 overexpression did significantly accelerate wound healing, decrease inflammation, and increase markers of angiogenesis and collagen I. Notably, they found that this beneficial effect was related to H19 interaction with miR-152-3p, the same microRNA that Tao et al. investigated above 86 . Chen et al., using urine-derived stem cells, instead of examining overexpression of factors, investigated whether silencing of DMBT1, an angiogenic factor, could influence results. As expected, exosomal treatment significantly improved wound closure, scar width, re-epithelialization and proliferation. Silencing DMBT1, however, significantly attenuated these positive effects, suggesting that this, too, may be a critical factor in mediating exosomal wound healing 87 . Wei et al. 88 similarly demonstrated that when LY294002, a PI3K-AKT-mTOR pathway blocker, was added to amniotic epithelial cell-derived exosomes, wound healing results were significantly decreased. These studies directly demonstrate factors that may be involved in diabetic wound healing mechanisms.
Finally, several more studies examined rarely used sources of exosomes, including fibrocytes, iPSCs or epidermal stem cells. Notably, the study using fibrocyte-derived exosomes, both subcutaneous injection and topical application was used. Furthermore, they found a dose-dependent increase in wound healing effect with increasing exosome concentration 89 . Further comparisons were made within studies, with comparing exosomes derived from epidermal stem cells versus exosomes derived from fibroblasts. Epidermal stem cells as well as exosomes derived from epidermal stem cells led to significantly better wound healing compared with fibroblast-derived exosomes 90 . Kobayashi et al. 91 also demonstrated that exosomes derived from iPSC culture media did not lead to the significant wound healing results that were found with direct iPSC-derived exosomes. These studies highlight the importance of exosomal source in treatment effect.
Thermal wounds
Thermal wounds are a complex type of wound, and severe burn injuries are associated with high morbidity and mortality, with over 100,000 deaths being linked to burns globally in 2019 115 . Moreover, the scars related to burn wounds can be debilitating with major pain, itch and impaired mobility. Therefore, a favorable scar formation during the regeneration phase is as important as fast closure and re-epithelialization of burn wounds with improvement during the proliferation phase. The local thermal wound is generally split into three zones, the zone of coagulation, the zone of ischemia and the zone of hyperemia. The central zone, which is exposed to the highest levels of heat and termed the zone of coagulation, represents an area of irreversible tissue injury. It involves high protein degradation and coagulation necrosis. Surrounding this zone, is the zone of ischemia, termed for its decreased perfusion, where tissue necrosis can be avoided if treated adequately and in a timely manner. The mechanism of tissue death in this area seems to involve oxidative stress, inflammation and impaired tissue perfusion. Finally, the zone of hyperemia is the most peripheral region of the wound, where inflammatory vasodilation leads to increased blood perfusion. Outside of the local injury, when burned body surface area exceeds 20%, excessive inflammation occurs and can lead to systemic shock, organ failure, and even death116,117. Outside of the acute complications, later stages of wound healing become particularly significant in burn wounds, as up to 33% of patients experience contracture and hypertrophic scarring, which can greatly limit quality of life 118 .
Exosomes also seem to play an important role in post-burn healing, with next-generation sequencing demonstrating that several miRNA expressed in exosomes of burn patients differed from healthy volunteers, many of which are involved in cell proliferation and tissue repair. This ultimately suggests that circulating exosomes following a burn may influence wound healing stages in burn wounds, and may therefore represent possible therapeutic targets 119 .
Exosome use in thermal wounds
See Table 3 for summary of results for studies on thermal wounds. Figure 4 shows a chronological summary of studies using thermal wounds. The most frequent source of exosomes used in thermal wounds were human umbilical cord mesenchymal stem cells. Most studies discussed second degree burns, with only one investigating a third degree burn model. Within the second degree burn models, several studies compared the efficacy of exosomes derived from human umbilical cord mesenchymal stem cells compared with exosomes derived from skin or lung fibroblasts. As expected, when derived from umbilical cord mesenchymal stem cells, the exosomes significantly increased markers of angiogenesis, increased re-epithelialization, proliferation and improved overall histological morphology92,93. Zhang et al. further demonstrated that the protein 14-3-3ζ seems to be involved in the wound healing process, particularly in controlling excessive proliferation and regeneration, as silencing of said protein increased proliferation (PCNA) and stem cell markers 94 . This is of course interesting when considering the risk of contracture and excessive scarring in burn wounds; however, they did not directly investigate collagen content or dermal proliferation in regard to scarring in their in vivo model. Finally, the only third degree burn model, and intravenous use of exosomes derived from human umbilical cord mesenchymal stem cells was examined by Li et al. Notably, they mostly investigated how these exosomes could attenuate excessive inflammation, and found that exosomes derived from the umbilical cord decreased white blood cell count, and inflammatory markers in general. Furthermore, histological analysis also demonstrated decreased neutrophil and macrophages in the exosome-treated group 95 . This is particularly important in preventing delayed healing due to excessive inflammation, as well as decreasing systemic complications in severe, large area burns.
In vivo use of exosomes in thermal wounds.

Note. Results are grouped by wound type, whether an in vitro or in vivo animal model was used, the source of exosomes, mode of delivery and a brief overview of the results. Studies that used the same source of exosomes are grouped together. Only statistically significant results from in vivo models were discussed.
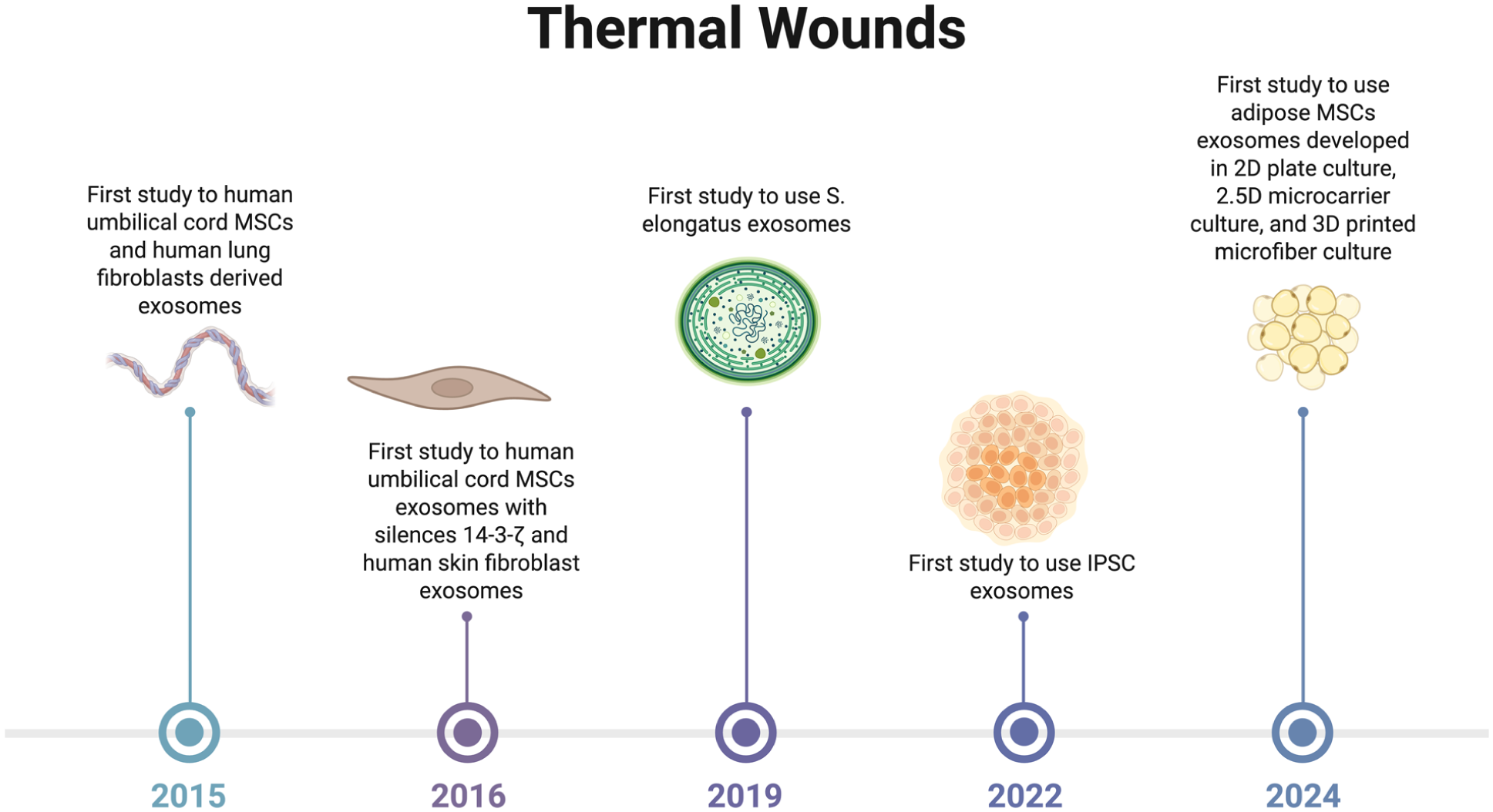
Summary of studies examining thermal wounds.
Other sources of exosomes have also been investigated, including adipose-derived mesenchymal stem cells. In this case, they compared how tissue repair was affected based on how the cells were cultured. While exosome treatment improved wound healing, angiogenesis and collagen synthesis, this effect was even more significant when exosomes were derived from cells cultured in a 3D culture, compared with a 2D or 2.5D microcarrier culture. Furthermore, they found that while blood vessel number was increased at day 7 in exosome-treated mice, at day 14, this number was significantly decreased in mice treated with exosomes from the 3D and 2.5D cultures compared with hydrogel alone or exosomes from 2D cultures. This may reflect that exosomes derived from 3D or 2.5D cultures accelerated wound healing, and that by day 14 these wounds have moved from the proliferation phase with granulation tissue onto the remodeling phase, while the other wounds experience lagging tissue repair 96 .
Interestingly, two further studies using exosome treatment in burn wounds found that wound closure rates, re-epithelialization and angiogenesis were improved compared with control, collagen deposition was not significantly different97,98. One of the studies used iPSCs, while the others used extracellular vesicles derived from Synechococcus elongatus. These extracellular vesicles were marked with PKH67, an exosome labeling dye98,120. They used this model, as S elongatus delivery has been shown to improve tissue oxygenation in ischemic heart disease, and this may represent an interesting and unconventional source of extracellular vesicles and exosomes 121 . However, further characterization would be required to better understand the mechanism of action in this case.
Studies on burn models predominantly utilized umbilical cord-derived EVs, demonstrating significant improvements in inflammation modulation, angiogenesis, and tissue remodeling. However, few studies assessed hypertrophic scar formation, a critical endpoint in burn wound healing. Comparative efficacy across EV sources remains underexplored in thermal wounds.
Other: ischemic, pressure, radiation wounds
Exosomes have also been used for some less studied types of wounds, which will be covered here. Figure 5 shows a chronological summary of studies using pther types of wounds. Table 4 covers study results.

Summary of studies examining other types of wounds.
In vivo use of exosomes in other, rarely studied wounds.
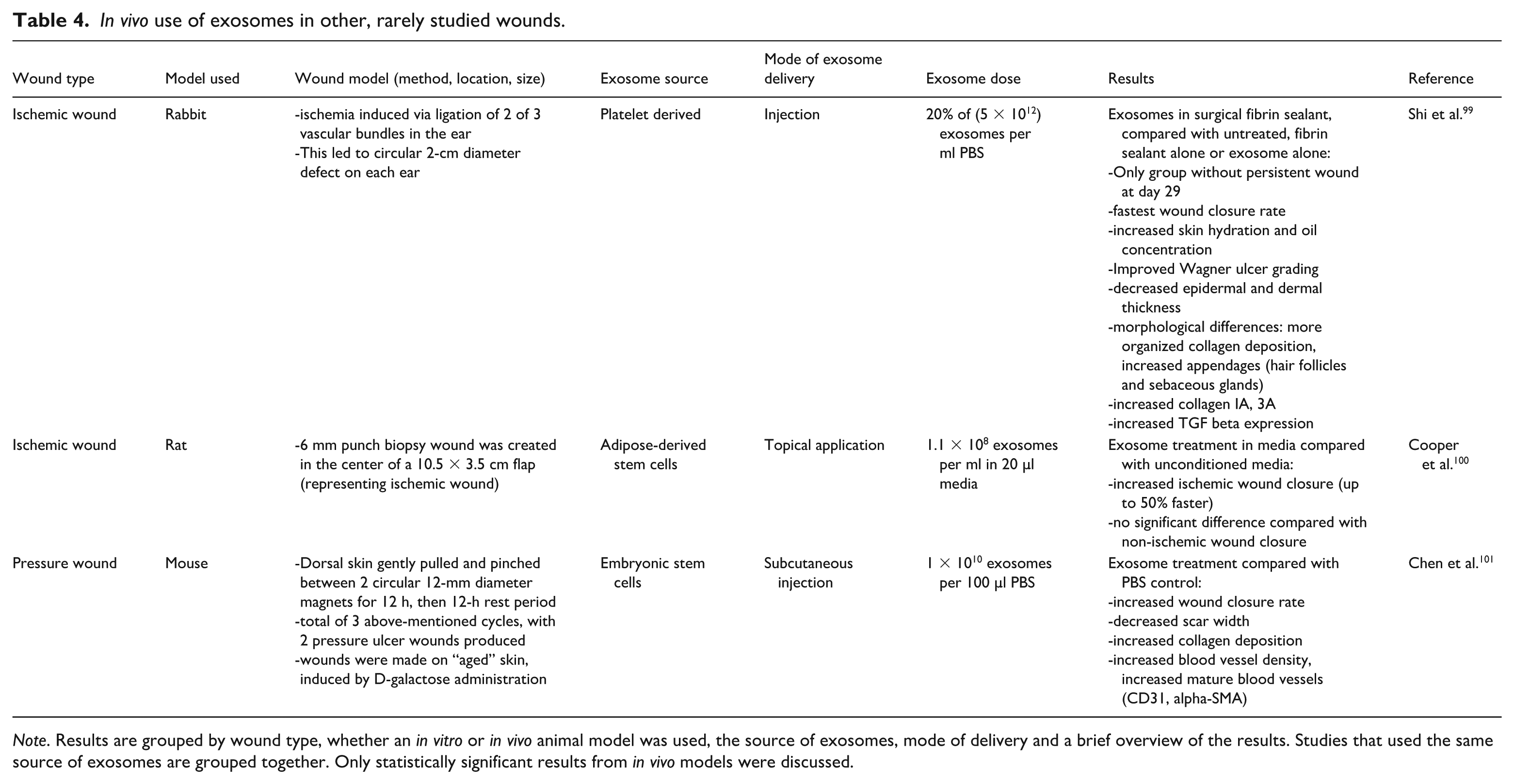
Note. Results are grouped by wound type, whether an in vitro or in vivo animal model was used, the source of exosomes, mode of delivery and a brief overview of the results. Studies that used the same source of exosomes are grouped together. Only statistically significant results from in vivo models were discussed.
First, several studies have demonstrated efficacy in using exosomes for ischemic injuries in several different organ models such as the brain or the heart122–124. Given these promising results in other organ systems, it is reasonable to investigate exosome use in ischemic skin wound models. Arterial ischemic leg ulcers, for example, make up approximately 5%–20% of all non-healing leg ulcers 125 . Given their ischemic nature, successful wound healing is all the more difficult. One study used injected platelet-derived exosomes on an ischemic wound model, whereby 2 of 3 vascular bundles were ligated. Exosomes were delivered within a surgical fibrin sealant to increase their in vivo half life, and they found that this significantly improved wound closure rate, morphological characteristics, collagen deposition and decreased epidermal and dermal thickness. This last observation, in particular, suggests that exosome treatment could also help avoid excessive scarring 99 . Topical application of adipose-derived stem cell exosomes also increased ischemic wound closure rate, with no significant difference compared with wound healing rate in non-ischemic wound closure rate 100 .
Another study examined the use of exosomes in the healing of pressure ulcers. While the etiology is different from arterial ischemic wounds, the mechanism also involves tissue ischemia due to increased pressure and subsequent microcirculatory occlusion. This serious complication has a large mortality and morbidity rate, and proper management is therefore critical126,127. Chen et al. found that embryonic stem cell-derived exosome treatment significantly improved wound closure rates, scar width, collagen deposition and angiogenesis. Interestingly, they used an “aged” skin model, using D-galactose administration to induce aging skin in mice, as pressure ulcers are even more difficult to treat with increasing age and declining tissue perfusion. They found that all the above-mentioned variables were not significantly different when exosome treatment in “old skin” was compared with “young” wound healing models, and may therefore not only improve wound healing but have an overall anti-aging effect 101 .
Finally, radiation wounds are another interesting wound type to examine. Radiation therapy remains a life-saving treatment for numerous types of cancer patients. However, up to 85% of these patients develop moderate-to-severe skin reactions, termed radiation dermatitis. Current understanding of radiation skin injury involves direct tissue damage via oxidative stress and free radical DNA damage, together with a subsequent inflammatory response128–130.
While no direct in vivo models were used, two studies will still be briefly discussed here but not included in the summary of results table due to their promising results in helping with radiation-induced wounds. First, one study found that exosomes derived from the irradiated cheeks of neonatal mice. Interestingly, only exosomes collected 6 h from irradiated, and not radiated cheek skin, showed radio-protective effect, with increased repair kinetics and growth in radiated fibroblast cell lines 131 .
Another study demonstrated that conditioned media derived from mesenchymal stem cells could alleviate radiation-induced skin wounds in rats. Wound area was significantly decreased with MSC-conditioned media treatment, and morphological characteristics were also improved (appendage number, vessel number) 132 . They hypothesized that this improvement may likely be due to paracrine mechanisms and secretion of growth factors. Given the knowledge that exosomes remain key components of conditioned medium, it would be interesting to investigate whether exosome treatment alone could also improve radiation-induced wounds.
Mechanism of extracellular vesicle use in wound healing
Given the promising results for in vivo EV use in wound healing, this section will focus on possible mechanisms that may further explain the results found. This section will be divided based upon wound type.
Table 5 and Figure 6 summarize the mechanisms of EV-mediated wound healing.
Mechanisms of EV-mediated wound healing.

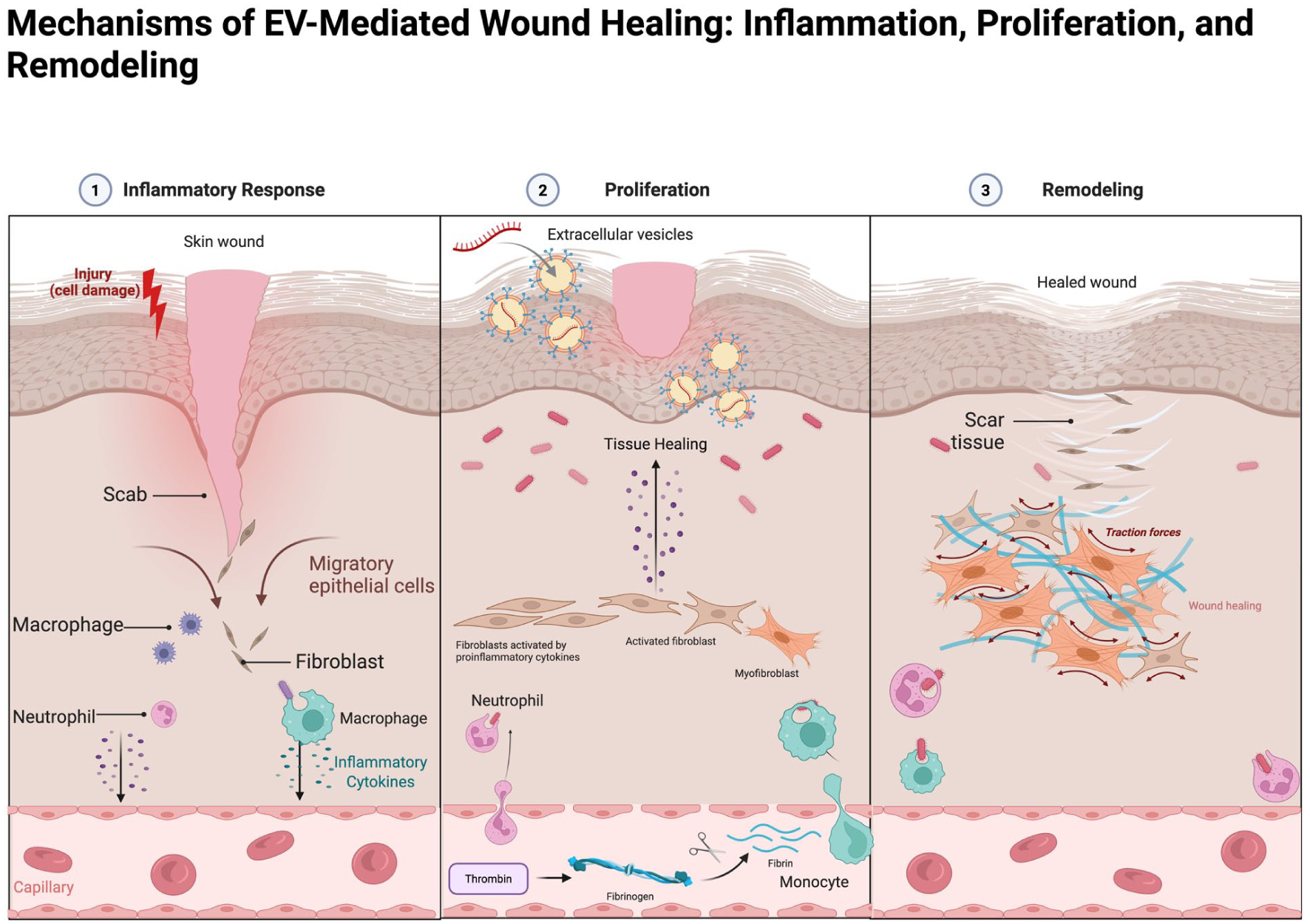
Figure of EV-mediated wound healing mechanisms.
Excisional wounds
Exosomes derived from various stem cells (adipose-, bone marrow-, umbilical cord-, amnion-derived, etc.) have shown potent pro-healing effects in acute excisional wounds. A key mechanism is the stimulation of fibroblast and keratinocyte proliferation and migration, primarily through activation of growth and survival pathways like PI3K/Akt and Wnt/β-catenin 15 . For example, adipose-derived MSC (ADMSC) exosomes are enriched in miR-21 and Wnt4; miR-21 targets Phosphatase and tensin homolog (PTEN) to unleash PI3K/Akt signaling, while exosomal Wnt4 directly triggers β-catenin activation 15 . Together, these signals drive cell cycle progression and migration in dermal cells, leading to faster re-epithelialization. In parallel, exosomes carry pro-angiogenic factors (e.g. VEGF-A, FGF2, and Angiopoietin) that stimulate new blood vessel growth to support the regenerating tissue 15 . Bone marrow MSC exosomes likewise promote angiogenesis and granulation tissue formation—for instance, by delivering miR-126 and other cargo that upregulate the HIF-1α/VEGF pathway 10 . Notably, MSC-exosomes can also modulate the Notch signaling involved in vessel maturation. ADSC exosomal miR-125a has been shown to target Delta-like ligand 4 (Dll4), an inhibitor of angiogenesis in the Notch pathway, thereby promoting more robust capillary sprouting 15 . Beyond mesenchymal cells, other cell sources contribute to wound closure: induced pluripotent stem cell (iPSC)-derived exosomes and epithelial cell-derived exosomes often contain abundant growth factors (FGF2, EGF, hepatocyte growth factor (HGF)) and matrix-remodeling enzymes, which accelerate keratinocyte proliferation and wound coverage 15 . These vesicles not only increase proliferating cell nuclear antigen (PCNA) levels in epidermal cells but also protect keratinocytes from apoptosis by inhibiting pro-apoptotic mediators in the wound edge 15 . The net effect in excisional wound models is a significantly faster closure with well-organized epidermal regeneration and a balanced deposition of new collagen. Studies report that exosome-treated wounds exhibit organized collagen fibers and appropriate transitions from collagen III to I during healing, which correlates with stronger yet supple scar tissue formation 10 . In summary, across multiple studies, exosomes act as paracrine boosters of the normal healing phases in acute wounds—they jump-start cell proliferation, enhance angiogenesis, and set the stage for effective tissue remodeling.
Diabetic wounds
Chronic diabetic ulcers present a hostile environment characterized by impaired angiogenesis, neuropathy, and persistent inflammation. Exosome-based therapy has emerged as a promising approach to overcome these barriers. A central benefit is the restoration of adequate angiogenesis under hyperglycemic conditions. Mesenchymal stem cell exosomes (from ADMSC or BM-MSC) can deliver microRNAs that upregulate the HIF-1α/VEGF signaling axis, a pathway often suppressed in diabetes. For instance, BM-MSC exosomal miR-126 and miR-24-3p have been shown to stabilize HIF-1α (by inhibiting VHL, the E3 ligase that degrades HIF) and consequently increase VEGF expression in ischemic diabetic tissue 10 . This leads to robust capillary growth and improved perfusion in the wound bed. Similarly, exosomes from umbilical cord MSCs may carry miRNAs (such as miR-21 or other PTEN-targeting miRs) that release the brake on the PI3K/Akt pathway, further amplifying downstream pro-angiogenic signals 15 . Besides neovascularization, immunomodulation is a critical mechanism in diabetic wound healing by exosomes. Chronic diabetic wounds are trapped in an inflammatory phase dominated by M1 macrophages and high levels of TNF-α, IL-1β, and IL-6. Exosomes help break this cycle by reprogramming immune cells: they promote a shift from M1 to M2 macrophage polarization 20 . ADMSC-derived exosomes, for example, significantly suppress pro- inflammatory cytokines (TNF-α, IL-6) while increasing markers of M2 macrophages when applied to diabetic wounds 20 . Key mediators include exosomal microRNAs like miR-223, which directly drives macrophages toward an anti-inflammatory M2 phenotype 18 , and miR-19b, which can reduce inflammatory chemokine (CCL1) levels via TGF-β-dependent pathways 18 . The result is a drop in chronic inflammation at the wound site and an increase in anti- inflammatory cytokines (e.g. IL-10, TGF-β), creating a milieu conducive to healing. In addition, some exosomes carry antioxidants and Nrf2 pathway activators that counteract the oxidative stress of hyperglycemia 10 . In diabetic models, treating with exosomes (especially those engineered to overexpress Nrf2) led to reduced ulcer area, increased granulation tissue, and higher expression of growth factors in the wound 10 . Taken together, exosomal therapy tackles diabetic wounds on multiple fronts: it enhances blood vessel formation, dampens prolonged inflammation, and even promotes collagen deposition and remodeling. By addressing both vascular insufficiency and inflammatory excess, exosomes markedly improve healing outcomes in diabetic foot ulcers and other chronic wounds.
Thermal wounds
In thermal injuries (burn wounds), exosomes contribute to healing in distinct ways, particularly by mitigating fibrosis and improving tissue regeneration. Burn wounds often heal with hypertrophic scarring due to prolonged inflammation and excessive myofibroblast activity. Exosome therapy can modulate the TGF-β/Smad signaling pathway that governs scar formation. Notably, mesenchymal exosomes have been reported to increase levels of Smad7 (an inhibitory Smad) while suppressing Smad2/3 activation in burn models 10 . This leads to a reduction in myofibroblast differentiation and a more controlled collagen deposition, ultimately limiting scar hypertrophy. For instance, ADSC-derived exosomes enriched in miR-192-5p were shown to attenuate collagen overproduction and contracture in a hypertrophic scar model by down-regulating TGF-β/Smad signaling 10 . Concurrently, exosomes promote dermal regeneration and angiogenesis in burn wounds. Human umbilical cord MSC exosomes, rich in Wnt4 protein, have been shown to activate Wnt/β-catenin signaling in recipient cells and are required for effective cutaneous wound repair in deep second-degree burns 15 . Activation of Wnt/β-catenin by exosomal Wnt4 encourages the proliferation of dermal cells and vascular remodeling in the injured tissue 15 . Indeed, treatment with hucMSC exosomes led to significantly greater angiogenesis in burn wounds, with increased CD31⁺ vessel density attributed to the delivered Wnt4 signal 15 . Exosomes also carry angiogenic growth factors like VEGF and angiopoietin-1, which support the formation of stable, perfused blood vessels in the burn area 15 . This neovascularization is crucial for supplying oxygen and nutrients to the regenerating epidermis and dermis. Another benefit observed in thermal wound models is the anti-apoptotic effect of exosomes on cells at the wound margins. By upregulating survival pathways (Akt/ERK) and anti-apoptotic proteins, exosomes help preserve more of the resident keratinocytes and dermal stem cells that can then proliferate to resurface the wound. Overall, in burn injuries exosomal therapy not only speeds up the closure of the wound but also improves the quality of healing—scars are flatter and more elastic due to controlled collagen remodeling, and the risk of contractures is reduced. This has been demonstrated in animal burn models where exosome-treated wounds showed better organized collagen fibers and less inflammation compared with controls 10 . The ability of exosomes to temper TGF-β-driven fibrosis while concurrently stimulating regeneration addresses the twin challenges of burn wound care: functional recovery of the skin and cosmetic minimization of scarring.
Other wounds (ischemic, pressure, radiation)
Ischemic wounds (such as chronic ischemic ulcers or poorly vascularized tissue injuries) benefit from exosome-driven improvements in blood supply and tissue survival. Exosomes from BM-MSCs, for example, can activate both VEGF and Notch signaling pathways in ischemic tissue, leading to the sprouting of new vessels and the maturation of those vessels into functional networks 25 . Interestingly, exosomal activation of Notch has been linked with enhanced fibroblast migration and secretion—one study using fetal dermal MSC-derived exosomes found that they accelerated wound closure by activating Notch signaling in adult dermal fibroblasts, effectively endowing adult cells with fetal-like regenerative capacity 25 . In addition, exosomes combat the high oxidative stress in ischemic wounds by delivering antioxidants or activating the cellular Nrf2 pathway. Embryonic stem cell-derived exosomes have been reported to activate Nrf2 in endothelial cells, thereby reducing oxidative damage and endothelial senescence in low-oxygen conditions 10 . This antioxidative action helps preserve tissue viability around the wound margin, setting the stage for more effective repair once perfusion is restored.
In pressure ulcers, which involve sustained mechanical stress, ischemia-reperfusion injury, and inflammation, exosome treatments focus on ECM remodeling and cell proliferation. Umbilical cord MSC exosomes have shown the ability to stimulate fibroblast proliferation in the ulcer bed and modulate collagen dynamics. By delivering enzymes and regulatory RNAs that influence collagen synthesis, exosomes help balance Collagen I/III ratios in the new tissue 10 . This balance is crucial: Collagen III provides initial flexibility, while Collagen I provides tensile strength. Properly timed transition from type III to type I collagen yields a scar that is strong but not overly stiff. Studies have found that exosome-treated pressure wounds exhibit improved tensile strength of the repaired skin, attributable to a more organized collagen architecture and reduced fibrosis. For example, in an animal pressure ulcer model, treatment with MSC exosomes led to higher collagen maturity and better biomechanical properties of the healed tissue compared with untreated wounds (likely due to upregulated Collagen I in later stages of healing) 10 . Exosomes also reduce the chronic inflammation in pressure ulcers by the same M2-polarization mechanisms noted above, which facilitates progression from the inflammatory phase to the proliferative phase of healing.
Radiation-induced wounds (from radiotherapy or nuclear accidents) are particularly challenging due to DNA damage, persistent oxidative stress, and impaired cell proliferation. Recent research indicates that exosome therapy can markedly improve healing in irradiated skin. One mechanism is the transfer of DNA repair proteins and miRNAs that enhance the survival of damaged cells. For instance, platelet-rich plasma-derived exosomes have been shown to activate pathways (like YAP and others) that bolster the DNA damage response in chronic radiation wounds, thereby accelerating re-epithelialization 21 . In addition, MSC-derived exosomes have demonstrated the ability to reduce radiation-induced cell death: they decrease markers of apoptosis (e.g. BAX and cleaved caspase-3) and pyroptosis in irradiated skin cells 23 . By inhibiting these cell death pathways, exosomes preserve more viable dermal and epidermal cells which can then participate in wound healing. Exosomal cargo such as anti-apoptotic miRNAs and heat-shock proteins likely contribute to this radioprotection. Another major issue in radiation wounds is chronic inflammation and fibrosis. Exosomes address this by modulating the immune response–treated wounds show reduced macrophage infiltration and a higher M1→M2 macrophage ratio 23 . Correspondingly, pro-inflammatory cytokines like IL-1β and IL-6 are suppressed in irradiated tissue after exosome treatment 23 . This creates an environment more permissive to healing, with less collateral tissue damage from inflammation. Finally, exosome-treated radiation wounds exhibit improved angiogenesis, as vesicles deliver pro-angiogenic factors to overcome radiation-induced vascular damage. Overall, the multifaceted actions of exosomes—enhancing cell repair, quelling inflammation, reducing oxidative stress, and stimulating angiogenesis—significantly promote the healing of otherwise refractory radiation injuries. Evidence from animal studies shows that wounds exposed to radiation heal faster and with less fibrosis when treated with ADSC exosomes, relative to controls 23 . This highlights the therapeutic potential of exosomes in managing complex wound types beyond the typical acute and diabetic wounds.
Challenges and conclusions
Comparison of studies and limitations
Overall, the many studies investigating exosome use for wound healing have shown promising results. And yet, it must be noted that only one study used a porcine model to examine wound healing with exosome treatment, while all others used mice or rats 38 . As has been mentioned, porcine skin is much more closely related to human skin, and investigating the effect of exosomes on a more closely related model will likely be of interest prior to testing on human subjects.
Furthermore, from the studies mentioned above, different methods of exosome use led to differing results, with unfortunately very few studies comparing different methods within the same study. The source of exosomes alone can have a distinct effect on wound healing results. Notably, one study did find that bone marrow-derived stem cells were less successful compared with adipose-derived stem cells for diabetic wound healing 61 . Interestingly, most other studies using bone marrow-derived stem cells as a source of exosomes for diabetic wound healing modified the cells prior to collecting exosomes67–71. Furthermore, when examining how plasma-derived exosomes performed when collected from healthy or diabetic patients, when exosomes were derived from diabetic patients, wound healing results were even worse than untreated groups. This was especially true at higher concentration 84 . This suggests that not only does cell source play a role, but the overall organism from which exosomes are derived must be considered.
Application methods were also rarely compared within individual studies; however, some comparison was made. For example, several studies found topical application to have greater effect on excisional wound healing than local injection43,82. Another study similarly found that subcutaneous exosome injection, while increasing wound closure rates, did not have as distinct an effect on morphological characteristics in excisional wounds when compared with topical or intravenous application 30 . Another study on excisional wounds also found that compared with subcutaneous injection, intravenous injection led to significantly improved results 29 . These results suggest that topical application and intravenous injection may be more beneficial than subcutaneous injection; however, further study may be required. Differing concentrations of exosomes also play a role, as increasing concentration of exosomes may be more effective72,77,89. Furthermore, within methods of application, when injecting or applying exosomes topically, exosomes may also be placed in a further scaffold or hydrogel prior to application. Indeed, two studies found that exosomes in hydrogel performed better than when in PBS37,73. In relation to topical application, one must also consider the type of dressing used when applying exosomes, which may further influence wound healing. There are several different types of wound dressings, and modern dressing may even include interactive or bioactive dressing with the addition of several ingredients such as silver/iodine, hydrocolloids, or more 133 . Given the above-mentioned differences, further investigation regarding methods of application is certainly required, as methods were very rarely standardized.
Several studies also modified or pre-treated cells prior to exosome collection, often with positive results50,51,62–71,85,86. This not only helps understand the mechanisms by which exosomes may improve wound healing but also adds another layer of complexity on how to best optimize wound healing, and what factors may further stimulate repair. Notably, when Ti et al. 74 used preconditioning cells with lipopolysaccharides prior to exosome collection; this led to improved wound morphology and decreased inflammation. However, directly adding lipopolysaccharides to exosomes during application to wounds led to increased inflammation and overall decreased wound healing 79 . While lipopolysaccharides are generally regarded as pro-inflammatory, several studies have shown that preconditioning cells or using low doses has the opposite effect and may be protective134,135. Furthermore, method of culture also seems to have an effect on exosome treatment impact, with one study demonstrating that exosomes derived from 3D cultures performed better than exosomes from 2D cultures 96 . Understanding these nuances therefore remains critical to maximize wound healing effect. Further culture conditions may also need to be considered, as one study has found that blue light exposure may influence the pro-angiogenic capabilities of exosomes 136 .
Finally, in terms of comparison between wound types; the most extensively studied were excisional and diabetic wound healing. Very few studies directly compared exosome treatment in different wound types. One study in particular demonstrated that exosome treatment did improve wound healing rate in both normal excisional and diabetic excisional mice. As expected, an excisional wound on normal mice showed improved wound healing rate. Interestingly, when comparing exosome treatment to exosome treatment, there was a greater increase in wound healing rate in normal mice than diabetic mice 78 . Furthermore, very little study has been done for other wound types, and whether exosomal treatment can have a benefit in other wound types, such as thermal, ischemic or pressure wounds will require further study.
Discrepancy in results
While most studies had similar findings, there were certain discrepancies. In particular, some studies found increased levels of alpha-SMA 39 , 43 , 102 , others found a decrease in this marker level 45 , 49 , 53 . This may be due to the timing of when wounds were analyzed. Given alpha-MSA is a marker of myofibroblast, their expression may be dependent on the stage of wound healing. While myofibroblasts are certainly required for early wound contraction, if they are overly present in later stages, they can be a marker of hypertrophic scarring 137 . As such, while increased alpha-SMA may be beneficial at earlier stages, later overexpression of this marker may not be desired. Notably, some studies also used alpha-SMA as a smooth-muscle marker to examine new vessel formation and angiogenesis, and in that context, exosome treatment often increased alpha-SMA expression. In line with the above-mentioned conflicts, while most studies found exosome treatment to lead to increased collagen deposition compared with untreated control, some studies observed decreased or no significant difference in collagen deposition36,49,53,68,97,98. This may once again be explained when considering the time point when collagen staining or analysis was done, as during late tissue repair, low collagen content correlates with decreased scarring. Furthermore, ratio of collagen I to III seems to be important in determining degree of scarring, with increased type III collagen compared with type I collagen being associated with scarless healing 138 . Several studies found that this ratio could be found with exosome treatment30,37,38,49,53. One study found contradicting results in terms of this ratio, however, and many others simply looked at overall increase in collagen without considering the ratio 93 . Finally, when discussing scarring in wound healing, some studies also found that exosome treatment could decrease scar width44,52,55,61,77,87,98,101. These findings are notable, as scarless healing is often a desired quality, and avoiding hypertrophic or excessive scarring is also an important characteristic.
Toxicity considerations
Although several studies have investigated the influence of exosomes concentration on wound healing and suggest a promising therapeutic profile, the field remains limited by a lack of rigorous exploration into dose-response relationships and safety parameters. No study to date has reported overt toxicity or adverse effects associated with exosomes administration, even at elevated doses. From both pharmacologic and clinical perspectives, understanding exosome kinetics (absorption, distribution, metabolism, and excretion) is essential for translating these therapies into safe and effective clinical protocols. Yet none of the reviewed studies conducted biodistribution analyses using labeled exosomes, nor did they evaluate potential off-target effects or systemic toxicity, despite the use of intravenous or repeated topical applications in several models. Furthermore, it is important to consider the possibility of exosome-mediated fibrosis, unwanted neoangiogenesis (in keloids or tumor environments), or bioaccumulation with repeated dosing. Therefore, before advancing to human trials, future in vivo studies must systematically assess dose optimization, conduct formal toxicological studies, and perform detailed biodistribution and saturation analyses; key components that are largely missing in the current literature. These steps are critical to define the therapeutic window, ensure patient safety, and meet translational regulatory standards.
Future perspectives and clinical application
Despite the positive results that have been observed with exosomal treatment, one must also discuss potential adverse effects. This becomes particularly important when considering any future clinical application. As previously discussed, the exact source of exosomes is crucial in determining their effect. Exosomes derived from diabetic patients led to decreased wound healing 84 . Similarly, other studies have found that exosomes derived from multiple myeloma bone marrow mesenchymal stem cells had increased oncogenic proteins, and cytokine and promoted multiple myeloma tumor growth in vivo 139 . Another study found that bone marrow mesenchymal stem cells alone have been shown to promote in vivo tumor growth 140 . This highlights the need to properly identify the source of exosomes, as well as the patients which will be using exosomes. If tumor promoting factors are found in exosomes, and given to patients with tumors, this may lead to much more severe complications down the line. This may be true for several pathologies, highlighting the need to better characterize and understand the factors found in exosomes. This will likely mean that exosomes will have to undergo thorough evaluation during manufacturing and prior to use in patients. One case series investigating the off-label use of exosomes injection for aesthetic purposes already demonstrated that this led to several patients developing granulomatous complications following treatment 141 . Properly characterizing exosomes, and taking into account their heterogeneous composition is also important for determining how they may be used and how regulatory agencies will determine their application 142 .
Of note, one of the first human, phase I clinical trials to examine the use of exosomes in healthy humans was done by Johnson et al. In this study, they used platelet derived extracellular vesicles. Importantly, these underwent ligand-based exosome affinity purification. Eleven healthy volunteers had two punch-biopsy wounds induced on separate arms. One wound was assigned to subcutaneous injection of extracellular vessels, while the other had subcutaneous placebo injection (isotonic solution). No adverse events, or clinically meaningful abnormalities were documented. Furthermore, no difference in healing time was found between treatment groups. The authors discussed that the lack of results may be as these represent healthy wounds, where no delayed wound healing is to be expected 143 . These results do represent a marked difference compared with the results seen throughout the numerous excisional wounds in mice. However, one must also observe that all the in vivo excisional wounds done on “normal” mice, as seen in this review, are still done on laboratory mice. It is known that the immune system of even wild-type and laboratory mice differ greatly 144 . As such, the difference in immune state in laboratory mice already presents a barrier to understanding how exosome treatment will be able to affect wound healing in a more clinical setting. Overall, whether exosomal treatment will lead to clinically significant improvement in wound healing in pathological wounds remains to be seen.
The major sources of therapeutic extracellular vesicles across the studies reviewed are from MSCs derived from a variety of tissue sources. These MSCs are monocellular cultures that secrete exosomes with signaling molecules that do not represent skin progenitor cells at any particular stage of development. Future studies of therapeutic exosomes should focus on the use of skin organoids that recapitulate the developmental process. These organoids consist of multiple cell types that are secreting exosomes for cell-to-cell communication in orchestrating the development of skin tissue. The content of these exosomes should therefore consist of signaling molecules that direct cell differentiation during the course of tissue development. A greater understanding of the expression of these signaling molecules at specific stages of skin development will enable the isolation of exosomes from skin organoids at appropriate stages to facilitate wound healing.
As interest in exosomes as a novel therapeutic for wound healing increases, there are several technical and translational barriers to be addressed before their clinical applications can be fully utilized. Just some of these challenges are associated with exosome isolation, scalability of such a product, the characterization, and the standardization of protocols, all of which significantly affect the reproducibility, efficacy and safety for such therapies.
There are a growing number of methods that are currently used to isolate exosomes, each having their own distinct advantages and limitations. Ultracentrifugation, has been considered the gold standard due to its accessibility and ability to separate vesicles based on size and density 145 . Although it remains the most widely employed technique, it is time-consuming, it can damage vesicles, and often co-isolates protein aggregates 146 . Some alternative methods include size-exclusion chromatography (SEC), polyethylene glycol-based precipitation, tangential flow filtration (TFF), and immunoaffinity capture using antibodies against exosomal surface markers147,148. Both SEC and TFF are methods that offer greater scalability and purity compared with ultracentrifugation, their cost and complexity can be limiting. As for immunoaffinity based methods, they provide high specificity but are not feasible for large scale production due to its lower yield of exosomes and higher cost 145 .
The majority of current isolation techniques are designed for small laboratory volumes, while clinical translation requires exosome production at scale. Tangential flow filtration and the use of bioreactor-based production systems is emerging as promising solutions for increased exosome collection from conditioned media 149 . However, even with advanced scalable systems, the yield of exosomes still remains relatively low, and extensive downstream purification will be required to remove possible contaminants like serum proteins, cytokines, and other extracellular vesicles150,151. In addition, to culture such large quantities while following good manufacturing practice conditions will also add to the complexity and cost moving forward.
Thorough characterization of exosome preparations is essential to ensure consistency, efficacy, and safety. Current standard methods for size and concentration include nanoparticle tracking analysis, dynamic light scattering, and tunable resistive pulse sensing 152 . In addition, transmission electron microscopy is often used to confirm morphology of the exosomes, and the surface protein markers such as CD9, CD63, and CD81 are typically assessed via western blot or flow cytometry. In addition, omics-based profiling, including proteomics and transcriptomics, can provide insight into the cargo composition, which is critical for understanding mechanisms of action in wound healing153,154.
The last significant barrier to address for clinical use is the lack of standardization in the isolation and purification of exosomes. Factors such as donor variability, cell passage number, culture conditions, and preconditioning regimens can all influence the composition and functionality of exosomes155,156. Moreover, regulatory agencies like the U.S. Food and Drug Administration (FDA) currently lack comprehensive guidelines for defining and approving exosome-based therapeutics. Standardized protocols are urgently needed to define critical quality attributes, including purity, potency, identity, and sterility 156 .
Taken together, these technical challenges highlight the importance of further method development and regulatory alignment before exosome therapies can be reliably integrated into clinical practice. Bridging these gaps will require multidisciplinary collaboration amongst a range of scientific fields.
In conclusion, while exosomes have been shown to be involved in wound healing, and may be beneficial for several types of wound models, further investigation will be required to better characterize exosomes, and identify the most efficient method of use for exosome application.
Footnotes
Acknowledgements
None.
Ethical considerations
Not applicable.
Author contributions
Conceptualization, methodology, formal analysis, E.B., W.C.L., and M.B.Q.; writing—original draft preparation, E.B.; figures, J.G. and D.N.: writing, review and editing, E.B., W.C.L., J.G., N.I., D.N., M.W., P.B., and M.B.Q. All authors have read and agreed to the final submitted version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
This narrative review does not report original research data. All sources cited are publicly available through the referenced literature.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
Not applicable.
Clinical trial registration
Not applicable.
