Abstract
We examined whether hypoxic exposure prior to the event of transplantation would have a positive or negative effect upon later islet graft function. Mouse islets exposed to hypoxic culture were transplanted into syngeneic recipients. Islet graft function, β-cell physiology, as well as molecular changes were examined. Expression of hypoxia-response genes in human islets pre- and posttransplant was examined by microarray. Hypoxia-preexposed murine islet grafts provided poor glycemic control in their syngeneic recipients, marked by persistent hyperglycemia and pronounced glucose intolerance with failed first- and second-phase glucose-stimulated insulin secretion in vivo. Mechanistically, hypoxic preexposure stabilized HIF-1α with a concomitant increase in hypoxic-response genes including LDHA, and a molecular gene set, which would favor glycolysis and lactate production and impair glucose sensing. Indeed, static incubation studies showed that hypoxia-exposed islets exhibited dysregulated glucose responsiveness with elevated basal insulin secretion. Isolated human islets, prior to transplantation, express a characteristic hypoxia-response gene expression signature, including high levels of LDHA, which is maintained posttransplant. Hypoxic preexposure of an islet graft drives a HIF-dependent switch to glycolysis with subsequent poor glycemic control and loss of glucose-stimulated insulin secretion (GSIS). Early intervention to reverse or prevent these hypoxia-induced metabolic gene changes may improve clinical islet transplantation.
Introduction
A major obstacle to clinical islet transplantation is the poor function of islets after engraftment. Poor function is evidenced by the usual requirement to transplant multiple islet preparations to achieve successful islet transplant outcomes (38). Also, surviving human islet grafts exhibit poor metabolic function in vivo (33) and demonstrate a steady decline in function over time (35). A number of processes may reduce the metabolic performance of transplanted islets, and in particular, hypoxia has been suggested to be a critical insult contributing to the poor performance of islet grafts (3,8,13). Islet grafts are known to have insufficient blood supply after transplantation (5) due to poor revascularization after engraftment (34) compared to the intimate spatial relationship pancreatic islets share with blood vessels in situ (17). Poor vascularization coupled with the implantation of islets into the relatively deoxygenated blood of the portal vein would result in reduced oxygen tension of islet grafts (5,29). Further, isolated islets cultured in standard conditions are exposed to dynamic changes in oxygen partial pressures ranging from normoxic, through hypoxic, to anoxic levels.
In the context of islet transplantation, it remains to be established if hypoxic exposure prior to the event of transplantation will result in impaired graft function. Many cell types exhibit a regulated hypoxic response, a common feature of which is the coordinated expression of hypoxicresponse genes mediated through the activity of the transcription factor hypoxia-inducible factor-1α (HIF-1α) (19). The induction of certain hypoxia-response genes may be considered beneficial for islet transplantation. Lack of nutrients and oxygenation as a result of inadequate vascularization is considered a major obstacle to early graft function (43). Murine islets respond to reduced oxygenation in culture and posttransplant by activating HIF-1α and vascular endothelial growth factor A (VEGFA) to enhance local angiogenesis (24,40,41). Moreover, increasing islet vascularization, by forced expression of VEGF, improves transplant outcomes in the early posttransplant period (46). In addition, some reports show that HIF-1α, together with its binding partner aryl hydrocarbon receptor nuclear translocator (ARNT), is required for maintaining the normal β-cell function (6,14). Increasing HIF-1α stabilization by administration of iron chelators (19) improves human islet function in rodent transplant model recipients (39). Thus, an increased HIF-1α activation, secondary to hypoxia during islet isolation, could be expected to improve graft function by acting to promote local angiogenesis and glucose sensitivity. Indeed, hypoxic preconditioning has a protective effect for heart transplants (26), and this involves mechanisms including the upregulation of HIF-1α (9). Some studies demonstrate that ischemic preconditioning can improve islet yield (15), and intermittent hypoxia can improve insulin secretion (20). Thus, hypoxic preconditioning of whole pancreas, or isolated islets prior to transplantation, may improve posttransplant graft function.
In contrast, other studies would predict hypoxic preexposure to have a negative impact upon subsequent islet graft function. Hypoxic culture conditions are detrimental to isolated islet glucose-stimulated insulin release in vitro due to reduced mitochondrial hyperpolarization, energy depletion, and induction of cell death (8,20,21); however, as this effect is reversible, its longer-term impact on postgraft function is not known. Following transplantation, islet grafts accumulate HIF-1α protein, which then attenuates with time (24,36). The period of high HIF-1α expression is associated with reduced insulin secretion and increased apoptosis, suggesting a link between HIF-1α accumulation and impaired graft function (24,25). Further to this, hypoxia induces a nuclear factor κ-light-chainenhancer of activated B-cells (NF-κB)-dependent transcriptional program in islets characterized by increased expression of proapoptotic genes and increased cell death (16). Studies also show that specific induction of HIF-1α in β-cells diverts glucose metabolism to favor glycolysis and lactate production, resulting in impaired ATP production, reduced insulin secretion, and systemic glucose intolerance (4,31,45). Thus, exposure of islets to hypoxia has been proposed as one explanation for impaired islet graft function (3,8,13).
Based on current literature, it is difficult to predict what the impact of hypoxia will be for subsequent islet graft function, and further definitive studies addressing this question are lacking. Therefore, in this study, we determined the impact of pretransplant hypoxic exposure on posttransplant islet graft function and whole body glucose homeostasis in mice. Hypoxia response-gene expression was also analyzed in isolated human islets prepared for transplantation.
Materials and Methods
Human Islets and Cadaver Pancreas Specimens
Eight human pancreatic islet isolates were prepared at the Centre for Transplant and Renal Research, Westmead Hospital (Westmead Hospital, NSW, Australia) by a closed loop method as previously described (27). Immediately after extraction, ~5,000 IEQ pure islets were resuspended in mRNA isolation buffer (RLT lysis buffer, Qiagen, Germantown, MD, USA) and immediately stored at −80°C. Seven cadaver pancreas specimens were obtained from the New England Organ Bank (Waltham, MA, USA) and processed in the Islet Cell Resource Center of the Joslin Diabetes Center (Boston, MA, USA). All donors were Caucasian, and their mean age and BMI was 50.4±11.4 years and 25.6±4.8 kg/m2, respectively. The donors who were hyperglycemic before the isolation of pancreas were excluded. Some of these specimens have been reported previously in other studies (23). Hyperglycemic donor samples were excluded. Frozen pancreatic tissue was sectioned at 8 μm in a cryostat, mounted on uncoated glass slides at −20°C, and immediately stored at −80°C. Information on the eight pancreatic islet isolates used for the microarray study are described in ref. (7).
Laser Capture Microdissection (LCM)
LCM was performed on frozen pancreatic sections, as described (22), using the PixCell II Laser Capture Microdissection System (Arcturus Engineering, Mountain View, CA, USA): laser power, 35 mW; pulse duration, 3.0 ms; spot size, 7.5 μm. Each section typically had 3-10 islets, and, on average, six to seven sections provided 800 pulses required for sufficient RNA for arrays. β-Cell enrichment was determined by the ratios of expression (RTqPCR) of glucagon or somatostatin to insulin in β-cell-enriched LCM samples; glucagon/insulin expression ratio for β-cell selected versus generalized nonselected islet tissue was 0.24±0.13.
RNA Extraction, Amplification and Labeling
LCM samples were prepared as described (22). For islet samples, total RNA was extracted using the RNeasyPlus Mini Kit (Qiagen), and single-stranded cDNA was produced using the Superscript III Reverse Transcriptase Kit (Invitrogen, Mount Waverley, VIC, Australia).
Microarray Analysis
Briefly, expression data from isolated islets (Affymetrix HGU133+2 arrays; Affymetrix, Santa Clara, CA, USA) and β-cell-enriched tissue (Affymetrix U133 X3P) were independently normalized using the robust multiarray average (RMA) algorithm and was collapsed from probe to gene level by picking the probe with the highest average expression level. The two datasets were quantile normalized, and those genes that were in common were selected for comparative analysis. Heatmaps were created in GENE-E (Broad Institute, Cambridge, MA, USA).
RTqPCR Analysis
Primers were designed with Primer3 (http://primer3.wi.mit.edu/) based on Genbank sequences (www.ncbi.nlm.nih.gov/genbank/) and synthesized by Sigma-Aldrich (St. Louis, MO, USA). PCR reactions were performed on the RG300 Real Time PCR System (Rotorgene, Qiagen) using FastStart SYBR Green Master Mix (Roche Diagnostics, Indianapolis, IN, USA). Human glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and mouse cyclophilin were used as the housekeeping genes, and data were analyzed using the ΔΔCt method. See Tables 1 and 2 for human and mouse primer sequences, respectively.
Human Primers Used for RTqPCR Analysis, Designed Using Primer 3, and With Sequences Obtained From Genbank and Synthesized by Sigma Aldrich (Australia)

SLC2A1 (GLUT1), solute carrier family 2 (facilitated glucose transporter), member 1 (glucose transporter 1); HK1, hexokinase 1, GCK, glucokinase (HK4); PDK1, pyruvate dehydrogenase kinase 1; LDHA, lactate dehydrogenase A; VEGFA, vascular endothelial growth factor A; INS, insulin; GCG, glucagon; SST, somatostatin; IAPP, islet amyloid polypeptide; PDX1, pancreatic and duodenal homeobox 1; MCT4 (SLC16A3), monocarboxylic acid transporter 4 (solute carrier family 16, member 3); IRS1, insulin receptor substrate 1; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Mouse Primers Used for RTqPCR Analysis, Designed Using Primer 3, and With Sequences Obtained From Genbank and Synthesized by Sigma Aldrich (Australia)

CPH2, cyclophilin.
Animal Studies
All animal studies were approved by the Garvan/St. Vincent's Animal Ethics Committee. Female (6–8 weeks of age) C57BL/6 and RAG-/- mice were obtained from Australian BioResource (Mossvale, NSW, Australia). Animals were housed no more than five mice per cage, with free access to standard mouse chow and water, and were maintained on a 12-h light/12-h dark cycle.
Mouse Islet Isolation
In brief, mouse islets were isolated by perfusion of distended pancreas with 25 mg/ml collagenase I/II blend research grade (Roche, Indianapolis, IN, USA). The tissue was then digested at 37°C for 13 min, washed in M199 + 10% bovine calf serum (BCS) (HyClone; Thermoscientific, Waltham, MA, USA), passed through a 425-μm sieve (US standard sieve series, A.S.T.E. E-11 specifications dual MFG, Co., Chicago, IL, USA), and the resultant tissue was separated on Ficoll-Paque Plus (GE Healthcare Australia Pty. Ltd., NSW, Australia). Islets were washed in M199 + 10% BCS and were ready for further treatment.
Hypoxic Treatment
Isolated murine islets of Langerhans were cultured in Roswell Park Memorial Institute 1640 (RPMI1640) media (Invitrogen) under normoxic (21% O2, 5% CO2, 74% N2) or hypoxic (1% O2, 5% CO2, 94% N2) conditions for 16–18 h as previously described (4) and indicated in text. To activate HIF-1α, islets were cultured with 0.5 mmol/L dimethyloxalylglycine (DMOG; Sigma-Aldrich) for 16 h (4). HIF-1α protein was assessed by Western blotting as per Cantley et al. (4) using anti-mouse HIF-1α (Novus Inc., Saint Charles, MO, USA) and anti-β-actin (BD Biosciences, San Jose, CA, USA).
In Vitro Insulin Secretion
Insulin secretion was determined as previously described (2). In brief, islets were cultured for 18 h under normoxic or hypoxic conditions in RPMI1640 media, then moved to Krebs-Ringer bicarbonate HEPES buffer (KRBH) for 30 min under normoxic conditions. KRBH buffer was composed of 129 mmol/L NaCl, 5 mmol/L NaHCO3, 4.8 mmol/L KCl, 1.2 KH2PO4, 1.2 mmol/L MgSO4, 10 mmol/L 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), 2.5 mmol/L CaCl2, 0.1% bovine serum albumin (BSA), and 2.0 mmol/L glucose at pH 7.4 (all from Sigma-Aldrich). Next, batches of five islets were handpicked, and cultured in 130 μl of KRBH containing 2 or 20 mmol/L glucose (Sigma-Aldrich) for 1 h under normoxia. Insulin secreted into the KRBH was quantified by radioimmunoassay (Linco/Millipore, Saint Charles, MO, USA).
Islet Transplantation
Isolated islets from C57BL/6 mice were transplanted into C57BL/6 mice, at a ratio of three donor pancreata per one recipient, as previously described (42). Recipient mice were rendered diabetic by streptozotocin (Sigma-Aldrich) injection (200 mg/dl, IP). Diabetes was determined as blood glucose levels ≥25 mM on two consecutive readings. Fed blood plasma was harvested by sampling ~20 μl of blood from the tail using plastic capillary tubes (Sarstedt, Ingle Farm, SA, Australia), followed by centrifugation (Eppendorf 5810R, Eppendorf South Pacific, NSW, Australia) at 12,000 × g for 5 min to remove erythrocytes and collection of the supernatant. Insulin levels were determined by insulin ELISA (Crystalchem Inc., Downers Grove, IL, USA). In some cases, the islet graft was harvested from the kidney at times indicated and immediately placed in lysis buffer (Qiagen) for mRNA extraction. In some experiments, insulin labeling within the graft was conducted as we have previously described (44).
Immunohistochemistry
Tissues were fixed in 10% buffered formalin (Sigma-Aldrich, Sydney, Australia), and paraffin sections were stained with hematoxylin-eosin (Sigma-Aldrich, Sydney, Australia) or were stained for insulin using polyclonal anti-rabbit (Cell Signaling Technology Danvers, MA, USA) with the secondary antibodies anti-guinea pig (Jackson ImmunoResearch Laboratories, West Grove, PA, USA). The Vectastain Elite ABC kit (Vector Laboratories, Burlingame, CA, USA) was used to amplify the signal, and diaminobenzidine (DAB; Sigma-Aldrich, Sydney, Australia) was used for visualization. Sections were counterstained with hematoxylin.
Intravenous Glucose Tolerance Testing
Intravenous (IV) glucose tolerance testing of mice was performed 14 days posttransplantation following an overnight (16 h) fast with ad libitum access to water. d-Glucose (20% solution) was injected into the tail vein of conscious mice at a dose of 1 g/kg. Blood glucose levels were determined using an Accu-Chek Performa glucometer (Roche), and 5-μl blood samples were collected using glass capillary tubes (Sigma-Aldrich, Sydney, Australia) at the indicated time points. Insulin levels were determined by insulin ELISA (Crystalchem Inc.).
Statistical Analysis
Statistical significance was determined using Mann-Whitney U tests or two-tailed Student's t tests as appropriate and as indicated in the figure legends. A value of p < 0.05 was regarded as statistically significant. Statistical analysis was performed in R [version 2.8.0; (12)] and Graphpad Prism (version 5.0a; Graphpad Software, San Diego, CA, USA).
Results
Expression of Hypoxic-Response Genes in Isolated Human Islets and Laser-Dissected β-Cells
Some studies indicate that isolated human islets express high levels of hypoxia-regulated genes (22), and it has been suggested that induction of this hypoxia response contributes to the poor performance of islet grafts (3,25). Indeed, from genome transcript profiling analysis, comparing the gene expression profile of eight independent human islet isolates prepared within our clinical islet transplant center, with seven preparations of laser-dissected human β-cells from human pancreata, we could identify increased expression of characteristic (19) hypoxia-response genes in isolated islets (Fig. 1). Increased genes included cAMP response element-binding protein (CREB) binding protein (Cbp)/P300-interacting transactivator, with Glu/Asp-rich carboxyterminal domain, 2 (CITED2), and DNA-damage-inducible transcript 4 (DDIT4) as well as pyruvate dehydrogenase kinase (PDK1), v-maf musculoaponeurotic fibrosarcoma oncogene homolog F (MAFF) and solute carrier family 7 (cationic amino acid transporter, y+ system), member 1 (SLC7A1). Therefore, as shown here and previously (22), isolated human islets exhibit a gene expression profile characteristic for that of cells responding to hypoxia stress. This gene signature is present prior to the event of transplantation.

Gene expression analysis of hypoxia response genes in laser dissected human β-cells and isolated human islets. (A) Heatmap of the global-normalized-log2 gene expression level for hypoxia-inducible factor (HIF)-target genes, islet factors, islet hormones, and non-islet factors as indicated by category. Each column represents a different sample of either laser-dissected human β-cells designated with “cadaver” or isolated human islets designated with “H.” Gene symbols are from the OMIM database (www.omim.org).
Effect of Pretransplant Hypoxic Stress on Islet Graft Function
To test how hypoxia preexposure would affect islet transplant outcomes, we established a model, whereby primary C57BL/6 mouse islets otherwise isolated under optimal conditions (anesthetized living donor), were then exposed to hypoxia by controlled culture in 1% O2 for 16 h and subsequently transplanted under the renal capsule of diabetic and syngeneic recipients. Islet grafts achieve near normal levels of oxygenation under the kidney capsule (29), so this approach would reveal how preexposure to hypoxia would impact later graft function. The results from these transplant studies are presented as both individual blood glucose plots (Fig. 2A) and pooled median values (Fig. 2B). Islet grafts prepared under normoxic (21% O2) conditions were able to rapidly restore metabolic control to normal levels, reliably keeping blood glucose in a tight range around 5 mM. The function of normoxic cultured islet grafts was no different to that of freshly isolated and immediately transplanted islet grafts (data not depicted). In marked contrast, mice receiving hypoxia-preexposed islet grafts exhibited poor glucose homeostasis after transplantation; indeed, the mean blood glucose reading for mice receiving hypoxia-preexposed grafts was >15 mM for the majority of the postoperative observation period (p < 0.05).
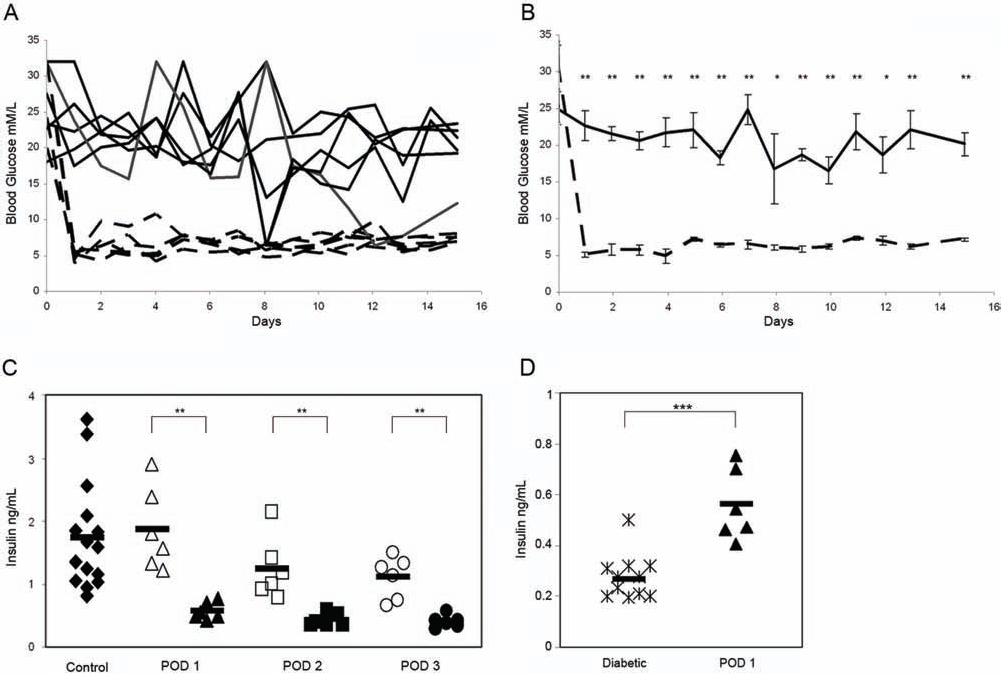
Maintenance of blood glucose homeostasis by hypoxia-preexposed islet grafts. (A) Blood sugar levels of individual mice receiving either control normoxic islet grafts (hatched line) or hypoxia-preexposed grafts (solid line); n = 6 per group. (B) Median blood sugar levels derived from (A) for mice receiving either control normoxic islet grafts (hatched line) or hypoxia-preexposed grafts (black line). Error bars indicate mean±SEM; n = 6 per group per time. (C) Fed plasma insulin levels for mice receiving either control normoxic islet grafts (open symbols) or hypoxia-preexposed grafts (closed symbols except diamonds) at postoperative days (PODs) 1, 2, and 3; n = 6 per group. “Control” indicates random feed blood insulin levels from a cohort (n = 14) of age- and sex-matched C57BL/6 mice provided for comparison (closed diamond symbols). Each point represents an individual mouse, and bars indicate median value; **p < 0.01. (D) Fed plasma insulin levels as in (C) for either streptozotocin-treated diabetic mice (stars) or mice receiving hypoxiapreexposed grafts (closed symbols). Each point represents an individual mouse, and bars indicate median value; ***p < 0.001.
Hypoxic Exposure Impairs Glucose-Stimulated Insulin Release In Vivo
Consistent with the significantly elevated fed glucose levels of mice receiving hypoxia-preexposed islet grafts, these mice also exhibited impaired insulin release in the fed state (Fig. 2C), whereas fed insulin levels for mice receiving control normoxia grafts where similar to that of nontransplanted control mice (p > 0.05; n > 6). Nonetheless, the fed insulin levels of mice receiving hypoxia-preexposed grafts was significantly higher than insulin levels seen for diabetic mice treated with streptozotocin but otherwise unmanipulated (Fig. 2D). These data suggested that mice receiving hypoxia-preexposed islet grafts harbored a β-cell defect. When challenged with an exogenous intravenous injection of glucose, mice receiving hypoxia pretreated grafts were found to be severely glucose intolerant, due to a highly significant defect in glucose-stimulated insulin secretion (GSIS) (Fig. 3A). The impaired GSIS related to a complete loss of both first- and second-phase insulin response to a glucose challenge (Fig. 3B). Thus, pretransplant exposure of islets to hypoxia is sufficient to severely impair graft function, resulting in the inability of a normally adequate mass of transplanted islets (three donors: one recipient) to control glucose homeostasis.
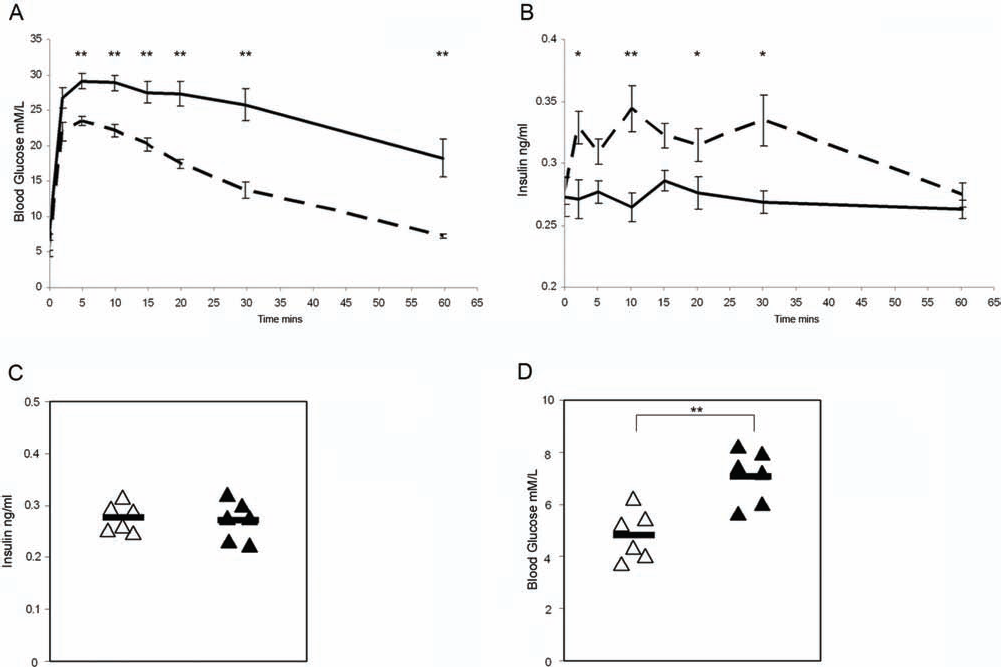
In vivo glucose-stimulated insulin secretion from hypoxia-preexposed islet grafts. (A) Blood glucose and (B) blood insulin levels; overtime for mice receiving control normoxic islet grafts (hatched line) or hypoxia-preexposed grafts (solid line) following a 16-h fast and intravenous glucose challenge (1 g/kg); *p < 0.05; **p < 0.01. (C) The 16-h fasting blood insulin levels and (D) fasting blood glucose from mice that received normoxic grafts (open triangles) or hypoxia-preexposed grafts (closed triangles). Data represents mean values±SEM, n ≥ 6 per time point; **p < 0.01.
Hypoxia-Preexposed Islet Grafts
It is significant to note that after a 16-h fast, mice receiving hypoxia-preexposed grafts had normal insulin levels (Fig. 3C) with only a slight elevation in fasting blood glucose (Fig. 3D). Together with our results from the intravenous glucose tolerance test, this suggests impaired function rather than a complete absence of β-cell function or, indeed, β-cells. Histological analysis of both hypoxia-preexposed islet grafts and normoxia grafts 10 days posttransplantation revealed evident islet structures and definitive insulin labeling (Fig. 4A–D). However, the insulin labeling did indicate regions impoverished for insulin protein within the hypoxia-preexposed grafts compared to the intense labeling revealed for normoxia grafts. These data point to a defect in β-cell function as opposed to an outright loss of β-cells as the main cause of poor glycemic control in mice receiving hypoxia-preexposed islet grafts. This interpretation is further supported by the finding of equivalent levels of pancreatic and duodenal homeobox 1 (PDX1) mRNA expression in both normoxia and hypoxia-preexposed grafts harvested at day 15 after transplantation (Fig. 4E). These data show that hypoxic preexposure has a deleterious effect upon later graft function, with depleted insulin content contributing to poor posttransplant glycemic control.

Hypoxia-preexposed islet grafts. Examples of hematoxylin and eosin staining for representative normoxic (A) and hypoxic-preexposed (B) grafts at day 10 posttransplantation. Examples of insulin labeling for corresponding normoxic (C) and hypoxic-preexposed (D) grafts in (A) and (B). (E) Expression levels for pancreatic and duodenal homeobox 1 (PDX1) in normoxic (open bar) and hypoxic-preexposed (closed bar) grafts as assessed by RTqPCR. For reference, PDX1 mRNA levels in freshly isolated islets are indicated by the broken line. Islet grafts were harvested at postoperative day 15. Data are pooled values from three islet grafts per group.
Hypoxia-Induced Molecular Changes in Isolated Rodent Islets
We next considered possible molecular mechanisms for the hypoxia-induced impairment in islet graft function. We noted that a common feature of the hypoxic response genes expressed by isolated human islets presented in Figure 1 was their coordinate regulation by the transcription factor HIF-1α (19). Some studies (4,31,45) show that stabilization of the HIF-1α protein in rodent β-cells impairs ATP production and insulin secretion by increasing the expression of genes that oppose glucose oxidation including lactate dehydrogenase (LDH), PDK1, and monocarboxylate transporter 4 (MCT4), whereas solute carrier family 2 (facilitated glucose transporter), member 2 (SLC2A2; GLUT2), and glucokinase (GCK) are negatively regulated. To determine whether hypoxic preexposure had triggered such an HIF-1α-dependent molecular switch to anaerobic glycolysis, primary mouse islets were exposed to hypoxia (1% O2, 16 h) and subsequently examined for HIF-1α accumulation and glycolytic gene expression. Western blot analysis demonstrated stabilization of HIF-1α protein levels in islets cultured under hypoxic conditions compared to control normoxic conditions (Fig. 5A). Further to this, and in association with HIF-1α stabilization, hypoxic islets exhibited increased mRNA levels for SLC2A1 (GLUT1), PDK1, LDHA, and MCT4 (Fig. 5B). In addition, SLC2A2 and GCK expression was downregulated by hypoxia. Thus, exposure of murine islets to hypoxia stabilized HIF-1α protein levels and altered the expression pattern of metabolic genes to favor anaerobic glycolysis and lactate production. This gene expression pattern was associated with poor posttransplant performance.

Molecular changes induced in primary mouse islets by hypoxia. (A) Western Blot analysis of HIF-1α protein in freshly isolated mouse islets cultured for 16 h under either normoxic (21% O2) or hypoxic culture conditions (1% O2), or in normoxic conditions supplemented with dimethyloxaloylglycine (DMOG; 0.5 mmol/L). Data from six independent islet batches per treatment. Two representative examples of each treatment are shown. (B) Gene expression levels for islets cultured as in (A) expressed as fold change comparing hypoxic versus normoxic culture conditions, n = 3 per group. (C) Gene expression levels for islets cultured as in (A) expressed as fold change comparing DMOG-treated versus normoxic culture conditions, n = 3 per group; *p < 0.05; **p < 0.01; ***p < 0.001. SLC2A1, solute carrier family 2 (facilitated glucose transporter), member 1; GCK, glucokinase; LDHA, lactate dehydrogenase A; PDK1, pyruvate dehydrogenase kinase, isozyme 1; MCT4, monocarboxylic acid transporter 4.
HIF-1α Stabilization is Sufficient to Alter Glycolytic Gene Expression in Rodent Islets
Hypoxia can activate multiple stress pathways including the NF-κB and HIF pathways (28). To test whether hypoxia-induced activation of the HIF-1α pathway alone was sufficient to induce changes in glycolytic gene expression, rodent islets were treated with the pharmacologic HIF-1α activator DMOG, and HIF-1α stabilization was determined. Isolated islets treated with DMOG showed increased HIF-1α stabilization (Fig. 5A). The accumulation of HIF-1α occurred with a concomitant increase in mRNA levels for SLC2A1, LDHA, and MCT4 (Fig. 5C). Also, SLC2A2 and GCK were downregulated, albeit nonsignificantly. This same pattern of HIF-1α stabilization and gene expression was similar to that seen for islets cultured under hypoxic conditions (Fig. 5B). Thus, stabilization of HIF-1α by hypoxic exposure altered the expression pattern to glycolytic genes that would favor anaerobic glycolysis.
Hypoxic Exposure Impairs Glucose-Stimulated Insulin Release In Vitro
Stabilization of the HIF-1α protein via gene mutation in rodent β-cells impairs ATP production and impairs insulin secretion by increasing the expression of metabolic genes favoring glycolysis and lactate production (4,31,45). We questioned whether hypoxic exposure of islets prior to transplantation also resulted in impaired β-cell function through a similar mechanism. Islets were cultured under normoxic or hypoxic conditions for 18 h before static incubations were performed for 1 h under normoxic conditions to measure insulin release (Fig. 6). These experiments revealed that hypoxic islets exhibited higher-than-normal insulin secretion at 2 mM glucose compared to islets cultured under normoxic conditions. Furthermore, islets exposed to hypoxia exhibited a reduced secretory response from approximately sixfold of basal following normoxic culture to ~2.5-fold following hypoxic culture. This indicates that hypoxia-pretreated islets have an impaired ability to sense and respond to changes in glucose concentration, consistent with our in vivo data demonstrating impaired GSIS in diabetic mice possessing hypoxia-pretreated grafts. Thus, stabilization of HIF-1α by hypoxic exposure altered the expression pattern of glycolytic genes to favor anaerobic glycolysis, resulting in impaired islet graft function.
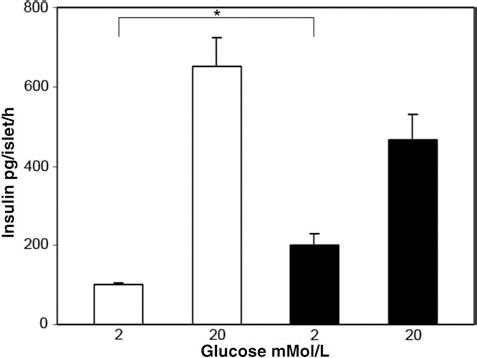
Effect of hypoxic exposure on in vitro GSIS. Glucose-stimulated insulin secretion (GSIS) response of mouse islets cultured for 18 h under either normoxic (21% O2; open bars) or hypoxic culture conditions (1% O2; closed bars) prior to static incubation for 1 h at 2 mM or 20 mM glucose. Data represent mean±SEM of eight replicates, from one of four independent experiments conducted; *p < 0.05.
Expression of HIF Target Genes in Isolated Human Islets
Given that human islets expressed characteristic HIF-1α-regulated genes (see Fig. 1), we next investigated whether isolated human islets exhibited a HIF-1α-dependent molecular switch to anaerobic glycolysis relative to β-cells in situ. This analysis revealed that relative to laser-dissected β-cells, isolated human islet preparations showed an ~18-fold increase in lactate dehydrogenase expression, approximately sixfold increase in hexokinase 1 (HK1) and HK2, threefold increase in SLC2A1, and 70% decrease in glucokinase levels (Fig. 7A). This pattern resembles mouse islets exposed to hypoxia (see Fig. 5B) as well as those expressing high levels of HIF (4,31,45).

Altered expression of hypoxia-regulated metabolic genes is associated with the isolation of human islets. (A) Using micro-arrays, gene expression levels of key metabolic hypoxia-response genes were measured and used to calculate fold changes in gene expression associated with human islet isolation (n = 8), when compared to human islets in situ recovered by laser microdissection (n = 7). Data represent mean gene expression changes±SEM. (B) Expression levels for key hypoxia-response genes in isolated human islets as assessed by RTqPCR (–ΔCt, n = 8). For reference, mRNA levels for insulin (INS), islet amyloid polypeptide (IAPP), PDX1, and IRS1 are plotted. Horizontal black bars show median values. (C) Hypoxia-response gene expression stability in human islet grafts 4 weeks after transplant into recombination activating gene knockout (RAG–/–) mice, assessed using RTqPCR and normalized to pretransplant levels. Individual grafts are plotted; black bars indicate the median; n = 6 per group; *p < 0.05, **p < 0.01, ***p < 0.001. HK1, hexokinase 1; VEGF, vascular endothelial growth factor.
We next determined whether the molecular profile of isolated human islets would correct after transplantation. We compared glycolytic gene expression levels in human islet preparations prior to, and 4 weeks after, transplantation under the renal capsule of euglycemic immunodeficient recombination-activating gene knockout (RAG-/-) mice by RTqPCR (Fig. 7B, C). The RTqPCR data showing baseline mRNA expression levels for the glycolytic genes in isolated islets are presented in Figure 7B, whereas comparisons between pre- and posttransplanted human islets are represented in Figure 7C as –ΔΔCt values. Note that LDHA mRNA levels in isolated human islets were consistently in the range of expression levels for islet amyloid polypeptide (IAPP) (Fig. 7B). Following transplantation, expression levels for LDHA and hexokinase were maintained at levels equivalent to that of newly isolated islets (see Fig. 7C). In contrast, SLC2A1 levels decreased approximately twofold. Thus, despite providing similar levels of oxygenation to that of the native pancreas (29), subcapsular transplantation could not reverse hypoxiaregulated metabolic gene expression to that of pancreatic islets in situ. In addition, HIF-1α protein was not detected in β-cell nuclei by immunostaining of islet grafts 4 weeks posttransplant (data not shown), suggesting that continual HIF-1α activation is not necessary to maintain the hypoxia-induced gene expression profile. Thus, isolated human islets express a molecular signature consistent with that of islets exposed to a hypoxic insult, which would, based on our animal modeling, predict impaired graft function and poor glycemic control. This profile is not corrected after transplantation into a relatively benign graft site—under the kidney capsule—and would be potentially further exacerbated by transplantation into the deoxygenated environment (29) of the portal vein.
Discussion
Many clinical studies indicate that, following isolation, human islets typically show poor function in vivo. The clearest evidence for this is seen in the need to transplant multiple islet grafts before adequate metabolic control can be achieved (33,35,38). Therefore, understanding the mechanisms responsible for poor islet graft function will allow the development of targeted strategies to improve the success of clinical islet transplantation.
Consistent with a previous report (22), we show that at the point of isolation, human islets express a molecular profile characteristic of cells exposed to a hypoxic environment. Given the clinical practice to transplant human islets immediately after isolation, without culture (38), and the speculation that hypoxia is detrimental to islet function (3,8,13), it would be important to determine the effect of hypoxic preexposure upon latter islet graft function. Therefore, we examined graft outcomes for islets prepared under optimal conditions versus those exposed to a hypoxic environment. Strikingly, hypoxic islet grafts failed to provide metabolic control to syngeneic recipients, demonstrating that hypoxic exposure prior to transplantation results in impaired islet graft function and whole-body glucose intolerance. Mechanistically, the failure of hypoxic grafts to regulate glucose homeostasis related to a major defect in the ability of β-cells to secrete insulin in response to a glucose challenge.
Different to many other cell types, β-cells express relatively low levels of lactate dehydrogenase, monocarboxylate transporters, and low Km hexokinases (30,32). We have identified the upregulation of these enzymes in hypoxic islets, along with increased PDK1, which act to reduce glucose oxidation: PDK1 inhibits PDH activity preventing pyruvate entry to the mitochondrial tricarboxylic acid (TCA) cycle, LDH converts pyruvate to lactate, and MCT4 enables export from the cell. In addition, we identified a reduction of the high Km glucokinase and increase in low Km hexokinases 1 and 2, potentially further disrupting glucose sensing in hypoxic β-cells. Normoxic β-cells rely on mitochondrial oxidative phosphorylation to generate sufficient quantities of ATP per molecule of glucose metabolized to trigger insulin secretion within the normal physiological range of glucose concentrations (37) and thereby maintain whole-body glucose homeostasis. Indeed, forced expression of LDHA (1) in β-cell lines impairs glucose sensing and insulin release, demonstrating that low expression of LDH is essential for normal β-cell function. Stabilization of HIF-1α protein, the principal hypoxia-induced transcription factor (19), in murine β-cells via genetic mutation alters β-cell metabolism to favor anaerobic glycolysis and leads to impaired glucose-stimulated insulin release and glucose intolerance in vivo (4,31,45). This functional impairment relates to a loss of β-cell specialization, due to specific molecular changes including increased expression of the normally suppressed genes LDHA, PDK1, and MCT4, whereas SLC2A2 and GCK are reduced (4,31,45). This HIF-1α-induced molecular reconfiguration severely dampens mitochondrial metabolism and increases lactate generation, resulting in impaired glucose-stimulated insulin release. These molecular and metabolic changes support the concept that islets exposed to low oxygen tension lose the specialization of gene expression responsible for normal β-cell physiology (32), resulting in graft dysfunction and poor transplant outcome.
Another finding of our study is that neither in vitro culture under normoxic conditions, nor transplantation into immunodeficient RAG-/- recipient mice, could restore the molecular profile of human islets to that of a nonhypoxic state. This is perhaps a rather unexpected observation.
Some evidence demonstrates that HIF-1α is activated in islets after transplantation (24), and HIF-1α may also be stabilized by inflammatory pathways (10,11). Thus, continued HIF-1α activation may explain the persistence of the altered expression pattern of glycolytic genes. Though not explored in this study, glucotoxicity may contribute to the persistence of graft dysfunction observed for mouse islets and for the altered glycolytic gene expression observed for human islets (18). Alternatively, the persistence of this molecular signature may be suggestive of epigenetic changes induced by organ harvesting and tissue isolation, which subsequently results in permanently altered gene expression patterns.
This study demonstrates that hypoxic exposure of islets prior to transplantation has an overall negative impact on graft function and glucose homeostasis. The induction of a hypoxia-like genetic program results in dysregulated expression of genes that control metabolic glucose sensing and suggests wider β-cell dedifferentiation. It is possible that the negative impact of the hypoxic-response program in islets is masking potential positive effects; therefore, further studies to specifically target positive consequences of HIF-1α activation, such as proangiogenic responses, may prove beneficial. Given that isolated human islets, like hypoxic-exposed rodent islets, show a loss of specialization with altered expression of genes regulating glucose-stimulated insulin release, this may explain by extrapolation why large numbers of islets and multiple transplants are required to reverse diabetes in clinical trials. Our findings also suggest that early intervention in reversing the hypoxic changes may be critical to improve graft function and transplant outcome.
Footnotes
Acknowledgments
The authors thank Associate Professor Sof Androkopoulos, Department of Medicine, University of Melbourne, Australia; as well as Dr. Ross Laybutt, Diabetes and Obesity Research Program, Garvan Institute, Australia, for the insightful critique and helpful discussion. The authors acknowledge the technical expertise of Helen Speirs from the Ramaciotti Centre University of New South Wales regarding microarrays; as well as Anita Patel and Lindy Williams from the clinical islet isolation team at Westmead Hospital, Westmead, Australia. Work by S.N.W., A.W., M.J.C., W.K., and S.T.G. was supported by the National Health and Medical Research (NHMRC) grants 427695 and 427828 to S.T.G. Work by G.C.W. and M.H.J. was supported by grants from the American Diabetes Association, the Juvenile Diabetes Research Foundation, the National Institutes of Health (RO1 DK66056 and P30 DK36836 – the Advanced Microscopy, Bioinformatics, and Genomics Cores of Joslin Diabetes and Endocrinology Research Center). J.C. holds the Viertel Fellowship, awarded by the Diabetes Australia Research Trust. S.N.W. is the recipient of a Ross Trust Fellowship. S.T.G. is an Australian Research Council Future Fellow and Honorary NHMRC Senior Research Fellow. Dr. Shane T. Grey is the guarantor of this work, had full access to all the data, and takes full responsibility for the integrity of data and the accuracy of data analysis. J.C., S.N.W., M.H., A.W., M.J.C., W.K., and S.T.G. researched the data. M.H.J. and G.W. conducted microarray laser-dissected human β-cell studies. W.J.H. and P.O.C. from the Australian Islet Transplant Consortium provided access to isolated human islets. J.C., S.N.W., G.W., and S.T.G. wrote and edited the manuscript. The authors declare no conflicts of interest.
