Abstract
Although innate immunity plays important roles in xenograft rejection, there have been few studies on the role of toll-like receptors (TLRs) in xenotransplantation. Furthermore, most studies focused on the recipient's TLRs. Therefore, we investigated whether TLRs in porcine islets can contribute to islet xenograft rejection. Adult porcine islets were isolated and stimulated by polyinosinic/polycytidylic acid (poly I:C) or lipopolysaccharide (LPS). Both poly I:C and LPS stimulation in porcine islets induced expression of chemokines (RANTES, MCP-1, IP-10, and IL-8), cytokines (IL-6 and type I interferons), and adhesion molecules (VCAM-1 and ICAM-1). Porcine islet supernatants stimulated by TLR agonists induced chemotaxis of human leukocytes. They also induced procoagulant activation (tissue factor and fgl-2). However, TLR stimulation did not influence insulin secretion. When porcine MyD88 was knocked down using shRNA lentivirus, TLR-mediated induction of proinflammatory mediators and procoagulants was attenuated. When LPS was injected to MyD88 or TLR4 knockout mice after porcine islet transplantation, LPS stimulation on donor islets interfered with islet xenograft tolerance induction by anti-CD154 antibodies. Inflammatory cell infiltration and expression of proinflammatory chemokines and cytokines in islet xenografts also increased. In conclusion, TLR activation in porcine islets induced both a proinflammatory and procoagulant response and thereby contributed to xenograft rejection.
Introduction
Xenotransplantation with porcine islets has emerged as a promising treatment for diabetes mellitus (11,15,24). However, pig islets are subject to vigorous xenogeneic rejection, although the degree of rejection in islet xenotransplantation is much lower than in vascularized solid organ xenotransplantation (32,41). Islet xenografts are followed by a specific rejection response called an instant blood-mediated inflammatory reaction (IBMIR), when islet xenografts are exposed to human blood directly. IBMIR consists of a thrombocoagulation response and a proinflammatory response by innate immunity such as complements and innate immune cells (38). Following IBMIR, acute cellular rejection occurs in islet xenografts more aggressively than islet allografts (6,46). Overall, innate immunity plays important roles in both IBMIR and acute cellular rejection in islet xenograft rejection (3,16,52,53).
Toll-like receptors (TLRs) are important innate immune receptors that recognize pathogen-associated molecular patterns (PAMPs) or danger-associated molecular patterns (DAMPs) (1,2,26). Many exogenous and endogenous TLR ligands are released when islet grafts are isolated and transplanted (22,55). Surgical trauma and ischemia-reperfusion injury cause the release of endogenous TLR ligands (55), and perioperative infection makes exogenous TLR ligands (22). TLRs, particularly TLR4 respond to endogenous ligands, such as heat shock proteins (5,39) and heparan sulfate (27) released in damaged tissues (28,42). Endogenous nucleic acids from necrotic tissues have been found to activate immune responses through TLR3 (9,12,29). TLR stimulation leads to activation of either nuclear factor κ-light-chain-enhancer of activated B-cells (NF-κB) or type I interferon (IFN). The final products of TLR signaling pathways are proinflammatory cytokines and chemokines (10,45). In addition, stimulation of TLR2 or TLR4 can induce a coagulation response (14,33,49).
Expression and roles of TLRs in parenchymal cells such as epithelial cells and endothelial cells have been demonstrated as well as antigen-presenting cells (26,40,56). Recently, murine and human islets were reported to express TLRs and in vitro TLR stimulation in islets induced chemokine production (20). Donor-derived monocyte chemoattractant protein-1 (MCP-1) plays an important role in the immediate islet graft function in a murine allograft model (44). Moreover, TLR stimulation broke tolerance induction in islet allografts and suppression of TLR4 in islets induced islet allograft tolerance (21,47). Considering the important roles of innate immunity in xenograft rejection, TLR activation could play an important role in islet xenograft rejection. However, there have been few studies on the role of TLRs in xenotransplantation, and most studies have focused on the recipient's TLRs (13,43,54). Therefore, we aimed to identify whether TLRs in porcine islets can contribute to islet xenograft rejection.
Materials and Methods
Animals
Porcine pancreases were harvested from adult female farm pigs at local slaughterhouses. Mice deficient in myeloid differentiation primary response 88 (MyD88) and TLR4 were the gift of Shizo Akira (Osaka University, Osaka, Japan). Male mice at 8–12 weeks of age were used as transplant recipients. The animal use protocol of this study was approved by the Institutional Animal Care and Use Committee of Seoul National University and Seoul National University Hospital.
Islet Isolation
Isolation and purification of porcine islets were performed as previously described (8), with minimal modifications. Briefly, the harvested pancreas was distended intraductally with University of Wisconsin solution containing Liberase DL (Roche Biochemicals, Basel, Switzerland) at a concentration of 2 ml/g of pancreatic tissue. The distended pancreas was digested in a modified Ricordi chamber (Biorep Technologies, Miami, FL, USA), and liberated islets were separated using density gradient on a Cobe 2991 cell separator (Gambro BCT Inc., Lakewood, CO, USA).
In Vitro TLR Stimulation
Porcine islets [1,000 islet equivalent (IEQ)] were grown in M199 media (Gibco) with 10% adult porcine serum (Gibco) and 1% penicillin/streptomycin solution (Gibco). Porcine islets were stimulated with polyinosinic/polycytidylic acid (poly I:C, 30 μg/ml, Sigma-Aldrich, MO, USA) or Escherichia coli 0111:B4 lipopolysaccharide (LPS, 0.5–1 μg/ml, Sigma-Aldrich) for 6–72 h.
Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted from islets using an RNA mini kit (Invitrogen, Carlsbad, CA, USA). The total RNA (1 μg) was reverse transcripted to produce cDNA using Superscript III reverse transcriptase (Invitrogen). Amplification was carried out using the PCR Thermal Cycler Dice™ (TaKaRa, Shiga, Japan). The β-actin gene was used for the quantitative titration of cDNA samples, and gene expression of target genes was presented as their expression relative to expression of β-actin gene. Groups without LPS/poly I:C stimulation were regarded as control groups.
After pig to mouse islet transplantation, frozen islet tissue was homogenized and grounded up by mortar and pestle. After RNA extraction, tissue expression of both murine and porcine chemokines/cytokines was analyzed using RT-PCR and was presented as relative expression normalized to expression of β-actin gene. Groups without LPS stimulation were regarded as control groups. We measured expression of chemokine C-X-C motif ligand 1 (CXCL1) as a functional homologue of interleukin-8 (IL-8) in mice, where there is no direct homologue of IL-8.
Primer sequences are available upon request.
Enzyme-Linked Immunosorbent Assay (ELISA) and Flow Cytometry
Protein levels of secreted chemokines in the culture supernatants of porcine islets were measured using cross-reactive human chemokine ELISA kits (RayBiotech Inc., Norcross, GA, USA).
For flow cytometric analysis, porcine islets were dissociated into single-cell suspensions by gentle agitation in accutase (Gibco, Gaithersburg, MD, USA). Islet cells were stained with anti-vascular cell adhesion molecule-1 (VCAM-1) or anti-intercellular adhesion molecule-1 (ICAM-1) antibodies generated in-house (35) and then stained with FITC-conjugated secondary antibodies (rabbit IgG, Invitrogen, Carlsbad, CA, USA). The stained cells were analyzed using a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA, USA).
Transmigration Assay
Human peripheral blood was taken from healthy donors (n = 3; age 32.3 ± 3.2 years; male/female, 2:1) after informed consent and ethical approval at Seoul National University Hospital. Peripheral blood mononuclear cells (PBMCs) were isolated from the blood by centrifugation using Ficoll-Plaque PLUS (GE Healthcare Life Science, Uppsala, Sweden). PBMCs (3×105)were added to the top of the insert in the Neuro Probe 48-Well Micro Chemotaxis Chamber (Neuro Probe, Inc., Gaithersburg, MD, USA). Culture supernatants (30 μl) were added below the culture insert. The transmigration assay was conducted for 2.5 h at 37°C. The number of transmigrated cells was calculated by counting the number of stained cells. Human PBMCs were pretreated with anti-RANTES (regulated on activation normal T-cell expressed and secreted) antibodies (4 μg/ml, R&D Systems, Minneapolis, MN, USA) or isotype control goat IgG (4 μg/ml, Sigma-Aldrich) for 30 min before transmigration assays in order to confirm the chemotactic effect of porcine RANTES.
Chromogenic Factor Xa Generation Assay and Thrombin Generation Assay
Tissue factor activity was analyzed by chromogenic factor Xa generation assay. Briefly, islets were incubated with activated factor VIIa (500 ng/ml; American Diagnostica, Stamford, CT, USA) and factor X (1 unit/ml; American Diagnostica) for 45 min at 37°C. The supernatant containing factor Xa was transferred to a new well and incubated with the chromogenic substrate S-2765 (DiaPharma, West Chester, OH, USA). The 96-well plates (BD, Franklin Lakes, NJ, USA) were read in an automated plate reader (Bio-Tek Instruments Inc., Winooski, VT, USA) at 405 nm for 10 min.
In order to measure the activity of Fgl-2 (fibrinogen-like protein 2), a thrombin generation assay was performed as described previously (19). Briefly, islets (500 IEQ) were incubated with an equal volume of human prothrombin (American Diagnostica) for 20 min at 37°C. After centrifugation, the supernatant was transferred to a new well, where Chromozym TH™ (Roche Biochemicals), a chromogenic substrate of human thrombin, was added. OD405 was measured using an automated plate reader.
Glucose-Stimulated Insulin Secretion Test
Porcine islets were incubated with poly I:C (30 μg/ml) or LPS (1 μg/ml) overnight (2.8 mM glucose). Insulin secretion was stimulated by treatment of cells with 20 mM glucose (Sigma-Aldrich) for 1 h at 37°C. Next, insulin concentration in the supernatant was determined by radio-immunoassay (Biosource, Nivelles, Belgium). The stimulation index was calculated as the ratio of stimulated (20 mM glucose) to basal (2.8 mM glucose) insulin release.
MyD88 Knockdown for the Porcine Islets Using shRNA Lentivirus
A shRNA sequence against porcine MyD88 mRNA (5′-GAGGACTGCCGAAAGTATA-3′) or the control sequence without any knockdown activity (5′-CTGCCCC AGCGATATCCAG-3′) was selected following a previous study (4) and was inserted into the expression vector, pDSL_ hpUGIP containing GFP (ATCC, Manassas, VA, USA). HEK-293T cells (ATCC) were transfected with the expression vectors together with packaging vectors (pMDLg/pRRE; pRSV-Rev, Addgene Inc., Cambridge, MA, USA) and enveloping vectors (pCMV-VSV-G, Addgene Inc.) by the calcium phosphate method for generation of shRNA lentivirus vector. The virus-containing supernatant was collected after 48 h and concentrated approximately 20-fold using Centricon Plus-70 (Millipore, Billerica, MA, USA). Porcine islets were infected with lentivirus [20 multiplicity of infection (MOI)] with the shMyD88 or control sequence for 8 h. After washing and an additional 16 h of culture, porcine islets were stimulated by LPS (1 μg/ml) for 6–48 h. The degree of MyD88 suppression was measured using both RT-PCR and Western blot (anti-MyD88 antibody, Abcam Inc., Cambridge, MA, USA). MyD88 knockdown-mediated suppression of LPS-induced inflammatory mediators and procoagulants was analyzed by RT-PCR.
Pig Islet to Mice Transplantation
MyD88 or TLR4 knockout C57BL6/J mice were rendered diabetic using a single intraperitoneal injection of streptozotocin (200 mg/kg, Sigma). Porcine islets (5,000–10,000 IEQ) were transplanted under the renal capsule of the recipients. Mice were treated intraperitoneally with anti-cluster of differentiation 154 (CD154) antibodies (0.5 μg, MR-1, BioXell, Lebanon, NH, USA) on days 0, 2, and 4 after transplantation. LPS (100 μg) was intraperitoneally administered on days 3, 5, and 7. Blood glucose concentration was measured using Accu-Chek® Performa® (Roche Diagnostics, Mannheim, Germany). Graft rejection was defined as the occurrence of a blood glucose concentration >350 mg/dl on two consecutive tests. Posttransplant normoglycemia was defined as random blood glucose concentration less than 200 mg/dl. In the case of islet recipients that were persistently normoglycemic by the end of the observation period (>200 days), the graft function was confirmed by unilateral nephrectomy of the kidney bearing the graft and documentation of the reappearance of diabetes.
Histological Analysis
Mice were sacrificed on day 8 after porcine islets were transplanted to TLR4 knockout C57BL6/J mice, and the kidneys with islet grafts were harvested. Grafts were embedded in Tissue-Tek® optimal cutting temperature (OCT) embedding compound (Sakura Finetek, Tokyo, Japan) and snap-frozen in liquid nitrogen for storage at −80°C. Guinea pig anti-insulin antibodies (DAKO, Carpinteria, CA, USA) and rat anti-mouse epidermal growth factor (EGF)-like module-containing mucin-like hormone receptor-like 1 (F4/80), rat anti-mouse CD4, rat anti-mouse CD8, and rat anti-mouse protein tyrosine phosphatase, receptor type C (CD45, B220; all e-Bioscience, San Diego, CA, USA) were used at 4°C overnight as the primary antibodies in immunofluorescent staining. Next, sections were stained with Alexa 488 goat anti-guinea pig IgG and Alexa 594 rabbit anti-rat IgG (Invitrogen) at room temperature for 1 h. Cell nuclei were counterstained with DAPI (Sigma-Aldrich) for 10 min. Cellular infiltration into the grafts was assessed using a Zeiss LSM 510 confocal microscope (Carl Zeiss, Inc., Thornwood, NY, USA).
Statistical Analysis
Most values were expressed as mean ± standard deviation. All analyses were performed using SPSS statistical software (version 17.0; Armonk, NY, USA). Continuous variables were compared using a t test, and categorical variables were compared using Fisher's exact test. Comparison between Kaplan–Meier survival curves was performed using the log-rank test. Values of p < 0.05 were regarded as statistically significant. For multiple comparison, values of p < 0.025 were considered statistically significant after Bonferroni adjustments for multiple comparisons.
Results
In Vitro TLR Stimulation in Porcine Islets Induced Expression of Proinflammatory Chemokines and Cytokines
Porcine islets expressed TLRs (TLR1–10) at resting status in RT-PCR (Fig. 1A). Both poly I:C and LPS stimulation induced RANTES (Fig. 1B), MCP-1 (Fig. 1C), interferon γ-induced protein 10 (IP-10) (Fig. 1D), and interleukin-8 (IL-8) (Fig. 1E) mRNA in porcine islets. Expression of interleukin-6 (IL-6) (Fig. 1F), interferon-α (IFN-α) (Fig. 1G), and interferon-β (IFN-β) (Fig. 1H) was also induced in porcine islets by poly I:C and LPS stimulation. Secretion of RANTES (Fig. 2A) and MCP-1 (Fig. 2B) as determined by the human chemokine ELISA kits was also increased in parallel with RT-PCR data. Overall, these results suggested that innate stimuli can stimulate TLR3 and TLR4 in porcine islets and thereby induce a proinflammatory response from porcine islets themselves.

TLR stimulation in porcine islets induced mRNA expression of proinflammatory chemokines and cytokines. (A) Toll-like receptor (TLR) expression in porcine islets at resting status. The expressions of TLR in porcine islets after overnight culture were analyzed by semiquantitative RT-PCR. (B–H) Induction of regulated on activation normal T-cell expressed and secreted (RANTES) (B), monocyte chemoattractant protein-1 (MCP-1) (C), interferon (IFN) γ-induced protein 10 (IP-10) (D), interleukin-8 (IL-8) (E), IL-6 (F), IFN-α (G), and IFN-β (H) in porcine islets in response to TLR stimulation. Expression of chemokines and cytokines after stimulation of polyinosinic/polycytidylic acid (poly I:C; 30 μg/ml) or lipopolysaccharide (LPS; 1 μg/ml) were analyzed by semiquantitative RT-PCR. The β-actin gene was used for the quantitative titration of cDNA samples, and gene expression of target genes was presented as their expression relative to expression of β-actin gene. Groups without LPS/poly I:C stimulation were regarded as control groups. *,#Significant differences (p < 0.01, p < 0.025, respectively) in comparison to control values at each time point. Each bar indicates the mean and standard deviation among triplicates. These results are representative of at least three independent experiments.
Porcine Islet Supernatants Stimulated by TLR Agonists Induced Chemotaxis of Human Leukocytes Across the Species Barrier
Islet supernatants cultured with poly I:C or LPS induced chemotaxis of human peripheral blood mononuclear cells (Fig. 2C). When transmigration assays were performed after pretreatment with anti-RANTES blocking antibodies, the chemotactic effects of the islet supernatant were significantly attenuated (p < 0.01), whereas isotype control goat IgG did not affect the chemotactic activity. These data indicated that porcine chemokines, especially RANTES, secreted from islets in response to TLR stimulation can recruit human leukocytes across the species barrier.

TLR stimulation in porcine islets induced secretion of porcine chemokines and chemotactic activity toward human leukocytes and upregulated expression of adhesion molecules. (A, B) Secretion of chemokines from porcine islets in response to TLR stimulation. After porcine islets were cultured with poly I:C (30 μg/ml) or LPS (1 μg/ml) for 48–72 h, concentrations of RANTES (A) and MCP-1 (B) in the supernatants were measured by ELISA. *Significant differences (p < 0.01) in comparison to basal values. (C) Chemotactic activities of supernatants from porcine islets stimulated by poly I:C or LPS toward human peripheral blood mononuclear cells. Porcine islets were cultured with poly I:C (30 μg/ml) or LPS (1 μg/ml) for 48 h, and the culture supernatants were used in transmigration assays. Anti-RANTES blocking antibodies decreased chemotactic activity significantly in transmigration assays (p < 0.01), whereas isotype control goat IgG did not affect the chemotactic activity. *Significant differences (p < 0.01) in comparison to the unstimulated control group. **Significant differences (p < 0.01) between the TLR stimulation groups and the anti-RANTES blocking groups. (D–E) Upregulation of vascular cell adhesion molecule-1 (VCAM-1) (D) and intercellular adhesion molecule-1 (ICAM-1) (E) in porcine islets by TLR stimulation. Porcine islets were cultured with poly I:C (30 μg/ml) or LPS (1 μg/ml) for 48 h. Surface expression of VCAM-1 and ICAM-1 in porcine islets was analyzed by flow cytometric analysis. Each bar indicates the mean with a standard deviation among triplicates. These results were representative of two to three independent experiments.
TLR Stimulation in Porcine Islets Induced Expression of Adhesion Molecules
Next, induction of adhesion molecules in porcine islets in response to TLR stimulation was investigated. Stimulation with either poly I:C or LPS increased expression of both VCAM-1 (p = 0.02 for poly I:C, p = 0.08 for LPS) (Fig. 2D) and ICAM-1 (p < 0.01 for poly I:C, p = 0.09 for LPS) (Fig. 2E) in porcine islets.
TLR Stimulation in Porcine Islets Induced Procoagulant Activation
Both poly I:C and LPS stimulation in porcine islets induced mRNA expression of tissue factor (Fig. 3A) and Fgl-2 (Fig. 3B). When functional activity of tissue factor was measured, tissue factor activity increased in parallel with RT-PCR data after stimulation by either poly I:C or LPS (Fig. 3C). Functional activities of Fgl-2 also increased in response to TLR stimulation in the thrombin generation assay (Fig. 3D). Procoagulant induction in porcine islets was more prominent in poly I:C stimulation than in LPS stimulation. Taken together, these data showed that TLR stimulation in porcine islets induced procoagulants as well as proinflammatory mediators.

TLR stimulation in porcine islets induced procoagulant activation. Porcine islets were cultured with poly I:C (30 μg/ml) or LPS (1 μg/ml) for 6–24 h. (A, B) Expression of tissue factor (A) and fibrinogen-like protein 2 (Fgl-2) (B) in porcine islets after coculture with poly I:C or LPS were analyzed by semiquantitative RT-PCR. The β-actin gene was used for the quantitative titration of cDNA samples, and gene expression of target genes was presented as their expression relative to expression of β-actin gene. Groups without LPS/poly I:C stimulation were regarded as control groups. *,#Significant differences (p < 0.01, p < 0.025, respectively) in comparison to unstimulated controls at each time point. (C, D) Functional activity of tissue factor (C) and Fgl-2 (D) in porcine islets by TLR stimulation was measured using a chromogenic factor Xa generation assay (C) and thrombin generation assay (D), respectively. *,#Significant differences (p < 0.01, p < 0.025, respectively) in comparison to unstimulated controls at each time point. Each bar indicates the mean and standard deviation among triplicates. The results were representative of three independent experiments.
TLR Stimulation Did Not Influence Insulin Secretion From Porcine Islets
When impact of TLR stimulation on insulin secretion was evaluated using a glucose stimulated insulin secretion test, stimulation with neither poly I:C nor LPS significantly influenced insulin secretary function, a parenchymal function of islets (p = 0.16 for poly I:C; p = 0.78 for LPS) (Fig. 4).

TLR stimulation in porcine islets did not affect insulin secretion from islets. When the impact of TLR stimulation on insulin secretion was evaluated using a glucose stimulated insulin secretion test, stimulation with neither poly I:C nor LPS influenced insulin secretion significantly (p = 0.78 for poly I:C; p = 0.16 for LPS). Each bar indicates the mean and standard deviation among triplicates. The results were representative of three independent experiments.
MyD88 Knockdown Suppressed the LPS-Induced Proinflammatory and Procoagulant Response of Porcine Islets
Infection of porcine MyD88-shRNA lentivirus (20 MOI) significantly attenuated expression of MyD88 mRNA in porcine islets in semiquantitative RT-PCR (p < 0.01) (Fig. 5A). The knockdown effect in protein expression of porcine MyD88 was about 80–90% according to the Western blot (p < 0.01) (Fig. 5B).
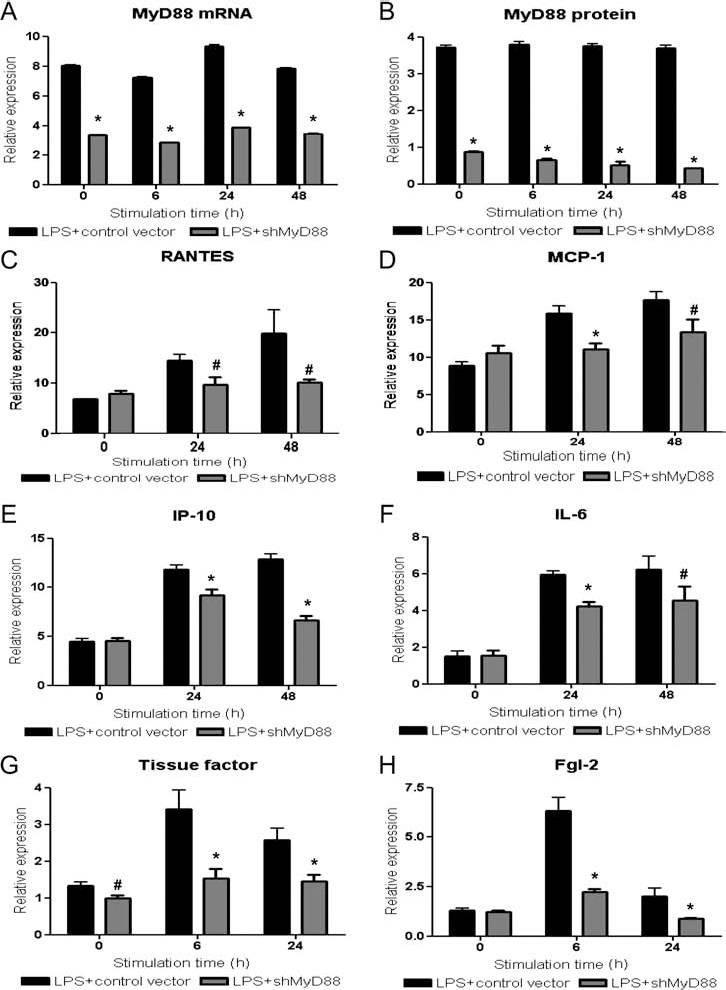
Knockdown of porcine MyD88 suppressed the LPS-mediated induction of chemokines and procoagulants in porcine islets. (A, B) Lentiviral shRNA knocked down expression of porcine myeloid differentiation primary response 88 (MyD88) in islets successfully. Porcine islets were transduced with 20 multiplicity of infection (MOI) of lentivirus (with a control or shMyD88 sequence). Expression of MyD88 in lentivirus-transduced, porcine islets with LPS (1 μg/ml) stimulation was analyzed by both semiquantitative RT-PCR (A) and Western blot (B). The β-actin was used for the quantitative titration of cDNA samples and as a quantity control in western blot analysis. Expression of MyD88 gene and protein was presented as their expression relative to expression of β-actin. (C–F) Knockdown of porcine MyD88 in islets attenuated LPS-induced expression of chemokines and cytokines significantly. shMyD88 lentivirus-transduced islets were cultured with LPS (1 μg/ml) for 24–48 h. Expression of chemokines (RANTES, MCP-1, IP-10) and IL-6 in porcine islets was analyzed by semiquantitative RT-PCR. The β-actin gene was used for the quantitative titration of cDNA samples. (G, H) Knockdown of porcine MyD88 in islets attenuated procoagulant induction by LPS stimulation significantly. shMyD88 lentivirus-transduced islets were cultured with LPS (1 μg/ml) for 6–24 h. Expression of tissue factor (G) and Fgl-2 (H) in porcine islets after lentivirus infection was analyzed by both semiquantitative RT-PCR. The β-actin gene was used for the quantitative titration of cDNA samples. Gene expression of target genes was presented as their expression relative to expression of β-actin gene. *,#Significant differences (p < 0.01, p < 0.05, respectively) in comparison to controls at each time point. Each value indicates the mean and standard deviation among triplicates. The results were representative of three independent experiments.
MyD88 knockdown suppressed LPS-mediated induction of chemokines (RANTES, MCP-1, IP-10) and cytokines (IL-6) in porcine islets (p < 0.05) (Fig. 5C–F). LPS-mediated induction of tissue factor and Fgl-2 from porcine islets was also suppressed by MyD88 knockdown in both RT-PCR (p < 0.05) (Fig. 5G–H). These data supported that TLRs in porcine islets played a significant role in induction of proinflammatory and procoagulant responses and had a functional significance.
TLR4 Stimulation in Islets Led to Xenograft Rejection
The impact of in vivo TLR stimulation in porcine islets while minimizing TLR stimulation on the recipient mice was investigated using MyD88 or TLR4 knockout mice. Porcine islets were transplanted into diabetic MyD88 or TLR4 knockout mice (Fig. 6A). Anti-CD154 treatment prolonged porcine islet xenograft survival with a median survival time (MST) longer than 200 days in both MyD88 knockout and TLR4 knockout recipients. LPS stimulation led to xenograft rejection in both MyD88 knockout (MST, 15 days for LPS group; LPS group vs. MR-1 group, p = 0.02) (Fig. 6B) and TLR4 knockout recipients (MST, 62.5 days for LPS group; LPS group vs. MR-1 group, p < 0.05) (Fig. 6C). Overall, LPS stimulation, mainly in porcine islets, interfered with islet xenograft tolerance induction by costimulatory blockade. These data suggested that TLR activation in donor islets plays a significant role in islet xenograft rejection.

TLR stimulation in porcine islets interfered with tolerance induction in pig to mice islet transplantation. (A) Porcine islets were transplanted to MyD88 knockout or TLR4 knockout mice. Mice were treated with anti-cluster of differentiation 154 (CD154) antibodies (MR-1) on days 0, 2, and 4 after transplantation. LPS was intraperitoneally administered to the experimental group on days 3, 5, and 7 after transplantation. (B, C) Anti-CD154 antibody (MR-1) treatment without LPS induced islet xenograft tolerance in more than half of MyD88 knockout (B) or TLR4 knockout (C) recipients. However, LPS stimulation in porcine islets led to xenograft rejection in both MyD88 knockout recipients (median survival time, 15 days for LPS group; LPS group vs. MR-1 group, p = 0.02, log-rank test) (B) and TLR4 knockout recipients (median survival time, 62.5 days for LPS group; LPS group vs. MR-1 group, p < 0.05, log-rank test) (C).
In Vivo TLR4 Stimulation Increased Proinflammatory Chemokines and Cytokines in Islet Xenograft
Consistent with in vitro data, expression of porcine chemokines (RANTES, MCP-1, IL-8) and cytokines (IL-6, IFN-α, IFN-β) in islet grafts was significantly increased by stimulation of LPS compared with the control group (p < 0.05) (Fig. 7A). Expression of murine chemokines (RANTES, MCP-1, IP-10) and cytokines (IL-6, TNF-α) from the recipients was also increased secondarily to LPS stimulation (p < 0.05) (Fig. 7B), whereas there was no significant difference in the expression of IFN-γ, IL-4, or IL-10 (Fig. 7B).

TLR stimulation in porcine islets increased the expression of proinflammatory chemokines and cytokines in both donors and recipients in pig to mouse islet transplantation. After porcine islets were transplanted to TLR4 knockout C57BL6/J mice, anti-CD154 antibodies were administered with or without LPS and islet xenografts were harvested on day 8. Tissue expression of both porcine (A, RANTES, MCP-1, IP-10, IL-8, IL-6, IFN-α, and IFN-β) and murine [B, RANTES, MCP-1, IP-10, chemokine C-X-C motif ligand 1 (CXCL1), IL-6, tumor necrosis factor (TNF)-α, IFN-γ, IL-4, and IL-10] chemokines/cytokines were analyzed using semiquantitative RT-PCR. The β-actin gene was used for the quantitative titration of cDNA samples, and gene expression of target genes was presented as their expression relative to expression of β-actin gene. Groups without LPS stimulation were regarded as control groups. *,#Significant differences (p < 0.01, p < 0.05, respectively) as compared with the control group without LPS. Each value indicates the mean and standard deviation among triplicates. The results were representative of two independent experiments.
In Vivo TLR4 Stimulation Increased Infiltration of Inflammatory Cells Into Islet Xenograft
When inflammatory cell infiltration around the islet xenograft was analyzed on day 8 after transplantation using confocal microscopy (Fig. 8), LPS stimulation increased infiltration of both innate (macrophages, p = 0.02) (Fig. 8A, B, F) and adaptive (CD4+ T-cells, p < 0.01) (Fig. 8C, F) (CD8+ T-cells, p = 0.03) (Fig. 8D, F) (B-cells, p < 0.01) (Fig. 8E, F) immune cells in parallel with higher expression of inflammatory chemokines by LPS stimulation.

TLR stimulation in porcine islets increased inflammatory cell infiltration into islet xenografts in pig to mouse islet transplantation. After porcine islets were transplanted to TLR4 knockout C57BL6/J mice, anti-CD154 antibodies were administered with or without LPS and islet xenografts were harvested on day 8. (A–F) When infiltration of macrophages (A, B), CD4+ T-cells (C), CD8+ T-cells (D), and B-cells (E) was analyzed using confocal microscopy, LPS stimulation increased the infiltration of all sets of inflammatory cells around islet grafts compared with the control group. Green indicates insulin staining, blue indicates DAPI staining, and red indicates staining for specific cell surface markers {F4/80 [epidermal growth factor (EGF)-like module-containing mucin-like hormone receptor-like 1] for macrophages, CD4, CD8, and B220 [protein tyrosine phosphatase, receptor type, C; CD45] for B-cells}. Original magnification: 400×. (F) Quantitative analysis of infiltrating cells into islet xenografts. Numbers of graft-infiltrating cells were counted and averaged in three high-power fields (400×) per each mouse. When the numbers of infiltrating cells were compared, infiltration of macrophages, T-cells, and B-cells increased significantly in the LPS group compared with the control group (*p < 0.01, #p < 0.05). Each value indicates the mean and standard error among three mice. The results were representative of three independent experiments.
Discussion
Innate immunity plays important roles in islet xenograft rejection such as IBMIR and acute cellular rejection. TLRs are an important sensor of innate immunity (1,26). Among various TLRs, we stimulated TLR3 and TLR4 in islets as representative TLRs, in the sense that they use different signal transduction pathways and have both exogenous and endogenous ligands. Poly I:C stimulates TLR3 and thereby activates the Trif-dependent pathway, in other words, the MyD88-independent pathway (50). LPS stimulation on TLR4 can trigger both MyD88-dependent and -independent pathways, although the MyD88-dependent pathway is predominant (31).
Both poly I:C and LPS stimulation markedly increased secretion of chemokines from porcine islets. Consistent with a previous study that showed the importance of donor endothelium-derived chemokines in allograft rejection (23), we demonstrated that in vivo LPS stimulation on donor islets increased intragraft chemokine expression and inflammatory cell infiltration into islet xenografts. Upregulation of intragraft murine chemokines in MyD88-or TLR4-deficient recipients must have been mainly derived from infiltrated leukocytes that were activated by amplified, non-TLR stimuli rather than direct TLR stimuli. Overall, these findings suggested that TLR stimulation by damaged cells could increase the secretion of chemokines from the islet xenograft itself and thereby contributed to xenograft rejection.
TLR stimulation in porcine islets also induced both IL-6 and type I interferon. Mice with IL-6 deficiency showed lower serum levels of immunoglobulin after transplantation of the fetal porcine islet-like cell cluster (7). Discordant pig-to-baboon cardiac xenografts showed more intense IL-6 responses than concordant rhesus monkey-to-baboon xenografts (36). Administration of IFN-β recapitulated TLR-mediated skin allograft rejection and the deficiency of type I interferon receptor abrogated TLR-mediated graft rejection (47). Therefore, TLR-mediated induction of IL-6 and type I interferon in porcine islets might contribute to islet xenograft rejection.
Stimulation by either LPS or HMGB-1 (high-mobility group box-1 protein) stimulation was reported to induce a coagulation response (33,49). Consistent with these data, we demonstrated that stimulation of either poly I:C or LPS induces activation of both tissue factor and Fgl-2 in porcine islets. Tissue factor induction on the islet surface triggers IBMIR (37), and induction of Fgl-2 contributes to thrombosis during acute vascular xenograft rejection (19). Therefore, TLR-mediated procoagulant induction in porcine islets might contribute to the IBMIR in islet xenograft. However, we used the subcapsular transplantation model instead of the intraportal transplantation model, because islet mass used in this study cannot be delivered through intraportal route to diabetic, immunocompetent mice for adequate control of blood glucose. Further studies using intraportal pig islet transplantation to nonhuman primates could confirm our in vitro findings.
In order to determine in vivo implications of TLR stimulation in porcine islets with minimal activation of TLRs in the recipient, we stimulated MyD88 or TLR4 knockout mice with LPS. LPS stimulation can use the MyD88-independent pathway together with the MyD88-dependent pathway, the main activation pathway. However, MyD88 deficiency impairs the macrophage response to several bacterial products (30) and in vivo LPS stimulation in MyD88 KO mice could not abrogate islet allograft tolerance (47). Therefore, the impact of LPS stimulation on the recipients via MyD88 independent pathways could be minimal in our model. On the other hand, poly I:C can stimulate the Mda5 (melanoma differentiation-associated gene 5)/IPS-1 (IFN-β promoter stimulator-1) pathway in addition to the TLR3-Trif (Toll/IL-1R domain-containing adaptor-inducing IFN-β)-dependent pathway (34). Poly I:C stimulation in TLR3 knockout mice still interfered with skin allograft tolerance (47). Therefore, it is very hard to minimize poly I:C stimulation on the recipient side.
The finding that LPS stimulation on donor islets interfered with xenograft tolerance induction in both MyD88 and TLR4 knockout mice confirmed the in vivo roles of TLR in porcine islets in xenograft rejection. There has been controversy over the in vivo role of TLR in donor islets in allotransplantation. Initially, TLR4 knockout in donor islets was reported to induce islet allograft tolerance (21); however, the following studies did not reproduce the finding of the initial study (25,47). Interestingly, another study used a syngeneic intraportal islet transplantation model, which focused on the innate immune response excluding the alloimmune response (18). TLR4 knockout on donor islets prolonged syngeneic islet graft survival. A stronger innate immune response in the intraportal transplantation model compared with the renal subcapsular transplantation model might also have contributed to these results. Taken together, our results could be attributed to stronger innate immune responses in islet xenograft rejection compared with islet allograft rejection.
In clinical islet transplantation, endogenous ligands could be dominant TLR stimuli over exogenous ligands such as LPS and poly I:C. TLR4 and its ligands such as HMGB1 and heat shock protein 70 (HSP70) were upregulated in islet allografts (57). HMGB1 stimulation on islets induced engraftment failure of islet graft from wild-type mice, whereas it did not induce failure of islet graft from TLR2/TLR4 double knockout mice (18). Furthermore, graft survival rates of islets from either TLR4 (21) or TLR2/4 double knockout (18) mice were prolonged in the absence of administration of TLR ligands. These results suggested that endogenous ligands might stimulate proinflammatory cascades in porcine islet cells and contribute to xenograft rejection in a fashion that is similar to LPS and poly I:C.
Islet xenograft rejection requires CD4+ T-cells and is accelerated by B-cells (48,51). Macrophages are the major effector cells in T-cell-mediated rejection (16,17). In parallel with the previous studies, we found that TLR stimulation increased infiltration of macrophages with CD4+ T-cells and B-cells. High intragraft expression of RANTES and MCP-1 in response to TLR stimulation must have contributed to predominant infiltration of the macrophages and CD4+ T-cells.
MyD88 knockdown in porcine islets by shRNA lentivirus attenuated upregulation of proinflammatory chemokines, cytokines, and procoagulants in response to in vitro TLR stimulation. These results support the active role of TLRs in porcine islets in islet xenograft rejection. However, we could not confirm role of TLRs in porcine islet, because we did not show that removal or blockade of TLRs in porcine islets attenuated islet xenograft rejection in vivo. Unfortunately, anti-porcine TLR4 blocking antibodies for in vivo experiments are not available, and decreased viability of islets by lentivirus infection process made it very hard to assess impact of MyD88 knockdown on islet graft survival in vivo (data not shown). Further in vivo studies using more advanced, in vitro MyD88 knockout or knockdown technologies are needed for determining whether MyD88 or TLR4 knockout pigs could be promising for clinical islet xenotransplantation.
Next, proinflammatory responses of islets in response to TLR stimulation might be attributed to small number of dendritic cells or endothelial cells among isolated islets rather than endocrine cells; however, it is not practically important, because the cell mixture instead of the purified endocrine cells is transplanted into recipients. Despite the limitations, we suggest for the first time that TLR stimulation in porcine islets could contribute to islet xenograft rejection, where innate immunity is strongly involved.
In conclusion, TLR activation in porcine islets induced both a proinflammatory and procoagulant response and contributed to islet xenograft rejection.
Footnotes
Acknowledgments
This study was supported by grants from the Ministry of Knowledge Economy, Republic of Korea (Grants 2009-67-10033838, 2009-67-10033805), a grant from the Korea Health 21 R&D Project, Ministry of Health and Welfare, Republic of Korea (Project No. A040004-010), and a grant from the SNUH Research Fund (No. 03-2012-0450). The authors declare no conflict of interest.
