Abstract
Peripheral nerve injuries involving nerve defects remain challenging to treat. Although autologous nerve grafting is considered the gold standard, it has notable limitations, including donor site morbidity. To address this, we developed a scaffold-free Bio 3D conduit composed of human umbilical cord–derived mesenchymal stromal cells (UC-MSCs) using bioprinting technology. In this study, we evaluated its efficacy and safety in a canine ulnar nerve defect model. At 10 weeks postoperatively, the Bio 3D group showed better motor and sensory recovery compared with the allograft group, as demonstrated by the pinprick test, electrophysiological studies, and hypothenar muscle wet weight (0.978 ± 0.100 vs. 0.637 ± 0.151, n = 3). Morphometric analysis revealed greater axonal regeneration, including larger myelinated axon diameters (4.27 ± 0.342 µm vs. 3.69 ± 0.161 µm, n = 3) and thicker myelin sheaths (0.621 ± 0.088 µm vs. 0.497 ± 0.021 µm, n = 3). Immunostaining showed that the number of transplanted UC-MSCs diminished over time, likely after exerting their therapeutic effects. No adverse events, systemic abnormalities, or distant human cell migration was observed. These findings suggest that UC-MSC–derived Bio 3D conduits are a promising alternative for peripheral nerve regeneration, especially for patients wishing to avoid donor nerve harvesting.
Keywords
Introduction
Despite advances in microsurgical techniques and the availability of various treatment materials, the management of peripheral nerve injuries remains challenging1,2. In particular, cases involving nerve defects in which tension-free suturing is not feasible present significant difficulties. For such injuries with large nerve defects, autologous nerve grafting is considered the gold standard 3 . However, autologous nerve grafting has inherent drawbacks, primarily donor site morbidity, including scarring and infection at the harvest site, loss of nerve function, and extended surgical time 4 . These limitations have led to extensive research into alternative treatment methods to replace autologous nerve grafts.
In previous studies, we utilized a Bio 3D printer to develop a scaffold-free nerve conduit (Bio 3D conduit) created from homogeneous spheroids of single cells 5 . Bio 3D printing is an innovative technology that arranges cell spheroids in a three-dimensional (3D) structure by placing them on fine needles based on predesigned 3D data 6 . Using dermal fibroblasts, we produced nerve conduits that demonstrated successful nerve regeneration in both a rat sciatic nerve defect model and a canine ulnar nerve defect model7–11. Furthermore, a clinical trial involving human subjects was conducted, in which autologous dermal fibroblasts harvested from patients were used to fabricate nerve conduits longer than 20 mm to treat chronic nerve injuries in the hand, yielding positive outcomes 12 . In that study, the Semmes–Weinstein monofilament test, a sensory assessment involving tactile stimulation by a filament, showed postoperative improvement in the innervated skin regions of study participants compared with preoperative values. In addition, the Quick Disability of the Arm, Shoulder, and Hand (QuickDASH) scores, a patient-reported outcome measure, also improved postoperatively. However, the use of dermal fibroblast-derived conduits has its own limitations, including donor site morbidity and the time-consuming process of culturing and expanding dermal fibroblasts, which makes it impractical for emergency surgeries.
To address these issues, we focused on mesenchymal stromal cells (MSCs) as a new cell source for creating Bio 3D conduits. Mesenchymal stromal cells are stem cells with tissue repair-promoting and immunomodulatory properties 13 . Allogeneic MSCs are clinically used as a treatment for graft-versus-host disease (GVHD) 14 and are considered promising cells in regenerative medicine. We previously reported successful nerve regeneration using a Bio 3D conduit created from human umbilical cord–derived MSCs (UC-MSCs), which were xenotransplanted into a rat sciatic nerve defect model without the use of immunosuppression 15 . UC-MSCs are harvested from the umbilical cords of newborns without the need for an invasive procedure. They are not affected by age-related changes and exhibit homogeneous properties. Furthermore, they can be stably cryopreserved for long periods 16 . Based on these characteristics, UC-MSCs are now considered the most promising cell source for the clinical application of Bio 3D conduits. In this study, we conducted a proof-of-concept study on UC-MSC–derived Bio 3D conduits using canines as a medium-sized animal model. The aim of this study was to confirm the efficacy of UC-MSC–derived Bio 3D conduits on nerve regeneration in a canine ulnar nerve defect model and to assess their safety. Unlike our previous study using autologous dermal fibroblast–derived conduits in the same canine ulnar nerve defect model 8 , this study employed UC-MSCs, which are considered a more promising cell source. Moreover, we newly evaluated the viability of conduit-forming cells using immunohistochemistry and assessed distant cell migration using Alu-PCR, providing a more comprehensive analysis of the transplanted conduits.
Materials and methods
Bio 3D conduits
The UC-MSCs used in this study were provided by IMSUT CORD (Cord Blood and Cord Bank) in The Institute of Medical Science, The University of Tokyo, Japan. IMSUT CORD activity was reviewed and approved by the institutional review board (No. 36–2). Using umbilical cords of healthy newborns, UC-MSCs were isolated, processed, and stored. Informed consent was obtained from the mothers, and the donations were made voluntarily. All studies were performed in accordance with the ISSCR guidelines (International Society for Stem Cell Research, 2021). The protocols were approved by an institutional animal care and use committee that included reviewers with expertise in stem cell biology. Donors were informed that organoids derived from their donated stem cells might be transplanted into animal models, as stated in the guidelines. Bio 3D conduits were assembled from UC-MSCs utilizing a Bio 3D printer (Regenova, Cyfuse, Tokyo, Japan) as described by Itoh et al 6 . Briefly, after detachment with trypsin, cells were seeded at 2 ± 1 × 104 cells per well into each well of a Low Cell Adhesion 96-well plate (Sumilon PrimeSurface, Sumitomo Bakelite, Tokyo, Japan). The purity of the collected cells was measured using a flow cytometer. Fluorescently labeled antibodies (CD34, CD45, CD73, CD90, and CD105) suitable for detecting MSC were used for the measurements. After 48 h, cells aggregated to form a spheroid in each well. Using a Bio 3D printer, they were sucked out of the 96-well plate into a fine suction nozzle, skewered into a circular stainless steel needle array, and allowed to develop into a tubular structure according to the predesigned pattern. Approximately 1 week after this procedure, the spheroids fused together to form a luminal structure and the needle arrays were removed. The obtained Bio 3D conduits were transferred to a perfusion bioreactor and cultured until they reached the desired mechanical strength. The final Bio 3D conduits had an inner diameter of 5 mm, a wall thickness of 1 mm, and a length of 10 mm (Fig. 1b). To measure the viability of the cells that compose Bio 3D Conduits, we stained the cells with a fluorescent reagent (Live/Dead Viability Kit, Thermo Fisher Scientific, Tokyo, Japan) and confirmed that more than 90% of the cells that compose Bio 3D conduits were viable.
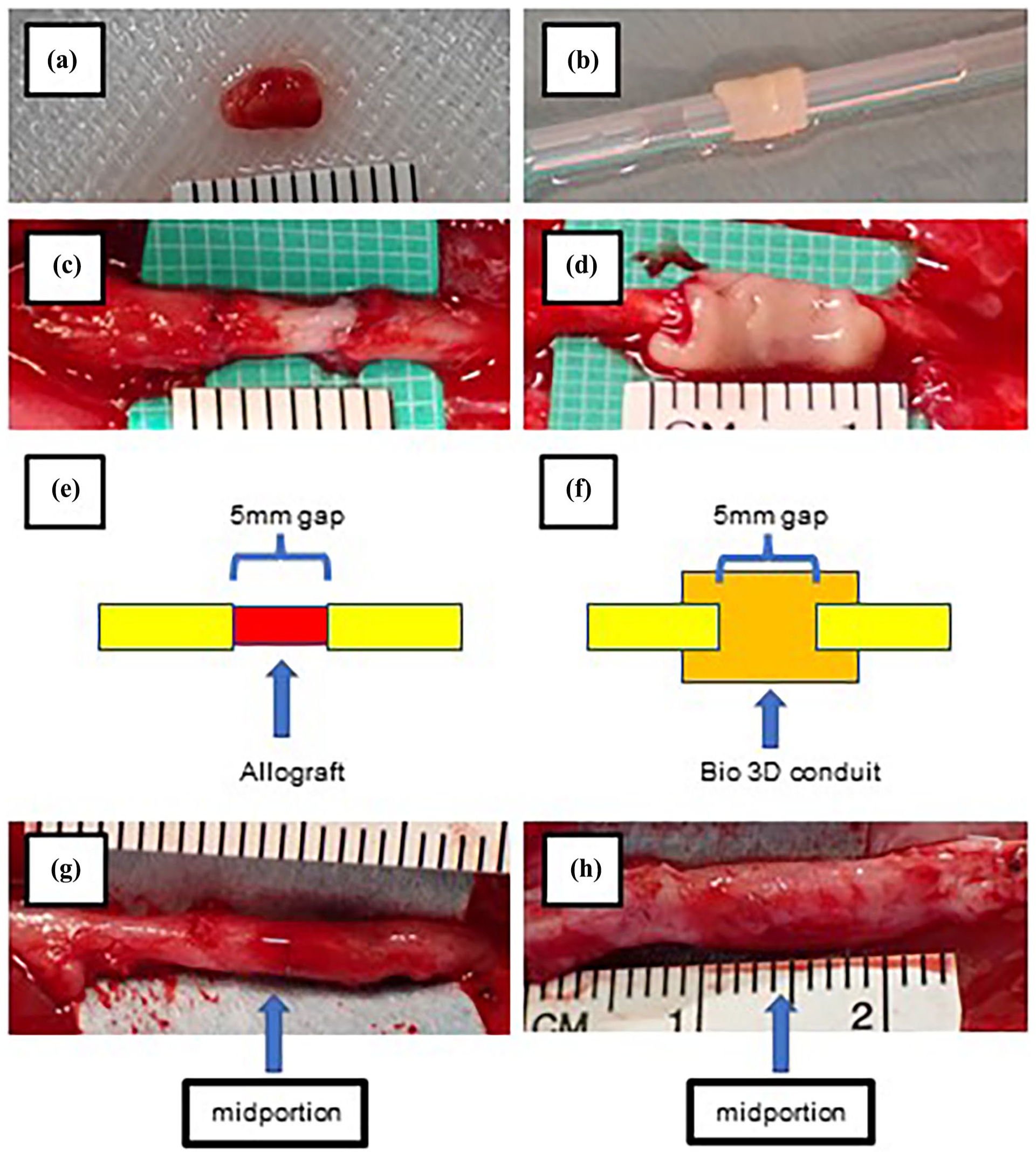
Intraoperative images and macroscopic findings of the ulnar nerve at 10 weeks postoperatively. (a) A 5-mm graft harvested from the ulnar nerve of an animal in the allograft group. (b) A Bio 3D conduit prior to implantation. (c, d) Intraoperative images following transplantation of an allograft (c) and a Bio 3D conduit (d). (e, f) Schematic diagrams of the surgical procedure, showing a 5-mm ulnar nerve gap bridged by an allograft (e) and by a Bio 3D conduit (f). (g, h) Macroscopic appearance of the ulnar nerve at 10 weeks postoperatively in the allograft group (g) and Bio 3D group (h). The midportion of the injury site, indicated by arrows, was evaluated with immunohistochemistry and morphometric analysis.
Experimental design
Six male beagle dogs (9–11 months old, weighing 8–12 kg; Nihon Bioresearch Inc., Gifu, Japan) were used in this study. We selected a relatively small sample size because the potential efficacy of UC-MSC–derived Bio 3D conduits has already been demonstrated in experiments using rats, and this study also aimed to evaluate their safety. All animals were housed individually in separate cages, provided with standard dog food, and given unlimited access to water. All procedures were conducted in accordance with the guidelines of the Animal Research Committee of Kyoto University and were reported in compliance with the ARRIVE guidelines 2.0 (National Center for the Replacement, Refinement and Reduction of Animals in Research [NC3Rs], 2020) for animal research. The animals were randomly assigned to two groups using computer software (Ibuki, Nihon Bioresearch Inc.): a Bio 3D group, in which the nerve defect was bridged with a Bio 3D conduit, and an allograft group in which the nerve defect was bridged with a nerve allograft. The average weight and variance between the groups were matched as closely as possible. At 10 weeks postoperatively, after conducting the pinprick test and electrophysiological study described below, all animals were euthanized under deep anesthesia by exsanguination from the axillary artery. Exclusion criteria for the experimental animals were not established prior to the experiment, and blinding of the groups was not possible due to the nature of the experiment.
Anesthesia
Animals were pretreated with a subcutaneous injection of 0.05 mg/kg atropine to reduce airway secretions, followed by intravenous administration of 25 mg/kg thiopental for induction. After intubation, general anesthesia was maintained with inhalation of 2% sevoflurane in oxygen.
Surgical procedures
The surgical procedures were based on those previously performed in our experiments using canines 8 . Each animal was anesthetized in the supine position and a 5-cm skin incision was made on the volar side of the distal ulnar forelimb. The left ulnar nerve was exposed between the flexor carpi ulnaris and the flexor digitorum. In the allograft group, the ulnar nerve was sharply transected at points 17.5 mm and 22.5 mm proximal to the wrist joint, and a 5-mm nerve segment was harvested (Fig. 1a). Nerve segments were exchanged between individuals and allotransplanted to bridge the 5-mm nerve defect (Fig. 1c, e). In the Bio 3D group, the ulnar nerve was sharply transected at a point 20 mm proximal to the wrist joint, and a Bio 3D conduit (Fig. 1b) was interposed between the proximal and distal stumps. Each stump was then pulled 2.5 mm into the conduit and anchored in place with epineural 9-0 nylon sutures, creating a 5-mm gap within the conduit (Fig. 1d, f). In both groups, the wound was closed in layers with 4-0 nylon sutures. Following the procedure, the left forearm was immobilized with a cast for 2 weeks.
Pinprick test
The pinprick test was performed on all animals to assess sensory recovery of the affected ulnar nerves at 10 weeks after surgery. A pinprick stimulus was applied with standardized forceps from the tip of the fifth digit to the ulnar side of the wrist on both forearms. The stimulus was applied by firmly pinching the skin with the forceps to deliver a nociceptive (pain-related) stimulus. Preliminary testing on the contralateral (uninjured) side confirmed that a light grasp of the skin did not elicit a withdrawal response, whereas a stronger pinch did. Based on these observations, we considered the test to be a valid method for nociceptive evaluation. The response of each animal to the pinprick test was graded from 0 to 3 as follows: grade 0, no response to stimulus; grade 1, withdrawal response to stimulus on the ulnar side of the wrist; grade 2, withdrawal response to stimulus on the fifth metacarpal joint; grade 3, withdrawal response to stimulus on the tip of the fifth digit.
Electrophysiological study
Electrophysiological studies of the bilateral ulnar nerves were performed at 10 weeks after surgery (Fig. 2). The compound motor action potential (CMAP) of the hypothenar muscle was measured, and the motor nerve conduction velocity (MNCV) of the ulnar nerves was calculated. The bilateral ulnar nerves were surgically exposed under general anesthesia. The ulnar nerves were stimulated at a point 40 mm proximal to the wrist joint (S1) and at the wrist joint (S2) with the electrode of a needle electromyography (EMG) device (Neuropack S1, Nihon Kohden, Tokyo, Japan). A pair of needle electrodes was inserted into the hypothenar muscle to record CMAPs. The amplitude of the CMAP (µV) in the hypothenar muscle was obtained by measuring the height of the EMG waveform evoked by stimulation at the S1 point. The MNCV (m/s) was calculated from the difference in latency (ms) between two EMG waveforms evoked by stimulation at the S1 and S2 points.

Electrophysiological studies of the ulnar nerves at 10 weeks postoperatively. (a) A representative photograph taken during electrophysiological testing. The ulnar nerves and hypothenar muscles were surgically exposed, and electrodes were directly inserted. The arrow indicates the site where a nerve defect was created 10 weeks earlier. (b) Results of electrophysiological testing of the affected ulnar nerves, including motor nerve conduction velocity (MNCV) and the amplitude of compound motor action potentials (CMAPs) of the hypothenar muscles. The symbol (−) indicates the absence of a detectable CMAP waveform. (c) Representative CMAP waveforms recorded in the Bio 3D group.
Wet weight of the hypothenar muscle
At 10 weeks postoperatively, the entirety of the bilateral hypothenar muscles, which are innervated by ulnar nerves, were harvested and immediately weighed on a digital scale. The wet weight ratio of affected: healthy hypothenar muscle was calculated for each animal. The ratio was then compared between the allograft group and the Bio 3D group.
Immunohistochemistry
At 10 weeks postoperatively, the regenerated ulnar nerves were harvested from each animal. The nerve samples were divided into two sections at the midportion of the injury site. The proximal sample was immunostained to qualitatively assess nerve morphology. The proximal samples were fixed in 4% paraformaldehyde (PFA) and then immersed in a 20% sucrose solution. After embedding in O.C.T. Compound, the frozen samples were horizontally sectioned at a thickness of 16 μm at the midportion. The nerve sections were washed with phosphate-buffered saline (PBS) and subjected to antigen retrieval using proteinase K (Sigma-Aldrich, St. Louis, MO, USA) for 10 min at room temperature. To block nonspecific binding, 2% donkey serum (ImmunoBioScience Corp., Davis, CA, USA) was applied to the slides and incubated for 1 h at room temperature. FLEX Polyclonal Rabbit Anti-S100 Ready-to-Use (1:2; Dako, Carpinteria, CA, USA) was used as a Schwann cell marker, and NF-H antibody (RNF402, 1:50; Novus Biologicals, Centennial, CO, USA) was used as a neurofilament marker for the primary antibody reaction. The sections were incubated with the primary antibodies for 24 h at 4°C. After incubation, the slides were washed with PBS, and anti-rabbit IgG (H+L), highly cross-adsorbed, CF 555 antibody produced in donkey, and anti-mouse IgG (H+L), highly cross-adsorbed, CF 488A antibody produced in donkey (both from Sigma-Aldrich) were added as secondary antibodies. The sections were incubated for 1 h at room temperature. After washing the slides with PBS, cover glasses were mounted on the slides using Fluoro-Keeper Antifade Reagent, Non-Hardening Type with DAPI (Nacalai Tesque, Kyoto, Japan). The sections were observed using a confocal microscope (BZ-X700; Keyence, Osaka, Japan). Red and green fluorescence intensities were quantified in representative images shown in Fig. 3 from both groups using ImageJ (National Institutes of Health, Bethesda, MD, USA) and expressed as integrated density (arbitrary units, a.u.).

Fluorescent immunohistochemistry of cross-sections of the ulnar nerve at 10 weeks postoperatively. (a, b) Representative fluorescent immunostained images of ulnar nerve cross-sections from the allograft group (a) and Bio 3D group (b), showing staining for Schwann cell marker (S100) and neurofilament marker (NF). (c, d) Representative fluorescent immunostained images of nerve sections from the Bio 3D group, showing staining with a human cell-specific marker. Merged images show fluorescence signals for the anti-human mitochondria antibody and DAPI. Two dots emitting fluorescence signals for both markers, indicated by an arrow, are observed in these images. Scale bars: 50 µm in a and b; 200 µm in c and d.
To evaluate the viability of the Bio 3D conduit, additional immunostaining was performed using the same samples. Anti-Mitochondria antibody [113-1], BSA and azide free (1:100, Abcam, Cambridge, UK) was used for the primary antibody reaction as a human cell–specific marker, and anti-mouse IgG (H+L), highly cross-adsorbed, CF 488A antibody produced in donkey (Sigma-Aldrich) was used as the secondary antibody. Immunostaining was also performed on samples from the allograft group as a negative control. One transverse section per animal was examined, and the number of fluorescent dots positive for DAPI and the human cell–specific marker was counted. The other protocols were the same as those described above.
Morphometric analysis of the regenerated nerve
The distal side of each two-part nerve specimen was used for morphometric analysis. Samples were fixed with 1% glutaraldehyde and 1.44% paraformaldehyde and postfixed with 1% osmic acid, and embedded in epoxy resin. Semi-thin transverse sections at the midportion were stained with toluidine blue solution, and one cross-section per animal was examined using a light microscope at a magnification of 400× (BZ-X700; Keyence). All myelinated fibers within the neural area were counted using ImageJ (National Institutes of Health).
In addition, using the same samples, two parameters of regenerated axons (myelinated axon diameter and myelin thickness) were examined, as reported in our previous studies. Ultra-thin transverse sections (60–80 nm) of each sample from the two groups were stained with lead citrate and uranyl acetate, and examined using a transmission electron microscope (TEM, JEM-1400Flash; JEOL, Tokyo, Japan). Ten randomly selected visual fields at 2000× magnification for each cross-section were analyzed. The myelinated axon diameter (a) and the bare axon diameter (b) were measured using ImageJ (National Institutes of Health). From these two measured values, myelin thickness ([a - b]/2) and the G-ratio (b/a) were calculated in each axon.
Assessment of distant human cell migration by Alu-PCR
To evaluate the presence of human cells in canine organs 10 weeks postoperatively, DNA was extracted from formalin-fixed paraffin-embedded (FFPE) tissue samples and analyzed using Alu-PCR. Tissue sections from canine organs, including the liver, gallbladder, pancreas, spleen, lymph nodes, thymus, kidneys, lungs, and trachea, were collected from the Bio 3D group and the allograft group. For DNA extraction, tissue blocks were divided into four groups: Group 1: liver, gallbladder, and pancreas; Group 2: spleen, lymph nodes, and thymus; Group 3: kidneys; and Group 4: lungs and trachea. Tissue blocks were sectioned to a total volume of 3.0 to 4.0 mm3 for each group. Positive control sections, consisting of the paraffin-embedded Bio 3D conduit, were prepared to a total volume of at least 0.5 mm3. DNA was extracted using the QIAamp DNA FFPE Advanced kit (Qiagen, Hilden, Germany) according to the manufacturer’s protocol, and the extracted DNA was stored at 4°C until further analysis.
Quantitative real-time PCR (qPCR) was performed using the TaqMan Gene Expression Master Mix (Thermo Fisher Scientific, Waltham, MA, USA) and custom-designed TaqMan Gene Expression Assays (Thermo Fisher Scientific) for human AluYb8 sequences (Supplementary Table 1). In addition, qPCR for canine GAPDH was conducted using the TaqMan Gene Expression Assays (Thermo Fisher Scientific, accession number: Cf04419463_gH); GAPDH was used as a housekeeping gene to normalize DNA levels across samples. The reaction mixture for both PCRs consisted of 10 µL of TaqMan Gene Expression Master Mix, 1 µL of primer/probe mix, and 7 µL of RT-PCR grade water (Thermo Fisher Scientific), for a total volume of 18 µL per reaction. DNA samples (2 µL) were added to the reaction mixture in a 96-well plate, bringing the final volume to 20 µL per well. Each DNA sample was analyzed in duplicate. The 96-well plates were briefly centrifuged and loaded into a real-time PCR system. The thermal cycling conditions are shown in Supplementary Table 2.
Safety evaluation
To evaluate the safety of the Bio 3D conduit, clinical signs, food intake, body weight, urinalysis, complete blood count, and blood biochemistry were assessed at defined time points. Postoperative complications and general clinical signs were monitored daily. Food intake was also recorded daily. Body weight was measured weekly. Urinalysis was performed preoperatively and at 9 weeks postoperatively. Complete blood count was conducted preoperatively, at 4 weeks, and at 9 weeks postoperatively. Blood biochemistry was evaluated at the same time points: preoperatively, at 4 weeks, and at 9 weeks postoperatively. In addition, an autopsy and pathological examination were performed on the major organs, including the liver, gallbladder, pancreas, spleen, lymph nodes, thymus, kidneys, lungs, and trachea.
Statistical analyses
All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). Specifically, it is a modified version of R commander designed to add statistical functions frequently used in biostatistics 17 . Data are presented as mean and standard deviations (SD). The t-test was used to compare the wet weight ratio of affected to healthy hypothenar muscles and the number of myelinated axons between the allograft group and the Bio 3D group. For the analysis of myelinated axon diameter, myelin thickness, and G-ratio, a linear mixed-effects model (LMM) was applied to account for the nested data structure, as multiple axons were measured per animal. The model was fitted using the lme4 package in R, with group (allograft vs. Bio 3D) as a fixed effect and animal ID as a random intercept to adjust for intra-animal correlation. Values were considered statistically significant at P < 0.05 for all data.
Results
Functional recovery of the affected ulnar nerves
At 10 weeks postoperatively, ulnar nerve function in the affected limb was assessed. In the pinprick test, two animals in the allograft group scored grade 1, and one scored grade 2. In contrast, all three animals in the Bio 3D group scored grade 3, indicating greater sensory recovery in the Bio 3D group. Electrophysiological analysis revealed significant EMG waveforms of the hypothenar muscles on the affected side only in the Bio 3D group; no significant EMG waveforms were detected in the allograft group (Fig. 2). The amplitude of CMAPs recorded from the hypothenar muscles in the Bio 3D group, expressed as a ratio of affected to healthy side, was 0.0187, 1.382, and 1.304 in order of individual animal numbers. The MNCV of the ulnar nerves, expressed as a ratio of affected to healthy side, was 0.641, 0.880, and 0.834. The mean amplitude of CMAPs was 5909 ± 4079 µV (n = 3), and the mean MNCV was 67.6 ± 5.7 m/s (n = 3). The mean wet weight ratio of the affected to healthy hypothenar muscle was 0.637 ± 0.151 in the allograft group (n = 3) and 0.978 ± 0.100 in the Bio 3D group (n = 3). The wet weight ratio was significantly higher in the Bio 3D group (P < 0.05). These findings indicate that motor and sensory function of the affected ulnar nerve improved to a greater degree in the Bio 3D group than in the allograft group.
Immunohistochemistry
At 10 weeks postoperatively, the affected ulnar nerve was harvested and subjected to immunohistochemistry. Macroscopic findings of the ulnar nerve at 10 weeks postoperatively are shown in Fig. 1g, h. Immunohistochemical examination of nerve sections at the midportion of the injury site in both the allograft and Bio 3D groups revealed positive staining for neurofilament and Schwann cell markers (Fig. 3a, b). The integrated density of red fluorescence (Schwann cell marker) was 1,277,357 a.u. in Fig. 3a and 943,775 a.u. in Fig. 3b, while that of green fluorescence (neurofilament marker) was 704,050 a.u. in Fig. 3a and 458,965 a.u. in Fig. 3b. To explore a potential indicator of myelination, the ratio of Schwann cell (S100) to neurofilament (NF) fluorescence intensity was calculated individually for each axon. Although the Bio 3D group showed a higher mean ratio (2.437 ± 0.838 vs. 2.107 ± 0.226, n =3), no statistically significant difference was observed (Supplementary Figure 1). In the Bio 3D group, the number of fluorescent dots positive for both DAPI and the human cell–specific marker was 1, 0, and 2, corresponding to individual animal numbers (Fig. 3C, 3D). No positive dots were observed in the allograft group, serving as the negative control. These findings suggest that nearly all transplanted Bio 3D conduits in the injury site had regressed within 10 weeks, with host Schwann cells and regenerated axons present in the area.
Morphometric analysis
At 10 weeks postoperatively, the affected ulnar nerve was harvested and subjected to morphometric analysis. Regenerated axons were counted in toluidine blue–stained nerve sections from both groups. The mean value of myelinated axon number was 4105 ± 1710 in the allograft group (n = 3) and 4558 ± 3429 in the Bio 3D group (n = 3) (Fig. 4b). To evaluate the quality of regenerated axons, ultra-thin nerve sections were observed by TEM. For this analysis, 10 randomly selected visual fields at 2000× magnification were examined per cross-section, and 104 axons from the allograft group and 87 axons from the Bio 3D group were included in the evaluation. The mean myelinated axon diameter was 3.69 ± 0.161 µm in the allograft group (n = 3) and 4.27 ± 0.342 in the Bio 3D group (n = 3). The mean myelin thickness was 0.497 ± 0.021 µm in the allograft group (n = 3) and 0.621 ± 0.088 µm in the Bio 3D group (n = 3) (Fig. 4d). The mean G-ratio was 0.713 ± 0.028 in the allograft group (n = 3) and 0.696 ± 0.043 in the Bio 3D group (n = 3). LMM analysis suggested that the Bio 3D group had a greater myelinated axon diameter (estimate = 0.617 µm, 95% CI: 0.269–1.000, P < 0.01) and myelin thickness (estimate = 0.122 µm, 95% CI: 0.076–0.171, P < 0.01) compared with the allograft group. The G-ratio showed no statistically significant difference between groups (estimate = −0.0150, 95% CI: −0.043–0.012, P = 0.281).

Morphometric analysis. (a) Representative images of nerve sections stained with toluidine blue from the allograft group and the Bio 3D group. The neural area is outlined. Images are shown at 40× and 400× magnification. (b) Myelinated axon number. The mean value was higher in the Bio 3D group than in the allograft group. Data are presented as mean ±SD (n = 3). (c) Representative images of ultra-thin sections observed with transmission electron microscopy. Scale bars: 2 μm. (d) Myelinated axon diameter and myelin thickness. Both parameters were greater in the Bio 3D group than in the allograft group, based on a linear mixed-effects model (LMM). Data are presented as mean ± SD based on animal-level means (n = 3).
Assessment of distant human cell migration
Alu-PCR targeting human AluYb8 sequences in major organs from all animals revealed nearly identical cycle threshold (Ct) values between the two groups, and these Ct values were distinctly higher than those of the positive control (Table 1). These results indicate that no distant migration of transplanted human cells was detected in the Bio 3D group at 10 weeks postoperatively.
(A) Alu-PCR targeting human AluYb8 sequences was used to investigate potential distant migration of transplanted human cells in animals from the Bio 3D group (n = 3, transplanted with human cell-derived constructs) and the allograft group (n = 3, without human cell-derived constructs). Cycle threshold (Ct) values for each animal are shown, based on samples from organs classified into Groups 1 through 4. The consistently high Ct values observed in both groups indicate that no evidence of distant human cell migration was detected in the Bio 3D group.

Safety evaluation
No postoperative complications were observed in any of the six animals. All animals consumed the full amount of food provided each day throughout the study period. Body weight remained stable, and no abnormalities were observed in urinalysis, complete blood count, or blood biochemistry. The results of body weight, urinalysis, hematological, and biochemical analyses are presented in Supplementary Table 3. Autopsy and pathological examination findings revealed no signs of tumor formation or other abnormalities in any of the major organs (data not shown).
Discussion
We previously reported successful nerve regeneration using UC-MSC–derived Bio 3D conduits transplanted into a rat sciatic nerve defect model 15 . The current study is a proof-of-concept study utilizing a medium-sized animal model, aiming to confirm the efficacy of Bio 3D conduits in promoting nerve regeneration in a canine ulnar nerve defect model, as well as to assess the systemic impact and viability of the transplanted cells. Compared with the rat sciatic nerve, the canine ulnar nerve has a larger diameter, which required the fabrication of larger Bio 3D conduits to match the nerve size. Demonstrating therapeutic efficacy in a nerve injury model with a larger diameter was a necessary step toward the clinical application of Bio 3D conduits. A comparative summary of the present study and our previous study using a rat model is provided in Table 2. When UC-MSC–derived Bio 3D conduits are xenotransplanted, rejection occurs, although it is anticipated to be milder than that of other xenoantigens due to the immunosuppressive substances released by MSCs15,18. Therefore, in this study, an allograft group with a certain level of antigenicity was selected as the control group. Nerve allografts are rich in extracellular matrix from the same species and function as a scaffold for nerve regeneration. However, an immune response to the cellular components within the graft occurs immediately after transplantation, followed by the progression of axonal regeneration 19 . In this study, nerve regeneration was observed in the histological evaluation of affected nerves in the Bio 3D and allograft groups (Fig. 3a, b), although the allograft group was inferior in both the quantity and quality of regenerated axons compared with the Bio 3D group (Fig. 4). To investigate a possible indicator of myelination, we calculated the fluorescence intensity ratio of S100 to NF for individual axons (Supplementary Figure 1). Although the Bio 3D group showed a higher mean ratio, no statistically significant difference was observed, which may be partly due to the intrinsic variability of immunofluorescence staining, including section-to-section differences in signal intensity and background. The mean G-ratio was also lower in the Bio 3D group; however, no statistically significant difference was observed between the two groups. In this study, morphometric analysis of the contralateral (noninjured) ulnar nerve was not performed, which is a limitation of the present study. In our previous research using the same animal species 8 , the mean G-ratio of the normal ulnar nerve was reported to be 0.462 ± 0.047, which is considerably lower than the mean G-ratios obtained in this study. This finding suggests that the remyelination process was still ongoing in both groups. In the Bio 3D group, all animals responded to pinprick testing for pain stimulation of the little finger, demonstrating superior sensory recovery compared with the allograft group. The superiority was also evident in electrophysiological studies, where the mean affected to healthy side ratios of ulnar nerve MNCV and amplitude of hypothenar muscle CMAPs in the Bio 3D group were 0.79 ± 0.10 and 0.90 ± 0.63, respectively. These values were remarkably favorable, even compared with our previous experiment in which Bio 3D conduits were created using canine autologous dermal fibroblasts 8 . We believe that the nerve regeneration-promoting effects of UC-MSCs contributed to this difference. Numerous previous animal studies have demonstrated the nerve regeneration-promoting properties of UC-MSCs 20 . In vitro, UC-MSCs release factors such as brain-derived neurotrophic factor (BDNF) and nerve growth factor β (NGFβ), and extracellular matrix-related proteins such as laminin, which enhance axonal growth21–23. Furthermore, it has been reported that culturing rat Schwann cells in human UC-MSC–conditioned medium enhances their viability and expression of BDNF and NGF 21 . Thus, UC-MSCs are promising cells for regenerative medicine targeting peripheral nerve injuries. However, effective transplantation methods for delivering UC-MSCs to the injury site—such as filling artificial nerve conduits or direct injection into nerves—have not yet been established 20 . Artificial nerve conduits are important therapeutic materials for peripheral nerve injuries; however, they are known to induce foreign body reactions. Moreover, direct injection into nerves does not retain cells at the injury site in cases of severe nerve damage involving defects. The Bio 3D conduit is an innovative delivery method for UC-MSCs that addresses these issues. The Bio 3D conduit is a scaffold-free tubular structure composed of a single type of cell, enabling the transplantation of a high-density cell population as a cohesive unit to the injury site. It is not only applicable to nerve defects, as demonstrated in this study, but can also be used for wrapping injured sites in cases of crush injuries and similar conditions. In this study, we presumed that the transplanted UC-MSCs secreted extracellular matrix components such as laminin, forming a scaffold for axonal growth. It is also presumed that they not only released neurotrophic factors to promote axonal elongation but also acted on host Schwann cells, which play a critical role in nerve regeneration 24 , thereby facilitating the overall nerve repair process. Considering the clinical application of UC-MSC–derived Bio 3D conduits in humans, it is likely that the anti-inflammatory properties of UC-MSCs would have an even greater positive impact on nerve regeneration compared with that observed in the current experiment using a xenotransplantation model. MSCs express major histocompatibility complex (MHC) class I antigens at low levels and do not express MHC class II antigens under basal conditions, which contributes to their low immunogenicity in allogeneic transplantation 25 . Moreover, allogeneic MSCs have been demonstrated to be effective in treating GVHD in transplantation, indicating that the immunogenicity of UC-MSC–derived Bio 3D conduits in allogeneic applications is likely minimal. Instead, they may regulate inflammation following nerve injury. Intravenous injection of MSCs is known to reduce inflammation and scar formation after spinal cord injury 26 . In addition, Ma et al 27 reported that intravenous injection of human UC-MSC–derived extracellular vesicles, which carry no risk of rejection, into a rat sciatic nerve defect model resulted in favorable nerve regeneration and a reduction in pro-inflammatory cytokines, such as interleukin (IL)-1 and IL-6, along with an increase in the anti-inflammatory cytokine IL-10 within the nerve tissue.
Comparison between our previous rat study and the present canine study.

In the safety evaluation, no postoperative wound complications were observed in any of the animals, and no abnormalities in systemic condition were detected. Neither urinalysis nor blood tests revealed any abnormalities. These findings indicate that the immunogenic transplant material used in this study did not cause significant inflammation that could lead to wound-related issues or systemic effects. This is an important observation when developing regenerative medicine for peripheral nerve injuries in humans. Immunostaining to assess the viability of the Bio 3D conduit revealed that by 10 weeks post-transplantation, very few cells remained at the injury site. In previous experiments using rats, the Bio 3D conduit maintained its tubular structure at 1 week postoperatively, albeit with a reduction in cell number 15 . To investigate whether human UC-MSCs had migrated to other organs, qPCR targeting the human-specific AluYb8 sequence was performed in this study. This method is highly sensitive for detecting xenotransplanted human cells28,29. The qPCR results targeting AluYb8 in specimens from major organs of both the Bio 3D group and the allograft group consistently showed higher Ct values compared with the positive control, indicating that no human cells were detected. Based on these findings, it is presumed that the UC-MSCs contained within the transplanted Bio 3D conduit exerted their therapeutic effects at the injury site and gradually diminished over a period of approximately 10 weeks.
This study has several limitations. First, in the electrophysiological examination, one animal had an extremely low CMAP amplitude in the hypothenar muscle. The most likely explanation is a technical error, such as nerve damage caused during exposure of the ulnar nerve, although the exact cause remains unclear. A larger sample size would likely yield more reliable results. Second, the results of the LMM analysis should be interpreted with caution. For parameters measured by electron microscopy (myelinated axon diameter, myelin thickness, and G-ratio) as well as the fluorescence intensity ratio, the data had a nested structure and within-animal variability because multiple axons were measured per animal. Therefore, a linear mixed-effects model (LMM) was employed. However, as only three animals were included per group (n = 3), the estimation of random effects may be statistically unstable. This limitation should be considered when interpreting the findings, in conjunction with other data presented in this study. Third, the control group in this study was limited to the allograft group. Despite the small sample size, nerve regeneration in the Bio 3D group appears favorable based on multiple measures presented in this study. In particular, the weight of the hypothenar muscle on the affected side at 10 weeks postoperatively was close to normal, suggesting that early reinnervation was achieved. However, the extent to which the treatment outcomes of the Bio 3D conduit differ from those of autografts remains unclear. Fourth, detailed functional evaluations of the forelimbs, such as gait analysis or locomotor assessments, were not performed in this canine model. Although the weight of the hypothenar muscle on the affected side was measured and may provide some indication of forelimb function, it does not fully substitute for direct locomotor assessments. Fifth, sensory recovery was assessed using a behavioral pinprick test, but no electrophysiological methods such as sensory nerve conduction studies (SNCS) or somatosensory evoked potentials (SEP) were employed. Sixth, while this study demonstrates the efficacy of UC-MSC–derived Bio 3D conduits in nerve regeneration and their safety for transplantation, the use of a xenotransplantation model provides only partial insight into the mechanisms by which Bio 3D conduits may influence nerve regeneration in allogeneic transplantation in humans. Lu et al 30 reported that xenotransplantation of mouse sciatic nerves into a rat sciatic nerve defect model resulted in intense mononuclear cell infiltration within the graft during the first 2 weeks, followed by a marked reduction in axon numbers, which became almost undetectable by 8 weeks. This degree of immune rejection was considerably stronger than that observed in previous studies involving allogeneic nerve transplantation in rodents19,31. Although the transplanted tissue differed in our study, it is reasonable to assume that xenogeneic immune rejection also occurred, and that this response was likely more severe than that expected following allogeneic transplantation of UC-MSC–derived Bio 3D conduits in humans. For this reason, an allograft group with intrinsic antigenicity was selected as the control, rather than an autograft group. In other words, predicting clinical outcomes of human UC-MSC–derived Bio 3D conduits relative to autologous nerve grafts would have been challenging using this xenotransplantation model. However, the use of a xenotransplantation model was necessitated by the premise of the Bio 3D conduit concept, which is based on the use of human UC-MSCs. Human UC-MSCs can be noninvasively collected during childbirth, and the cells used in this study were indeed obtained in this manner and banked at medical institutions. Moreover, the paracrine effects of UC-MSCs are reported to be superior to those of bone marrow- or adipose-derived MSCs 21 . For these reasons, experimental systems utilizing other cell sources, such as canine MSCs, were not considered. Finally, this study does not provide clear information regarding their potential immunogenicity in long-term clinical applications. Because of the immunomodulatory properties of UC-MSCs, allogeneic cells may remain in the host longer than initially expected, which could increase the risk of chronic immune responses over time. Therefore, careful and continuous monitoring of clinical signs will be essential in future clinical trials involving Bio 3D conduits. However, findings from already established clinical applications of MSCs, such as in the treatment of GVHD, suggest that intravenously administered allogeneic MSCs tend to disappear from the host shortly after infusion and do not exhibit long-term engraftment 32 , potentially indicating a lower risk of long-term immunogenicity of the Bio 3D conduits.
Conclusion
In this study, we demonstrated the efficacy and safety of a human UC-MSC–derived Bio 3D conduit in peripheral nerve regeneration. The Bio 3D conduit is an innovative delivery method for stem cells, enabling the transplantation of a large number of MSCs as a cohesive unit to the site of peripheral nerve injury. The differences in treatment outcomes between this conduit and other therapeutic materials, including autologous nerve grafts, require further investigation. Nevertheless, this approach is expected to become a safe alternative treatment, particularly for patients who wish to avoid sacrificing their own nerves.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251361711 – Supplemental material for Efficacy and safety of Bio 3D conduits composed of human umbilical cord–derived mesenchymal stromal cells: A proof-of-concept study in a canine ulnar nerve defect model
Supplemental material, sj-docx-1-cll-10.1177_09636897251361711 for Efficacy and safety of Bio 3D conduits composed of human umbilical cord–derived mesenchymal stromal cells: A proof-of-concept study in a canine ulnar nerve defect model by Kazuaki Fujita, Ryosuke Ikeguchi, Tomoki Aoyama, Takashi Noguchi, Koichi Yoshimoto, Daichi Sakamoto, Terunobu Iwai, Tetsuya Miyamoto, Yudai Miyazaki, Shizuka Akieda, Tokiko Nagamura-Inoue, Fumitaka Nagamura, Koichi Nakayama and Shuichi Matsuda in Cell Transplantation
Supplemental Material
sj-xlsx-2-cll-10.1177_09636897251361711 – Supplemental material for Efficacy and safety of Bio 3D conduits composed of human umbilical cord–derived mesenchymal stromal cells: A proof-of-concept study in a canine ulnar nerve defect model
Supplemental material, sj-xlsx-2-cll-10.1177_09636897251361711 for Efficacy and safety of Bio 3D conduits composed of human umbilical cord–derived mesenchymal stromal cells: A proof-of-concept study in a canine ulnar nerve defect model by Kazuaki Fujita, Ryosuke Ikeguchi, Tomoki Aoyama, Takashi Noguchi, Koichi Yoshimoto, Daichi Sakamoto, Terunobu Iwai, Tetsuya Miyamoto, Yudai Miyazaki, Shizuka Akieda, Tokiko Nagamura-Inoue, Fumitaka Nagamura, Koichi Nakayama and Shuichi Matsuda in Cell Transplantation
Footnotes
Acknowledgements
We thank the Division of Electron Microscopic Study, Center for Anatomical Studies, Graduate School of Medicine, Kyoto University for technical assistance in electron microscopic analysis.
Ethical considerations
The UC-MSCs used in this study were provided by IMSUT CORD (Cord Blood and Cord Bank) in The Institute of Medical Science, The University of Tokyo, Japan. IMSUT CORD activity was reviewed and approved by the institutional review board (No. 36–2). All experimental procedures were conducted in accordance with the guidelines of the Animal Research Committee of Kyoto University.
Consent to participate
Informed consent was obtained in writing, and the donations were made voluntarily.
Consent for publication
This study does not include any data from individual persons who provided UC-MSCs; therefore, consent for publication was not required.
Author contributions
K.F., R.I., T.A., S.A., T.N., F.N., K.N., and S.M. designed the experiments. T.N. and F.N. supervised the cultivation and handling of UC-MSCs. S.A. and Y.M. were responsible for creating the Bio 3D conduits. K.F., R.I., and D.S. performed all other experiments. T.N., K.Y., T.I., and T.M. supported the interpretation of the data analysis. K.F. prepared the original draft of the manuscript, while R.I. and T.A. reviewed and edited it. S.M. gave final approval of the manuscript.
Ethical approval
This study was approved by our institutional review board.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Japan Agency for Medical Research and Development, Grant Number 22bk0104155h0001.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: There are no patents or marketed products to declare. K.N. is the co-founder and shareholder of Cyfuse Biomedical K.K., Tokyo, Japan (Cyfuse). Y.M. is an employee, and S.A. is the representative director of Cyfuse. They contributed to the manufacturing of 3D conduits, and Cyfuse provided the bioprinter to manufacture the conduit. The company has the industrial rights related to the bioprinting method used to construct the 3D conduit in this work. Cyfuse provided support in the form of salaries for authors Y.M. and S.A., and provided research grants to T.A., K.N., and S.M. These competing interests do not alter the authors’ adherence.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
