Abstract
Porcine islets are a promising resource for xenotransplantation. However, low efficacy of islet isolation because of their marked fragility remains a problem. Recently we found that the standard purification method using COBE 2991 cell processor (COBE) with Ficoll density gradient solution damaged islets mechanically by high shearing force. In this study, we evaluated our new purification method using large plastic bottles for the efficacy of islet purification. Ten porcine pancreata were used. The average warm ischemic time was over 40 min; therefore, these pancreata were considered to be in a marginal condition. After digestion, the digested tissue was divided into three groups. Each group was purified using either top loading method with bottle (top group) or bottom loading method with bottle (bottom group) or standard COBE method (COBE group). Islet yield per pancreas weight (IEQ/g) and the rate of postpurification recovery in the top group were significantly higher than the COBE group (top: 8060 ± 1652 IEQ/g, bottom: 4572 ± 614 IE/g, COBE: 3900 ± 734 IE/g. p < 0.02 in top vs. COBE; top percentage of recovery: 99.3 ± 12.3%, bottom: 62.6 ± 8.8%, COBE: 49.5 ± 6.7%, p < 0.02 in top vs. bottom and COBE). The average sizes of purified islets in the top and bottom groups were significantly larger than COBE group (Average diameter top: 156 ± 8 μm, bottom: 147 ± 6 μm, COBE: 119 ± 6 μm, p < 0.01 in top vs. COBE and in bottom vs. COBE), which indicated that bottle method can reduce shear force during purification. Our new purification using top loading bottle method enabled us to obtain a high yield of porcine islets from marginal pancreata.
Introduction
Islet transplantation is a promising treatment for type 1 diabetic patients (19,20). However, given the shortage of donor pancreata, alternative sources have been sought. Porcine islets are considered to be an unlimited source for xenotransplantation (1,13,16,18). However, xenotransplantation has been limited by low efficacy of islet isolation because of the marked fragility and rapid dissociation of porcine islets. Islet isolation process consists of pancreas distension, pancreas digestion, and islet purification (2,4). Purification process is one of the most difficult procedures for islet isolation; however, research into optimal purification methods is limited.
Recently we found that the standard purification method using COBE 2991 cell processor with Ficoll density gradient solution damaged islets mechanically by high shear force (21). Especially the unique design and shape of the COBE bag with narrow segment caused strong shear forces that made the islets come apart (21). On the other hand, our preliminary study indicated that using large hard cylindrical plastic bottles with less viscosity gradient solution improved the efficacy of islet purification substantially (24). The shear force could be minimized or eliminated because the bottles do not have a narrow segment and no centrifugation is required during loading and collecting processes. In addition, we have established a top loading method with continuous density gradients instead of the traditional bottom loading method using discontinuous density gradients, which has been a standard bottle method.
In this study, we performed experiments for head to head comparison of COBE versus bottle method (top or bottom loading) using marginal porcine pancreata.
Materials and Methods
Porcine Islet Isolation
Porcine pancreata were obtained at a local slaughterhouse. About 30 min after the cessation of heart beating, the operation was started. After removing the duodenum from the pancreas, we immediately inserted a cannula into the main pancreatic duct and infused about 200–250 ml of extracellular-type trehalose-containing Kyoto (ET-Kyoto) solution (Otsuka Pharmaceutical Factory Inc., Naruto, Japan) (3,9). We removed fatty tissues and put the pancreas into the two-layer [ET-Kyoto/oxygenated perfluorochemical (PFC)] preservation container (7). Warm ischemic time (WIT) was defined as the time between the cessation of heart beating and the placement of the pancreas into the preservation solution. Cold ischemic time (CIT) was defined as the time between the placement of the pancreas into the preservation solution and the start of islet isolation.
Islet isolation was conducted based on the method described in the Edmonton protocol (5,6,14,15,19). Briefly, after decontamination of the pancreas, the ducts were perfused in a pressure-controlled fashion with cold Liberase HI (1 mg/ml; Roche Molecular Biochemicals). After distention, the whole pancreas was cut into about nine pieces, placed in a sterilized Ricordi chamber (1 L), and shaken gently. While the pancreas was being digested by recirculating the enzyme solution through the Ricordi chamber, we monitored the extent of digestion with dithizone staining by taking small samples. Once almost all digest had been collected, RPMI-1640 medium (Gibco) with 10% fetal bovine serum was added into the system for dilution, and then the system was cooled to stop further digestive activity. The digested tissue was collected and washed with University of Wisconsin (UW) solution. The phase I period was defined as the time between the placement of the pancreas in the Ricordi chamber and the start of collecting the digested pancreas. The phase II period was defined as the time between the start and end of the collection.
Islet Purification
The pancreatic digest was separated into exact halves. One half was purified using COBE 2991 cell processor (COBE), (CaridianBCT, Denver, CO) as described previously (8,10,12). The other half was separated into two parts again, and one quarter was purified with the “top loading” bottle method (top) and the other quarter was done with the “bottom loading” bottle method (bottom). All these methods were conducted with a continuous density gradient with iodixanol-ET-Kyoto solution. Since iodixanol has low viscosity, it is possible to apply slower centrifugation for the dispersion than Ficoll. For the continuous density solution, low-density (density 1.077) and high-density (density 1.095–1.125) iodixanol-ET-Kyoto solutions were produced by changing the volumetric ratio of iodixanol and ET-Kyoto solution. A schematic of the bottle method is shown in Figure 1. For the bottle purification, we used two 500-ml flat-bottom plastic containers; one was used for “top” and the other was used for “bottom.” In the top method, the gradient consisted of 50 ml of high density at the bottom layer and then 300 ml of continuous density gradient in the second layer and 50 ml of low-density including the pancreatic digest in the third layer and 50 ml of MEM solution on the top as a cap solution. In bottom method, 100 ml of high density including the digest was at the bottom and 300 ml of continuous density gradient was in the second layer and 50 ml of cap solution was on the top. The gradient was produced with a gradient maker (Fig. 1A). For the bottle method, we invented and used a “tip-bent” candy cane-shaped stainless pipe (Fig. 1B) (Umihira Works Co., Kyoto, Japan) to pour the solution into the bottle. That form enabled us to minimize the convection current and hold the gradient. All systems were centrifuged at 1000 rpm for 5 min at 4°C. After centrifugation, islets were seen in the upper layer of the bottles (Fig. 2).

Our new purification method with large bottles. (A) The schema of the bottle purification method. First the continuous gradient solution including the digested tissue was generated in large bottles with a gradient maker. Then they were centrifuged at 1000 rpm for 5 min at 4°C and the solution was collected into 10 tubes. (B) A “tip-bent” candy cane-shaped stainless pipe used for making the gradient. (C) A picture during making the gradient (bottom loading).
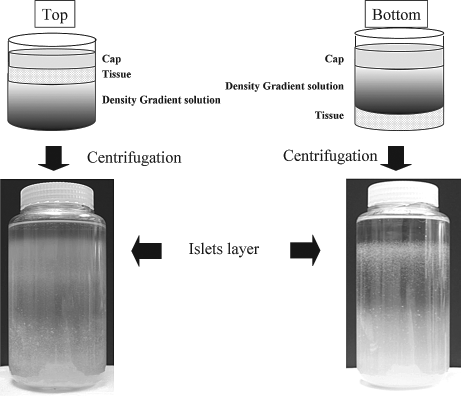
Pictures of the bottles after centrifugation. Left: Top loading. Right: Bottom loading. Islets exist in the upper layer in both methods.
After centrifugation, the gradient solution was collected into 10 tubes from the top to the bottom (Fig. 1A). To confirm stability of continuous density gradient, we measured the density of each 10 fractions before applying this method for islet purification. In the COBE method, about 80 ml of the solution was left in the COBE bag structurally. On the other hand, whole solution was collected in the bottle method, which can prevent the loss of islets. Samples were taken from each tube, stained with dithizone (Sigma Chemical Co., St. Louis, MO), and assessed for islet purity. Islet-rich tubes were selected and the tissues were collected.
Islet Evaluation
Islet preparations were evaluated for yield, purity, and morphology by using dithizone (3 mg/ml) staining. The crude number of islets in each diameter class was determined by counting islets using an optical graticule. The crude number of islets was then converted to the standard number of islet equivalents (IEQ; diameter standardizing to 150 μm) (17). Gross morphology was qualitatively assessed by scoring the islets for shape (flat vs. spherical), border (irregular vs. well rounded), integrity (fragmented vs. solid/compact), uniformity of staining (not uniform vs. perfectly uniform), and diameter (least desirable: all cells <100 μm; most desirable: more than 10% of the cells >200 μm) (6). Each parameter was graded from zero (the worst) to 2 (the best), so that the worst islet preparations were given a cumulative score of zero and the best a score of 10. Spherical, well-rounded, solid/compact, uniformly stained, and large islets were characterized as the best islets.
Islet viability after purification was assessed using trypan blue staining. Fifty islets were inspected and their individual viability was determined visually, followed by calculation of their average viability.
In vitro islet function was assessed by monitoring the insulin secretory response of the islets during glucose stimulation as described previously (19). Briefly, 5,000 IEQ were incubated with either 2.8 or 25 mM glucose in RPMI-1640 for 2 h at 37°C in a 5% CO2 atmosphere. The supernatant was collected and insulin levels were determined using an enzyme-linked immunosorbent assay kit (ALPCO Diagnostics, Windham, NH). The stimulation index was calculated by determining the ratio of insulin released from islets in the high-glucose concentration to the low-glucose concentration. The data were normalized by total DNA.
For in vivo assessment of the islet function, nude mice (Harlan, Houston, TX) rendered diabetic by a single injection of streptozotocin (STZ) at a dose of 120 mg/kg were used. When the nonfasting blood glucose level exceeded 350 mg/dl for 2 consecutive days, the mice were considered to be diabetic. The 10,000 IEQ porcine islets obtained from each group were transplanted into the renal subcapsular space of the left kidney of a diabetic nude mouse immediately after the isolation. During the 30-day posttransplantation period, the nonfasting blood glucose levels were measured at three times per week. Normoglycemia was defined when two consecutive blood glucose level measurements showed less than 200 mg/dl. No statistical differences in either pretransplantation blood glucose levels or body weight were observed among the three groups. These experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of Baylor Health Care System.
Statistical Analysis
All results were expressed as the means ± SE. Differences among the three groups were analyzed by ANOVA followed by Student's t-test with Bonferroni correction. Values less than p < 0.05 were considered significant in ANOVA and in Student's t-test with Bonferroni correction. Differences of ratio among the three groups were analyzed by chi-squared test with Ryan method.
Results
Stability of the Density Gradient in the Bottle Purification Methods
After centrifugation, the solution was collected into 10 tubes. We measured the density of each tube, and confirmed that the continuous density gradients were maintained during the whole purification procedure in both top and bottom methods (Fig. 3).

Continuous density gradient was maintained in both top and bottom loading methods during the purification. After centrifugation, the solution including the digested tissue was collected into 10 tubes and the density of each tube was measured. The density of the low-density solution was 1.077 while the high-density solution was 1.095. The experiments were performed three times independently.
Porcine Islet Isolation Characteristics
A total of 10 porcine pancreata were used in the present study. The islet isolation variables are shown in Table 1. The average of the weight of used pancreata was 136 ± 17 g. The average of the warm ischemic time was 46 ± 2 min; therefore, these pancreata were in a marginal condition.
Porcine Islet isolation Variables

Data are expressed as mean ± SE. IEQ, islet equivalent.
Postpurification variables are shown in Table 2. Islet yield (IEQ) per pancreas weight (IEQ/g) after purification was significantly higher in the top group than the COBE group (top: 8060 ± 1652 IEQ/g, bottom: 4572 ± 614 IEQ/g, COBE: 3900 ± 734 IEQ/g; p < 0.02 in top vs. COBE). The rate of postpurification recovery was significantly higher in the top group than in both the bottom and COBE groups (percentage of recovery top: 99.3 ± 12.3%, bottom: 62.6 ± 8.8%, COBE: 49.5 ± 6.7%, p < 0.02 in top vs. bottom and p < 0.001 in top vs. COBE, respectively).
Postpurification Variables
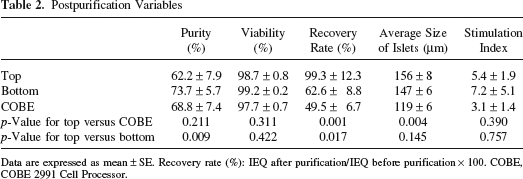
Data are expressed as mean ± SE. Recovery rate (%): IEQ after purification/IEQ before purification × 100. COBE, COBE 2991 Cell Processor.
The Size of Isolated Islets After Purification
The average diameter of purified islets and the rate of more than 200 μm size islets were significantly higher in the top and bottom group than in the COBE group (average of the diameter of islets top: 156 ± 8 μm, bottom: 147 ± 6 μm, COBE: 119 ± 6 μm, p < 0.01 in top vs. COBE and in bottom vs. COBE; percentage of >200 μm size islets top: 26.0 ± 3.2%, bottom: 20.0 ± 3.0%, COBE: 10.8 ± 2.6%, p < 0.02 in top vs. COBE and in bottom vs. COBE) (Figs. 4 and 5).

Morphology of postpurification islets. The same volume samples after purification were taken from three methods. In the top method, large islets were seen. COBE, COBE 2991 Cell Processor. Original magnification: 40×. Scale bars: 150 μm.

Postpurification islet yield and the size of islets. Left: Postpurification IEQ/g in the three groups. Right: Ratio of the number of more than 200 μm size islets in the three groups. *p < 0.02.
Assessment of Islet Function In Vitro and In Vivo
Stimulation index in the bottom group was higher than the other two groups, although there was no significant difference (top: 5.4± 1.9, bottom: 7.2± 5.1, COBE: 3.1 ± 1.4, respectively) (Table 2).
To assess the islet graft function in vivo, 10,000 IEQ islets of each group were transplanted under the kidney capsule of STZ-induced diabetic nude mice. The blood glucose levels in 7 of 11 mice (64%) in the top group, 5 of 11 mice (45%) in the bottom group, and 2 of 5 mice (40%) in the COBE group decreased and reached normoglycemia (Table 3). The ratio of the cured mice in the top group was the highest among the three groups, although there was no statistical significance.
In Vivo Functional Assay
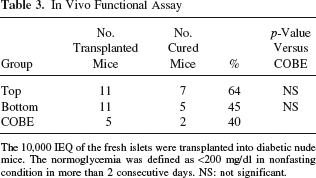
The 10,000 IEQ of the fresh islets were transplanted into diabetic nude mice. The normoglycemia was defined as <200 mg/dl in nonfasting condition in more than 2 consecutive days. NS: not significant.
In summary, the purification with top loading method resulted in the highest yield and the largest size of islets. These data suggest that top loading was the best purification method for porcine islet isolation among the three groups.
Discussion
Islet allotransplantation is a promising therapeutic method for diabetes. However, this relatively safe and minimally invasive therapy still faces the difficult problem of a limited supply of human pancreas donors. Porcine islet cells are a good substitute for human islet cells. However, porcine islets are known to be difficult to isolate because of a lack of a surrounding islet capsule (11) and the fragility to be easily fragmented (22) during isolation.
We recently demonstrated that the purification method using large bottles could improve the islet isolation outcome for porcine pancreas (24). In that study, the bottom loading method and discontinuous density solution were used for the purification. However, continuous density gradient should separate islets from other cells more efficiently than discontinuous solution. In addition, the previous study was not a direct head-to-head comparison. Therefore, the present study is not only the first report of a direct comparison of porcine islet purification methods between COBE and large bottles, but also a direct comparison between top loading and bottom loading methods with continuous density gradient.
Another modification used in this study is that the digested tissue was mixed with low-density solution instead of preservation solution such as UW solution, which is the standard method in the case of COBE. When we first started the top loading method, we mixed the digest in UW solution and loaded it to the upper layer, which made a wall of tissue between the density gradient solution and the tissue solution because the density of UW solution is much lower (1.040 g/ml) than light solution (1.077 g/ml). The tissue was precipitated at the bottom of UW solution before the centrifugation and this condensed tissue may cause a lot of islets to be trapped within the acinar tissue and dragged into the bottom. This should not occur in COBE, since the COBE bag is centrifuged during the tissue loading. On the other hand, when the tissue was mixed with the low-density solution, there was not such a wall because we used the same density of solution for the tissue and light solution. Then the relatively heavy acinar tissues started to move to the bottom of the bottle soon after they were loaded, before the centrifugation. This may minimize the dragged islets.
With top loading method, we were able to isolate large quantities of islets from porcine pancreata. Several factors may be considered to be important. First, bottle methods can minimize the shearing force during the centrifugation for the purification. As shown previously, the COBE bag has a narrow segment and islets suffered significant shearing force during passage through this narrow segment. Since the bottle does not have such a narrow segment, the shear stress could be substantially reduced. In fact, this could be the reason why the size of purified islets in both top and bottom method was significantly larger than COBE. Since porcine islets are fragile, minimizing the mechanical damage during the isolation is critical.
Second, the pancreata used in this study were in a marginal condition. The average warm ischemic time was more than 45 min, due to the speed of dressing the pigs at a local slaughterhouse. Even 30 min of warm ischemic condition can cause severe apoptosis in porcine islets (23); therefore, islets in this study are considered to be significantly damaged. Indeed, for the in vivo functional analysis, 10,000 IEQ islets were required to cure diabetic mice. To put it differently, however, our modified isolation method enables us to obtain functional islets from porcine pancreata under such severely adverse conditions. Although the differences did not reach statistical significant, the islets in the top group had the highest quality according to the in vitro and in vivo evaluation.
Third, top loading has several advantages over bottom loading. In the case of bottom loading, the purity of islets was the highest among the three groups but IEQ/g was less than top loading. We assume that it is partially because many islets could be entrapped by the vicinal acinar tissue during making the density gradient solution, and they could not float up to the upper layer. In addition, the relatively longer time when the loaded islets were exposed to the high-density solution during bottom loading might damage the islets, whereas islets were exposed to the low-density solution in top loading method.
Moreover, our bottle purification method is simple and easy to perform. It can eliminate the cost and experience of the COBE 2991 Cell Processor, and it may contribute to the spread of islet isolation to many institutes. We plan to apply this method to human islet isolation, and we believe that it will also improve human islet isolation.
In conclusion, we developed a new purification method using large bottles with a top loading. This method enabled us to obtain an extremely high yield of porcine islets.
Footnotes
Acknowledgments
The authors wish to thank Ms. Yoshiko Tamura and Mr. Greg Olsen (Baylor University Medical Center) for their technical supports. This research is partially supported by All Saints Health Foundation and Otsuka Pharmaceutical Factory Inc. The authors declare no conflict of interest.
