Abstract
Mesenchymal stem cell (MSC) transplantation has been shown to be beneficial in treating cerebral ischemia. However, such benefit is limited by the low survival of transplanted MSCs in an ischemic microenvironment. Previous studies showed that melatonin pretreatment can increase MSC survival in the ischemic kidney. However, whether it will improve MSC survival in cerebral ischemia is unknown. Our study examined the effect of melatonin pretreatment on MSCs under ischemia-related conditions in vitro and after transplantation into ischemic rat brain. Results showed that melatonin pretreatment greatly increased survival of MSCs in vitro and reduced their apoptosis after transplantation into ischemic brain. Melatonin-treated MSCs (MT-MSCs) further reduced brain infarction and improved neurobehavioral outcomes. Angiogenesis, neurogenesis, and the expression of vascular endothelial growth factor (VEGF) were greatly increased in the MT-MSC-treated rats. Melatonin treatment increased the level of p-ERK1/2 in MSCs, which can be blocked by the melatonin receptor antagonist luzindole. ERK phosphorylation inhibitor U0126 completely reversed the protective effects of melatonin, suggesting that melatonin improves MSC survival and function through activating the ERK1/2 signaling pathway. Thus, stem cells pretreated by melatonin may represent a feasible approach for improving the beneficial effects of stem cell therapy for cerebral ischemia.
Introduction
Stem cell therapy has shown great potential for repairing and remodeling the neurovascular structure after ischemic brain injury. Mesenchymal stem cells (MSCs) can be easily harvested from autologous donors without ethical concerns and rapidly expanded in vitro (30). Studies have demonstrated that MSCs can differentiate into various somatic cells and can improve functional recovery after ischemic brain injury (2,19,41). Transplantation of MSCs into ischemic brain induces tissue regeneration and improves functional recovery (28). Furthermore, administration of MSCs enhances focal angiogenesis in the ischemic perifocal region after ischemic brain injury in rats (7). Despite these promising initial results, there are several problems that limit the development of MSC therapy. Previous studies have demonstrated that more than 80% of grafted cells died within 72 h after infusion (35). Most engrafted MSCs do not survive after implantation, presumably due to cell death (including apoptosis) induced by the ischemic microenvironment (34). Strategies to increase MSC survival are necessary to improve the effectiveness of MSC therapy for cerebral ischemia.
Melatonin, a hormone secreted by the pineal gland, is deemed as one of the most powerful endogenous antioxidant agents (32). Melatonin is used in treating circadian rhythm disorders, insomnia, and other sleep disorders (1). It is also reported to attenuate ischemic brain injury by decreasing oxidative damage (6) and protecting neurons from ischemia/reperfusion injury (6,8). It exerts antioxidative effects both directly via scavenging reactive oxygen species (ROS) and indirectly by stimulating several antioxidative enzymes, such as superoxide dismutase and catalase (21). Recently, numerous studies showed that melatonin receptors MT-1 and MT-2 were expressed in MSCs and neural stem cells (27,31). Melatonin could also exert its antioxidant function in stem cells via its receptors. For example, melatonin pretreatment improves MSC survival in ischemic kidney (24). Melatonin also influences the proliferative and differential activity of neural stem cells (25). However, whether melatonin pretreatment can protect the cell death of MSCs against the negative impacts of ischemic microenvironment in the brain remains unknown.
In this study, we used a rat model of middle cerebral artery occlusion (MCAO) in vivo and hydrogen peroxide (H2O2) or oxygen-glucose deprivation (OGD)-induced oxidative stress models in vitro to investigate whether melatonin pretreatment could promote MSC survival and promote therapeutic efficiency. The mechanism by which melatonin pretreatment affects MSC function in focal cerebral ischemia was also investigated.
Materials and Methods
MSC Culture and Characterization
The use of animals was approved by the Institutional Animal Care and Use Committee of Shanghai Jiao Tong University. MSCs were isolated from the femurs and tibias of 10 adult male Sprague-Dawley rats (Sippr-BK Co., Shanghai, China) weighing 250-300 g (3). Briefly, the femurs and tibias of rats were flushed with sterile phosphate-buffered saline (PBS; Sinopharm Chemical Reagent Co., Shanghai, China) and resuspended with high-glucose Dulbecco's modified Eagle's medium (DMEM; Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS, Gibco). Bone marrow MSCs were cultured in a 10-cm dish (Corning Incorporated, Corning, NY, USA) at 37°C with 5% CO2 in an incubator (Thermo Scientific, Barrington, IL, USA). Nonadherent cells were removed after 48 h. MSCs between passages 3 and 6 were used in the study. MSCs were characterized by immunocytochemistry and flow cytometry (FACS Arial II; BD Falcon, San Jose, CA, USA) using cluster of differentiation 29 (CD29; 1:100; eBioscience, San Diego, CA, USA), CD90 (1:100; eBioscience), CD105 (1:300; R&D Systems, Tustin, CA, USA), CD31 (1:50; BD Falcon), CD45 (1:100; eBioscience), and CD34 (1:300; BD Falcon) antibody markers, according to the manufacturer's instruction. The cell nuclei were stained using 4′,6-diamidino-2-phenylindole (DAPI; Vector Labs, Burlingame, CA, USA).
Cell Viability Analysis
To evaluate MSC viability under oxidative stress, MSCs were seeded at a density of 1 × 104 per well in 96-well culture plates (BD Falcon) and subjected to different concentrations of H2O2 (10 to 1,000 mM; Sinopharm Chemical Reagent Co.) for 1 h. MSCs were then washed three times with PBS and cultured for another 24 h in high-glucose DMEM with 10% FBS. These MSCs were then examined by cell counting kit 8 (CCK-8; Dojindo, Kumamoto, Japan). Data were acquired using a microplate reader (Synergy2 BioTek, Winooski, VT, USA). All experiments were performed in triplicate, and data are presented as mean ± SD.
For OGD study, MSCs were washed twice in glucose-FBS-free DMEM (Gibco) and cultured in glucose-FBS-free DMEM and seeded in 96-well culture plates. The plates were placed into an anaerobic chamber using a well-characterized, finely controlled modular chamber system (Billups-Rothenberg, Inc., Del Mar, CA, USA), then flushed with a gas mixture of 5% CO2/95% N2 at room temperature for 30 min at 5 L/min. The chamber was then sealed and placed at 37°C in a conventional cell incubator for 12 h. Then the MSCs were returned to their normal culture conditions (high-glucose/FBS-containing DMEM) in a 37°C incubator with 20% O2 and 5% CO2 for the reoxygenation procedure for 24 h. Untreated cultures were maintained for the same period of time under a normoxic atmosphere in the glucose and 10% FBS-containing DMEM solution. MSC viability was examined by CCK-8. Data were acquired using a microplate reader (Synergy2 BioTek). All experiments were performed in triplicate, and data are presented as mean ± SD.
Melatonin Pretreatment of MSCs In Vitro and In Vivo
To determine the effect of melatonin on MSC survival under oxidative stress, MSCs were pretreated with various concentrations of melatonin (0.05 to 1 mM; Sigma, St. Louis, MO, USA) for 24 h. The cells were then washed three times with PBS and subjected to 1,000 mM H2O2 for 1 h or OGD for 12 h, followed by CCK-8 assay 24 h later. To confirm the functional specificity and mechanism of melatonin, MSCs were preincubated with 10 mM melatonin receptor antagonist luzindole (Sigma) or 10 mM extracellular signal-regulated kinase (ERK) phosphorylation inhibitor U0126 (Sigma) for 1 h prior to melatonin treatment in a parallel experiment.
According to a previous study, the expression of the antioxidant enzyme catalase and basic fibroblast growth factor (bFGF) in MSCs reaches a maximum between 16 and 24 h after treatment with 5 mM melatonin (24), so an incubation time of 24 h was selected in our study.
For in vivo transplantation experiments, MSCs were pretreated with 5 mM melatonin for 24 h and washed with PBS three times to completely remove melatonin from the MSC suspension prior to transplantation.
MSC Labeling
MSCs were trypsinized (Gibco), washed with PBS, and resuspended in 4 μM CM-DiI dye (Invitrogen, Carlsbad, CA) at 37°C for 15 min followed by 15 min at 4°C in the dark, which is an appropriate method for stem cell tracking as previously described by others (9,18) and for DiI by us (11). Then unincorporated dye was removed by centrifugation at 300 × g for 5 min. MSCs were resuspended in PBS prior to transplantation.
Western Blot Analysis
MSCs and brain samples (from the striatum of ipsilateral hemisphere) were resuspended in lysis buffer [a kit from Pierce, Rockford, IL, USA, including 1% Triton X-100; 20 mM hydroxyethyl piperazineethanesulfonic acid (HEPES; pH 7.5); 150 mM NaCl; 1 mM ethylenediaminetetraacetic acid (EDTA); 1 mM ethylene glycol tetraacetic acid (EGTA); 1 mM dithiothreitol (DTT); 1 mM β-glycerol phosphate; 1 mM Na3VO4; 1 mM phenyl-methylsulfonyl fluoride (PMSF); with 10 mg/ml each of leupeptin, aprotinin, pepstatin, and phosphatase inhibitor] for 30 min and centrifuged at 13,000 × g for 25 min at 4°C. The supernatants were collected and quantified with bicinchoninic acid (BCA) protein assay (Pierce). To analyze protein levels, equal amounts of total proteins were subjected to 10% (w/v) sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE; Bio-Rad, Hercules, CA, USA) and transferred to nitrocellulose membranes (Whatman, Piscataway, NJ, USA). Filters were then blocked with 5% skim milk (Sinopharm Chemical Reagent Co.) for 1 h at room temperature and probed with anti-phospho ERK (1:1,000; Cell Signaling Technology, Beverly, MA, USA), anti-ERK (1:1,000, Cell Signaling Technology), anti-vascular endothelial growth factor (VEGF; 1:500; Santa Cruz Technology, Santa Cruz, CA, USA), and anti-β-actin (1:2,000, Santa Cruz Technology) antibodies at 4°C overnight. Membranes were washed three times in Tris-buffered saline and Tween-20 (TBST; Sinopharm Chemical Reagent Co.), incubated with horseradish peroxidase-conjugated goat anti-rabbit or rabbit anti-goat IgG (HangZhou HuaAn Biotechnology Co., HangZhou, China) for 1 h at room temperature, and then reacted with an enhanced electrochemiluminescence (ECL) substrate (Pierce). The result of chemiluminescence was recorded with an imaging system and semiquantified using Quantity One software (Bio-Rad).
Middle Cerebral Artery Occlusion (MCAO) and MSC Transplantation
Forty adult male Sprague–Dawley rats weighing 250 to 300 g were used. Rats were anesthetized with ketamine/ xylazine (100 mg/10 mg/kg, Sigma) intraperitoneally. Body temperature was maintained at 37 ± 0.5°C using a heating pad (RWD Life Science, Shenzhen, China). Animal surgery was performed as previously described (42). Briefly, a 4-0 suture (Covidien, Mansfield, MA, USA) with round tip and silicon coating was inserted from the left external carotid artery (ECA) into the internal carotid artery (ICA) and reached the circle of Willis to occlude the origin of the middle cerebral artery (MCA). The success of occlusion was determined by monitoring the decrease of surface cerebral blood flow (CBF) to 10% of baseline CBF using a Laser Doppler flowmetry (Moor LAB, Moor Instruments, Devon, UK). Reperfusion was performed by withdrawing the suture 2 h after MCAO.
Twenty-four hours after transient MCAO, 40 rats were randomly divided into four groups (10 rats per group): 1) PBS group, 2) MSC group, 3) melatonin-pretreated MSC group (MT-MSC), and 4) luzindole-pretreated melatonin-treated MSC group (Lu-MT-MSC). Melatonin-pretreated or untreated DiI-labeled MSCs (1 × 106) were stereo-tactically injected into the striatum of the ipsilateral hemisphere with the following coordinates: AP −0.5 mm; L −3.0 mm; V −5.0 mm. The same amount of PBS (25 μl) was injected as the control. The wound was closed, and the animal was returned to its cage for follow-up experiments. 5-Bromo-2′-deoxyuridine (BrdU, Sigma; two 50 mg/kg intraperitoneally doses given 8 h apart) was administered from 3 to 9 days poststroke, and rats were sacrificed 14 days after stroke.
Behavior Tests
Rats were trained for 3 consecutive days prior to surgery. Neurobehavioral tests were performed before MCAO and at 1, 3, 7, and 14 days after MCAO by an investigator who was blinded to the experimental groups. Modified neurologic severity score (mNSS) of the animals was graded on a scale of 0 to 14 (19), which is a composite of motor, reflex, and balance tests.
Rotarod Test
An accelerating rotarod (Zhenghua, Anhui, China) provides an index of fore- and hindlimb motor coordination and balance (14). The duration that rats remained on the accelerating rotating rod was measured. The velocity was slowly increased from 20 to 40 rpm in 5 min (300 s). Each animal was given three trials, and the time that the animals spent on the rungs or gripped the device and spun around for two consecutive revolutions was recorded.
Elevated Body Swing Test
The elevated body swing test was used to test asymmetrical motor behavior (4). Rats were held by the base of the tail and raised 10 cm above the testing surface. The initial direction of swing, defined as the turning of the upper body by >10° to either side, was recorded in 20 trials in each rat, performed over 5 min. The number of turns in each (left or right) direction was recorded for each rat.
Immunohistochemistry and MSC Counting
Rat brains were fixed by transcardial perfusion with 4% paraformaldehyde (Sinopharm Chemical Reagent Co.) in PBS buffer. After perfusion, the brain was removed and frozen. Cryosections, 20 μm thick, from anterior commissure to hippocampus were collected. The sections were stained with 0.1% cresyl violet (Sinopharm Chemical Reagent Co.). Infarct area was determined by subtracting the area of cresyl violet staining in the ipsilateral hemisphere from that of the contralateral hemisphere (20).
Apoptosis of MSCs was evaluated by terminaldeoxynucleotidyl transferase mediated nick-end labeling (TUNEL) staining according to the manufacturer's protocol (Chemicon, Temecula, CA, USA). Briefly, frozen coronal sections were fixed in 4% paraformaldehyde, washed with PBS, and incubated in terminal deoxynucleotidyl transferase (TdT enzyme) at 37°C for 1 h. The sections were stained with FITC-conjugated anti- digoxigenin antibody and visualized under a fluorescent microscope at 20× objective lens (Leica, Solms, Germany). The apoptotic MSCs and the total number of MSCs were calculated from five random areas per section (200 μm apart) and nine sections per rat, containing the graft region, by two observers blinded to the experimental group.
Immunohistochemistry and double immunostaining were performed as previously described (11) and briefly below. After blocking with bovine serum albumin (Sigma), brain sections were incubated with affinity purified goat anti-doublecortin (DCX, 1:100; Santa Cruz Technology), rabbit anti-VEGF (1:100; Santa Cruz Technology), and mouse anti-glial fibrillary acidic protein (GFAP; 1:100; BD Falcon) antibodies at 4°C overnight. Secondary antibodies were Alexa Fluor 488- or 594-labeled donkey anti-goat, goat anti-rabbit, or goat anti-mouse IgG (Invitrogen).
To determine the proliferation of endothelial cells, brain sections were incubated in 2 M HCl (Sinopharm Chemical Reagent Co.) at 37°C for 30 min and rinsed with 0.1 M boric acid (pH 8.5; Sinopharm Chemical Reagent Co.) at room temperature for 10 min. After blocking with 10% bovine serum albumin, the sections were stained with primary antibodies goat anti-CD31 (1:100; R&D systems) and mouse anti-BrdU (1:100; Santa Cruz Technology). Sections were then incubated with Alexa 488- and 594-conjugated secondary antibodies and visualized under a confocal microscope (Leica, Solms, Germany). A negative control was performed by omitting the primary antibody.
Microvessel Counting
Microvessel counting through CD31 staining microvessel density (MVD) was a simple and reproducible way to morphologically identify the number of microvessels (5,15,39). Numerous studies of manual and computer-assisted methods confirm minimal interobserver variability in the microvessel counting (12). Two brain coronal sections from the CD31 and CD31/BrdU double fluorescein staining brain, 1 mm frontier and 1 mm posterior from the ischemic core, were chosen. Three areas of microvessels, just in the perifocal region, including cortex and striatum, were chosen at low-power objective lens (20×), and microvessel counting was carried out on these pictures. Two investigators blinded to the experimental group assessed microvessel counts separately. Only vessels with a clearly defined lumen or a well-defined linear vessel shape will be taken into account. Single endothelial cells will be ignored. The number of microvessels was calculated as the mean of the blood vessel counts obtained from the three pictures (43).
Statistical Analysis
Values are presented as mean ± SD. For behavioral data, two-way repeated-measures analysis of variance was performed to analyze the overall difference between treatment groups over time, and then Bonferroni-corrected post hoc comparisons were used to analyze the difference between treatment groups at each time point. Other data were analyzed by one-way ANOVA, followed by Tukey post hoc comparisons. Two-tailed p < 0.05 were considered statistically significant. All statistical analyses were performed using SPSS software (v18.0) (SPSS, Inc., Chicago, IL, USA).
Results
Characterization of MSCs
MSCs were generated from rat bone marrow and characterized by immunocytochemistry and flow cytometry. Results showed that most cultured cells were positive for CD105 and CD90 while negative for CD45 and CD34 in immunostaining (Fig. 1A). When analyzed by flow cytometry, over 90% of these cells were revealed to be CD29/CD90 positive and CD31/CD45 negative (Fig. 1B), indicating that these cells are MSCs. To track MSCs in vivo, MSCs were labeled by CM-DiI and injected into the ischemic rat brain. The CM-DiI-labeled MSCs were detected at least 14 days after labeling via fluorescent microscope. About 90% of non-melatonin-pretreated cells and melatonin-pretreated cells could be labeled by CM-DiI (Fig. 1C, D).
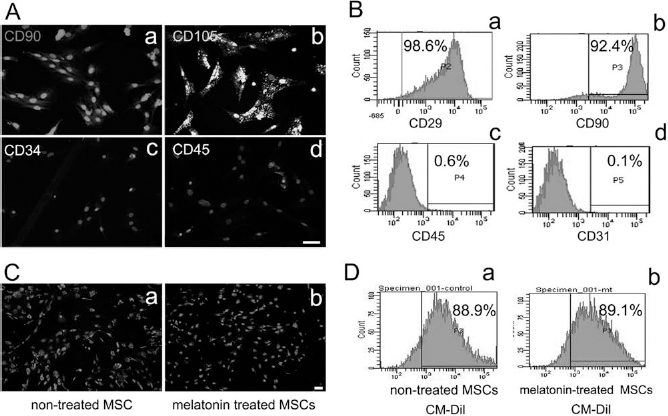
Characterization of rat mesenchymal stem cells (MSCs). (A) Fluorescent photomicrographs of cultured MSCs. Cells were positively immunostained for cluster of differentiation 90 (CD90) (a) and CD105 (b) while negative for CD34 (c) and CD45 (d). Light gray staining is the 4′,6-diamidino-2-phenylindole (DAPI) staining for cell nucleus. Scale bar: 50 mm. (B) Flow cytometry analysis for CD29 (a), CD90 (b), CD45 (c), and CD31 (d). (C) Fluorescent photomicrographs of untreated MSCs (a) and melatonin-treated MSCs (b) after 14 days of CM-DiI labeling. Scale bar: 50 μm. (D) Flow cytometry analysis of untreated MSCs (a) and melatonin-treated MSCs (b) after CM-DiI labeling.
Melatonin Pretreatment Improves the Survival of MSCs Under H2O2 Stress or OGD
To evaluate the effect of melatonin treatment on MSC viability in vitro, we used hydrogen peroxide oxidative stress as well as OGD models. MSC viability was reduced to approximately 50% after incubation in 1,000 mM H2O2 (Fig. 2A). In contrast, the viability was 85% for melatonin-pretreated MSCs (Fig. 2B). Melatonin receptor antagonist luzindole almost completely blocked the protective effect of melatonin (Fig. 2C). With OGD stimulation, melatonin also improved MSC viability, and luzindole reversed the effect of melatonin (Fig. 2D). Our in vitro results showed that melatonin improved the survival of MSCs against oxidative stress, and this effect was receptor mediated.

Melatonin pretreatment attenuated apoptosis and cell death of MSCs after H2O2 stimulation or oxygen-glucose deprivation. (A) MSC viability after exposure to various concentrations of hydrogen peroxide (H2O2) (mM). Data are mean ± SD, n = 3 per group, and experiments were performed in triplicate (same as follows); ***p < 0.001. (B) Cell viability of melatonin-pretreated MSCs after 1 h of 1,000 mM H2O2 exposure. Cells were pretreated with 5, 10, 100, or 1,000 mM melatonin for 24 h. *p < 0.001, control group was MSCs free of H2O2 and melatonin. (C) MSC viability under conditions of H2O2 alone, melatonin pretreatment plus H2O2, or luzindole (10 mM; 1 h prior to melatonin) plus melatonin treatment plus H2O2. ***p < 0.001. (D) MSC viability after OGD treatment, melatonin pretreatment plus OGD, or luzindole treatment plus melatonin treatment plus OGD. **p < 0.01; ***p < 0.001.
Melatonin-Treated MSCs Further Reduced Brain Infarct Volume and Improved Neurobehavioral Outcome of MCAO Rats
Brain infarct volume was determined by cresyl violet staining at 14 days after MCAO (Fig. 3A). Results showed that MT-MSCs greatly reduced infarct volume compared to groups of animals receiving PBS, untreated MSCs, or Lu-MT-MSCs (p < 0.05) (Fig. 3B). There was no significant difference in neurobehavior among the four groups in all three tests prior to cell transplantation. In contrast, neurological score (Fig. 3C), rotarod test (Fig. 3D), and elevated body swing test (Fig. 3E) showed that the neurobehavioral outcomes were greatly improved in the MT-MSC-treated group compared to PBS, untreated MSC, or Lu-MT-MSC groups at 7 and 14 days after MCAO (p < 0.05).
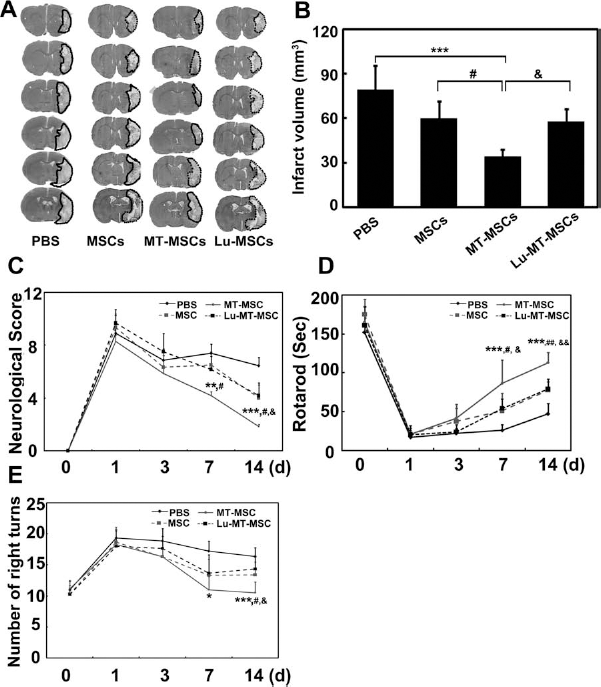
Melatonin-pretreated MSCs further attenuated cerebral infarct volume and facilitated functional recovery after stroke. (A) A representative set of coronal brain sections (cresyl violet staining) from rats treated with phosphate-buffered saline (PBS), untreated MSCs, melatonin-treated (MT-)MSCs or luzindole pretreated (Lu-)MT-MSCs 14 days following transient middle cerebral artery occlusion (tMCAO). Dotted line shows the border of infarct area. (B) Infarct volume in MCAO rats treated with PBS, untreated MSCs, MT-MSCs, and Lu-MT-MSCs. Data are mean ± SD, n = 10 per group. Functional recovery was evaluated using neurological score (C), rotarod test (D), and elevated body swing test (E). The behavioral tests were performed at 1 day before MCAO and 1, 3, 7, and 14 days after MCAO. Data are mean ± SD, n = 10 per group. Black full line: PBS group; gray full line: MT-MSC group; black dash line: Lu-MT-MSC group; gray dash line: MSC group. *p < 0.05, **p < 0.01, and ***p < 0.001, MT-MSCs versus PBS; #p < 0.05 and ##p < 0.01, MT-MSCs versus MSCs; &p < 0.05, and &&p < 0.01, MT-MSCs versus Lu-MT-MSCs.
Melatonin Pretreatment Reduced the Apoptosis and Death of Transplanted MSCs in MCAO Rats
CM-DiI-labeled MT-MSCs, untreated MSCs, and Lu-MT-MSCs were injected into the ischemic rat brain. TUNEL staining of brain sections revealed that MSC survival was increased in the MT-MSC-treated animal brain compared to the untreated MSC or Lu-MT-MSC groups (p < 0.05) (Fig. 4A, B). In addition, the number of endogenous apoptotic cells was reduced in the MT-MSC-treated group compared to the PBS, untreated MSC, or Lu-MT-MSC-treated groups (p < 0.05) (Fig. 4C). By counting the number of MSCs in five random fields per section and nine sections (200 μm apart) per animal in the MSC, MT-MSC, and Lu-MT-MSC groups (Fig. 4D), we found that the number of MSCs was increased threefold in the MT-MSC group compared to the MSC and Lu-MT-MSC groups (p < 0.05) (Fig. 4E), indicating melatonin pretreatment greatly increased MSC survival after transplantation into the brain.

Melatonin pretreatment attenuated MSC apoptosis and improved MSC survival in ischemic rat brain. (A) Representative photographs of cresyl violet-stained coronal section of ischemic rat brain. White color indicates the infarct area. Needle track was 5 mm under the cortex. Red box is the area for imaging analysis. Terminal-deoxynucleotidyl transferase mediated nick-end labeling (TUNEL) staining of the brain sections of MCAO rats that received PBS (a), untreated MSC (b), MT-MSCs (c), and Lu-MT-MSCs (d) reveals apoptotic cells in the ischemic perifocal (arrowheads, green). TUNEL and Dil double staining of the brain sections of MCAO rats that received MSCs (b), MT-MSCs (c), and Lu-MT-MSCs (d) detects apoptotic MSCs (arrows, yellow). Scale bar: 50 mm. Double-positive cells in the MSC, MT-MSC, and Lu-MT-MSC groups were counted and the data presented in (B) **p < 0.01. (C) Bar graph shows the number of apoptotic host cells in the PBS group, MSC group, MT-MSC group, and Lu-MT-MSC group. Data are mean ± SD, n = 10 per group. **p < 0.01; ***p < 0.001. (D) Photomicrographs illustrated the DiI-labeled MSCs (red) in MSC (a), MT-MSC (b), and Lu-MT-MSC (c) groups. (E) Bar graph shows the number of MSCs in the MSC, MT-MSC, and Lu-MT-MSC groups. Data are mean ± SD, n = 10 per group. ***p < 0.001.
Melatonin Improves the Viability of MSCs via ERK1/2 Pathway
Western blots revealed that the levels of p-ERK1/2 in melatonin-treated MSCs were twofold greater than that in untreated MSCs. Pretreatment with luzindole reduced p-ERK1/2 level to baseline (p < 0.05) (Fig. 5A, B). Incubation with 10 mM ERK phosphorylation inhibitor U0126 completely blocked the protective effect of melatonin against H2O2 oxidative stress (p < 0.05) (Fig. 5C), suggesting that melatonin pretreatment enhanced MSC survival under oxidative stimulation mainly via the ERK1/2 pathway.

Melatonin pretreatment improved the therapeutic efficiency of MSCs through p-ERK1/2. (A) Expression of phosphorylated extracellular signal-regulated kinase (p-ERK)1/2 and ERK1/2 in MSCs treated with H2O2 (lane 2), melatonin pretreatment plus H2O2 (lane 3), and luzindole (10 mM; 1 h prior to melatonin) plus melatonin treatment plus H2O2 (lane 4). Lane 1 was the control with no H2O2 stimulation. (B) Bar graph showing the semiquantitative data from (A). Data are mean ± SD, n = 6 per group. *p < 0.05. (C) MSC viability under normal conditions, H2O2 stimulation, H2O2 with melatonin, and H2O2, melatonin and ERK phosphorylation inhibitor-U0126 treatment. Data are mean ± SD, n = 3 per group, and experiments were performed in triplicate. *p < 0.05.
Melatonin-Pretreated MSC Transplantation Increased Angiogenesis and Neurogenesis 14 Days After Stroke
The number of microvessels was analyzed in the perifocal cortex and striatum 14 days after MCAO. Endothelial cells were identified by CD31, and neuroblasts were stained with DCX (Fig. 6A). Statistical results showed that the number of CD31-positive microvessels in the cortex was significantly increased in the MT-MSC group compared to PBS, untreated MSC, or Lu-MT-MSC groups (p < 0.05) (Fig. 6B). Similar results were observed in the striatum where angiogenesis was increased in the MT-MSC group compared to the other groups (p < 0.05) (Fig. 6C), as well as in the subgranular zone (SVZ) region of the ipsilateral hemisphere, where neurogenesis was also increased in the MT-MSC transplantation group (p < 0.05) (Fig. 6D). Newly formed microvessels were identified by CD31 and BrdU double staining (Fig. 6E). More newly formed microvessels were observed in the MT-MSC group in the cortex and striatum (p < 0.05) (Fig. 6F, G), indicating that MT-MSCs promoted angiogenesis and neurogenesis in ischemic rat brain.
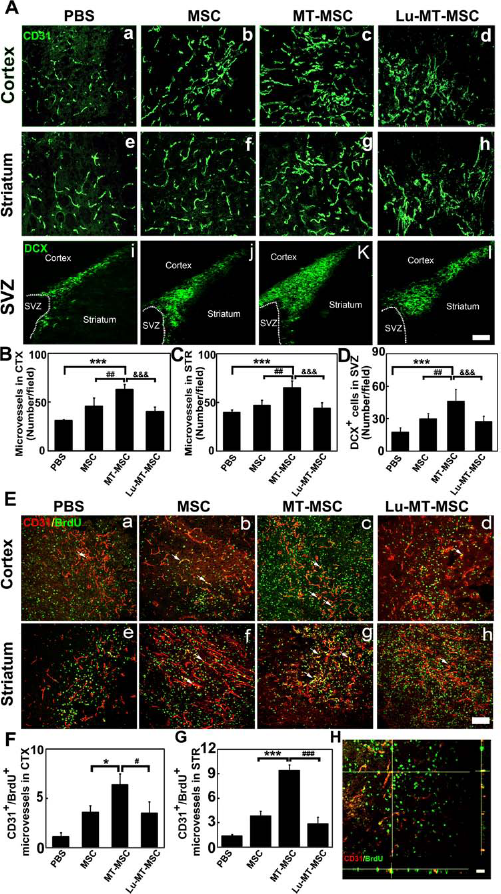
Cerebral angiogenesis and neurogenesis were increased in MT-MSC-treated rats after 14 days of MCAO. (A) Representative photograph of CD31 and doublecortin (DCX) immunostaining in the PBS group (a, e, i), MSC group (b, f, j), MT-MSC group (c, g, k), and Lu-MT-MSC group (d, h, l) in the ischemic perifocal region of cortex and striatum. Scale bar: 50 mm. Bar graphs showing the number of microvessels in the ischemic perifocal region of cortex (CTX) (B), striatum (STR) (C), and neuroblasts in the subventricular zone (SVZ) region (D). Data are mean ± SD, n = 10 per group. ***p < 0.001; **p < 0.01. (E) Confocal images of CD31 (red) and bromode-oxyuridine (BrdU) (green) double staining in the PBS (a, e), MSC (b, f), MT-MSC (c, g), and Lu-MT-MSC groups (d, h) in the ischemic perifocal region of cortex and striatum. Arrows show newly formed microvessels (yellow). Scale bar: 50 mm. Bar graphs showing the number of newly formed microvessels in the ischemic perifocal region of cortex (F) and striatum (G). Data are mean ± SD, n = 10 per group. *p < 0.05; ***p < 0.001. (H) A representative confocal image of BrdU and CD31 colocalization in three-dimensional space.
Melatonin-Pretreated MSC Transplantation Increased VEGF Expression in Cortex and Striatum 14 Days After Stroke (Fig. 7)
Double immunostaining showed that VEGF was mainly expressed in GFAP-positive cells and transplanted MSCs in the striatum (Fig. 7A, F), and Western blot analysis showed that melatonin-pretreated MSCs enhanced the expression of VEGF in stroke rat brain, compared with the PBS and untreated MSC group (p < 0.05) (Fig. 7B, C). Additionally, some transplanted MSCs migrated to the cortex, and they also expressed VEGF (Fig. 7D, E).
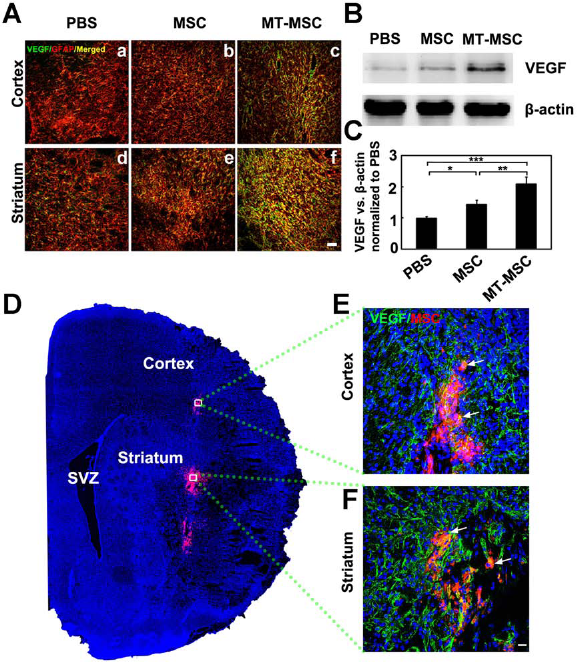
Transplantation of MT-MSCs enhanced VEGF secretion after 14 days of MCAO. (A) Confocal images of vascular endothelial growth factor (VEGF) (green) and glial fibrillary acidic protein (GFAP) (red) expression in cortex and striatum in PBS (a, d), MSC (b, e), and MT-MSC (c, f) groups. Most VEGF was expressed in GFAP-positive cells (yellow). Scale bar: 50 μm. (B) Western blot analysis showed expression of VEGF and β-actin in the ipsilateral striatum of the PBS, MSC, and MT-MSC groups. (C) Bar graph showing the semiquantitative data from panel. Data are mean ± SD, n = 10 per group. *p < 0.05, **p < 0.01, ***p < 0.001. (D) Confocal image showed distribution of DiI-labeled MSCs (red) in cortex and striatum in the ipsilateral hemisphere. Blue is DAPI nuclear staining. Photograph showed VEGF staining (green) in DiI-labeled MSCs (red) in cortex (E) and striatum (F). Arrows indicate merged VEGF and MSC staining (yellow). Scale bar: 20 μm.
Discussion
Our present study demonstrated that melatonin pre-treatment enhanced MSC survival under H2O2 oxidative stress or OGD in vitro. Furthermore, transplantation of MT-MSCs into the ischemic rat brain significantly increased the survival of transplanted MSCs and improved its therapeutic efficacy. Compared to the untreated MSC transplantation group, infarct volume was reduced, and neurobehavioral outcomes were improved in MT-MSC-treated rats. Mechanistic study demonstrated that melatonin improved MSC survival via the ERK1/2 signaling pathway. MT-MSCs demonstrated higher therapeutic potential through upregulating VEGF, reducing ischemia-induced cell death including apoptosis, and promoting focal angiogenesis and neurogenesis. Our results provide strong evidence that increasing MSC survival is an important step to enhancing the benefits of MSC therapy.
Stem cell therapy has shown great potential to restore neurological function after ischemic brain injury (10,17,23,29,37,44). However, one of the main hurdles to significant functional improvement after stem cell transplantation is that more than 80% of transplanted stem cells die within 3 days after transplantation in ischemic organs (26,35). Therefore, it is crucial to improve stem cell viability under ischemic conditions. Other studies have shown that the survival of grafted stem cells is improved by preconditioning with hypoxia after transplantation into ischemic rat brain (36,38). Hypoxia or melatonin pretreatment greatly increased both survival of MSCs and their therapeutic efficiency in the ischemic brain. However, the mechanisms of the two strategies are different. Hypoxia upregulates growth factors including brain-derived neurotrophic factor (BDNF) and VEGF, and downregulates proinflammatory cytokines in MSCs, while melatonin pretreatment activates the ERK1/2 signaling pathway. In addition, melatonin is widely used as a diet complement and antioxidant agent with no reported side effects (33). It is therefore readily available to be used in clinical trials to improve the efficiency of MSC therapy.
The results of our study indicate that melatonin pretreatment enhanced MSC survival under oxidative stimulation via the ERK1/2 pathway. In previous studies, it has been noted that MSCs express melatonin receptors MT1 and MT2 (45). Melatonin receptor antagonist luzindole completely reversed the protective effect of melatonin, suggesting that the protective effect is receptor mediated. Our results showed that melatonin pretreatment greatly increased ERK1/2 phosphorylation in MSCs under H2O2 stimulation. Luzindole blocked ERK1/2 phosphorylation, further indicating that the function of melatonin is achieved via ERK1/2 pathway activation. In addition, ERK1/2 inhibitor U0126 could also completely reverse the protective effect of melatonin on MSCs under oxidative stimulation. Taken together, these results demonstrate that melatonin improved MSC survival mainly through activating the ERK1/2 pathway.
We prelabeled MSCs with CM-DiI dye, which is a carbocyanine membrane probe with strong photo-stable fluorescence, long cellular retention, and minimal cytotoxicity (13). Previous studies have demonstrated that it is useful for long-term MSC labeling (9,11). In addition, DiI is retained in cells throughout fixation, permeabilization, and paraffin-embedding procedures (16).
MSCs are able to differentiate into neurons, endothelial cells, and osteoblasts in vitro (40). MSCs can also secrete nerve growth factor (NGF), BDNF, bFGF, and other factors that may enhance cell survival in the ischemic microenvironment (22). Consequently, remodeling processes occur, with endogenous stem cells now surviving and migrating toward the ischemic region, leading to new neurovascular unit formation (46). Our study demonstrated that increased survival of transplanted MSCs reduced infarct volume and improved neurobehavioral outcomes for at least 14 days. It is notable that though melatonin promoted MSC survival under ischemic conditions, it did not affect MSC differentiation. Neither melatonin-treated nor untreated MSCs injected into ischemic brain differentiated into mature neurons or astrocytes in vivo (data not shown), suggesting that the therapeutic effects of MSCs may be mediated by the secretion of favorable paracrine factors. Indeed, we measured the expression of VEGF and found that VEGF was mainly secreted by astrocytes 14 days after stroke, and transplantation of melatonin-treated MSCs could enhance VEGF secretion. Increased expression of VEGF in rats transplanted with MT-MSCs subsequently induced angiogenesis and neurogenesis in the ischemic perifocal region, which are beneficial to the repairing and remodeling after ischemic brain injury. In our study, we injected MSCs into the striatum; however, some MSCs migrated to the cortex, enhanced VEGF expression, and increased angiogenesis in the cortex.
In summary, our study demonstrated that melatonin pretreatment promoted MSC survival under ischemia-related conditions in vitro and augmented the therapeutic efficiency of MSCs in transient focal ischemia in vivo. The protective effect of melatonin is achieved through activation of the ERK1/2 pathway. This strategy of pretreating stem cells may represent a safe approach for improving the beneficial effects of stem cell therapy for cerebral ischemia.
Footnotes
Acknowledgment
This study was supported by research grants from the National Natural Science Foundation of China (81070939 and 81100868), National Basic Research Program of China (973 Program 2011CB504405), the Science and Technology Commission of Shanghai Municipality (10JC1408100), and KC Wong Foundation (G.Y.Y.). We thank Dr. Richard F. Keep from the University of Michigan and Tiffany Dai from the University of California Berkeley for their valuable discussion and kind editorial assistance. The authors declare no conflicts of interest.
