Abstract
Allogeneic islet transplantation becomes a viable option for patients with unstable type 1 diabetes. However, considering the huge number of patients with type 1 diabetes, human donor shortage is a serious issue. To overcome the donor shortage issue, xenotransplantation is an attractive option. In fact, clinical islet xenotransplantation has been conducted since 1990s. The first clinical trial was performed using fetal pigs and demonstrated the porcine pancreatic tissue could survive in human body with immunosuppressive strategies. To scale up the islet production, Canadian group established a method for islet isolation from neonatal pigs. Their method has been used for clinical islet xenotransplantation in New Zealand, Russian, Mexico, Argentina, and China. Recently Korean group published a clinical protocol for islet xenotransplantation using adult pigs. For the next generation of islet xenotransplantation, gene-modified pigs were created. Especially “superislets” created by Belgian group demonstrated promising preclinical outcomes. With advanced donor pigs, islet xenotransplantation might become a suitable treatment for the majority of type 1 diabetic patients.
Pig Pancreas from
Introduction
Allogeneic islet transplantation becomes a viable treatment for type 1 diabetic patients who experience severe hypoglycemia despite intensive diabetes management and education 1 . However, considering a limited number of pancreatic donors, xenogeneic islet transplantation is an attractive option 1 . In fact, the first islet xenotransplantation was performed in 1990 2 .
The ideal donor pig for islet xenotransplantation is a fundamental question. In this review article, we assess the clinically used donor pigs and discuss future possibilities of a next generation of donor pigs.
Fetal porcine pancreas for islet xenotransplantation
Swedish group
The first clinical islet xenotransplantation was conducted by Swedish group using fetal pancreata. Fetuses ranging in age from 51 to 77 days were used 3 . The crown-rump length of the fetuses ranged from 10 to 20 cm, and the body weight from 40 to 260 g. The fetuses were immediately collected from the uterus and placed on ice during transportation to the laboratory. Removed pancreata were put on chilled Hanks’ balanced salt solution (HBSS) supplemented with benzylpenicillin, and streptomycin within 1 h after killing the saw.
Their islet isolation method was as follows. The pancreata were minced and transferred to a glass vial containing collagenase. Then digestion was conducted by vigorous shaking in a 37°C water bath. After visual confirmation of disintegration, digestion was arrested by adding HBSS. Then digested pancreata were washed with HBSS and resuspended in RPMI 1640 culture medium with fetal calf serum or heat-inactivated human serum. The culture dishes were maintained at 37°C in a gas phases of 5% CO2 in humidified air. The culture medium was changed every 2 days until the sixth or seventh day. This method provided average 10,000 islet like cell cluster per a pancreas 4 . They mentioned optimal age of the piglets, and they preferred as old as possible due to larger glands with more differentiated endocrine properties. However, with their method, late gestational (2–3 days before term) or newborn piglets were less effective. The group tested the effect of nicotinamide and sodium butyrate during culture. They found the optimal maturation of porcine fetal pancreatic cells was obtained in serum-free medium supplemented with nicotinamide. Sodium butyrate was a potent stimulus for beta cell differentiation but leaded to increase apoptotic cell death 5 . They transplanted the fetal porcine pancreatic cells into type 1 diabetic patients who had already received kidney transplantation. They successfully demonstrated positive porcine C-peptides for 200–400 days, however no apparent clinical benefits were shown.
New Zealand group
New Zealand group used fetal pancreata for islet isolation using the Korsgren islet isolation method 6 . They used term-gestation piglets delivered by hysterotomy from healthy farm stock 7 . Pigs in New Zealand are remarkably free of many porcine endemic viruses as a result of isolationist agricultural policies. A standard collagenase digestion of the minced pancreata according to Korsgren protocol was carried out. The digested pancreatic tissue was cultured in RPMI-1640 medium containing 10mM nicotinamide 7 . They encapsulated the isolated islets then transplanted the encapsulated islets into two type 1 diabetic patients without immunosuppressive drugs 7 . They successfully demonstrated positive porcine C-peptides. In addition, reduction of insulin dose and HbA1c were shown.
Neonatal porcine pancreas for islet xenotransplantation
Canadian group
Korbutt and his colleague demonstrated a large-scale isolation method using neonatal porcine pancreas
8
. They used 1-day-old to 3-day-old Landrace Yorkshire neonatal pigs (1.5–2.0 kg body weight) of either sex. Their islet isolation method was as follows. Procured pancreata were cut into fragment of 1–2 mm3, then digested with a collagenase. The digested pancreatic tissue was filtered through a nylon screen (500 μm) then cultured in Ham’s F10 tissue culture medium (10 mmol/l glucose, 50 mmol/l IBMX, 0.5% BSA, 2 mmol/l
Six of the seven macaques receiving neonatal porcine islets with the costimulation blockade regimen achieved a sustained period of insulin independence for the length of survival of the recipient.
New Zealand group
In 2009, The International Xenotransplantation Association published consensus statement for source pig on clinical trials of porcine islet products in type 1 diabetes 10 . In the statement, donor pigs need to fulfill the designated pathogen-free (DPF) status. Related to the DPF, Food and Drug Administration (FDA) describes DPF as follows. This term is used to describe animals, animal herds, or animal facilities that have been rigorously documented to be free of specified infectious agents and that are maintained using well-defined routines of testing for designated pathogens and utilizing rigorous Standard Operating Procedures (SOPs) and practices of herd husbandry and veterinary care to assure the absence of the designated pathogens 11 .
To achieve DPF status, New Zealand group established the multilevel testing system consisting of testing donor herd, donor sows. They created a DPF facility, and the donor pig herd was maintained in the facility and tested regularly using a combination of culturing, serological, and polymerase chain reaction (PCR) method. Ten bacterial species, 15 viruses, and one protozoa were routinely tested 12 (Table 1). They also screen sows at both prefarrowing and postfarrowing, donor piglets, and islet cells as shown in Table 2.
Testing schedule for the NZ donor pig herd 12 .

PCV2: Porcine Circovirus Type 2, PCV1: Porcine Circovirus Type 1, PLHV2: Porcine Lymphotropic Herpesvirus Type 2, PCMV: Porcine Cytomegalovirus, Rota V A-C: Rotavirus A, Rotavirus B and Rotavirus C, Reo V: Reovirus (all types), PTV: Porcine Teschovirus, PEVB: Porcine Hemagglutinating Encephalomyelitis Virus, HEV: Hepatitis E Virus, BVD: Bovine Virus Diarrhea, AujD: Aujeszky’s Disease, PPV: Porcine Parvovirus, PRRSV: Porcine Reproductive and Respiratory Syndrome Virus, EMCV: Encephalomyocarditis Virus. MAT: microscopic agglutination test, ELISA: enzyme-linked immunosorbent assay, LAT: latex agglutination test, PCR: conventional nested PCR, RT-PCR: reverse transcriptase PCR, rt-PCR: real-time PCR, rtRT-PCR: real-time reverse transcriptase PCR, VNT: virus neutralization test.
Multilevel screening system for sows, donor pigs and islet cells 12 .
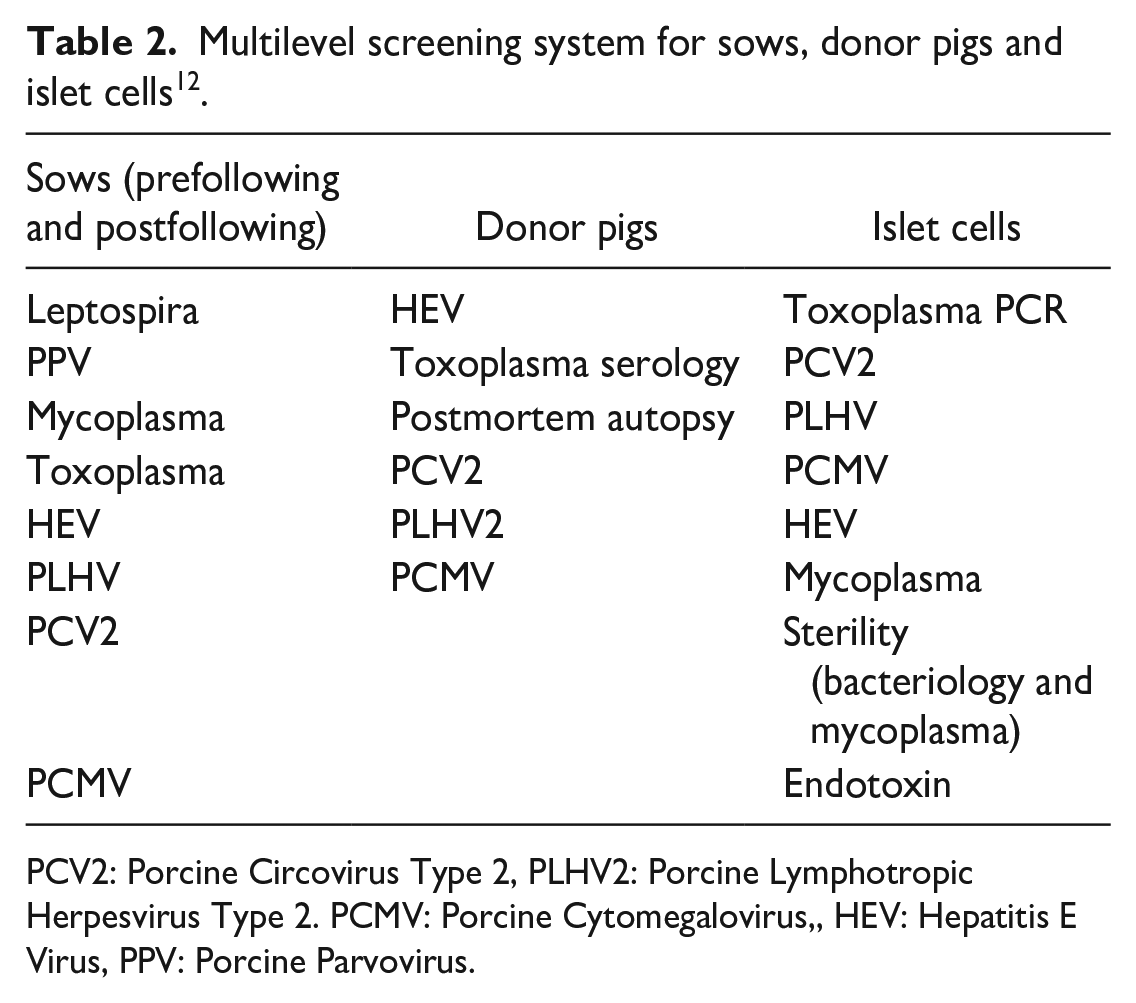
PCV2: Porcine Circovirus Type 2, PLHV2: Porcine Lymphotropic Herpesvirus Type 2. PCMV: Porcine Cytomegalovirus,, HEV: Hepatitis E Virus, PPV: Porcine Parvovirus.
They modified the Korbutt isolation method for the large scale neonatal porcine islet isolation 13 . They used 7-day-old to 17-day-old neonatal piglets. Digested pancreata were filtered through a 300 μm stainless-steel screen. Then the filtrated tissue was cultured in 500 mL disposable spinner flask at a concentration of 40 mL per gram of pancreatic tissue in RPMI-CAN serum-free medium (RPMI-1640, 0.3g/l ciprofloxacin, 1% human serum albumin, 0.12% wt/vol nicotinamide). The flasks were stirred on a four-position slow speed magnetic stirrer at 80–90 rpm in a humidified 5% CO2 incubator at 37°C. Isolated islets were encapsulated using alginate and poly L ornithine (PLO) to avoid immunological rejection. The encapsulated neonatal porcine islets were transplanted into 16 type 1 diabetic patients in New Zealand and 22 type 1 diabetic patients in Argentina 14 . Importantly, there were no xenogeneic infection in all patients 14 .
In New Zealand clinical trial, majority of patients improved the number of unaware hypoglycemia 15 . In Argentine trial, majority of patients improved HbA1c, reduced insulin requirements and reduced the number of unaware hypoglycemia 16 . Especially in Argentina trial, majority of patients were positive attitude even 10 years after receiving encapsulated neonatal porcine islet transplantation 17 .
Chinese group
Between 2013 and 2017, Dr Wei Wang and his team transplanted newborn pig islets into 10 patients with type 1 diabetes 18 (ClinicalTrials.gov ID NCT03162237). The pigs were from the first Asian Designated Pathogen Free Donor Facility established by the Third Xiangya Hospital and Human XENO Biological Technology Co,. Ltd.
The neonatal pig islets (10,000 IEQ/kg) and patients’ autologous T regulatory cells (2 × 106/kg) were transplanted. They used tacrolimus, mycophenolate mofetil, belatacept as immunosuppression. The condition of these patients improved substantially 18 .
Spring Point Project
Spring Point Project (Minneapolis, MN, USA) created a Source Animal Facility (SAF) for generating DPF pigs for clinical xenotransplantation 19 .
To enable DPF pigs should be reared within biosecure barrier facilities, so-called Source Animal Facilities (SAFs), isolated from potential pathogen sources via filtered air, filtered and disinfected water, sterilized fee, show-in and show-out access for staff, and hygienic measures including through screening of employees for potentially transmissible pathogens prior to hiring.
Origin of animals for SAF was populated with caesarian derived colostrum deprived (CDCD) piglets derived from vaccinated specific pathogen free (SPF) sows. The founder animals were selected based on the criteria that high islet of Langerhans yield from adult donors 20 .
The CDCD piglets were housed for the first 3 weeks in the quarantine nursery in isolated nursery carts outfitted with tenderfoot flooring. After 3 weeks, the animals were moved into animal holding rooms outfitted with stainless steel penning affixed to epoxy sealed concrete flooring and walls. The Designated Pathogen Free (DPF) listing of excluded pathogens was shown in Table 3.
Designated Pathogen Free (DPF) listing of excluded pathogens at Spring Point Project.

Medical porcine development organization
In Japan, currently no DPF pig for xenotransplantation. To accelerate xenotransplantation the “Medical Porcine Development Organization” was established in Japan on December 1, 2023, at the Kobe University Graduate School of Medicine in the department of hepatobiliary pancreatic surgery 1 . This organization aims to create new medical care protocols and contribute to the health and welfare of the population by establishing and improving the technology to produce pigs than can be used as medical raw materials.
Adult porcine pancreas for islet xenotransplantation into nonhuman primates
Adult porcine pancreata were not used clinically. However, these pigs were used for preclinical nonhuman primate studies.
Minnesota group
In 2016, Hering et al. 21 published long-term insulin free in diabetic nonhuman primate after intraportal adult porcine islet transplantation. They used genetically unmodified wild-type adult pigs as pancreas donors. They transplanted isolated islets 25,000 IEQ/kg into streptozotocin induced diabetic nonhuman primates. They used three types of immunosuppression protocols (Table 4).
Immunosuppression protocol for porcine islet xenotransplantation into diabetic nonhuman primates by Minnesota group.

At 100 days after transplant islet xenograft survival rates were 0% in group A, 50% in group 5, and 100% in group C. However, eight out of nine recipients showed thromboembolic lesions in group B and C, probably related to ABI793 therapy. Therefore, this protocol was not proceeded in clinical trials.
Seoul group
Seoul National University group used adult miniature pig for islet xenotransplantation. They used Seoul National University miniature pig (SNU miniature pig) for their study. At first, the Seoul group conducted 68 porcine islet isolation to identify important parameters for successful islet isolation. The pigs with a mean age 25 months, median age of 24.3 months and a mean weight of 88 kg were used. The number of male and female pigs was the same. The male pigs were bred without castration. The complete pregnancy related history was recorded for the female pigs. The general condition of all pigs was carefully examined and noted. After transfer to the operative unit a week before operation, the pigs’ adaptation to their new environment was monitored by observing their feeding by a veterinarian.
They divided the isolation outcomes based on islet yield, then compared the outcomes between high yield group and low yield group. The total islet yield in high yield group was 304,962 ± 25,014 IEQ and in low yield group was 142,672 ± 12,493 IEQ. They identified good or moderate pancreatic distention with collagenase, older age (>2yr), and male gender are significant parameters to obtain high total islet yield 22 .
The group demonstrated long-term control of diabetes in immunosuppressed nonhuman primate by the transplantation of adult porcine islets 23 . The immunosuppression protocol is shown in Table 5.
Immunosuppression protocol by Seoul group.

In addition to immunosuppression, they used cobra venom factor and anti-TNF-alpha monoclonal antibody to reduce instant blood mediate reaction (IBMIR). For induction immunosuppression, they used anti-thymoglobulin, for maintenance immunosuppression they used sirolimus. Three out of five cases, they used own Treg as shown in Table 5.
They transplanted 81,000 IEQ/kg in the first case and 100,000 IEQ/kg in all the other cases. The diabetic nonhuman primates could maintain an insulin free status more than 603 days in the first case, 167 days in the second case, 512 days in the third case, 303 days in the fourth case, and 180 days in the fifth case. This protocol used anti-CD154 mAb which is not clinical available due to the side effects. Therefore, this protocol was not proceeded in clinical trials.
Since anti-CD154 mAb is not clinically available, the Seoul group assessed anti-CD40 antibody-based immunosuppression despite anti-CD154 mAb, in the pig to nonhuman primate islet xenotransplantation model 24 . They conducted nine cases, and their immunosuppression protocol was described in Table 6.
Anti-CD40 mAb based immunosuppression protocol by Seoul group.

The streptozotocin induced diabetic nonhuman primates received approximately 100,000 IEQ/kg islets in their portal veins. The nonhuman primates could maintain insulin free periods for 4 and 5 days in the first and second cases. The insulin free periods were prolonged for 25 and 8 days in the third and fourth cases. The insulin free periods were more than 60, 266, 10, 14, and 3 days in the fifth, sixth, seventh, eighth, and ninth cases, respectively. They concluded that anti-CD40 mAb combined with tacrolimus was effective in prolonging porcine islet graft survival, but anti-CD40 mAb was not as effective as anti-CD154 mAb in terms of preventing early islet loss.
The Seoul group created a new immunosuppression protocol using clinically available immunosuppression 25 . The protocol is shown in Table 7.
Clinically available immunosuppression protocol by Seoul group.

They transplanted 55,600 to 102,000 IEQ/kg porcine islets into streptozotocin induced diabetic nonhuman primates (n = 7). The durations of normoglycemia with less than half doses of exogenous insulin were more than 200, 45, 194, 80, 210, 93, and more than 222 days. They concluded that the study could contribute greatly to the initiation of islet xenotransplantation clinical trials.
The Seoul group published “Clinical Trial Protocol for Porcine Islet Xenotransplantation in South Korea” in 2024 26 . A clinical trial, titled “A single-center, open-label, sponsor-initiated clinical trial to assess the safety and efficacy of porcine islet transplantation on diabetes patients” has received approval by the Korean Ministry of Food and Drug Safety.
The donor pig is the DPF grade Seoul National university (SNU) miniature pigs aged more than 2 years. Preceding transplantation, porcine islets sourced from DPF pig will undergo isolation, commencing 6 days prior to the procedure, continuing until the total islet count (10,000 islet equivalent /kg of the patient body weight) is achieved.
They explained that the review process by Korean Ministry were categorized into the source of pigs and the definition of DPF status, islet product release criteria, concerns about zoonosis, the use of immunosuppressants (Table 8), patient selection and inclusion/exclusion criteria, and the sample archive. Thus, islet xenotransplantation using adult pigs are ready for the clinical trial.
Clinical protocol for porcine islet xenotransplantation by Seoul group.

Dresden group
In 2017, Dresden Group demonstrated favorable outcome of experimental islet xenotransplantation without immunosuppression using a unique device in a nonhuman primate model of diabetes 27 . The donor pigs were female retired breeder Goettingen minipigs (Ellegard) with an average of 3–4 years and average pregnancies of three to four were used 27 . Before organ retrieval, the animals were housed under standard conditions (19°C–23°C; 40%–70% relative humidity; 12:12 h day: night cycle) and fed twice daily with special minipig pelleted food (Ellegard). Water was provided ad libitum.
Before this nonhuman study, they conducted 21 porcine islet isolations 28 . They used four young minipigs and 17 retired breeder minipigs and found that the usage of retired breeder animals resulted in high quality and quantity of islets. Therefore, for the nonhuman primate study, female retired breeder minipigs with an average age of 3–4 years and average pregnancies of three to four times were used. They isolated islets using Ricordi method and purified islets with discontinuous gradient centrifugation. Isolated islets were immobilized in alginate and put into the unique device (Fig. 1). They demonstrated that a diabetic nonhuman primate maintained excellent blood glucose levels more than 6 months with positive porcine C-peptide.

The chamber system for islet macroencapsulation and encapsulated porcine islets described by Dresden team. The system is built from two islet modules containing the islets immobilized in alginate, and central oxygen module. Oxygen-enriched gas mixture (95% oxygen) is directly injected into central oxygen module, and oxygen is delivered into islet modules. This system allows islet survival due to avoiding the ischemic issue.
Gene modified pigs: next generation of donor pigs
Australian group
Australian group used gene modified neonatal pig islets for the treatment of diabetic baboons
29
. They used alpha 1,3-galactosyltransferase knock out (GTKO) pigs and CD55-CD59-HT triple-transgenic pigs. CD55-CD59-HT triple transgenic pigs express human CD55, CD 59 and alpha 1,2-fucosyltransferase. These two kinds of pigs were mated to produce GTKO/CD55-CD59-HT piglets which are used as donors. 1-day-old to 3-day-old newborn piglets of either sex obtained from a registered pig supply unit were used
30
. They used the modified Korbutt method for islet isolation
30
. The digested tissue was washed thrice in ice-cold HBSS, filtered through a 500 µM sieve and plated into Petri dishes with culture media. Culture media consists of Ham’s F10 medium, 10mM glucose, 50 mM isobutylmethylxanthine, 10% pooled autologous pig sera, 2mM
Pittsburg group
Pittsburg group used gene modified pig for islet xenotransplantation 31 . They compared islets from two wild type pigs, two female GTKO pigs, and seven female human complement regulatory protein (hCD46) transgenic pig. The wild type pigs were female outbred large white pigs. The body weight of all pigs were more than 180 kg and ages were between 7 months and 2 years. Islets were isolated using Ricordi method followed by purification using discontinuous gradient centrifugation 32 .
A total of 85,000 to 100,000 IEQ/kg islets were transplanted into nine streptozotocin induced diabetic nonhuman primates (Table 9). In all diabetic nonhuman primate, ATG was administered for induction immunosuppression and mycophenolate mofetil (MMF), anti-CD154 were used for maintenance immunosuppression. Aspirin was also used to minimize coagulation related islet damages. At the time of transplantation, low molecular weight dextran sulfate, prostacyclin, and methyl prednisolone were administered to prevent IBMIR 33 .
Outcomes of preclinical study for gene modified porcine islet xenotransplantation into nonhuman primate by Pittsburg group.

Majority of transplanted nonhuman primates achieved insulin free status (Table 9). However, nonhuman primates which received islets from the gene modified porcine showed longer insulin free durations. The authors mentioned no complement activation were observed in nonhuman primates which received islets from the gene modified porcine.
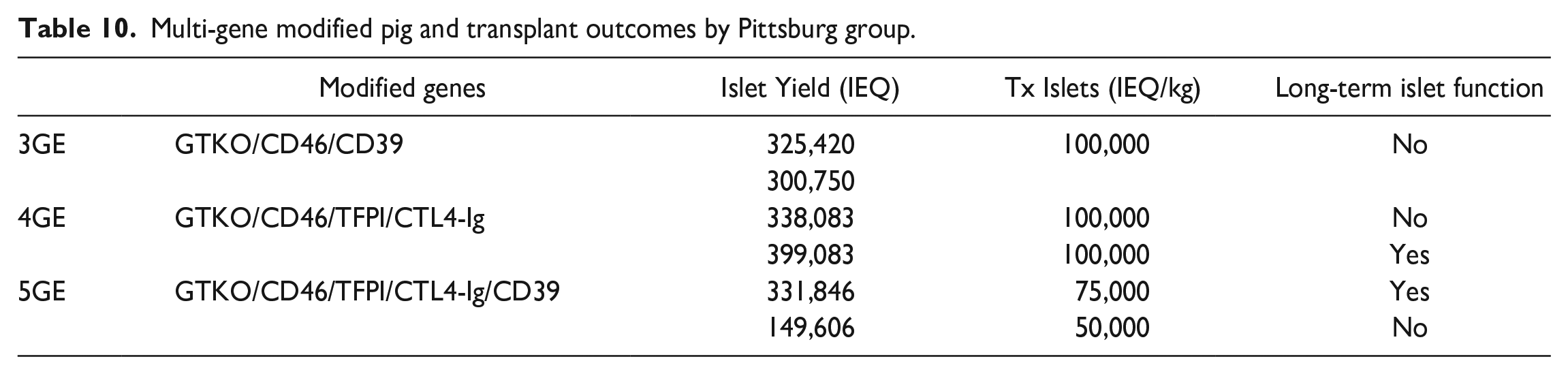
The Pittsburgh group further tested gene modified pigs for islet transplantation into streptozotocin induced diabetic nonhuman primates (cynomolgus monkeys) 34 . In addition to GTKO/CD46, they further modified with human CD39, human tissue factor pathway inhibitor (hTFPI), and porcine CTLA4-Ig. Human CD 39 was shown to decrease platelet activation. hTFPI was aimed at inhibition of IBMIR. The porcine CTLA4-Ig was used to inhibit the cellular immune response.
Three groups were created for this study and six female pigs aged 16 to 36 months, weighing 159–181 kg were sources of islets (Table 10). Islets were transplanted into streptozotocin induced five diabetic nonhuman primates. One nonhuman primate received isolated islets from two 3GE pigs. The other four nonhuman primates received islets from one 4GE or 5GE pigs respectively. All nonhuman primate received prostacyclin, methylprednisolone, dextran sulfate, and aspirin for anticoagulant and/or anti-inflammatory effects; ATG for induction, and MMF and anti-CD154mAb for maintenance immunosuppression.
Multi-gene modified pig and transplant outcomes by Pittsburg group.
To monitor early islet destruction after transplantation, porcine C-peptide levels were measured at 1, 2 and 24 h. Porcine C-peptide levels after 1-h transplantation were significantly lower in all nonhuman primate with 3-4-5 GE islets compared with previous nonhuman primates with no or one GE pig islets. This demonstrated islets from 3-4-5 GE pigs caused less immediate islet destruction which could reduce the IBMIR.
However, only two nonhuman primates demonstrated long-term islet function. They concluded that despite encouraging effect on early isle loss, GE islet grafts did not demonstrate consistency regarding a long-term success.
Belgian group “superislets”
Belgian group demonstrated that beta cell specific gene modified pig islets using adenoviral vectors to transduce modified GLP-1 and activated type 2 muscarinic receptor (M3R) enhanced insulin secretary ability 35 . They successfully transduced GLP-1 and M3R into both neonatal and adult porcine islets. Adult pigs were Belgian Landrace pigs more than 200 kg body weight. Insulin secretary abilities were 12.5-fold increase in neonatal islets and 6.1-fold increase in adult porcine islets.
More recently they created transgenic pig expressing the GLP-1M3R cassette under the porcine insulin promoter (InsGLP-1M3R) 36 . They used genetically modified both adult and neonatal porcine for islet isolation. The body weights of adult pigs were more than 200kg. Islets were isolated using collagenase digestion and discontinuous gradient purification for the adult pigs and Korbutt method for the neonatal pigs. They described islet yield from one pig was between 25,000 to 40,000 IEQ from neonatal pigs and 50,000 to 80,000 IEQ from adult pigs. However, they also mentioned islet isolation is reproducible/easy in neonatal and variable/difficult in adult pigs.
InsGLP-1M3R islets isolated from neonatal and adult pigs secrete up to 15-fold more insulin in response to glucose stimulation compared with wild-type islets. InsGLP-1M3R islets also proved to be more efficient in treating diabetes in a rodent model as shown by a significantly higher percentage of normoglycemic recipients and higher porcine C-peptide levels. Due to high insulin secretary ability, they named InsGLP-1M3R islets as “superislets.”
Conclusion
Fetal pigs were used for the first clinical trial of islet xenotransplantation by Swedish group followed by New Zealand group. To scale up islet isolation, Korbutt and his colleagues used neonatal pigs and developed “Korbutt neonatal islet isolation method” which is a breakthrough development. In fact, recent clinical trials in New Zealand, Argentina, Mexico, and China were all used neonatal pigs with the Korbutt method. Due to reproducibility and easiness of islet isolation, neonatal pigs have been used for the clinical trials. However, Seoul group developed an excellent protocol using adult pigs and ready for clinical trials.
For the next generation of donor pig, gene modification seems attractive approach. Islets from gene modified pigs can cause less IBMIR and less immunogenic reaction which should contribute clinical outcomes. Eventually, “superislets” should improve the efficacy of islet transplantation which might more effective than pancreas transplantation.
With advanced donor pigs, islet xenotransplantation might become a suitable treatment for the majority of type 1 diabetic patients.
Footnotes
Acknowledgements
No acknowledgment.
Ethical Considerations
This is a review article, and all information was gathered from published literatures. Therefore, no ethical assessment was required.
Informed Consent/ Patient Consent
This study did not conduct any clinical trials, therefore, no informed consent existed.
Author Contributions
SM conceptualized and designed the review, conducted the literature search and analysis, interpreted the findings, and prepared the manuscript. All other authors conceptualized and designed the review, conducted interpreted the findings and reviewed the manuscript. All authors approved the final manuscript.
Trial Registration Number/Date
This study did not conduct any clinical trials, therefore, no registration number existed.
Grant Number
There is no grant number.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Japan IDDM network, type 1 diabetes research fund
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All information was gathered from published literatures. Therefore, all data are available.
