Abstract
Osteoarthritis (OA) is degenerative disease, leading to pain and functional disability. It is reported that polydeoxyribonucleotide (PDRN) is a suitable therapy for OA. However, the therapeutic mechanisms of PDRN in OA are not fully understood. To investigate the effect of PDRN in an in vitro model of OA, interleukin (IL)-1β or phosphate-buffered saline (PBS) was used to treat a human chondrocytic cell line in hypoxic conditions for 24 h (IL-1β group or control group). PDRN was then used to treat IL-1β group cells for 24 h (PDRN group). By Label-Based Human Antibody Array 1000, angiopoietin-2 (ANG-2), platelet-derived growth factor (PDGF), angiostatin, and endostatin, which were related to angiogenesis, were chosen for further validation studies. Quantitative real-time reverse transcription polymerase chain reaction and western blot analysis validated that the levels of PDGF and ANG-2, which were related to pro-angiogenesis, were significantly increased in the PDRN group compared with those in the control group or the IL-1β group. However, the levels of endostatin and angiostatin, which were related in anti-angiogenesis, were significantly decreased in the PDRN group compared with those in the control group or the IL-1β group. In the same manner, vascular endothelial growth factor, which was a mediator of angiogenesis, was significantly increased in the PDRN group compared with those in the control group or the IL-1β group. Furthermore, wound closure was significantly increased in the PDRN group compared with the control group or the IL-1β group by in vitro scratch assay. Moreover, PDRN decreased expression of metalloproteinase 13, as a catabolic factor for OA, but increased expression of aggrecan, which was an anabolic factor for OA. These data suggest that PDRN may promote angiogenesis and wound healing via down-regulation of catabolism and up-regulation of anabolism in an in vitro model of OA.
Introduction
Knee osteoarthritis (OA) involves degeneration of the articular cartilage and meniscus in the knee joint, synovial hyperplasia, subchondral bone sclerosis, and edema, leading to joint pain, swelling, stiffness, muscle atrophy, and functional disability 1 . To date, the pharmacological armamentarium for OA has been limited to symptomatic treatments with a goal to diminish functional impairments and pain severity 2 . Currently, corticosteroids and hyaluronic acid (HA) are used for intra-articular injection therapy of OA. However, corticosteroid injections only provide short-term pain relief by modulating inflammation. They can lead to further joint destruction 3 . HA, a large non-sulfated glycosaminoglycan found in synovial fluid, has been injected into arthritic knees to aid and improve articular cartilage lubrication 4 . Nevertheless, recent clinical practice guidelines, issued by the American Academy of Orthopedic Surgeons in 2013, stated that they were unable to recommend HA for treatment of symptomatic OA in the knee based on the lack of evidence of its effectiveness from high and moderate quality research studies 5 . Thus, the ideal intra-articular treatment for OA should not only mechanically protect the damaged cartilage surface, but also restore chondrocyte homeostasis by reestablishing the physiological articular micro-environment.
Polydeoxyribonucleotide (PDRN) is extracted from the sperm of trout bred for human food purposes. It is regarded as a source of pyrimidines and purines, thereby stimulating nucleic acid synthesis through the salvage pathway 6 . By activating the purinergic A2A receptor, PDRN enhances cell proliferation and promotes rapid healing process via stimulating angiogenesis in experimental models of autologous skin graft donor sites, diabetic pressure ulcers, and varicoceles 6 –9 . An in vivo study has investigated the effect of PDRNs on collagen-induced arthritis in mice 10 . Results of that study showed that PDRN treatment improved clinical signs of arthritis and histological damage, reduced cartilage expression and inflammatory cytokine production from stimulated human chondrocytes, thus representing a valid alternative for treatment of arthritis 10 .
Angiogenesis, defined as blood vessel outgrowth from pre-existing vasculature, is essential for growth and development, the reproductive cycle, and tissue repair. However, unlike skin wounds or diabetic pressure ulcers, angiogenesis is known to play a key role in the progression of cartilage degradation in OA 11 . Angiogenesis contributes to synovitis, osteochondral damage, osteophyte formation, and meniscal pathology in patients with OA. Nerve growth along new blood vessels into structures normally not innervated could also contribute to pain in OA 11 . Hence, whether PDRN exerts an effect on angiogenesis on the chondrocytes of OA is a conflicting issue in the consideration of PDRN as a treatment option of OA.
To our knowledge, the effect of PDRN on angiogenesis in OA has not yet been reported. Therefore, the aim of this study was to investigate the effect of PDRN on factors associated with angiogenesis and determine changes of pro-angiogenic and anti-angiogenic factors after administration of PDRN in an in vitro model of OA.
Materials and Methods
Cell Culture and IL-1β Stimulation
SW1353 cells, which have been reported as human chondrocytic cell line 12 –14 . They were obtained from were obtained from American Type Culture Collection (ATCC; HTB-94; Manassas, VA, USA) and maintained in Dulbecco’s modified Eagle medium high glucose (DMEM-HG; Gibco, Carlsbad, CA, USA) containing 10% fetal bovine serum (FBS; Gibco) and 1% 60 U/ml penicilline (Gibco) at 37%. After reaching 80% confluence, cells were harvested using 0.05% trypsin-ethylenediaminetetraacetic acid (EDTA; Gibco). Cells were washed, centrifuged, resuspended and seeded onto new plates. Culture medium was replaced every 2 to 3 days. In each experiment, cells were rendered quiescent for 12 hours by adding DMEM-HG without FBS under hypoxic condition. Cells were stimulated with phosphate-buffered saline (PBS, WELGENE, Gyeongsangbuk-do, South Korea) or 10 ng/ml of interleukin (IL)-1β (R&D Systems, Minneapolis, MN, USA) for 24 hours to establish an in vitro OA model 13,15 –18 . After stimulation with IL-1β, cells were treated with 100 μg/ml of PDRN (Placentex Integro, Mastelli Srl, Italy) for 24 hours and harvested using 0.05% trypsin-EDTA (Gibco) as described above. Three experimental groups were used in this study as follows: PBS treated group (control group); IL-1β treated group (IL-1β group); IL-1β followed by PDRN treatment group (PDRN group). The experimental scheme of this study was shown in Fig. 1A.

Experimental scheme and L1000 array analysis. (A) Seeded SW1353 cells were serum-starved under hypoxic conditions for 12 hours and then stimulated with 10 ng/ml of IL-1β or PBS for 24 hours followed by treatment with 100 μg/ml of PDRN. After 24 hours, cells were harvested for the L1000 array. To validate the L1000 array study and OA pathogenesis, qRT-PCR and a western blot were conducted. To examine the migration efficiency, an in vitro scratch assay was performed. (B) The number of enriched proteins is indicated by bar graphs. The number of up-regulated proteins is indicated by a red bar while, the number of down-regulated proteins is indicated by a green bar.
Label-Based Human Antibody Array 1000
To prepare the protein for human L1000 array, whole cell lysates from experimental groups were homogenized and dissolved in radioimmunoprecipitation assay buffer (Thermo Scientific, Rockford, IL, USA) with protease inhibitor (Abcam, Cambridge, MA, USA) and Xpert phosphatase inhibitor cocktail (GenDEPOT, Houston, TX, USA). A human L1000 glass slide array (RayBiotech, Norcross, GA, USA) was performed by eBiogen Inc. (Seoul, South Korea).
Bioinformatics Analysis
To identify protein function, enriched protein-coding genes were used with biological processes in the Database for Annotation, Visualization and Integrated Discovery (DAVID) 6.8 annotation tool. Within several biological processes, enriched protein-coding genes involved in angiogenesis, were identified for further validation studies.
Quantitative Real-Time Reverse Transcription Polymerase Chain Reaction (qRT-PCR)
Total RNA was reverse-transcribed into cDNA using ReverTra Ace® qPCR RT Master Mix with gDNA Remover (Toyobo, Osaka, Japan) according to the manufacturer’s instructions. Expression levels of genes of interest were determined using qPCRBIO SyGreen Mix Hi-ROX (PCR BIOSYSTEMS, London, UK) in a StepOnePlus RT-PCR System (Applied Biosystems, Foster City, CA, USA). Data analysis was performed using the 2-ΔΔ cycle threshold (CT) method 19 . Primers used for qRT-PCR are listed in Table 1.
Primers Used for qRT-PCR.

ANG-2: angiopoietin-2; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; PDGF: platelet-derived growth factor; qRT-PCR: quantitative real-time polymerase chain reaction.
Western Blot Analysis
To assess the protein levels of platelet-derived growth factor (PDGF), angiopoietin-2 (ANG-2), endostatin, angiostatin, vascular endothelial growth factor (VEGF), matrix metalloproteinase 13 (MMP13), and aggrecan, proteins from cell pellets were harvested as described above. Total proteins were quantified using BCA™ Protein Assay Kit (Thermo Scientific). Samples were denatured and separated by 4–12% Bis-Tris gels in 1× NuPage MOPS SDS running buffer (Invitrogen, Eugene, OR, USA). Proteins were transferred onto polyvinylidene difluoride membranes (Invitrogen) in 20% (vol/vol) methanol in NuPage Transfer Buffer (Invitrogen) at 4°C. Membranes were blocked and then incubated at 4°C overnight with the following antibodies: anti-PDGF, anti-ANG-2, anti-endostatin, anti-angiostatin, anti-VEGF, anti-MMP13, anti-aggrecan (1:1000, Abcam), and anti-glyceraldehyde-3-phosphate dehydrogenase (GAPDH; 1:3000, Santa Cruz Biotechnology, Dallas, TX, USA). The next day, blots were washed three times with tris-buffered saline (TBS) plus Tween 20 and incubated with horse-radish peroxidase-conjugated secondary antibodies (1:4000; Santa Cruz) at room temperature for 1 hour. After washing three times with TBS plus Tween 20, proteins were visualized with AmershamTM ECL Western Blotting Detection Reagent (GE Healthcare, Little Chalfont, UK) and West-Q Pico ECL solution (GenDEPOT) systems. The Multi Gauge (version 3.0) software (Fujifilm, Tokyo, Japan) system was used to quantify relative protein expression for the western blot.
In Vitro Scratch Assay
To determine the effect of PDRN treatment on cell spreading and migration capabilities, cells were seeded into six-well tissue culture dishes. A linear wound was then generated in the monolayer of cells with a 200 μl plastic pipette tip. Any cellular debris was removed by washing with PBS (WELGENE), DMEM with PBS (control group), or DMEM with IL-1β (IL-1β group). After 24 hours, PDRN was added (IL-1β+PDRN group). After 24 hours of incubation, images of migrated cells were taken using a digital camera connected to an inverted microscope to observe the closure of wound area. The final gap width of scratch was measured and calculated compared with the initial gap. Wound area was quantified with ImageJ software (NIH, Baltimore, MD, USA). All scratch assays were performed in quadruplicates.
Statistical Analysis
All results are expressed as mean ± standard error of the mean (SEM) from at least three independent experiments. Statistical analyses were conducted using the Statistical Package for Social Sciences, version 23.0 (SPSS Inc, Chicago, Illinois, USA). A one-way analysis of variance followed by a post-hoc Bonferroni comparison was performed to confirm statistical results. P-value < 0.05 was considered statistically significant.
Results
L1000 Array Analysis
Enriched proteins were categorized according to fold change ratio ≥ |1.5|. Up- and down-regulated proteins at ≥1.5-fold were counted and summarized in Fig. 1B. Enriched protein-coding genes from the IL-1β group compared with the PDRN group were categorized according to the biological process of gene ontology term analysis using the DAVID 6.8 annotation tool and listed in Table 2. These biological process are statistically significant when the false discovery rate (FDR) was less than 0.001. Within several biological processes, proteins coding genes, involved in angiogenesis such as ANG-2 20,21 and PDGF 22,23 related to pro-angiogenesis and angiostatin and endostatin related to anti-angiogenesis, were chosen for further validation studies. These proteins are presented in Table 3.
Biological Process of Gene Ontology Analysis From the IL-1β Group Compared with the PDRN Group.
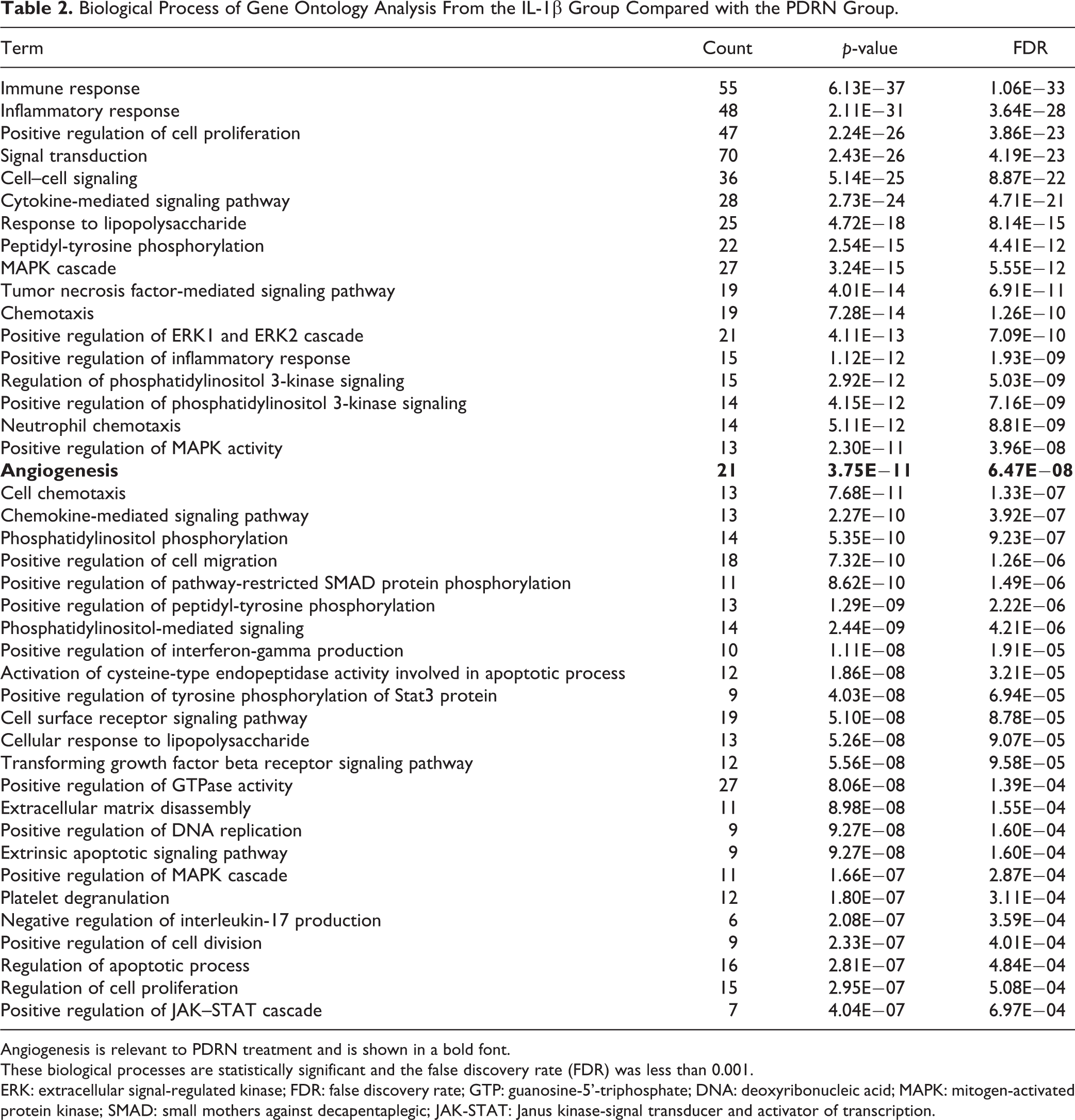
Angiogenesis is relevant to PDRN treatment and is shown in a bold font.
These biological processes are statistically significant and the false discovery rate (FDR) was less than 0.001.
ERK: extracellular signal-regulated kinase; FDR: false discovery rate; GTP: guanosine-5’-triphosphate; DNA: deoxyribonucleic acid; MAPK: mitogen-activated protein kinase; SMAD: small mothers against decapentaplegic; JAK-STAT: Janus kinase-signal transducer and activator of transcription.
L1000 Array Analysis of Protein Related to Angiogenesis.

ANG-2: angiopoietin-2; IL: interleukin; PDGF: platelet-derived growth factor; PDRN: polydeoxyribonucleotide.
Validation for L1000 Array
The qRT-PCR and western blot analysis were conducted to validate L1000 array results for proteins involved in angiogenesis. By qRT-PCR, there were no significant changes in expression in the IL-1β group compared with the control group (Fig. 2A). Expression change results for the four selected proteins were as follows: PDGF, 0.887-fold; ANG-2, 1.089-fold; endostatin, 0.842-fold; and angiostatin, 0.937-fold. In the PDRN group, PDGF and ANG-2 were significantly increased, whereas endostatin and angiostatin were significantly decreased compared with those in the control group. Their expression changes were as follows: PDGF, 1.259-fold (p < 0.05); ANG-2, 1.454-fold (p < 0.05); endostatin, 0.654-fold (p < 0.01); and angiostatin, 0.746-fold (p < 0.05). The PDGF level was significantly increased (p < 0.01) whereas the angiostatin level was significantly decreased (p < 0.05) in the PDRN group compared with those in the IL-1β group.

Effects of PDRN on mRNA and protein levels of angiogenesis. (A) PDGF, ANG-2, endostatin, and angiostatin expression levels were validated by qRT-PCR. Relative expression levels of target genes were calculated with the 2−ΔΔCt method. (B) Western blot analysis was performed with anti-PDGF, anti-ANG-2, anti-endostatin, anti-angiostatin, and anti-GAPDH antibody (as a control). (C) Relative protein expression levels in the IL-1β group and PDRN group compared with those in the control group by western blot. All results are expressed as mean ± SEM. *p < 0.05, **p < 0.01, and ***p < 0.001 vs. The control group. #p < 0.05, ##p < 0.01, and ###p < 0.001 vs. The IL-1β group.
Expression levels of these proteins in the IL-1β group compared with those in the control group by western blot were as follows: PDGF, 4.444-fold (p < 0.01); ANG-2, 0.965-fold; endostatin, 3.526-fold (p < 0.001); and angiostatin, 1.754-fold (p < 0.05; Fig. 2B and 2C). In the PDRN group, PDGF and ANG-2 levels were significantly increased whereas endostatin and angiostatin levels were significantly decreased compared with those in the control group. Their expression values were as follows: PDGF, 8.659-fold (p < 0.001); ANG-2, 1.320-fold (p < 0.05); endostatin, 0.041-fold (p < 0.001); and angiostatin, 0.165-fold (p < 0.05).
In the PDRN group, PDGF and ANG-2 levels were significantly increased whereas endostatin and angiostatin levels were significantly decreased compared with those in the IL-1β group. Their expression values were as follows: PDGF (p < 0.001), ANG-2 (p < 0.05), endostatin (p < 0.001), and angiostatin (p < 0.001).
Effects of PDRN on Expression of VEGF and Wound Healing
It has been reported that PDRN, an adenosine receptor A2A agonist, can induce stimulation of VEGF expression and facilitate wound healing 24 . VEGF is reported as one of the potent pro-angiogenic growth factors and can affect wound healing as a mediator of angiogenesis 25 . To determine the effect of PDRN on wound healing, the expression of VEGF was examined by western blot and in vitro scratch assay.
In the IL-1β group, the VEGF expression was not significantly different compared with the control group by western blot as follows: VEGF (0.964-fold; Fig. 3A and B). In the PDRN group, the VEGF expression was significantly increased compared with the control group and the expression value as follows: VEGF (1.319-fold, p < 0.05; Fig. 3A and B). In the PDRN group, the VEGF expression was also significantly increased compared with the IL-1β group (p < 0.05; Fig. 3A and B). In vitro cell migration ability was determined by the number of cells that migrated across scratch parts of cells (Fig. 3C and D). The percentage of wound closure was as follows: Control group, 4.68%; IL-1β group, 5.51%; and PDRN group, 41.62% (Fig. 3C). As shown in Fig. 3C and D, the wound closure ratio of the PDRN group was significantly increased compared to the IL-1β group (p < 0.001) or the control group (p < 0.001). As shown in Fig. 3D, a significant increase in cell migration was observed in the PDRN group compared with the control group or the IL-1β group.

Effects of PDRN on protein levels of VEGF and wound healing. (A) Western blot analysis was performed with anti-VEGF and anti-GAPDH (as a control). (B) The relative protein expression in the IL-1β group and PDRN group compared with the control group by western blot. (C) The area of the wound closure was quantified, and the ratio of wound closure was expressed as a percentage of recovered wound compared with the area at 0 h of each group. All results are expressed as mean ± SEM. *p < 0.05, and ***p < 0.001 vs. The control group. #p < 0.05, and ###p < 0.001 vs. The IL-1β group. (D) Representative data of wound healing experiment. The beginning of the experiment is before treatment with PDRN and indicated as 0 h in the figure. After treatment with PDRN for 24 hours is indicated as 24 h in the figure.
Effect of PDRN on Expression of MMP13 and Aggrecan
It has been reported that imbalance of anabolic and catabolic activity is crucial for OA pathogenesis 26 –29 . Thus, we examined the effect of PDRN on expression of anabolic and catabolic factors.
MMP13 and aggrecan, which were components of cartilage and can regulate cartilage destruction. It is reported that the expression of MMP-13, one of the catabolic genes for OA, was increased, whereas the expression of aggrecan, one of the anabolic genes for OA, was down-regulated in IL-1β treated chondrocytes 30 . Thus, we examined the expression levels of MMP13 and aggrecan in this study.
In the IL-1β group, MMP13 expression was statistically increased compared with that in the control group (Fig. 4A). On the other hand, aggrecan expression was significantly decreased in the IL-1β group compared with that in the control group (Fig. 4A). Their expression values were as follows: MMP13, 1.359-fold (p < 0.05); and aggrecan, 0.765-fold (p < 0.05; Fig. 4B). MMP13 expression was significantly decreased while aggrecan expression was significantly increased in the PDRN group compared with those in the control group (Fig. 4A). Their expression values were as follows: MMP13, 0.589-fold (p < 0.05); and aggrecan, 2.211-fold (p < 0.001; Fig. 4B). MMP13 expression was significantly (p < 0.001) decreased while aggrecan expression was significantly (p < 0.001) increased in the PDRN group compared with those in the control group (Fig. 4A and B).

Effects of PDRN on protein levels of MMP13 and aggrecan. (A) Western blot analysis using anti-MMP13, anti-aggrecan, and anti-GAPDH (as a control). (B) Relative protein expression in the IL-1β group and the PDRN group compared with the control group by western blot. All results are expressed as mean ± SEM. *p < 0.05, and ***p < 0.001 vs. The control group. ###p < 0.001 vs. The IL-1β group.
Discussion
Findings from this study indicate that PDRN could promote the expression of pro-angiogenic factors and inhibit the expression of anti-angiogenic factors in an in vitro model of OA. This is the first study that compares changes in factors influencing angiogenesis after PDRN treatment in this model.
To detect relative expression levels of 1000 human proteins including angiogenic factors, the Label-Based Human Antibody Array 1000 was used. Among the angiogenic factors evaluated, PDGF, ANG-2, and VEGF levels were significantly increased, whereas endostatin and angiostatin levels were significantly decreased in the PDRN group compared with those in the IL-1β group, based on western blot analysis. These factors play important roles in angiogenesis. Activation of angiogenesis is basically the result of an imbalance between pro- and anti-angiogenic factors 31 . PDGF 32,33 , ANG-2 34 , and VEGF 34 are known as pro-angiogenic factors whereas endostatin 35,36 and angiostatin 37,38 are known as anti-angiogenic factors.
The mechanism underlying preservation of avascularity in articular cartilage remains unclear. However, it has been suggested that hyaline cartilage contains high concentrations of endogenous inhibitors of angiogenesis 39 . Angiogenic and anti-angiogenic factors might be up-regulated in the osteoarthritic joint. However, when vascular growth predominates, articular cartilage will lose its resistance to vascularization 11 . Blood vessel and nerve growth are linked by common pathways involving the release of pro-angiogenic factors. As sensory nerves grow along new blood vessels in osteoarthritic joints, they eventually penetrate non-calcified articular cartilage, osteophytes, and inner regions of menisci. Consequently, angiogenesis can contribute to structural damage and pain in OA, thus providing potential targets for new OA treatments 40 .
Considering these points, it is very difficult to interpret whether the angiogenic effect of PDRN has a positive effect on OA. Angiogenic effects of PDRN on the recovery of chondrocytes in OA cannot be determined by the results of this study. At the present time, there are two reasonable hypotheses. First, as is known, increased angiogenesis by PDRN may adversely affect chondrocytes in OA. Nevertheless, the positive effects of PDRN on OA patients such as the anti-inflammatory and cell repair effects are greater than those of angiogenesis. Consequently, PDRN may be a valuable regenerative medicine to help wound healing in OA patients. Second, it can be assumed that the angiogenesis effect of PDRN itself does not have a bad influence on OA, in contrast with what was known in the past. If the first hypothesis is correct, PDRN will be considered as a treatment for OA without violating the existing theory that angiogenesis plays a key role in the progression of cartilage degradation in OA 11 . For the second hypothesis to be persuasive, it is necessary to redefine the concept of angiogenesis in OA, especially in the early stages of OA.
The phenomenon of OA may be defined clinically, radiologically, and pathologically. However, its etiology remains poorly understood. The precise contribution of angiogenesis to symptoms and pathology of OA is currently unclear. Angiogenesis is a complex multistep process controlled by a wide range of positive and negative regulatory factors. Angiogenesis occurs during essential physiological processes such as embryogenesis and wound repair. However, angiogenesis can also contribute to a variety of pathological conditions, including unwanted vessel growth in chronic inflammatory diseases and growth or metastasis of tumors 41 . In OA, angiogenesis interacts closely with inflammation, compressive forces, and hypoxia. However, studies regarding the role of angiogenesis in early stages of OA, especially as a defense mechanism against the degenerative process, are insufficient.
Recently published studies have reported that PDRN may represent a new and safe injection therapy for knee OA. Gennero et al. 42 have estimated the efficacy of PDRNs on cartilage degradation and found that PDRNs are suitable for long-term cultivation of in vitro cartilage, with therapeutic effects on chondrocytes by protecting cartilage. A randomized and double-blind clinical trial published in 2014 on 75 patients assessed the efficacy and safety profiles of intra-articular PDRN injection in the treatment of knee OA associated with persistent pain and showed a reduction of pain and an increase in the function in daily living and sport activity from the baseline values comparable to those obtained with the use of HA 43 . Another study that followed up 95 patients with knee OA or chondropathy (grade III or IV) for 60 days after injecting PDRN found that intra-articular administration of PDRN in patients with both severe knee arthritis and chondropathy can reverse the short and medium-term symptoms and function, with a significant improvement in quality of life 44 . Chondrocytes have a pivotal role during OA. They are mainly responsible for the anabolic–catabolic balance required for matrix maintenance and tissue function 41 . On the other hand, OA chondrocytes are characterized by accelerated catabolic processes as well as suppression of anabolic processes. An imbalance in the expression of catabolic and anabolic factors can eventually lead to osteoarthritic cartilage destruction 45 . In the in vitro scratch assay in our study, a significant increase in cell migration was observed in the PDRN group compared with either the control group or the IL-1β group. The percentage of wound closure values was as follows: control group, 4.68%; IL-1β group, 5.51%; and PDRN group, 41.62%. These results also support that PDRN might have therapeutic effect on OA chondrocyte regardless of whether the effect of angiogenesis on OA is positive or negative. Apart from the effect of PDRN on angiogenesis, the possibility that PDRN has therapeutic effect on OA cartilage is still high. Therefore, angiogenesis induced by PDRN on OA cartilage should be clarified.
This study has some limitations. Observations reported here should be interpreted with caution since factors involved in angiogenesis were not demonstrating real angiogenesis. However, the activation of angiogenesis is basically the result of an imbalance between pro- and anti-angiogenic factors. Increased expression of angiogenesis activators can lead to angiogenesis switch and act positively on neovascularization 46,47 . In addition, this study was based on cell line model of OA instead of human patients. The cell line model of OA might differ from the chronic OA condition. We conducted human chondrocytic cell line with the addition of DMEM-HG under hypoxic condition for 12 hours and stimulation with 10 ng/ml of IL-1β for 24 hours. This process might not be sufficient enough to reflect chronic status of OA. Additional study on chronic in vitro or in vivo model of OA is required.
In conclusion, PDRN may promote angiogenesis in an in vitro OA model. PDRN significantly increased the levels of pro-angiogenic factors (PDGF, ANG-2, and VEGF) and decreased the levels of anti-angiogenic factors (endostatin and angiostatin) in an in vitro model of OA. PDRN also significantly facilitated cell migration regardless of angiogenesis through up-regulation of anabolism and down-regulation of catabolism (Fig. 5). Upon these findings, the role of PDRN, as novel regenerative medicine for wound healing in OA, needs to be considered and verified in various aspects.

Effects of PDRN on in vitro OA model. (A) IL-1β induces the pathogenesis of OA in chondrocytes through up-regulation of catabolism and down-regulation of anabolism. (B) PDRN inhibit the pathogenesis of OA via up-regulation of angiogenesis and wound healing.
Footnotes
Author Contributions
Ahreum Baek and Yoon Kim contributed equally to this work.
The contributions to this study were: AB, conception and design, collection and assembly of data, data analysis and interpretation, manuscript writing; YK, data analysis and interpretation, manuscript writing; JWL, data analysis and interpretation; SCL and SRC conception and design, data analysis and interpretation, manuscript writing, final approval of the manuscript. All authors read and approved the manuscript.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
Statement of Human and Animal Rights is not applicable for this article.
Statement of Informed Consent
Statement of Informed Consent is not applicable for this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a new faculty research seed money grant of Yonsei University College of Medicine for 2015 (32-0025); the National Research Foundation (NRF-2017R1D1A1B03028855 and 2018R1A6A3A01013415); and the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health and Welfare, Republic of Korea (HI16C1012).
