Abstract
Isolation of islets from market-sized pigs is costly, with considerable islet losses from fragmentation occurring during isolation and tissue culture. Fetal and neonatal pigs yield insulin unresponsive islet-like cell clusters that become glucose-responsive after extended periods of time. Both issues impact clinical applicability and commercial scale-up. We have focused our efforts on a cost-effective scalable method of isolating viable insulin-responsive islets. Young Yorkshire pigs (mean age 20 days, range 4–30 days) underwent rapid pancreatectomy (<5 min) and partial digestion using low-dose collagenase, followed by in vitro culture at 37°C and 5% CO2 for up to 14 days. Islet viability was assessed using FDA/PI or Newport Green, and function was assessed using a glucose-stimulated insulin release (GSIR) assay. Islet yield was performed using enumeration of dithizonestained aliquots. The young porcine (YP) islet yield at dissociation was 12.6 ± 2.1 × 103 IEQ (mean ± SEM) per organ and increased to 33.3 ± 6.4 × 103 IEQ after 7 days of in vitro culture. Viability was 97.3 ± 7% at dissociation and remained over 90% viable after 11 days in tissue culture (n = ns). Glucose responsiveness increased throughout maturation in culture. The stimulation index (SI) of the islets increased from 1.7 ± 2 on culture day 3 to 2.58 ± 0.5 on culture day 7. These results suggest that this method is both efficient and scalable for isolating and maturing insulin-responsive porcine islets in culture.
Introduction
Allotransplantation of islets is partially or totally curative of type 1 diabetes mellitus (T1DM) for periods of months to years, yet utilization remains limited because of the need for permanent immunosuppression and shortage of pancreas donors (19,43,45,52). Xenotransplantation of porcine islets within biocompatible immunoisolation devices has the potential to address both issues (10,31,34,44). Pigs are considered an appropriate islet source because of their similar size and physiology to humans, nearly identical molecular structures of human and porcine insulin, a long history of safe use of porcine insulin in treatment of human diabetes, large litter size, and relative ease of husbandry. Recent work in streptozotocin-diabetic immune-deficient mice suggests that deposition of the islet amyloid polypeptide, which promotes β-cell apoptosis, is seen at 4 weeks and worsens by 8 weeks when human islets were transplanted. However, no islet amyloid polypeptide deposition was seen when neonatal or adult porcine islets were transplanted for up to 195 days. These data support the potential of porcine islet transplantation as therapy for human diabetes (39).
Transplantation of alginate-encapsulated pig islets without pharmacologic immunosuppression has reversed diabetes in rodent and nonprimate animal models (5,8, 9,11,33,41) and improved metabolic control in a human subject for 14 months with the viability of transplanted islets confirmed, 9.5 years posttransplant (12). With continued encouraging progress in transplantation of encapsulated porcine islets into nonimmunosuppressed recipients, the need for improved and less costly methods of islet isolation and culture becomes more important for widespread utilization of the therapy in humans to commence.
Harvesting and processing of islets from market-sized pigs are costly and difficult. Multiple staffs are required for each organ harvest and digestion, and isolation takes several hours and requires extensive equipment and supplies (e.g., variable batches of effective collagenase enzymes, Ricordi chamber, organ preservation, isolation process, and culture media and testing reagents.) Adult porcine islet isolation is accomplished with enzymatic and mechanical digestion of the pancreas followed by endocrine tissue purification using Ficoll gradient centrifugation. Several factors affect the final yield of porcine islets from adult pigs including breed, quality of organs, and the inherent fragility of adult porcine islets that results in substantial fragmentation during isolation, storage at low temperature, and culture (47,51). Consequently, the islet yield from each pancreas is substantially lower than the theoretical yield based on the intrinsic islet count. The overall cost of using market-sized animals includes the substantial resources required to raise animals to an appropriate size.
Use of neonatal or fetal pigs presents different problems because their pancreases contain indistinctly demarcated islets and are structurally delicate. Culture of fetal and neonatal porcine pancreas following partial collagenase digestion and without mechanical maceration or endocrine tissue concentration yields viable islet-like cell cultures (ICCs) (14,21,25). While neonatal and fetal ICCs proliferate in culture, they are nonfunctioning at the time of isolation and do not develop an insulin secretory response to glucose challenge for 2 weeks or more. Indeed, ICCs from fetal pigs may take as long as 3 months posttransplant to develop function (26). Consequently, maturation and insulin responsiveness occurs after encapsulation and transplant making dosing imprecise at best.
The possibility of disease transmission from grafted pig tissue to humans is a concern. Establishment and maintenance of pathogen-free breeding stock in specialized facilities with strict hygiene and sterility protocols is foundational to achieving a scalable supply of pig islets (7,24,49). Despite such precautions, exposure to porcine endemic retrovirus (PERV), which is present in all pig strains, cannot be prevented (36). The data pertaining to PERV transmission are inconclusive. Reports have shown human and other animal cells infected with PERV from pig cells in vitro as well as in vivo (37,53). Therefore, use of porcine islets has theoretical risk of cross-species PERV infection. In other studies, no PERV cross-species infection was detected in human, monkey, or rodent studies lasting 140 days to 7 years with and without immunosuppression using nonencapsulated or encapsulated fetal, neonatal, or adult porcine islets (4,18,35,38,48. A study by Garkavenko et al. looked at 18 human recipients of porcine islets who had been monitored up to 9 years and found no evidence of PERV or any other viral infection transmission (15). Data show that natural antibodies found in human sera may prevent in vivo transmission of PERV (32).
An isolation and culture method that produces high counts of porcine IEQs with high viability and insulin responsiveness to glucose prior to transplant is desirable. The purpose of this study is to develop a reproducible and scalable method for porcine islet isolation and culture that produces large numbers of viable islets that are glucose responsive before transplant.
The aims of this study are to demonstrate the following:
Young preweaned pigs (aged 4–30 days) are a more effective source of viable porcine islets for transplants. Determine optimum age range for islet isolation.
Partial digestion islet isolation method enhances islet survival and is not enzyme dependent.
Insulin-producing tissue matures in tissue culture, and glucose responsiveness progressively improves as acinar tissue simultaneously necroses and sloughs off during culture.
Proliferation and glucose responsiveness during culture of young porcine (YP) islets enable more informed dosing decisions and confirm insulin secretory function prior to transplant.
Materials and Methods
Islet Isolation Process and Maturation of Porcine Islets
Pancreases from young male Yorkshire pigs (4–30 days, n = 65, S&S Farms, Ramona, CA, USA) were removed using rapid surgical procurement (<5 min) and placed in Organ Preservation Solution (OPS, Mediatech, Manassas, VA, USA); cold ischemia time was limited to <30 min. All animal procedures including but not limited to monitoring, surgery, and euthanasia were performed with approval from the University of California Institutional Animal Care and Use Committee (IACUC) at the University of Irvine. All efforts were made to reduce the quantity of animals required for this study. Pancreases were then washed in cold (4°C) Hank's balanced salt solution (HBSS; Corning Cellgro, Manassas, VA, USA) supplemented with 2-[4-(2-hydroxyethyl)piperazin-1-yl] ethanesulfonic acid (HEPES; Corning Cellgro) and trimmed of surrounding adipose and lymphatic tissue in a laminar flow hood. Pancreatic tissue was then enzymatically digested from the majority of surrounding acinar tissue by mincing in either a low-dose CIzyme™ collagenase MA/BP protease enzyme mixture (0.25 mg/ml collagenase MA; 0.04 mg/ml BP protease in HBSS, VitaCyte LLC, Indianapolis, IN, USA) or a low-dose Sigma Type V (2.5 mg/ml in HBSS, Sigma Aldrich, St. Louis, MO, USA).
Tissue was then allowed to digest in 100 ml of enzyme in HBSS in a 250-ml flask (Greiner, Monroe, NC, USA) for 17 ± 2 min at 37°C, while shaking at 60 rpm. Digestion process was then stopped using cold (4°C) HBSS supplemented with HEPES and 10% bovine serum albumin (BSA; Sigma-Aldrich) added to the digestion flask. Digested tissue was then filtered through a 500-μm metal mesh funnel (TWP Inc., Berkeley, CA, USA) using HBSS supplemented with 10 mM HEPES and 10% BSA (HBSS-Sup) into two to three 250-ml conical tubes (Corning Lifesciences, Tewksbury, MA, USA) to remove undigested tissue greater than 500 μm. Amount of undigested tissue was weighed, and the percentage lost was calculated. Tissue was then centrifuged for 2 min at 200 × g and washed twice using 200 ml of HBSS-Sup. Islet tissue clusters were then washed twice in Recovery Maturation Media (Optatio LLC, Chino, CA, USA).
Islet tissue clusters (50–500 μm) were then cultured and allowed to mature into complete islets. Islets were cultured at 37°C, 5% CO2 in T-175 suspension flasks (Corning Inc.) at a tissue density of approximately 80 μl of tissue in 40 ml of culture media. For the first 48 h postisolation, islet tissue clusters were matured in a novel Recovery Maturation Media supplemented with 215 mM aprotinin (Sigma-Aldrich), 0.5 mM Pefabloc (Sigma-Aldrich), 417 mM dornase alfa (Genentech, San Francisco, CA, USA), and 10% porcine serum (Lampire Biological Laboratories Inc., Ottsville, PA, USA). After 48 h, islet tissue clusters were removed, centrifuged for 2 min at 200 × g, and resuspended in a novel maturation media (Optatio LLC) supplemented with 10% porcine serum (Lampire Biological Laboratories Inc.) for 8–9 days in order to fully mature. Every 48 h, 50% media changes, using supplemented maturation media, were performed to replace media and remove debris.
Characterization of Porcine Islets
Aliquots of tissue were evaluated and enummerated, checked for viability and function as well as for amount of apoptosis every 48 h during media changes. Islet count (IC) and IEQ were determined by staining a 100-μl aliquot with 1 ml dithizone (DTZ; MP Biomedicals), waiting for 5 min, and observing islets at 25× on a standard stereomicroscope (Max Erb, Santa Ynez, CA, USA) containing a 10× eye piece graticule (29,40,42). DTZ staining was also used to determine percentage of purity over the maturation period. Percentage of purity was determined by the amount of positive DTZ staining divided by the amount of unstained regions within each cluster of tissue. Percentage of viability was analyzed using either fluorescein diacetate (FDA; Thermofisher Scientific, Swedesboro, NJ, USA) and propidium iodide (PI; MP Biomedicals) or Newport Green (Invitrogen, Carlsbad, CA, USA)/PI, imaged using confocal microscopy (Zeiss LSM510; Thornwood, NY, USA), and quantified with a Microplate reader (Tecan Infinite F200, Magellan V7; Tecan, Männedorf, Switzerland) (20,28,30).
Islet function during maturation was determined using glucose-stimulated insulin release (GSIR) (13,27,46). Porcine islets were incubated for 1 h in corresponding order in low glucose (2.8 mM, Fisher Scientific, Fairlawn, NJ, USA), high glucose (28 mM), high glucose (28 mM) plus 3-isobutyl-1-methylxanthine (IBMX, 50 μM, Sigma Aldrich), and then low glucose (2.8 mM). Stimulation index (SI) was calculated as the ratio of insulin [determined by enzyme-linked immunosorbent assay (ELISA); Mercodia, Winston Salem, NC, USA] secreted in high glucose over the amount of insulin secreted in low glucose. Maximum stimulation index (MX) was calculated as the amount of insulin secreted in high glucose plus IBMX divided by the amount of insulin secreted in high glucose (13,27,46).
Porcine islet apoptosis was analyzed by incubation of porcine islets for 10 min in YO-PRO-1 and PI (Membrane Permeability/Dead Cell Apoptosis Kit; Invitrogen) and imaged using confocal microscopy (2,17).
Pancreas structure and composition was analyzed using standard hematoxylin and eosin (H&E, Fisher Bioreagents, Waltham, MA, USA) staining. Islet cellular composition was determined using immunohistochemistry for insulin (Abcam, Cambridge, MA, USA) (22).
Statistical Analysis
Statistical analysis was carried out by Student's t test. Data are presented as mean plus or minus the standard error of the mean (mean ± SEM). ANOVA and either Dunnett's post hoc, for adult porcine comparisons, or Tukey's test, for comparisons of YP characteristics, were used to determine significance of differences between experimental groups with the level of significance set at p < 0.05.
Results
Comparison of Adult and Young Pig Pancreas and Islet Structure
For tissue structural comparison, tissue samples stained with H&E were compared between an adult market weight pigs' (25–35 kg) pancreases and YP pancreases. Histology revealed YP pancreases were less dense and had diffusely scattered insulin-positive cells without well-demarcated islet structures, in contrast to the well-defined islets found in the adult pig pancreas (Fig. 1). These results demonstrate that the YP pancreas is less developed than the adult pancreas, allowing for islets to be isolated and matured in tissue culture, rather than in vivo, which results in fragmented adult islets post isolation.
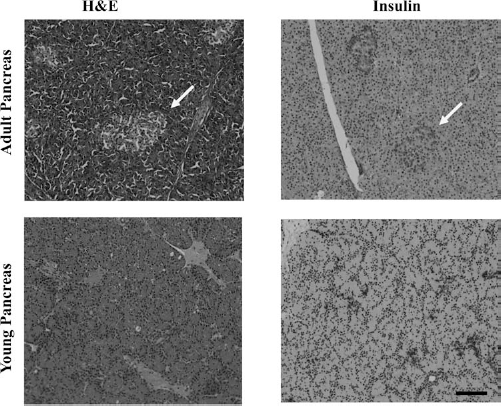
Histological comparison of adult porcine pancreas versus YP pancreas. Samples were taken from splenic end of the pancreases from either adult (market weight) or young pigs (15 days old). Pancreas samples were fixed and either stained with hematoxylin and eosin stain (H&E) or anti-insulin and hematoxylin (Insulin). Adult pancreas showed complete islet formation, denoted by arrows, and overall denser cell populations (denser volume of nuclei), while the YP pancreas showed no visible intact islet formations and unstructured insulin and glucagon staining when compared to adult porcine pancreas. All images taken at 20×. Scale bar: 100 μm.
YP Islet Isolation and Maturation
During islet tissue culture (37°C/5% CO2), we observed a progressive loss of acinar tissue surrounding the islets, which resulted in an overall increase in islet purity (Fig. 2) Immediately following isolation, islets were on average 8.5 ± 1.1% pure and increased during maturation to 78 ± 1.5% after 7 days of culture (Table 1, Fig. 2B). IC and IEQ each displayed approximately an approximate threefold expansion during maturation. ICs increased from 10.4 ± 3.3 × 103 postisolation to 32.0 ± 5.8 × 103 after 7 days of culture; IEQ increased from 12.6 ± 2.1 × 103 postisolation to 33.3 ± 6.4 × 103 after 7 days of culture. Full maturation of YP islets was reached on day 8 of in vitro tissue culture, with no YP islets exceeding 350 μm in diameter and approximately 90% of islets were between 50 and 150 μm (Table 2).

Islet characterization: IEQ, purity, viability, and function over maturation in vitro. Islet samples taken at 0, 1, 3, 5, 7, 9, and 14 days postdigestion, during islet maturation. (A) Samples stained with DTZ for IEQ, calculated as percentage of DTZ-positive staining, during in vitro tissue culture. (B) DTZ-stained samples were also used to assess purity, and FDA/PI was used to assess viability. Purity (B, ×) was calculated as percentage unstained tissue, and islet viability (B, ▪) was calculated as percentage alive (FDA, green)/percentage dead (PI, red). (C) For islet function 100 islets were incubated at 37°C, 5% CO2, for 1 h, in ▪ 2.8mM glucose then ▪ 28mM glucose, followed by ▪ 28mM glucose plus 50 μM IBMX and then □ 2.8 mM glucose. Samples were analyzed for insulin release using a standard porcine insulin ELISA. (D) Image of islet sample stained with FDA/PI after 5 days of culture shows primarily cell death of surrounding tissue, while internal islet remains viable. Graphed results are displayed as mean ± SEM per pancreas.
Islet Maturation Over 14 Days in Tissue Culture

Islets were partially enzymatically dissociated and allowed to mature in tissue culture (37°C, 5% CO2) for 14 days. Islet samples were taken directly following enzymatic digestion, after 3, 7, and 14 days in culture. Islets were stained with DTZ to quantity number of islets (IC), percentage of stained versus unstained islets (IEQ), and islet purity (percentage of DTZ-positive vs. nonstaining tissue). Percentage of islet sizes was determined using a 10 × 10 eyepiece graticule in a stereomicroscope (magnification: 25×, n = number of pancreases). No islets greater than 300 μm in diameter were observed. Results displayed as mean ± SEM.
Comparison of Porcine Islet Isolation Models

YP islets procurement and islet characteristics were compared to other possible sources for clinical islet transplant. Fetal pigs were obtained by cesarean, while neonatal pigs and young were obtained using natural birth. Table adapted from Dufrane et al. (7). Parameters and characteristics for partially digested YP islets have been added (Young) to compare with current techniques. GTP, Good Tissue Practice; GMP, Good Manufacturing Practice.
Age Comparison During Islet Isolation and Maturation
Islet isolations were separated into the following age categories: 4–10 days, 11–18 days, 20–30 days, and adult. Islets isolated from YP islets showed an increase in IEQ, purity, and viability when compared to islets isolated from a 5-g piece of the tail of the adult market weight pig (6 months) (Table 3). Islets isolated from 4-to 30-day-old pigs had an average of 5,343 ± 297 IEQ/g of pancreas, while islets isolated from adult pigs had an average of 1,500 ± 126 IEQ/g of pancreas. There was no significant difference (p < 0.05) between groups with pigs younger than 30 days for IEQ per gram of pancreas, but there was an increase in IEQ for the islet preparation for islets isolated from 11- to 18-day-old and 20- to 30-day old pigs due to their larger pancreas size (5.25 ± 1.6 g and 6.65 ± 0.75 g, respectfully).
Porcine Age Comparison for Islet Isolation

Pancreases were removed surgically and isolated from varying ages. For adult pancreas, a 5-g piece was removed from the splenic side for age comparison. Isolated islets were allowed to mature in tissue culture (37°C/5% CO2) for 8 days. Results presented as mean ± SEM. All groups showed significant (p < 0.05) increase in IEQ, purity, and viability when compared to islets isolated using the same technique with an adult pancreas.
Islet Apoptosis
Islets displayed progressive decrease in degree of apoptosis during in vitro culture maturation, which occurred primarily in outer cell layers with an inner core of viable cells (Fig. 2D). The percentage of apoptotic cells were 22.5 ± 1.0% after 3 days of tissue culture, which decreased to 4.2 ± 0.5% on day 8 of tissue culture (Table 1, Fig. 2D).
Islet Function
Isolated islets were glucose responsive after 1 day of isolation and increased throughout tissue maturation, demonstrating sequential increases on culture days 3, 5, 7, and 11. The SI of the islets increased from 1.9 ± 2 (low: 0.93 μg/L; high: 1.77 μg/L) on culture day 3 to 2.59 ± 0.5 (low: 1.28 ± 0.1 μg/L; high: 3.32 ± 0.41 μg/L) on culture day 7 and maintained an SI of 2.87 ± 1.2 (low: 0.93 μg/L; high: 2.67 μg/L) on day 11 of in vitro tissue culture. GSIR during islet function displayed the increase of islet function obtained during islet and maintenance of islet functional ability during the entire 11 days of culture displayed by the return to basal insulin production level (Fig. 2C).
Discussion
The data indicate that large numbers of YP islets of high purity, viability, and function can be successfully isolated using pancreatic partial digestion in purified collagenase followed by in vitro culture for 7–9 days. When enzymes were compared, unpurified Sigma V versus purified collagenase MA/protease mixture, using our isolation method there was no significant difference, indicating that a stable, well-defined purified enzyme can be used for these isolation methods (Table 4).
Enzyme Comparison for Partial Digestion of a YP Pancreas

Islets were isolated from 12 YP pancreases after rapid surgical removal and either partially digested in Sigma Type V collagenase (Sigma-Aldrich) or CIzyme™ collagenase MA (VitaCyte). Samples of isolated islets were characterized during in vitro maturation and stained with either DTZ for IEQ or FDA/PI for viability. IEQs were calculated as the percentage of DTZ-positive staining tissue. Percentage of viable islets was calculated as percentage alive (FDA)/percentage dead (PI). Islet function was determined by SI. Results displayed as mean ± SEM per pancreas (where applicable).
Similar to the findings of Korbutt, Elliott, and others, who studied fetal and neonatal pigs, the young pig pancreas (1,11,12,25,26 lacks well-defined islets and has a random distribution of endocrine cells, making it unsuitable for methods conventionally used to isolate adult mammalian islets.
Islets from market weight and young adult pigs are well demarcated in situ but fragile, resulting in substantial islet losses during enzymatic and mechanical digestion and in vitro culture (3,50). Pancreases from neonatal or fetal pigs are structurally delicate; they contain indistinctly demarcated islets, and following partial collagenase digestion, the insulin secretory function of resultant islet-like cell clusters (ICCs) in response to glucose challenge requires 2 weeks or more to develop. Their β-cell mass is maximal at approximately 7–9 days of culture and declines relatively rapidly thereafter. Hence, encapsulation and transplantation of fetal and neonatal islets is performed prior to maturation with insulin responsiveness occurring after transplantation. ICCs from fetal pigs may take as long as 3 months posttransplant to function (7,25). Use of nonpurified collagenase adds additional variability to the isolation process, reducing the ability to control and predict the outcome, and presents additional challenges in creating a procedure suitable for commercial scale-up (6,16,23,25).
The present study examined the use of alternate methodologies to address shortcomings in current techniques. A number of issues appear to be favorably impacted (Table 2).
Ease of Procurement, Isolation, and Decreased Islet Stress
Rapid (<5 min) pancreatectomy of young preweaned piglets permits multiple pancreases to be harvested, quickly enabling pooled processing of multiple organs. Fine mincing of the organs, in a purified collagenase, on ice (4°C) permits warm digestion time to be limited while achieving sufficient initial release of YP islets from acinar stroma. Keeping digestion time as brief as possible aids in maintenance of nascent islet architecture as their collagen matrix is poorly developed and subject to easy disruption. The ability to achieve satisfactory isolation of ICCs without the need for purification using centrifugation and Ficoll gradients reduces loses associated with toxic effects and mechanical stress. This protocol's partial digestion omits the need for the Ricordi chamber and several centrifugal steps, which ultimately avoids the potentially damaging effect Ficoll gradient purification can have on islets during purification.
Deterioration of exocrine acina, shown by a decrease in cellular content of acinar tissue and an increase in islet purity, takes place during culture instead of during the islet isolation process (Table 1). The time lapse from organ harvest to isolation to start of culture is substantially reduced from approximately 3–5 h to less than 1 h including organ harvesting, thus further increasing ease.
Increased Islet Yields, Purity, Viability, and Glucose Sensitivity in Culture
Microscopic examination during in vitro culture confirms continued acinar deterioration and an approximately threefold increase in YP islet numbers. In contrast to fetal and neonatal ICCs, YP islets have immediate glucose sensitivity that increases during the culture period.
Reduced Time in Culture to Optimal Conditions for Transplant
Coincidental improvements in IEQ count, viability, and insulin secretory response to glucose stimulation indicate YP islets are transplant ready by days 7–9 of culture compared to 14 days for fetal and neonatal ICCs (11,12,25). Hence, transplantation of young piglet islets processed and cultured in this manner promises superior β-cell numbers and function at time of transplant when compared to the present art (7,25).
Reduced Costs
Multiple factors contribute to likely savings with the use of young adult over adult pigs as islet sources: partial digestion without the need for the Ricordi chamber or Ficoll gradient separation; less husbandry and resources expended in raising animals to appropriate size prior to harvest; reduced use of enzymes and reagents; reduced labor requirements.
Scalability
The study methodology suggests the feasibility of a scalable “on demand” islet transplantation program that is cost-effective and requires approximately 1 week, with optimum YP islet purity, viability, and function after 7–9 days in tissue culture (37°C/5% CO2) from pancreas procurement to clinical transplant of functioning islets. Such synchronicity makes scale-up to larger production of transplant-ready islets feasible.
The results of this study are encouraging; appropriate follow-on studies include 1) further characterization of the islets isolated and matured using this method; 2) development of scale-up methods to enable future transplant trials; and 3) determination if cryopreservation of YP islets is a suitable means of storage to enable an on-demand supply of transplant-ready islets. During that process, development of protocols of GTP (Good Tissue Practice) and GMP (Good Manufacturing Practice) should be a focus.
Footnotes
Acknowledgments
This work was funded with research funds from the Department of Surgery, University of California, Irvine. The authors acknowledge the support and technical assistance from the Department of Pathology and Sue and Bill Gross Hall Core Facility, University of California Irvine. All authors declare no conflicts of interest.
