Abstract
Delayed and insufficient revascularization during islet transplantation deprives islets of oxygen and nutrients, resulting in graft failure. Vascular endothelial growth factor (VEGF) could play a critical role in islet revascularization. We aimed to develop pharmacological strategies for VEGF overexpression in pancreatic islets using the iron chelator deferoxamine (DFO), thus avoiding obstacles or safety risks associated with gene therapy. Rat pancreatic islets were infected in vivo using an adenovirus (ADE) encoding human VEGF gene (4.108 pfu/pancreas) or were incubated in the presence of DFO (10 μmol/L). In vitro viability, functionality, and the secretion of VEGF were evaluated in islets 1 and 3 days after treatment. Infected islets or islets incubated with DFO were transplanted into the liver of syngenic diabetic rats and the graft efficiency was estimated in vivo by measuring body weight, glycemia, C-peptide secretion, and animal survival over a period of 2 months. DFO induced transient VEGF overexpression over 3 days, whereas infection with ADE resulted in prolonged VEGF overexpression lasting 14 days; however, this was toxic and decreased islet viability and functionality. The in vivo study showed a decrease in rat deaths after the transplantation of islets treated with DFO or ADE compared with the sham and control group. ADE treatment improved body weight and C-peptide levels. Gene therapy and DFO improved metabolic control in diabetic rats after transplantation, but this effect was limited in the presence of DFO. The pharmacological approach is an interesting strategy for improving graft efficiency during transplantation, but this approach needs to be improved with drugs that are more specific.
Keywords
Introduction
The success of clinical islet transplantation for curing patients with type 1 diabetes mellitus is limited, despite recent progress made with improved islet isolation or immunosuppressive regimens (35,39–41,43). The widespread application of this attractive alternative to insulin therapy or pancreas transplantation is limited by donor organ shortage and the requirement of two to three donor pancreata per recipient to achieve insulin independence. In fact, reports demonstrated that only a small fraction of transplanted islets successfully engraft, results confirmed by experimental islet transplantation where 50–70% of the transplanted islets are lost in the immediate posttransplantation period (10,37,40). A main challenge for long-term insulin independence following islet transplantation in type 1 diabetes is occult hypoxic, inflammatory, and immune graft loss (11,41,44). Anoxia from initial avascularity of the islet is particularly stressful to islet functions in a hyperglycemic context of a diabetic recipient (4,10). In fact, native islets in the pancreas have a rich glomerular-like vascular system that consists of fine capillaries supplied by one to five feeding arterioles (30,32). Such a rich microvasculature in pancreatic islets provides efficient delivery of oxygen and nutrients to islet cells and at the same time ensures a rapid dispersal of pancreatic hormones into circulation. However, islets are avascular after isolation process. For this reason, after transplantation, the survival and function of islets must rely on the reestablishment of new vessels in the grafts, to derive blood flow from the host vessel system (2,50). There is evidence that freely transplanted islets are associated with significantly lower islet revascularization than native islets in the pancreas, and that this problem occurs irrespective of whether islets are transplanted intraportally in the liver (25). Thus, it is critically important to maintain adequate microvascular perfusion of oxygen and nutrient supplies to islets.
However, adequate microvascular perfusion to islets does not resume immediately after islet transplantation. It can take up to between 7 and 10 days for intragraft microvessels to form after islet transplantation; moreover, the reestablishment of intragraft blood perfusion can take even longer (>14 days) (18,50). This delay in reestablishing functional microvasculature in newly grafted islets starves islet cells of oxygen and nutrients. Indeed, several studies have shown that newly transplanted islets are hypoxic, causing islet cells to undergo apoptosis and/or necrosis, which affects the loss of functional β-cell mass posttransplantation (18,50). In fact, despite transplanting a large number of islets (11,000 islets/kg body weight) per diabetic recipient, only about 30% of transplanted islets become stably engrafted, corresponding to a total loss of about 70% of the functional islet mass in the early posttransplantation phase (2).
The molecular mechanisms that underlie pancreatic islet revascularization remain poorly understood, but seem to be a complex multistep process regulated by several angiogenic factors (42,50,51). Among these factors, vascular endothelial growth factor (VEGF) appears to be the most potent growth factor regulating blood vessel growth (15,52). Indeed, VEGF acts in the early phase to stimulate the formation of new blood vessels from preexisting ones (52); thereafter, it initiates the revascularization of islets during transplantation (21). VEGF is mitogenic for endothelial cells in various in vitro and in vivo systems (5,8), increases permeability of microvessels (22), and is expressed in islets (49). However, VEGF expression in islet cells is transiently induced, followed by significant decline 2–3 days after transplantation (50,51). Moreover, there is an impaired angiogenesis of islet grafts in diabetic patients, which contributes to the absence of adequate islet revascularization under hyperglycemic conditions. Thus, rapid and adequate islet revascularization would protect islet grafts from hypoxia-induced inflammation and necrosis, thereby improving long-term graft survival and providing better preservation of functional mass. Sigrist et al. (46) demonstrated that local VEGF delivery to islet grafts improved the outcome of islet transplantation by enhancing islet revascularization. To improve islet graft vascularization, other studies have delivered human VEGF cDNA by adenoviral gene transfer to mouse islets, followed by transplantation under the renal capsule in streptozotocin-induced diabetic mice (53) and provided the proof-of-principle that VEGF overexpression stimulated graft angiogenesis and augmented islet revascularization. Moreover, VEGF increased functional β-cell mass by improving angiogenesis of isolated human and murine pancreatic islets (22). Therefore, exploring strategies of VEGF overexpression in pancreatic islets, to improve angiogenesis and graft function, may prove interesting.
We aimed to develop strategies of VEGF overexpression in rat pancreatic islets using gene therapy, with an adenovirus encoding the VEGF 165 gene, or a pharmacological approach with deferoxamine (DFO), an iron chelator used in the treatment of acute iron poisoning, chronic iron or aluminium overload, and diseases like thalasemia (29). Previous in vitro studies have shown that these two approaches allowed an efficient overexpression of VEGF in the β-cell line RINm5F and in pancreatic islets (23,24,48). However, the adenoviral infection was proapoptotic and amounts of insulin mRNA in β-cell lines (24) were lower than with DFO, which preserved β-cell line RINm5F viability and presented an antiapoptotic effect (24). Therefore, these data obtained in vitro in β-cells have suggested that DFO could be a new alternative for increasing VEGF overexpression and improving islet revascularization during transplantation.
Materials and Methods
Animals
Experiments were performed in accordance with the principles and guidelines of the French legislation on animal welfare: Journal Official number 87–848. Wistar and Lewis rats were supplied by Janvier laboratory (Le Genes St Isle, France). Rats were housed in standard collective cages, in a temperature-controlled room (23 ± 1°C) with a 12-h light:12-h darkness cycle. They were fed with SAFE-A04 (Villemoisson-sur-Orge, France). Food and water were available ad libitum.
In Vitro Study
Islet Isolation and Purification
Pancreatic islets were isolated from 250–300 g male adult Wistar rats for the in vitro study and from syngeneic Lewis rats for the in vivo study, to sustain immune reactions. The islet isolation procedure was performed according to Sutton et al. (47). Then islets were cultured at 37°C in humidified air with 5% CO2 in 48-well culture plates (CELL-STAR®, Greiner Bio-one, Kremsmünster, Austria) for viability, functionality, and VEGF secretion studies, or in 60 × 15-mm culture dishes (Becton Dickinson, Meylan, France) for mRNA and protein extraction. Islets were cultured in medium M199 (Lonza) supplemented with 10% heat-inactivated fetal calf serum (FCS, Cambrex Bio Science, Verviers, Belgium), 1% of a mixture of penicillin (10,000 UI/ml), streptomycin (10 mg/ml), and amphotericin B (25 μg/ml) (AB/AM, Sigma).
Adenoviral Infection
Two adenoviruses (Ad VEGF and Ad β-galactosidase, Cell Biolabs, San Diego, CA, USA) were used to infect rat pancreatic islets. The adenoviral vector, Ad VEGF, was built from human adenovirus serotype 5 deleted for the E1 and E3 gene. Ad VEGF contained an expression including the mouse VEGF 164 gene. The viruses produced were thus non-replicative.
Rat pancreatic islets were infected in vivo to spread the infection to all islet cells as briefly described below. After distending the pancreatic duct with Hank's solution (Lonza, Saint Beauzire, France) the pancreas was isolated from other organs by several ligatures on the spleen, stomach, lung, and duodenum. Adenoviral preparation [100 multiplicity of infection (MOI)] was injected under the portal vein. The pancreas was then removed and placed in Hank's solution at 4°C. After 45 min, islets were purified after collagenase digestion.
The percentage of infected islets was determined using a β-galactosidase reporter gene staining kit (Sigma) 1 and 3 days after infection.
Deferoxamine
Deferoxamine mesylate (Sigma) was solubilized in ultrapure water at 100 mmol/L. Islets were incubated in 10 μmol/L of DFO diluted in culture medium for 1 day and 3 days.
Islet Viability
The current standard method, involving fluorescein diacetate/propidium iodide (FDA/PI, Sigma), was used for assessing islet viability. Ten islets for each condition were randomly selected by two independent investigators. The ratio between green cells and red cells provides the percentage of viability.
Islet Functionality
One and 3 days after treatment, 10 islets from each experimental condition were washed extensively and incubated in Krebs Ringer bicarbonate (KRB) solution with 0.5% FCS and 2.5 mmol/L glucose (Sigma). Islets were then stimulated with KRB solution containing 0.5% FCS and 25 mmol/L glucose. Each incubation step was performed for 90 min at 37°C in humidified air with 5% CO2. Supernatants were collected after each incubation and stored at −20°C. Insulin measurements were performed using a rat insulin enzyme linked immunosorbent assay (ELISA) kit (Mercodia, Uppsala, Sweden). The stimulation index was calculated as a ratio of stimulated insulin secretion to basal insulin secretion. Results are representative of three separate experiments.
Study of Apoptosis by Evaluating Bax/Bcl-2 mRNA Expression Using RT-PCR
After 1 and 3 days of treatment, total cellular RNA was extracted from islets using Trizol Reagent (Sigma) and converted to cDNA using Moloney murine leukemia virus (M-MLV) reverse transcriptase and random hexamers (Fermentas GmbH, St. Leon-Rot, Germany). Quantification of β-actin, Bax, and Bcl-2 mRNA was performed according to Langlois et al. (24 Enzyme Linked Immunosorbent Assay) and was given as the ratio of integrated optical density with Bax or Bcl-2 to that with β-actin.
VEGF Protein Secretion
The culture medium was collected 1 and 3 days later and was stored at −20°C. VEGF measurements were performed using the rat VEGF elisa kit (Tebu-bio, Le Perray en Yvelines, France) for the pharmacological approach and the human VEGF elisa kit (Bender MedSystems, Austria) for the infection approach. Total protein concentration was quantified according to Henkel and Bieger (16). The results were expressed in pg/μg of total protein.
Hypoxia-Inducible Factor-1α, (HIF-1α,) Analysis Using Western Blotting
Protein extract was obtained from 200 islets and Western blotting was performed according to Langlois et al. (24). HIF-1α was studied using an anti-rat HIF-1α monoclonal antibody (1/1000, R&D Systems, Minneapolis, NM, USA). The integrated optical density of bands was quantified using the ImageJ v.1.34 software and normalized against β-actin (anti-mouse β-actin monoclonal antibody, 1/5000, Santa Cruz Biotechnology Inc., CA, USA).
Immunohistochemistry of Insulin
Immunohistochemical staining was performed on paraffin-embedded sections using the extravidin-biotin-peroxydase method (Sigma), with monoclonal antibodies specific for insulin (Sigma), according to Maillard et al. (28). Appropriate positive and negative controls were used. Analysis involved about 20 islets per condition.
In Vivo Study
Islet Transplantation
Islet transplantation was carried out in diabetic male Lewis rats weighing about 200 g and was induced by a single intraperitoneal injection of streptozotocin (75 mg/ml, Sigma). Animals were considered diabetic after two consecutive blood glucose measurements of ≥ 3 g/L using the Glucose RTU® test (Biomérieux SA, Craponne, France). Immunohistology of the pancreas of streptozotocin rats confirmed that no insulin secreting cells are viable. This study consisted of four animal cohorts: diabetic rats that underwent a laparotomy and an intraportal injection of CMRL (Gibco, Illkirch, France) (sham group), transplanted diabetic rats (control), transplanted diabetic rats with infected islets (ADE), and diabetic rats transplanted with islets preincubated with 10 μmol/L DFO. Before transplantation, untreated and infected islets were incubated in medium M199 + 10% FCS + 1% AB/AM for 1 day and islets subjected to DFO treatment were incubated for 3 days. After the incubation, 1,000 islet equivalents per kilogram of body weight were counted, washed, and prepared in 1 ml of CMRL before intraportal transplantation.
Metabolic Follow-up
Metabolic control was monitored for 60 days after transplantation. Before each measurement, rats were fasted overnight. Animal survival and body weight (expressed in grams) were first assessed. Then graft efficiency was determined using the ratio between fasting C-peptide level and glucose level (pmol of C-peptide/g of glucose). Blood fasting glycemia, measured using the glucose RTU® test, was expressed in g/L, and C-peptide levels in plasma using a rat C-peptide ELISA kit (Mercodia) were expressed in pmol/L.
Statistical Analysis
Statistical in vitro analysis was performed with Sigmastat 3.10 (Systat Software, Richmond, CA, USA). Levels of significance for influence of treatments on pancreatic islets and for influence of the duration of experiment were assessed by least significant difference ANOVA test with a post hoc Bonferroni correction. t-Test was performed to assess the significance of influence of DFO treatment on HIF-1 expression. Then, for the in vivo study, repeated data on body weight and the ratio C-peptide/glycemia were analyzed using linear mixed models with SAS software. Variance-covariance models were used and the choice of the best-fitting variance-covariance matrix was based on the AIC goodness of fit measure. Data are reported as mean ± SD. The influence of the duration of the experiment and that of the treatment are represented, respectively, with # and *. Finally, values of p < 0.05 were considered significant.
Results
Adenoviral Infection Efficiency
Islet infection with adenoviral vectors proved efficient for β-galactosidase transduction (Fig. 1). The quantification of adenoviral infection after X-gal labeling showed that 90% of islet cells were infected with 100 MOI (Fig. 1A) after 1-day treatment. This percentage was maintained after 3 days (Fig. 1B). However, only 20% of islet cells were infected using the ex vivo method and the labeling was localized to the periphery of islets.
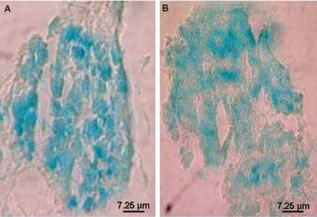
Adenoviral infection efficiency in rat pancreatic islets using β-galactosidase staining on sections of 4 μm, 1 day (A) and 3 days (B) after infection with 100 multiplicity of infection (MOI) of adenovirus.
Evaluation of Islet Viability and Functionality
Islet viability was preserved with 10 μmol/L DFO treatment (Fig. 2A, c and d) or adenoviral infection (Fig. 2A, e and f) compared with control (Fig. 2A, a and b) after 1 day (90.82 ± 6.05% with DFO, n = 11; 93.00 ± 8.19% after adenoviral infection, n = 8; vs. 92.09 ± 6.83%, n = 11 (Fig. 2A, a, c, e) and 3 days of culture (91.38 ± 6.95% with DFO, n = 8; 89.71 ± 8.30% after adenoviral infection, n = 7; vs. 87.86+11.50%, n = 7 (Fig. 2A, b, d, f).
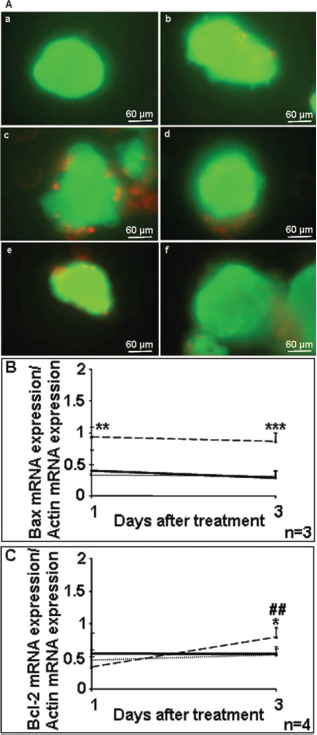
Analysis of rat pancreatic islet viability. (A) Fluorescein diacetate/propidium iodide (FDA/PI) staining of control islets (a, b), deferoxamine mesylate (DFO) 10 μmol/L (c, d), or adenovirus (ADE) 100 MOI (e, f) 1 day (a, c, e) and 3 days (b, d, f) after culture. (B) Bax and Bcl-2 and (C) mRNA amounts in control (solid line), DFO 10 μmol/L (dotted line), and ADE 100 MOI (dashed line) conditions after 1 and 3 days of treatment. Results are expressed as mean ± SD. The influence of the duration of the experiment (#) and that of the treatment (*): ***p < 0.001; **,##p < 0.01; *p < 0.05.
Amounts of proapoptotic (Bax, Fig. 2B) factor mRNA were significantly greater in infected islets than control and DFO-treated islets 1 day (0.93 ± 0.15, n = 4 vs. 0.40 ± 0.32, n = 5 and 0.33 ± 0.16, n = 3; p < 0.01) and 3 days after treatment (0.86 ± 0.14, n = 4 vs. 0.28 ± 0.11, n = 5 and 0.31 ± 0.09, n = 3; p < 0.001). mRNA amounts of the antiapoptotic factor Bcl-2 (Bcl-2, Fig. 2C) remained unchanged 1 day and 3 days after treatment in controls (from 0.54 ± 0.28 to 0.54 ± 0.17, n = 5) and DFO-treated islets (from 0.44 ± 0.15 to 0.52 ± 0.14, n = 4). However, Bcl-2 amounts were significantly greater after adenoviral infection in control and DFO-treated islets (n = 4; p < 0.05); however, levels of Bcl-2 remained lower than those of Bax 3 days after treatment.
An immunohistochemical study of insulin showed similar staining 1 day after treatment, whatever the experimental condition (Fig. 3A, a, c, e). However, insulin staining in control islets (Fig. 3A, b) and infected islets (Fig. 3A, f) decreased after 3 days of culture; necrotic areas were also visible, although these were greater in number in islets infected with ADE. Insulin staining and islet structure were maintained throughout the experiment for islets treated with DFO (Fig. 3A, c and d).

Evaluation of insulin secretion of rat pancreatic islets. (A) Insulin immunostaining of rat pancreatic islets 1 day (a, c, e) and 3 days (b, d, f) in control (a, b), DFO 10 μmol/L (c, d), or 100 MOI of ADE (e, f, insulin secreting cells appears in red) conditions.(B) Index of stimulation of rat pancreatic islets after 1 and 3 days of culture in control islets (black bar), after treatment using DFO 10 μmol/L (gray bar) or ADE (100 MOI) (lined bar). Results were expressed as mean ± SD. The influence of the duration of the experiment (#) and that of the treatment (*): ***,###p < 0.001; **p < 0.01.
We also investigated islet function (Fig. 3B): the stimulation index was significantly higher 1 day after culturing in the presence of 10 μmol/L DFO than that observed with control and ADE-infected islets, from 3.74 ± 0.58 to 1.96 ± 0.83 and 2.03 ± 1.06, respectively (p < 0.001). However, islet function for those treated with 10 μmol/L DFO decreased during the experiment to levels that were similar to control conditions after 3 days of culture (1.91 ± 0.22 vs. 2.02 ± 0.45, n = 6). Moreover, adenoviral infection induced a significant decrease in function 3 days after treatment (1.15 ± 0.13, n = 4; p < 0.01).
VEGF and HIF-1α Analysis in Islets
No difference was observed in VEGF secretion (Fig. 4A) between experimental conditions after 1 day of culture (control: 2.42 ± 1.09, DFO: 2.21 ± 1.84, ADE: 2.49 ± 1.25 pg/μg total protein, n = 3). However, 3 days after treatment, amounts of VEGF were significantly higher in DFO and ADE conditions than in control conditions [7.95 ± 0.84 pg/μg total protein for DFO (n = 3), 11.26 ± 0.82 for ADE pg/μg total protein (n = 3) vs. 1.80 ± 1.10 pg/μg total protein for control (n = 4); p < 0.001]. We measured the amount of HIF-1α, a transcriptional factor involved in VEGF expression, to understand the signaling pathway for VEGF overexpression with DFO. Western blotting analysis revealed significant amounts of HIF-1α in the presence of DFO after 3 days of treatment (1.51 ± 0.31 vs. 0.42 ± 0.20 in control, n = 3; p < 0.01) (Fig. 4B).

Analysis of vascular endothelial growth factor (VEGF) secretion in rat pancreatic islets (A) after treatment (black bar: control, gray bar: 10 μmol/L DFO, lined bar: ADE 100 MOI). (B) Analysis of hypoxia-inducible factor-1α (HIF-1α) protein expression by Western blotting 3 days after treatment (black bar: control, gray bar: 10 μmol/L DFO). Results were expressed as means ± SD. The influence of the duration of the experiment (#) and that of the treatment (*): ***,###p < 0.001; **,##p < 0.01; *p < 0.05.
Metabolic Assessment
Analysis of rat survival showed that the transplantation of islets treated with ADE or DFO did not induce animal death (n = 9), whereas 50% (3/6) and 18% (2/11) of rats died in the sham and control groups, respectively. However, no animal deaths were observed in the control group until 30 days after transplantation, whereas deaths were recorded as early as day 7 in the sham group (1/6).
Analysis of body weight over the course of the experiment (Fig. 5A) for each studied group showed an increase in body weight in controls (from 182 ± 11 g at 3 days to 200 ± 13 g at 60 days, n = 11), DFO (from 217 ± 6 g at 3 days to 247 ± 12 g at 60 days, n = 9), and ADE groups (from 200 ± 8 g at 3 days to 255 ± 19 g at 60 days, n = 9) compared with the sham group in which a decrease was observed (from 185 ± 6 g at 3 days to 158 ± 20 g at 60 days, n = 6). To confirm these results, mean rat body weights were studied for each group over the course of the whole experiment (Fig. 5B). The mean values were compared, showing that body weight measured in animals infected with ADE was significantly higher than in the other groups, with 212.28 ± 2.45 g (n = 9) versus 201.78 ± 2.50 g for control (n = 11; p < 0.05), 199.67 ± 2.83 g for DFO (n = 9; p < 0.01) and 193.63 ± 4.07 g for sham animals (n = 6; p < 0.001). No significant difference was observed between control, DFO, and sham groups.

Change in body weight after islet transplantation in diabetic rats. (A) Body weight in SHAM (open circle), control (filled square), DFO 10 μmol/L (open triangle), and ADE (filled diamond) groups. (B) Analysis of the means of body weight for each group during the experiment. Results were expressed as means ± SD (***p < 0.001; **p<0.01; *p < 0.05).
Finally, we evaluated the insulin secretion capacity of transplanted animals during a fasting period, by measuring the ratio C-peptide/glycemia. The analysis of insulin secretion during the experiment (Fig. 6A) showed that, whatever the treatment used, the ratio C-peptide/glycemia was twofold higher in transplanted animals than in sham animals immediately after transplantation [115.31 ± 62.50 for control (n = 7), 131.35 ± 31.43 for DFO (n = 5), and 147.27 ± 45.34 for ADE (n = 6) versus 41.39 ± 7.60 pmol of C-peptide/g of glucose (n = 6) after 3 days of study]. Before transplantation, the ratio measured in control, DFO and ADE groups was similar to that in sham rats. This improvement in insulin secretion was maintained throughout the experiment in DFO and ADE groups, with 113.27 ± 50.79 (n = 5) and 127.01 ± 19.99 pmol/g (n = 6), respectively, at 60 days. However, insulin secretion in control rats decreased to a similar level to that of the sham group 60 days after transplantation (52.24 ± 9.83 (n = 6) versus 36.89 ± 6.43 pmol/g (n = 6), respectively). Statistical analysis of the C-peptide/glycemia ratio for each group over the entire experiment (Fig. 6B) showed that insulin secretion was significantly greater in the ADE group (148.89 ± 10.59 pmol/g, n = 6) than in control (92.62 ± 10.34 pmol/g, n = 7; p < 0.01) and sham (51.89 ± 12.53 pmol/g, n = 6; p < 0.001) rats. Finally, improvement in graft function in the DFO group was significantly greater than that observed in the sham group (109.81 ± 11.79 vs. 51.89 ± 12.53 pmol/g, n = 5; p < 0.01), whereas no significant differences were obtained with control group.
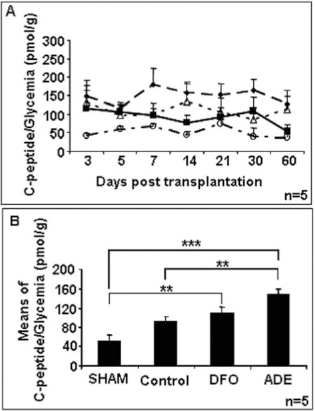
Change in insulin secretion after islet transplantation in diabetic rats. (A) Fasting ratio C-peptide/glycemia in sham (open circle), control (filled square), DFO 10 μmol/L (open triangle), and ADE (filled diamond) groups. (B) Analysis of the means of the fasting ratio C-peptide/glycemia for each group during the experiment. Results were expressed as means ± SD (***p < 0.001; **p < 0.01; *p < 0.05).
Discussion
The formation of new microvasculature by capillaries sprouting at the site of islet transplantation is crucial for the long-term survival and function of the graft (4,53). This study confirmed the key role of VEGF in the improvement of islet engraftment efficiency (3,46,53). Several authors have thus focused on developing strategies to overexpress VEGF in pancreatic islets using viral infection (6,7). However, VEGF is efficiently overexpressed by adenoviral infection (6,12), but there are several obstacles or safety risks associated with gene therapy, such as the transduction of unwanted cells, which is particularly relevant here, thus requiring fine-tuned transgene expression. A pharmacological approach in which DFO is used to enhance VEGF expression is more feasible for human use than gene therapy. However, the mechanism of DFO is a chelator of iron with a lot of metabolic effect into the metabolism. DFO mimics iron deficiency. Therefore, the utilization of DFO to enhance islet transplantation survival in vivo as a treatment is not relevant and the studies focus on the action of DFO in vitro before islet transplantation.
This in vitro study has confirmed that adenoviral infection represents an efficient process for overexpressing the VEGF gene in islets (9,26,33,45). However, it is essential to infect islets using an in vivo method, to ensure that the gene is present in all islets, as the efficiency of an ex vivo infection of islet cells enables only the transduction of peripheral cells (33,45). However, adenoviral infections were toxic, as shown by the loss of islet function and the destruction of cells in the core of islets, probably via an apoptosis process. These data confirmed results previously obtained in our laboratory on the β-cell line RINm5F (24). Consequently, the use of an adenoviral infection to overexpress VEGF may be deleterious for islet viability and function in vitro.
Therefore, the development of a pharmacological approach, using DFO, constituted a new and easy method for VEGF overexpression in all islet cells. In accordance with results previously obtained in β-cells (24), 10 μmol/L DFO preserved islet viability and stimulated islet function. We demonstrated that the presence of DFO increases VEGF release by islets, due to an increase of hypoxia characterized by overexpression of HIF-1α protein. Indeed, the expression of HIF-1α is regulated by prolyl hydroxylase (PHD) that requires molecular oxygen as substrate and iron as a cofactor to function (13). In normoxia, the alpha subunit of HIF-1 is a target for prolyl hydroxylation by HIF prolyl-hydroxylase, which makes HIF-1α a target for degradation by an E3 ubiquitin ligase, leading to quick degradation by the proteasome (17,19). Thus, the DFO, in chelating iron, prevents the proper functioning of the PHD and therefore the degradation of HIF-1α. Finally, the accumulation of HIF-1α stimulates the expression of VEGF and therefore angiogenesis.
The benefic effect of DFO is not only due to the improvement of islet revascul:arization. Vaithilingam et al. (48) have demonstrated that DFO treatment improved the outcome of encapsulated transplanted human islets with independence of the revascularization process. Indeed, the anti-oxydant (36) and the anti-inflammatory (31) properties of DFO are well described in the literature and could explain the cumulative benefic effect of DFO during free islet transplantation. However, although VEGF overexpression was similar between the DFO and ADE groups, we observed transient overexpression of VEGF, lasting only 3 days, in islets treated with DFO (Langlois et al., unpublished data). By contrast, using adenovirus, VEGF overexpression was maintained for 14 days (Sigrist et al., submitted). Several studies have demonstrated that the revascularization of islets occurs in the 7–10-day posttransplantation period. The transient overexpression period using DFO may then be insufficient to establish and maintain islet vascularization. However, it has been well documented that adenoviruses are characterized by complement activation, cytokine release, and subsequent vascular damage leading to a systemic inflammatory response (27,34,38), whereas DFO is known to protect cells against immune destruction (31), oxidative stress (36), and enhanced insulin internalization and biological actions (14,24), indicating that the pharmacological approach is less risky.
We first demonstrated that the transplantation of 1,000 islet equivalents per kg body weight into the liver of diabetic rats constituted a good model for engraftment, because this relatively low quantity of islets allowed the differentiation of treatment effects. A larger number of islets may have blurred treatment differences, with normoglycemia observed in all groups.
A study of rat survival, associated with glycemia findings, tended to confirm the beneficial effects islet transplantation, despite the eventual toxicity of ADE on metabolic control in type 1 diabetes. Moreover, DFO and adenoviral infection may improve the long-term efficiency of the graft, due to VEGF overexpression.
The increase in body weight in the adenoviral infection may be explained by greater insulin secretion than that observed with DFO treatment and standard transplantation, which is confirmed by the C-peptide/glycemia ratio. Body weight gain is a good marker for insulin secretion and action, occurring through its well-known and crucial role in glucose storage in muscle and liver cells and its role in increasing fatty acid synthesis (20). Thus, the gene therapy approach was significantly more efficient than the pharmacological approach.
Finally, this study confirmed the advantages of islet transplantation and the important role of VEGF in improving islet viability. Moreover, despite adenovirus toxicity, a prolonged overexpression of VEGF leads to the most efficient grafts. However, our pharmacological approach with DFO improved the metabolic control of diabetic rats after transplantation, but to a lesser extent than with gene therapy. Histological studies performed on the liver of the transplanted rat (data not shown) showed that while DFO induced short-term secretion of VEGF at the beginning of the study, adeno maintained this overexpression all along the experiment. Moreover, this result was confirmed with the immunohistochemistry of the vessels (lectin staining). Finally, functionality of islet using insulin staining showed that insulin secretion was maintained with DFO and ADE during the study while no insulin secreting cells are present in control.
A pharmacological approach inducing VEGF overexpression may be an interesting tool to improve graft efficiency. However, DFO may not be the best molecule to maintain successful islet transplantations long-term, as VEGF was not overproduced for the same duration as during adenoviral infection. Moreover, DFO acts via a hypoxia pathway. Therefore, it will be interesting to identify other molecules that sustain VEGF overexpression already in use in humans and which could then be easily applied to human islet transplantation with no safety risks. Other molecules overexpressing VEGF in islets and not employing a hypoxia pathway, like mersalyl (1), should be considered.
Footnotes
Acknowledgments
The authors declare no conflict of interest associated with this manuscript.
