Abstract
In this study, we evaluated the effects of exendin-4 on free and encapsulated islet grafts in a rodent model. We also investigated the role of a transcription factor, hypoxia-inducible factor-1 (HIF-1), in mediating the beneficial effects of exendin-4. Diabetic athymic mice were transplanted with free rat islets under the kidney capsule or with macroencapsulated rat islets SC with or without exendin-4, islet preculture (exendin-4 0.1 nM for 20 h), and/or recipient treatment (IP 100 ng/day, day 0–7). The mice were followed for 4 weeks and the graft function and β-cell volume were evaluated. The effects of exendin-4 on islet HIF-1α mRNA and protein expression and on ATP content in a rat insulinoma cell line (INS-1E) were also examined. Preculture with exendin-4 followed by recipient treatment improved the outcome of both free (73% graft function vs. 26% in controls, p = 0.03) and macroencapsulated islet grafts (100% vs. 25% in controls, p = 0.02). In macroencapsulated grafts, the exendin-4-treated group had significantly larger endocrine volume, less graft necrosis, and more blood vessels around the capsule. In rat islets cultured with exendin-4, HIF-1α mRNA and protein expression were significantly enhanced. ATP content was increased in exendin-4-treated INS-1E cells under hypoxic conditions. The improved functional outcome after transplantation of a marginal islet mass with a brief initial treatment with exendin-4 is related to a larger surviving endocrine cell volume. Exendin-4 may improve islet graft resistance to hypoxia during the peritransplant period by increasing the expression of HIF-1α.
Keywords
Introduction
The loss of functional β-cell mass has emerged as one of the major factors limiting the efficacy of islet transplantation in selected patients with type 1 diabetes (2,11,34). Exendin-4, a glucagon-like peptide-1 (GLP-1) receptor agonist, is one of several drugs that have been shown to improve the outcome of islet transplantation (19,20,35,40,42,48). This drug has shown promising results in type 2 diabetic animal models (18) and in patients with type 1 and 2 diabetes (10,41). It has a number of effects including glucose-dependent stimulation of insulin secretion (1), the suppression of glucagon secretion (8), the slowing of gastric emptying, the inhibition of food intake, and the modulation of glucose trafficking in the peripheral tissues. Of special interest is its ability to inhibit β-cell apoptosis and increase β-cell proliferation (26,27,47).
The period immediately after islet isolation and transplantation is particularly crucial for long-term survival of the islets. In an earlier study, we demonstrated that a combination of islet preculture and recipient treatment with exendin-4 improves the metabolic outcome of a suboptimal number of rat islets transplanted to diabetic athymic mice (35). However, that study was not designed to compare the individual impact of islet preculture or recipient treatment on graft outcome. This is important for clinical use as side effects of exendin-4, such as severe nausea and vomiting, can affect patient compliance (25).
After clinical islet transplantation, the islets are at risk of being destroyed not only by the recipient's immune system but also by toxic damage caused by immunosuppressive drugs (33). Encapsulation of the islet graft inside an immunoprotective membrane device is an attractive option to avoid the need for long-term immunosuppression. However, until such an implantable device is revascularized, the islet graft is exposed to nutrient deprivation (i.e., hypoxia that may induce apoptosis of the β-cells) (28,29). During the early posttransplant period, the antiapoptotic effects of exendin-4 could improve islet survival inside these immunoprotective devices. To our knowledge, the effects of exendin-4 on macroencapsulated islets have not yet been studied.
The transcription factor hypoxia-inducible factor-1 (HIF-1) is expressed by cells in response to hypoxia and consists of α and β subunits. While HIF-1β is expressed constitutively, HIF-1α is degraded by an oxygen-mediated pathway. Normally, mammalian cells lack HIF-1α and HIF-1. HIF-1 influences glucose metabolism adaptation, angiogenesis, apoptosis, and cell growth (31). Following HIF-1α expression, glycolysis is increased to produce more ATP and avoid energy deprivation in hypoxic cells. Recently, it has been reported that HIF-1α is an important molecule to regulate β-cell function in pancreatic islets even in normoxic conditions (6). We hypothesized that exendin-4 may have a cytoprotective effect on β-cells during the hypoxic phase in the peritransplant period.
The aim of this experimental study was to evaluate three different treatment protocols of exendin-4 by determining the metabolic outcome of rat islets transplanted under the kidney capsule of diabetic athymic mice. The effects of exendin-4 on the outcome after transplantation of a marginal mass of macroencapsulated islets were also evaluated. The mechanisms of action of exendin-4 on rat islets in culture were investigated with special attention to HIF-1α expression. Effect of exendin-4 on energy production was evaluated on a single cell level using a rat insulinoma cell line (INS-1E) under hypoxic conditions.
Materials and Methods
Animals
The local Animal Ethics Committee approved the study. The requirements of the Animal Welfare Act and the National Institutes of Health guidelines for the care and use of laboratory animals were followed.
Rat Pancreatic Islet Preparation
Male Sprague-Dawley rats (Scanbur, BK AB, Stockholm, Sweden) weighing 300 g were used as pancreatic islet donors. The method of isolating rat islets has been described previously (35). Briefly, the rat pancreas was digested using a collagenase solution (Collagenase-P, 0.7 mg/ml, Roche Diagnostics GmBh, Mannheim, Germany) and the islets were purified using density gradient centrifugation (Histopaque-1119, Sigma-Aldrich, St. Louis, MO, USA; Lymphoprep™, Axis-Shield PoC AS, Oslo, Norway; Hanks balanced salt solution, Karolinska University Hospital, Stockholm, Sweden). Isolated islets were cultured in RPMI-1640 medium (Gibco, BRL, Life Technology Ltd., Palisly, Scotland) supplemented with 10% fetal bovine serum (FBS), 2 mM l-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin (all provided by Gibco) at 37°C in a humidified atmosphere (5% CO2 and 95% air).
Induction of Diabetes in Athymic Mice
Diabetes was induced in male inbred athymic mice (nu/nu Black 6, Taconic M&B, Ry, Denmark, weighing 25 g) by a single IV injection of streptozotocin (Sigma-Aldrich Co., 200 mg/kg body weight). An animal was considered to be diabetic if its blood glucose levels exceeded 20 mM for 2 consecutive days.
Islet Transplantation Under Kidney Capsule
The rat islets were handpicked after 20-h culture and transplanted under the kidney capsule as previously described (35). Diabetic athymic mice were used as islet recipients. Four study groups are shown in Table 1. In our model, the diabetic athymic mice were repeatedly cured by 50 rat islets but not by 30 islets, as shown in a previous study (35). To mimic the clinical situation, we used a marginal mass of 30 free islets in this study.
Exendin-4 Treatment Protocols for Mice Transplanted With 30 Free Islets Under the Kidney Capsule

From day 0 to 7, recipient mice were treated (IP) with 100 ng exendin-4 (+) or with vehicle only (-).
Macroencapsulated Rat Islet Transplantation
TheraCyte™ devices (TheraCyte, Inc., Laguna Hills, CA, USA) were utilized for the macroencapsulation study. The internal volume of the device used for this study was 4.5 μl. We have observed that transplantation of 1,000 macroencapsulated rat islets regularly resulted in normalization of blood glucose levels 2–3 weeks after transplantation in diabetic athymic mice, while 500 rat islets, which constitute a marginal mass in macroencapsulated situation, were usually insufficient for curing the diabetic mice. The isolated rat islets were cultured for 20 h in either the presence or absence of exendin-4 (0.1 nM) (AnaSpec, San Jose, CA, USA). Five hundred rat islets were handpicked and pelleted before being loaded into the device. The methods for loading islets and transplantation of the membrane were previously described (37). In this study, a combination of preculture and recipient treatment with exendin-4 was compared to the control group without exendin-4 treatment.
Posttransplant Management
The mice were observed for 4 weeks. The nonfasting blood glucose levels and weights of the animals were measured daily during the first week. Thereafter, mice with blood glucose levels below 10 mM for 2 consecutive days were assessed twice a week. The mice with hyperglycemia (≥ 11 mM) were checked daily. The mice with blood glucose levels above 20 mM received a mixture of short- and long-acting human recombinant insulin (2 units each of Actrapid and Ultratard, Novo Nordisk AS, Bagsvaerd, Denmark) subcutaneously as previously described (35). Mice with nonfasting blood glucose levels of 10 mM or below were defined as cured, 11–20 mM as partial graft function and above 20 mM as graft failure. An intraperitoneal glucose tolerance test (IPGTT) was performed in cured mice after 4 weeks as previously described (35). Spontaneous recovery of native pancreas function was ruled out by graftectomy that resulted in recurrence of diabetes in all cured and partially cured animals. Body weight gains were calculated by subtracting the body weight at day 0 from the body weight at day 28 prior to graftectomy. When the mice were killed before day 28, body weight values of the last day were used.
Measurement of Insulin Content of the Islets Transplanted Under Kidney Capsule
The total amount of insulin in the islet grafts at 4 weeks after transplantation was compared between the control and study groups. Renal subcapsular grafts in the exendin-4-treated animals were easily identified macroscopically. However, in the nontreated animals, the grafts were usually invisible; therefore, the whole graft-bearing kidneys were used for the insulin content analysis. Both kidneys were removed immediately after the mice were sacrificed and were quickly frozen in liquid nitrogen and kept at −80°C until use. The frozen kidneys were homogenized in acid–ethanol solution (0.1 M HCl in 75% alcohol). After centrifugation, the supernatant was used to measure insulin content by a Rat insulin ELISA kit (Mercodia AB, Uppsala, Sweden).
Histological Studies
The methods used for histological analyses were previously described (30,37). Briefly, formalin-fixed explanted devices were divided centrally, embedded in paraffin, and serially sectioned into 5-μm thick sections using a microtome (Microm International, Walldorf, Germany). Sections with a distance of 74 μm were collected and stained with hematoxylin and eosin. Volume densities (i.e., fractional volumes and absolute volumes) were evaluated by morphometry. The total volume of tissue inside the device was measured using digital photos taken of all tissue-containing sections. In the photos, the area of tissue was measured using a semiautomatic interactive image analyzer (Videoplan, Zeiss, Oberkochen, Germany) and the volumes were calculated according to Cavalieri's principle (38). To estimate the volume density of the various tissue components, every fourth section from the same graft was photographed at 10× magnification. Because one section contained two different levels of the encapsulated graft, at least eight levels of the graft were examined in an individual specimen. Point counting was performed using a square lattice (1 cm). Conventional stereological principles were used to estimate volume density of viable endocrine cells, necrosis, and fibrosis. The absolute volume of each tissue type was then calculated by multiplying the volume density with the total tissue volume (45). One device in the exendin-4-treated group had to be excluded from the evaluation due to technical problems during dehydration.
The numbers of vascular profiles adjacent to the membrane surface were evaluated using the histological samples described above. In this study, we chose six levels from the specimen of the same grafts systematically. From the central area of the membrane surface, micrographs were taken, with an original magnification of 20x, 8 mm along the membrane at both sides. Vascular profiles within 100 μm from the membrane surface were counted as previously described (36). All examinations were performed blindly on coded specimens by the same investigator (M.K.). The micrographs were counted twice and the intraobservation variability was found to be less than 10%.
Immunofluorescence Staining
To estimate the amount of insulin-positive and glucagons-positive cells inside the device, every fifth section was chosen for immunofluorescence staining. Sections were deparaffinized and boiled with Tris buffer for 14 min. The sections were blocked with 1% rabbit sera for 30 min at room temperature, and then incubated with mouse anti-glucagon antibody (clone k796B10, 1:100 dilution, Sigma) for 1 h. For the secondary antibody, fluorescein isothiocyanate (FITC) rabbit anti-mouse IgG antibody (1:1000, Jackson Immune Lab) was used. For insulin staining, rhodamine-conjugated anti-insulin antibody (clone 2D11-H5, 1:50, a kind gift from Mabtech AB, Sweden) was added on the slide and incubated for 60 min. Nuclei were stained with DAPI (4,6-diamidino-2-phenylindole hydrochloride; Pierce, Rockford, IL, USA) at the concentration of 100 ng/ml in PBS containing 0.5% FBS for 2 min. Finally the slides were mounted with DAKO fluorescent mounting medium (DAKO Corporation). Slides were examined with an Olympus fluorescent microscope and pictures were taken using Cell^F Imaging Software.
In Vitro Study with Rat Pancreatic Islets or INS-1E Cells
Protein Extraction and Western Blot for Determination of HIF-1α, and Active Caspase-3
Four hundred purified rat islets were cultured in RPMI-1640 media with 0, 0.1, and 10 nM exendin-4 for 20 h. The nuclear and cytosolic proteins were sequentially extracted according to the protocol of Schreiber et al. (32). Briefly, the rat islets were suspended in lysis buffer A [10 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid and potassium hydroxide (HEPES-KOH; pH 7.8), 10 mM KCl, 0.1 mM EDTA (pH 8.0), and 0.1% NP40], incubated on ice for 20 min, and then vigorously mixed. After centrifugation at 1,500 rpm (Eppendorf 5403 Refrigerated Centrifuge) for 10 min, the supernatants were recovered as cytoplasmic proteins. The pellets were resuspended in lysis buffer C [50 mM HEPES-KOH (pH 7.8), 420 mM KCl, 0.1 mM EDTA (pH 8.0), 5 mM MgCl2, and 20% glycerol] and incubated on ice for 30 min. After centrifugation at 15,000 rpm at 4°C for 30 min, the supernatants were recovered as nuclear proteins. Protein concentrations were measured using a BCA Protein Assay kit (Pierce, the Thermo Scientific Life Science Research, Rockford, IL, USA). Nuclear and cytosolic extracts (30 μg) were resolved on 8% and 12% sodium dodecyl sulfate polyacrylamide gels (SDS-PAG) for HIF-1α and active caspase-3 blots, respectively. HIF-1α protein was detected using a 1:2000 dilution of rabbit antibody specific for HIF-1α (Novus Biologicals, Littleton, CO, USA). Active caspase-3 protein was detected using rabbit anti-active caspase-3 antibody (1:200, Chemicon international, Billerica, MA, USA). As the loading control, rabbit anti-topoisomerase-1 (Topo1; 1:10,000, TopoGen, Columbus, OH, USA) and rabbit anti-β-actin (1:5000, Abcam, Cambridge, UK) were used, respectively. Primary antibodies were detected with peroxidase-conjugated donkey anti-rabbit immunoglobulin antibody (Amersham, GE Healthcare, Buckinghamshire, UK) and visualized with ECL reagent (GE Healthcare) on a high performance chemiluminescence film (GE Healthcare). Densities of the immunoblot were quantified using ImageJ software (version 1.62; National Institutes of Health, Bethesda, MD, USA). The relative expression of HIF-1α and active caspase-3 were determined by fold changes compared to each loading control.
Semiquantitative Real-Time PCR
Total RNA was extracted from purified rat islets incubated with 0, 0.1, and 10 nM exendin-4 for 20 h using RNeasy Mini kit (Qiagen, Hilden, Germany) according to the manufacturer's instructions. RNA integrity and concentration was analyzed using a NanoDrop® Spectrophotometer ND-1000 (Saveen & Werner AB, Limhamn, Sweden) and 1 μg of RNA was reverse transcribed into cDNA using QuantiTect® Reverse Transcription Kit (Qiagen). Real-time PCR was performed in the ABI Prism 7000 instrument using SYBR® Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA) and predesigned primer sets (QuantiTect® primers, Qiagen) for rat Hif-1α, pancreatic and duodenal homeobox 1 (Pdx-1), B-cell lymphoma 2 (Bcl2), and 18S ribosomal RNA. Data were analyzed by ABI Prism 7000 SDS software. Results were expressed relative to the endogenous control 18S ribosomal RNA and then expressed as fold changes above the respective control samples in the same PCR. All experiments were repeated for validation.
INS-1E Cells
INS-1E cells were a kind gift from Dr. P. Maechler (Department of Cell Physiology and Metabolism, Centre Medical Universitaire, Geneva, Switzerland) (24). The cells were cultured in RPMI-1640 medium supplemented with 10% FBS, 10 mM HEPES, 100 U/ml penicillin, and 100 μg/ml streptomycin, 1 mM sodium pyruvate, and 50 μM β-mercaptoethanol at 37°C in a humidified atmosphere containing 95% air and 5% CO2.
Hypoxic Incubation and ATP Assay
Before ATP measurement, the cells were seeded in 96-well tissue culture plates at a density of 6 × 104 cells/well in serum-free RPMI-1640 with various exendin-4 concentrations (0, 0.1, and 10 nM). Triplicates were made in each condition. The plates were incubated for 20 h at 37°C under normal conditions for cell culture, and then the medium was changed to serum-free medium containing the same concentration of exendin-4. The plates were then placed in a sealed incubator chamber (Billups-Rothenberg, Del Mar, CA, USA) filled with 1% O2, 5% CO2, and 94% N2 at 37°C as hypoxic conditions. ATP assays were performed on cells after the hypoxic incubation using an ATP bioluminescent somatic cell assay kit (Sigma-Aldrich) according to the manufacturer's instructions.
Statistics
Data are shown as means ± SD. When blood glucose levels and body weight gain were compared between the four groups, an analysis of variance followed by the Bonferroni posttest was used. The unpaired Student's t-test was used when two groups were compared. The proportions of animals with a graft function in each group were compared using the two-tailed Fisher's exact test. The differences were considered as statistically significant if values were p < 0.05.
Results
Islet Preculture and Recipient Treatment with Exendin-4 Improves the Metabolic Outcome of Free and Macroencapsulated Islet Grafts
Free Islet Grafts
To compare the individual effect of islet preculture and recipient treatment on free islet graft outcome, three different treatment protocols of exendin-4 were used in this study. The islet preculture with exendin-4 alone (group 2) did not improve the graft outcome in comparison to controls (28% vs. 26%, ns). Of the recipients treated with exendin-4 (group 3), 54% had functional grafts as compared to 26% of the controls (ns). Only the combination of islet preculture and exendin-4 treatment of mice (group 4) resulted in a significantly better graft outcome (73% vs. 26% in controls, p = 0.03) (Table 2). Mice given combined exendin-4 treatment gained significantly more body weight than untreated controls (3.2 ± 1.7 vs. 1.5 ± 2.1 g, p = 0.04) (Table 2).
Effect of Different Exendin-4 Treatment Protocols on Functional Outcome of Free Islet Grafts

Graft function: cure + partial function. For definition, see Materials and Methods.
In the IPGTT, all the cured animals (except one in group 2) showed a quick recovery of blood glucose comparable to the healthy mice.
Macroencapsulated Islet Grafts
The effect of islet preculture and recipient treatment with exendin-4 was examined in a macroencapsulated islet transplantation model. The metabolic outcome was improved significantly in combined treatment group (Table 3). Three weeks after transplantation, 100% of animals in the treated group had functioning grafts compared to 20% in the control group (p = 0.02). The treated animals also gained significantly more weight than their untreated counterparts (3.0 ± 1.5 vs. −1.0 ± 2.1 g, p = 0.01) during the 4-week follow-up period. Daily nonfasting blood glucose levels in the two groups are shown in Figure 1A. In the exendin-4-treated group, blood glucose began to decline 14 days posttransplantation and was significantly lower than controls after 3 weeks. All cured animals showed a response comparable to that of healthy mice in the IPGTT (Fig. 1B).

(A) Nonfasting blood glucose after transplantation of a marginal number (500) of macroencapsulated islets in exendin-4-treated mice (n = 6, diamonds) and untreated controls (n = 5, squares). *p < 0.05. (B) Blood glucose concentrations during intraperitoneal glucose tolerance test (IPGTT) in exendin-4-treated mice transplanted with macroencapsulated islets (n = 6, open circles) and healthy nondiabetic mice (n = 4, closed squares).
Effect of Exendin-4 Treatment on the Functional Outcome of Macroencapsulated Islet Grafts

Graft function: cure + partial function. For definition, see Materials and Methods.
Larger Endocrine Cell Volume in Exendin-4-Treated Group Than in Control Group
Total Insulin Content of the Free Islet Graft 4 Weeks After Transplantation
We assessed the total insulin content of islet graft in groups 1 and 4 to further investigate beneficial effect of combined exendin-4 treatment on islet survival. As shown in Figure 2, the group given combined exendin-4 treatment (group 4) showed a significantly greater insulin content (650 ± 466 ng/kidney, n = 9) than the control group (220 ± 148 ng/kidney, n = 9) (p < 0.05).

Insulin content of graft-bearing kidneys at 4 weeks after transplantation, control versus exendin-4-treated groups (n = 9 in each group). *p < 0.05.
Macroencapsulated Islet Graft Volume
The absolute volumes of different tissues of the macroencapsulated grafts were estimated using serial sections of paraffin-embedded grafts 1 month after transplantation. One example of the histology from each group is shown in Figure 3A (control) and 3B (exendin-4 treated). Consecutive sections were stained for insulin and glucagon (Fig. 3C, D, respectively). In the control device (Fig. 3A), most of the endocrine tissue was necrotic with condensed nuclei and broken cells (Fig. 3C). Only a few cells were stained positive for insulin (Fig. 3C, red color). In the device from an exendin-4-treated animal the majority of the cells were insulin positive with normal nuclei (Fig. 3D, red color), and a single glucagon-positive cell was found (green color, white arrow). All insulin stained sections were carefully compared to adjacent sections stained by H&E, confirming that the majority of the endocrine cells were insulin positive.

(A, B) Hematoxylin and eosin staining of macroencapsulated islet grafts 4 weeks after transplantation. In the control group (A), black arrows indicate necrotic area with condensed nuclei and broken cells. In the exendin-4-treated group (B), blue arrows indicate viable endocrine tissue with round nuclei and a homogenous shape of cells. The star indicates the inner membranes. Original magnification: 40x. (C, D) Immunofluorescence staining for insulin (red color, indicated by yellow dotted arrows) and glucagon (green, indicated by white arrow), consecutive sections to (A) and (B), respectively. Original magnification: 20x. (E) Absolute volumes of endocrine tissue (stripes), fibrous tissue (white), necrotic tissue (black), and others (dots) in macroencapsulated islet grafts 4 weeks after transplantation compared between the two groups (n = 5 each group). Error bars indicate SD of total volumes.
The total tissue volumes inside the devices and the volumes of fibrotic tissue were similar in the exendin-4-treated and control groups (total volume 0.89 ± 0.36 vs. 0.83 ± 0.28 mm3 and fibrotic tissue volume 0.54 ± 0.25 vs. 0.39 ± 0.19 mm3). The volumes of endocrine tissue were greater in the exendin-4-treated group than in the control group (0.25 ± 0.11 vs. 0.10 ± 0.04 mm3, p < 0.05), and the volumes of necrotic tissue were less (0.02 ± 0.02 vs. 0.23 ± 0.15 mm3, p < 0.05, respectively). Other structures within the tissue inside the device (e.g., small cystic structure) were categorized as “others” (exendin-4-treated group 0.08 ± 0.05 mm3 vs. control group 0.09 ± 0.05 mm3, ns). Results are summarized in Table 4 and Figure 3E.
Effect of Exendin-4 Treatment on Tissue Volumes of Macroencapsulated Islet Grafts at 4 Weeks Posttransplantation

One graft was damaged during fixation and was therefore excluded from the graft volume analysis. ns, not significant.
The numbers of vascular profiles within 0.1 mm from membrane surfaces were significantly higher in the exendin-4-treated group than in the control group (221 ± 67 vs. 90 ± 70, p < 0.05) (Fig. 4A). A higher number of vascular profiles was observed close to the membrane surface in the areas with viable endocrine cells. One example from each group is shown in Figure 4B and C.

(A) Numbers of vascular profiles around the device are compared between two groups (n = 5 control group, n = 6 exendin-4-treated group). *p < 0.05. (B, C) H&E-stained picture from a membrane in each group (control and exendin-4-treated groups, respectively). Arrows indicate vascular profiles.
Mechanisms of Exendin-4 Effects on Rat Islets in Culture
Protein Expression of HIF-1α and Active Caspase-3
The mechanisms behind the beneficial effects of exendin-4 treatment were investigated in vitro using rat islets cultured for 20 h in various exendin-4 concentrations. We compared HIF-1α expression in the islets cultured with exendin-4, using TOPO1 as a loading control. As shown in Figure 5A, exendin-4 enhanced HIF-1α expression in isolated rat islets compared to the control group. Figure 5B shows the relative expression of active caspase-3/β-actin with different exendin-4 concentrations. Expression of proapoptotic protein, active caspase-3 was reduced in the exendin-4-treated group.
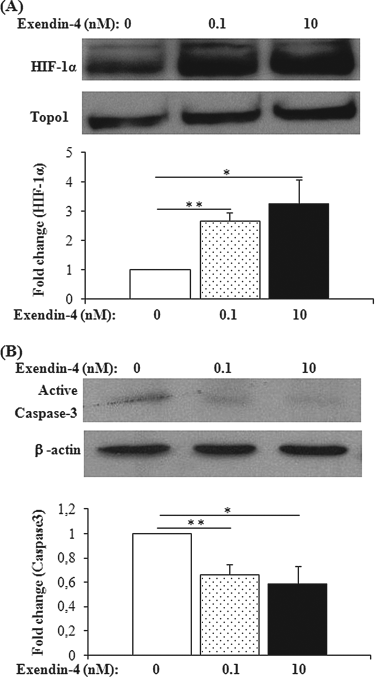
Western blot analyses assessing hypoxia inducible factor (HIF)-1α (A) and active caspase-3 (B) expression levels with/without exendin-4 treatment. The upper part of each panel shows specific bands of indicated proteins, whereas the lower part shows the fold change of target protein level/loading control protein level compared to the respective control samples (n = 6 in each group). **p < 0.01, *p < 0.05.
Detection of Hif-1α, Pdx-1, and Bcl2 mRNA Expression by Real-Time PCR
The mRNA expression levels of Hif-1α, Pdx-1, and Bcl2 were determined in order to study the proliferative and antiapoptotic effect of exendin-4 on cultured islets, using real-time PCR. Hif-1α mRNA increased in exendin-4-treated groups (Fig. 6A). Islets cultured with 10 nM exendin-4 showed significantly higher expression of Hif-1α mRNA than control islets (fold increase 6.70 ± 0.59, p < 0.05). Pdx-1 mRNA, which encodes a proliferation and differentiation factor for pancreatic β-cells, was enhanced by exendin-4 treatment (fold increase 0.1 nM exendin-4: 2.54 ± 0.47, p < 0.05 and 10 nM exendin-4: 3.15 ± 0.37, p < 0.05) (Fig. 6B). The mRNA expression of antiapoptotic gene Bcl2 was increased by exendin-4. However, a significant increase was found only in 0.1 nM exendin-4-treated islets (fold increase 2.93 ± 0.36, p < 0.05) (Fig. 6C) due to high variation in gene expression in 10 nM exendin-4-treated islets.

Real-time PCR analysis of Hif-1α (A), pancreatic and duodenal homeobox 1 (Pdx-1) (B), and B cell lymphoma 2 (Bcl2) (C) gene transcripts in isolated islets with/without exendin-4 treatment. Fold changes of target gene mRNA level/the endogenous control mRNA level are compared to the respective control samples (n = 3–6). *p < 0.05.
ATP Production in INS-1E Cells Under Hypoxic Conditions
To investigate the effect of exendin-4 on energy production in islets, the amount of ATP produced in INS-1E cells was determined following subjection to hypoxia and nutrient depletion. The cells cultured under hypoxic conditions for 2 h with exendin-4 had more ATP than cells cultured without exendin-4. Using ATP content of control cells as a baseline, ATP content was increased when cells were cultured with 0.1 nM exendin-4 (fold increase: 1.73 ± 0.13, p < 0.05, n = 8) and 10 nM exendin-4 (fold increase: 1.49 ± 0.12, p < 0.05, n = 8) (Fig. 7).

ATP content in rat insulinoma (INS-1E) cells. Fold changes in ATP content (ATP in exendin-4-treated cells/ATP in nontreated control cells) were calculated and means of eight experiments ± SE are shown. *p < 0.05.
Discussion
This study further supports a beneficial effect of exendin-4 in islet transplantation. Islet preculture with exendin-4 followed by a short-term recipient treatment improved the metabolic outcome when a marginal mass of islet graft was transplanted under the kidney capsule. Similar effects of exendin-4 treatment were observed when islets were transplanted in an immunoprotective device subcutaneously. In the exendin-4-treated group, endocrine cell volume in the macroencapsulated grafts was much higher, indicating that the positive metabolic effect at least partly was related to improved β-cell survival. A protective effect of exendin-4 for islets under hypoxic conditions was supported by higher HIF-1α expression and lower active caspase-3 expression seen in islets cultured with exendin-4. Exendin-4-treated islets contained higher levels of HIF-1α, Pdx-1, and Bcl2 mRNAs. Furthermore, exendin-4-treated INS-1E cells produced more ATP than control cells under hypoxic conditions, indicating that exendin-4 increases glycolysis and thereby improves energy production.
Islet culture prior to transplantation provides a possibility to optimize islet graft viability and function. Before the vascularization in the grafts is established, islets are damaged by severe inflammation and hypoxia. A previous in vitro study indicates that the administration of exendin-4 can partially protect β-cells from cytokine-induced apoptosis and necrosis (22). Our in vitro results also show that a 20-h incubation of islets with exendin-4 improved islet viability. In the in vivo study, however, we observed that only the preculture of islets with exendin-4 (without recipient treatment) did not improve metabolic control after islet transplantation. We showed that short-term recipient treatment with exendin-4 improved metabolic control after islet transplantation when evaluated a few days after the discontinuation of the treatment (35), although this study showed that this positive effect was not sufficient to improve graft outcome at 4 weeks after transplantation. A combination of in vitro and short-term posttransplantation exendin-4 treatment was required to improve metabolic control significantly for longer periods after islet transplantation. We propose that the antiapoptotic effects of exendin-4 initiated in vitro may be complemented by a direct effect of exendin-4 on transplanted grafts as well as indirect effects of exendin-4 on the recipient mice such as glucose-dependent stimulation of insulin secretion (1) and the suppression of glucagon secretion (8).
Four weeks after transplantation, renal subcapsular grafts in the exendin-4-treated animals were easily identified macroscopically, indicating a larger surviving islet graft mass. In the nontreated animals, the grafts were usually invisible. The higher insulin content of the graft-bearing kidney in the combined treatment group also indicates a better maintained β-cell mass in the presence of exendin-4 and suggests that this effect is related to improved β-cell survival. This hypothesis is further supported by our findings derived from the experiment in which combined exendin-4 treatment was evaluated in macroencapsulated islets. The majority of endocrine cells were positive for insulin, while only a few cells were glucagon positive.
The majority of the encapsulated cells are lost early after implantation probably due to the poor physiological milieu. Hypoxia, malnutrition, and inflammatory responses around the devices are thought to be major factors contributing to failure of macroencapsulated islets (9). Although these factors improve with time after implantation, β-cell mass lost in the early posttransplantation period does not recover even after the device is revascularized (7). We have shown that full vascularization of these devices takes approximately 3 months (30) and that preimplantation of the devices allowing vascularization to occur before transplantation, is one way to reduce the curative dose of islets (30,37). While a freshly loaded and implanted device would prevent islets from immune damage, it also worsens hypoxia and nutrient deprivation. It was our goal to determine how much of this damage could be reduced using exendin-4 treatment in this model. In our study, cure rates were higher in the group treated with exendin-4 than in untreated controls. By using exendin-4 treatment, cure was achieved by transplanting only 500 encapsulated islets compared to the 1,000 encapsulated islets usually required in this diabetic mouse model using the Theracyte Device (39). A significant reduction in islet requirements by 50% supports the continued use of exendin-4 in clinical islet transplantation.
We also found that the volume of endocrine cells in grafts in exendin-4-treated animals was twice as large as that in the control group. The number of vascular profiles around the devices was increased in the exendin-4-treated group and seemed related to the amount of endocrine tissue within the section. The initial treatment with exendin-4 may well have conferred the β-cell resistance to injury during this period. Viable β-cells or the intraislet endothelial cells that have been shown to be involved in promoting graft vascularity (4,21,23) could produce VEGF (vascular endothelial growth factor) possibly stimulated by HIF-1 (see below) and induce vascularization of the device surface (5,17). In contrast to our results, exendin-4 was unable to show any effects on graft outcomes in a recent study of syngeneic microencapsulated mouse islets (3). The differences in types of encapsulation, sites of transplantation, exendin-4 treatment protocols, and donor species may have contributed to the diversity in results.
Under hypoxic conditions, a number of genes that are essential for cell survival are upregulated by HIF-1 (44,46). Our in vitro study showed increased mRNA and protein expression of HIF-1α in rat islets cultured with exendin-4. A simultaneous decrease in the protein expression of the proapoptotic enzyme caspase-3 was also observed. The antiapoptotic gene Bcl2 and a β-cell differentiation factor encoded by Pdx-1 were increased on the mRNA expression level in the exendin-4-treated islets compared to control islets. These findings indicate that HIF-1 upregulation by exendin-4, in concentrations used here, confers a protective effect in islets. Two recently published studies support the importance of HIF-1α molecule for islet function. One showed that even the low levels of HIF-1α expression present in normoxia is necessary to maintain β-cell function (6) and the other showed that the induction of HIF-1α/VEGF by desferrioxamine could improve microencapsulated islet graft function (43).
The degree of hypoxia varies within the islets and is more severe in cells located in the core of the islets. Thus, intact islets are not an ideal model for studies investigating the molecular mechanisms of exendin-4 in hypoxic β-cells. Therefore, we used INS-1E cells to study the effects of exendin-4 on energy production in insulin-producing cells in this study. The cells treated with exendin-4 in culture showed more ATP after short-term hypoxia (2 h) compared to control cells. This result suggests that the treated cells may have a better energy production. The improved energy production and associated HIF-1 expression may be the mechanism by which exendin-4 induces its beneficial effects on pancreatic β-cells.
When clinical protocols using exendin-4 in islet transplantation are being designed, it is important to discuss the optimal treatment period. In recent clinical trials, exenatide treatment combined with tumor necrosis factor (TNF) receptor antagonist, etanercept, from the time of initial islet transplantation showed promising effects for engraftment and long-term graft survival (12–15). However, these studies do not clarify whether exenatide alone may have any beneficial effect on outcome of islet transplantation. Furthermore, exenatide treatment of islet recipients who received a marginal mass of grafts or had progressive graft dysfunction did not restore the islet graft function (15,16). In all cases, nausea and vomiting were major side effects of exendin-4 therapy that limited patient compliance (14,16). Our data from this rodent study indicate that a shorter initial treatment may be of value to preserve islet graft mass. In conclusion, the pretreatment of islets in culture medium with exendin-4 followed by recipient treatment during the crucial first week improves the survival of both free and encapsulated islets. This study has indicated for the first time that the protective effects of the GLP-1 receptor agonist exendin-4 may be mediated via the HIF-1 pathway.
Footnotes
Acknowledgments
We thank Anneli Hansson for excellent technical assistance and Dr. Ganeshkumar Munnorcode for statistical advice. Financial support was provided through the regional agreement on medical training and clinical research (ALF) between Stockholm County Council and Karolinska Institutet (project number 20060710, 20080443), Swedish Research Council (52120083389), research grants from Karolinska Institute, the Foundation for International Surgical Cooperation, the Foundation for Oncological Cooperation and Barndiabetesfonden (2007, 2008, and 2009). The macroencapsulation devices used for this study were kindly provided by TheraCyte, Inc., Laguna Hills, CA, USA. The authors declare no conflicts of interest.
