Abstract
Cell replacement therapy by intracerebral transplantation of fetal dopaminergic neurons has become a promising therapeutic option for patients suffering from Parkinson's disease during the last decades. However, limited availability of human fetal tissue as well as ethical issues, lack of alternative nonfetal donor cells, and the absence of standardized transplantation protocols have prevented neurorestorative therapies from becoming a routine procedure in patients suffering from neurodegenerative diseases. Improvement of graft survival, surgery techniques, and identification of the optimal target area are imperative for further optimization of this novel treatment. In the present study, human primary fetal ventral mesencephalon-derived tissue from 7- to 9-week-old human fetuses was transplanted into 6-hydroxydopamine-lesioned adult Sprague–Dawley rats. Graft survival, fiber outgrowth, and drug-induced rotational behavior up to 14 weeks posttransplantation were compared between different intrastriatal transplantation techniques (full single cell suspension vs. partial tissue pieces suspension injected by glass capillary or metal cannula) and the intranigral glass capillary injection of a full (single cell) suspension. The results demonstrate a higher survival rate of dopamine neurons, a greater reduction in amphetamine-induced rotations (overcompensation), and more extensive fiber outgrowth for the intrastriatally transplanted partial (tissue pieces) suspension compared to all other groups. Apomorphine-induced rotational bias was significantly reduced in all groups including the intranigral group. The data confirm that human ventral mesencephalon-derived cells serve as a viable cell source, survive in a xenografting paradigm, and functionally integrate into the host tissue. In contrast to rat donor cells, keeping the original (fetal) neuronal network by preparing only a partial suspension containing tissue pieces seems to be beneficial for human cells, although a metal cannula that causes greater tissue trauma to the host is required for injection. In addition, homotopic intranigral grafts may represent a complimentary grafting approach to the “classical” ectopic intrastriatal target site in PD.
Introduction
Transplantation of fetal dopaminergic (DAergic) neurons has become a promising and novel therapeutic approach for patients suffering from Parkinson's disease (PD). Convincing data have been reported in several clinical and preclinical studies showing survival and functional integration of grafted ventral mesencephalon (VM)-derived neurons (16,28) [see Strömberg et al. (43) for review]. Unwanted side effects such as graft-induced dyskinesia, however, have been reported in other studies (18,27,36,53). Surprisingly, most of the clinical studies lack a standardized transplantation protocol, which might be one of the reasons for varying outcome (53). Tissue preparation and surgical procedures are crucial parameters for the success of fetal transplantation (3,34), which will be investigated in the present study.
PD is one of the most common neurodegenerative diseases caused initially by a pathological loss of DAergic neurons in the substantia nigra. This loss and consequently some of the motor impairments can be induced in rodents by the injection of the neurotoxin 6-hydroxydopamine (6-OHDA). The rodent DA system, its depletion, and related impairments have been widely studied and proven to be a viable model for PD (15,55). Vital improvements in functional restoration in parkinsonian rats have been made over the last decades, resulting in good graft survival and functional integration in an allograft paradigm (7,25,33,47). Yet, only a few research groups have examined the behavior and functional integration of human fetal VM-derived (hVM) cells transplanted into a rodent host (9,10,13,41—45,50). Limited access to human fetal tissue and the necessity for immunosuppression (8,11) are the main obstacles in these studies. Experiments using hVM-derived tissue, however, are crucial for a better understanding and characterization of the grafted cells' biology and for optimization of clinical transplantation procedures. So far, no study has systematically investigated the influence of the tissue preparation technique of hVM (full single cell suspension vs. partial tissue pieces suspension) on graft survival, fiber outgrowth, and drug-induced rotational bias. Redmond and colleagues (38) investigated the effects of single cells versus tissue pieces and transplantation sites within the striatum in monkeys. The authors report no significant differences between suspension preparations and injection sites; based on histological analyses of postmortem tissue, however, they discovered an increased glial response to grafts derived from tissue pieces suspensions. This and the fact that single cell suspensions are easier in use and easier in translation to clinical trials made them favor the single cell suspension over suspensions derived from tissue pieces.
The most commonly used transplantation strategy in experimental studies and clinical trials has been the reinnervation of the DA-depleted striatum by graft placement into the dorsolateral tier. The main reason for the ectopic transplantation is that—traditionally—it is believed that intranigral grafts cannot reconstruct the nigrostriatal pathway and are unable to reconnect target areas in an adult striatum (17). Functionally, however, they can induce partial behavioral recovery by reducing the apomorphine- and not amphetamine-induced rotational bias (31), confirming that the nigrostriatal DA system fulfils its functions not only via synaptic transmitter release in the striatum, but also via dendritic DA release in the substantia nigra (2,22,26,29). Interestingly and in contrast to previously published data, two recent studies (19,46) elegantly showed that VM-derived intranigral grafts in mouse models of PD are actually capable of sending out long-distance fibers that seemed to be targeted to striatal areas. In fact, these mice showed functional restoration after apomorphine- and, surprisingly, after amphetamine-induced rotation. Another study (37) presented evidence that embryonic VM grafts, transplanted into the rostral mesencephalon of adult MPTP-lesioned monkeys, have the capacity of long-distance neuronal outgrowth. This was, however, only true when a GDNF (glial cell line-derived neurotrophic factor) signal was present (e.g., by means of viral vector-based GDNF overexpression in striatal target areas).
The present study was designed to investigate (i) the influence of hVM tissue preparation (full single cell suspension vs. partial tissue pieces suspension) and the influence of the surgery technique (metal cannula vs. glass capillary) on intrastriatal graft survival and functional recovery and (ii) the survival rate and functional significance of intranigral hVM grafts.
Materials and Methods
Experimental Design
The current study compares the transplantation of human ventral mesencephalon (hVM) into the striatum and into the substantia nigra in hemiparkinsonian rats using either the microtransplantation technique (glass capillary, full single cell suspension) or the macrotransplantation technique (metal cannula injecting either a full single cell suspension or a partial tissue pieces suspension).
Animals were allocated into six groups (Table 1): (1) transplantation of a partial (tissue pieces) suspension using a metal cannula for injection into the striatum (Part/metal/CPu, n = 7), (2) transplantation of full (single cell) suspension using a metal cannula for injection into the striatum (Full/metal/CPu, n = 8), (3) transplantation of a full (single cell) suspension using a glass capillary for injection into the striatum (Full/glass/CPu, n=9), (4) transplantation of cell culture medium (vehicle) using a metal cannula for injection into the striatum (Sham/metal/CPu, n = 8), (5) transplantation of a full (single cell) suspension using a glass capillary for injection into the substantia nigra (Full/glass/SN, n = 9), and (6) transplantation of cell culture medium (vehicle) using a glass capillary for injection into the substantia nigra (Sham/glass/SN, n = 8). Figure 1 illustrates the experimental design. Validation of lesion deficits (4 weeks postlesion = baseline) and functional graft integration into the host tissue (6, 10, and 14 weeks posttransplantation) were measured by drug-induced rotation. Fifteen weeks posttransplantation, all rats were perfused, and their brains were subjected to histological analyses.

Experimental design. LX, unilateral 6-OHDA medial forebrain bundle (MFB) lesion; TX, transplantation of either hVM cell suspension (full or partial suspension) or vehicle (sham); rot, drug-induced rotation (apomorphine and amphetamine).
Experimental Groups

Animals
Adult female Sprague–Dawley rats (Charles River Laboratories, Sulzfeld, Germany), weighing 200–250 g at the start of the experiment, were used. Animals were housed in cages of two to five in a 12-h light/12-h dark cycle with free access to food and water. Room temperature and humidity were kept constant at 21°C and 55%, respectively. All animal experiments were performed in full compliance with the local ethical board of the University of Freiburg and in accordance with the guidelines of the Regierungspraesidium Freiburg, Germany.
6-OHDA Lesion Surgery
Animals were anesthetized by intraperitoneal injection of ketamine (10%, 0.1 mg/kg body weight; Essex, Germany) and rompun (2%, 0.1 mg/kg body weight; Bayer, Germany). Two stereotaxic injections of 6-hydroxy dopamine HCl (6-OHDA; 3.6 μg 6-OHDA/μl in 0.2 mg/ml l-ascorbate-saline; Sigma-Aldrich, Germany) into the right medial forebrain bundle (MFB) were performed using a 10-μl Hamilton syringe. The following coordinates (in mm with reference to bregma and dura) and injection volumes were used: (1) 2.5 μl at tooth bar (TB) −2.3, anterior–posterior (AP) −4.4, lateral (L) −1.2, dorsoventral (DV) −7.8; (2) 3.0 μl at TB +3.4, AP −4.0, L −0.8, V −8.0. The injection rate was 1.0 μl/min, and the cannula was left in place for another 3 min before slowly retracting it.
Drug-Induced Rotational Behavior
Four weeks after lesion surgery and 6, 10, and 14 weeks after the transplantation, drug-induced rotational behavior was assessed using automated rotometer bowls according to Ungerstedt and Arbuthnott (48): apomorphine-induced rotation (0.05 mg/kg subcutaneously; Sigma-Aldrich, Germany) was monitored over a period of 40 min, and 3–5 days later d-amphetamine (2.5 mg/kg intraperitoneally; Sigma-Aldrich, Germany) was administered and rotational behavior was recorded over a period of 90 min. Results are expressed as revolutions/full body turns per minute (rpm; net rotation divided by 90 min). Based on the results of the 4 weeks postlesion, drug-induced rotation (baseline) groups were matched for transplantation. Only rats with >4 full rpm contralateral to the lesioned hemisphere after apomorphine-injection and >6 rpm ipsilateral to the lesion after amphetamine-injection were included in the study (25% of all lesioned animals were excluded for having only a partial lesion, that is, not having met the aforementioned thresholds).
Collection, Dissection, and Preparation of Human Embryonic VM Tissue
Human embryonic tissue was collected by transvaginal low pressure aspiration or curettage at elective routine surgical termination of pregnancy (sTOP), performed according to the regulations of the local research ethics committee of the University of Freiburg, Germany, the regulations of German law (§218), and the guidelines established by the Network of European CNS Transplantation and Restoration (6,24). Full informed consent was obtained from the maternal donors. Human DAergic tissue from embryos ranging between 7 and 9 weeks of age (postconception; based on calculation and preoperative ultrasound examination) was obtained by dissection of the ventral mesencephalon. Tissue was stored overnight in hibernation medium (HBSS, Gibco, UK) at 4°C.
Because of the limited amount and the irregular availability of human tissue donations, the transplantation had to be split into multiple surgery sessions. Depending on embryo age, a total amount of four to eight animals were transplanted from one human VM during each session. To overcome differences in viability, embryo age, and handling between the groups, animals of two different experimental groups were transplanted during each grafting procedure.
Preparation of full (single cell) suspensions for the Full/metal/CPu, the Full/glass/CPu, and the Full/glass/SN groups was performed according to a modified microtransplantation approach (33,35), based on the cell suspension technique described by Bjorklund et al. (5). In brief, tissue was coarsely cut into pieces, incubated in 0.1% trypsin and 0.05% DNase (both Sigma-Aldrich, Germany) in Dulbecco's modified Eagle's medium (DMEM, Gibco, UK) at 37 °C for a period of 20 min and afterwards rinsed four times in 0.05% DNase in DMEM. The following mechanical dissociation was performed by repeated trituration beginning with a 1-ml pipette and subsequently using a 200-μl pipette with a smaller tip in order to achieve a homogenous full (single cell) suspension. Cells were centrifuged at 600 rpm for 5 min, and the supernatant was removed. The pellet was resuspended with 0.05% DNase/DMEM to achieve a cell density of 100,000 cells/μl in the final cell suspension. Viability of cells pregrafting was >80% during all transplantation surgeries as measured by the trypan blue dye (Sigma-Aldrich, Germany) exclusion method.
On order to prepare the cell suspensions for the Part/metal/CPu and Full/metal/CPu groups, the VM was split into two halves: one half was used for the preparation of the full (single cell) suspension (see above); the other half was used for preparation of the partial (tissue pieces) suspension. To prepare the partial (tissue pieces) suspension for the Part/metal/CPu group, the tissue was incubated in trypsin and DNase at 37°C for a period of 10 min. The mechanical dissociation was performed by gentle trituration with a 1-ml pipette, resulting in a partial suspension with macroscopically visible small tissue pieces. After centrifuging (600 rpm for 5 min), the required volume of 0.05% DNase/DMEM for resuspension of the pellet of tissue pieces was assumed to be the same as for the other half used for the full (single cell) suspension as the VM was cut into two equal pieces during each dissection. This method was used to assure that there were approximately as many cells in the partial suspension as in the full suspension (100,000 cells/μl).
Cell Transplantation Surgery and Immunosuppression
Transplantation surgery was performed 5–12 weeks postlesion (based on availability of human tissue). Starting 1 day prior to transplantation, all rats were put on a daily immunosuppressive regime (see below). On the day of surgery, rats were anesthetized and placed in a stereotaxic frame (Stoelting, USA) as described above. Each animal received four deposits of 1-μl cell suspension (100,000 cells/μl; all transplanted groups) or 1 μl of cell culture medium (Sham/metal/CPu and Sham/glass/SN) using two injection tracts. Over the course of the transplantation session, partial and full suspensions were homogenized intermittently by gently retriturating. Two types of injection cannulae were used: (1) a metal cannula (500 μm outer diameter; 2 μl Hamilton microsyringe, Hamilton Europe, Switzerland) or (2) a glass capillary (50 μm outer diameter; made in-house; attached to 2 μl Hamilton microsyringe via polyethylene tubing) [for further details, see Nikkhah et al. (34)]. All grafts were placed ipsilaterally to the side of lesion. The coordinates for the implantation were as follows: intrastriatal transplantation TB 0.0, AP +1.0, L −1.8/-2.6, DV −4.0/-5.0; intranigral transplantation TB −3.3, AP −6.0/-6.5, L −1.9, DV −6.7/-7.0.
All rats were immunosuppressed by daily intraperitoneal injections of cyclosporine A (10 mg/kg; Sandimmun, Sandoz, Switzerland) for the duration of the experiment, commencing the day prior to transplantation (8,10). Antibacterial prophylaxis was administered by adding Borgal 24% to the drinking water (dilution 4 ml/L, equals 0.16 g trimethoprim/L and 0.8 g sulfadoxin/L; Intervet, Germany).
Immunohistochemistry
Fifteen weeks following transplantation, animals were deeply anesthetized by an overdose of ketamine and rompun and perfused transcardially with 300 ml 0.1 M phosphate-buffered saline (PBS), followed by 300 ml ice-cold 4% paraformaldehyde (PFA) in 0.1 M PBS. After postfixation in 4% PFA for 2 h, brains were transferred in 20% sucrose in 0.1 M PBS for dehydration. Coronal sections were cut in a series of 5 on a freezing sledge microtome (Leica, Germany) at 40 μm thickness. One series of sections was stained for tyrosine hydroxylase (TH; 1:500, rabbit anti-TH, PRD-515P, Covance, USA) and another series for human neurofilament 70 (hNF70; 1:100, mouse anti-hNF70, MAB5294, Serotec, UK) using the ABC-Kit (Vector Laboratories, Canada) and 3,3′-diaminobenzidine (DAB; Merck, Germany) method for visualization. Sections from selected brains were additionally double stained for TH (1:2,500; mouse anti-TH T-1299, Sigma-Aldrich, USA) and the vesicular monoamine transporter (VMAT; 1:1,000; rabbit anti-VMAT-2 PRB-329P, Covance, USA).
TH-immunoreactive (TH+) grafted neurons were counted under brightfield condition (Leica DRMB, Leica, Germany) using the StereoInvestigator Software (MicroBrightField, USA). A meander-like scan supported by the microscope's motor stage was undertaken to count all TH+ cells within the graft. The total TH+ cell number within each graft was calculated according to the Abercrombie formula (1).
Graft volume was measured in each section containing TH+ cells using the analySIS Software [Soft Imaging Systems, Germany; graft volume (mm3) = grafted area on sections × 40 μm section thickness × 5 series].
Statistical Analysis
Histological data were subjected to a one-way analysis of variance (ANOVA) and Newman–Keuls post hoc tests (GraphPad Prism5, GraphPad Software, Inc., USA). Repeated drug-induced rotation was analyzed by a two-way ANOVA and Bonferroni post hoc analysis. Significance level for all statistical tests was set at p < 0.05. Results are expressed as means ± SEM.
Results
Behavioral Tests
All animals were tested 4 weeks postlesion and 6, 10, and 14 weeks posttransplantation on both the amphetamine and the apomorphine rotation tests. Results of the 4-week postlesion test were used to allocate animals to the different groups, and these values were used as baseline results in the subsequent analysis for evaluating the effects of transplant treatments.
Apomorphine-Induced Rotation
All animals exhibited strong contralateral rotation on the first apomorphine test conducted 4 weeks following unilateral nigrostriatal lesion, but prior to any transplantation surgery (Fig. 2A). The transplantation of hVM cells had a significant effect on apomorphine-induced rotation [Group: F(5, 167) = 16.29, p < 0.01; Group × Test: F(15, 167) = 1.76, p < 0.05]. As shown in Figure 2A, this is attributable to both sham-transplant groups exhibiting a progressive increase in apomorphine-induced rotation over the successive four tests, whereas the four transplant groups all exhibited a progressive reduction in apomorphine-induced rotation following transplantation. Post hoc tests indicated no significant difference in the rate of reduction of rotation between the different transplant treatments, and a post hoc analysis restricted to just these four groups indicated a significant difference across tests, but no significant interaction between transplant treatments [Test: F(3, 112) = 7.85, p < 0.01; Group × Test: F(9, 112) = 0.32, p = 0.97, n.s.]

Results of the apomorphine- and amphetamine-induced rotation. (A) The transplantation of hVM cells had a significant effect on apomorphine-induced rotation [GROUP: F(5, 167) = 16.29, p < 0.01]. The Part/metal/CPu, Full/glass/CPu, and Full/glass/SN groups developed significant less rotational behavior compared to the sham-transplanted Sham/metal/CPu group 6, 10, and 14 weeks posttransplantation. The Full/metal/CPu group showed a significant difference to the Sham/metal/CPu group 10 and 14 weeks posttransplantation. All grafted groups displayed fewer rotations than sham-transplanted rats from the Sham/metal/CPu group 6, 10, and 14 weeks posttransplantation. (B) The Part/metal/CPu group demonstrated a significant reduction of amphetamine-induced asymmetry compared to the Full/glass/SN group and the sham-transplanted groups (Sham/metal/CPu, Sham/glass/SN) 10 and 14 weeks posttransplantation. Please note that the Part/metal/CPu group overcompensated to the contralateral side 14 weeks posttransplantation and became significant different to the Full/metal/CPu and Full/glass/CPu groups [Part/metal/CPu vs. all other groups, F(5, 169) = 12.45, p < 0.05]. Post hoc comparisons between the groups on the final 14-week test confirmed that net ipsilateral rotation in the Part/metal/CPu group < Full/glass/CPu = Full/metal/CPu groups < Full/glass/SN = Sham/glass/SN = Sham/metal/CPu groups (Newman–Keuls, criterion p < 0.05). See Table 1 for group ID.
Amphetamine-Induced Rotation
All animals exhibited strong ipsilateral rotation on the first amphetamine test conducted 4 weeks following unilateral nigrostriatal lesion, but prior to any transplantation surgery (Fig. 2B). In contrast to the apomorphine tests, the amphetamine tests indicated clear differences between the transplant groups in their efficacy to alleviate amphetamine-induced rotation. As indicated in Figure 2B, there was a significant Group × Test interaction [F(15, 169) = 12.45, p < 0.05], which remained significant even when the analysis was restricted to just the four transplantation groups [F(9, 113) = 2.88, p < 0.05]. In essence, the intranigral graft group and the two sham-transplant groups all exhibited a stable level of amphetamine-induced rotation across all four amphetamine tests. By contrast, all three intrastriatal graft groups exhibited a progressive decline in rotation across tests posttransplantation, albeit to varying degrees. In particular, the greatest recovery was seen in the Part/metal/CPu group, which actually exhibited an overcompensation (rotation towards the contralateral side by the last test), whereas both the Full/metal/CPu and Full/glass/CPu groups exhibited a partial compensation to an intermediate degree. Post hoc comparisons between the groups on the final 14-week test confirmed that net ipsilateral rotation in the Part/metal/CPu < Full/glass/CPu = Full/metal/CPu < Full/glass/SN = Sham/glass/SN = Sham/metal/CPu (Newman–Keuls, criterion p < 0.05).
Histological Analysis
Lesions
TH immunohistochemistry indicated a complete or almost complete loss of endogenous TH+ neurons in the striatum and the substantia nigra on the lesioned side in all animals, confirming the efficacy of the lesions (Fig. 3).
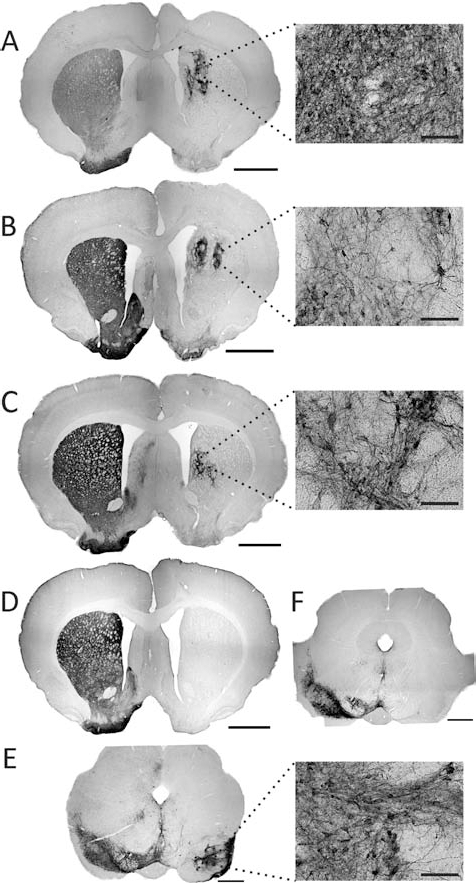
Photomicrographs of coronal sections stained for tyrosine hydroxylase (TH) from all groups. (A) Intrastriatal graft (Part/metal/CPu). (B) Intrastriatal graft (Full/metal/CPu). (C) Intrastriatal graft (Full/glass/CPu). (D) Intrastriatal sham transplantation (Sham/metal/CPu). (E) Intranigral graft (Full/glass/SN). (F) Intranigral sham transplantation (Sham/glass/SN). Scale bar on low magnification sections: 2 mm (A–D) or 1 mm (E, F); scale bar on magnified fields (inset): 100 μm.
Graft Survival
The Part/metal/CPu and the Full/glass/SN groups formed large and spherically shaped grafts (Fig. 3A, E), whereas grafts in the Full/metal/CPu and in the Full/glass/CPu groups formed smaller grafts that were either oblong pencil-like tissue strands along the needle tracts or small rounded deposits (Fig. 3B, C). The intrastriatal grafts (Part/metal/CPu, Full/metal/CPu, Full/glass/CPu) were all located centrally within the medial striatum. The intranigral grafts (Full/glass/SN) not only covered the area of the substantia nigra pars compacta but also invaded the pars reticulata. Sham animals (Sham/metal/CPu and Sham/glass/SN) were lesioned but received only a sham transplantation of cell suspension vehicle. No TH+ neurons or fibers in the lesioned hemisphere were found in these two groups (Fig. 3D, F).
The numbers of surviving TH+ neurons 15 weeks posttransplantation differed between the four VM transplant groups [F(3, 29) = 3.72, p < 0.05]. Assuming a 10% ratio (10) of DAergic cells to total cell number (which was 400,000 cells in 4 μl cell suspension) the Part/metal/CPu (2,183 ± 449 cells) had the best survival rate of 5.5% (Full/metal/CPu: 2.2%; Full/glass/CPu: 1.6%; Full/glass/SN: 3.9%). As shown in Figure 4A, post hoc comparisons indicated that there were significantly higher numbers of surviving TH+ cells in the Part/metal/CPu group compared to either of the other two intrastriatal VM transplant groups (Full/metal/CPu, Full/glass/CPu; both p < 0.01; Newman–Keuls, criterion p < 0.05), whereas the animals of the Full/glass/SN group receiving intranigral VM grafts exhibited an intermediate level of TH+ cell survival, which did not differ from any of the intrastriatally transplanted groups Part/metal/CPu, Full/metal/CPu, and Full/glass/CPu (all n.s.).
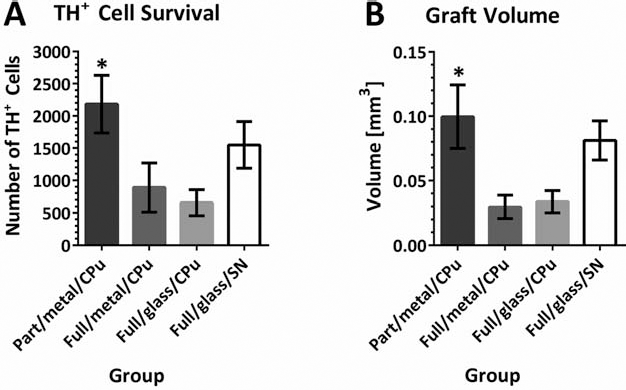
(A) The number of surviving TH+ neurons 15 weeks posttransplantation was significantly higher (∗) in the Part/metal/CPu group (2,183 ±449 cells) compared to the Full/metal/CPu group (892 ± 378 cells) and the Full/glass/CPu group (657 ± 199 cells). Animals receiving intranigral VM grafts (Full/glass/SN) showed no significant difference in the number of surviving TH+ cells (1548 ± 362 cells) compared to the intrastriatally transplanted groups. (B) The volume of the TH+ grafts was significantly higher (∗) in the Part/metal/CPu compared to the Full/metal/CPu group and the Full/glass/CPu group. Animals transplanted into the substantia nigra (Full/glass/SN) showed significantly higher graft volumes than the Full/metal/CPu group and the Full/glass/CPu group. There was no significant difference in the graft volume of the Part/metal/CPu group compared to the Full/glass/SN group. See Table 1 for group ID.
Similar differences were seen in the volumes of the VM-derived grafts between the four transplanted groups [F(3, 29) = 5.30, p < 0.05] (Fig. 4B). Again, the volume was highest in the Part/metal/CPu group, which post hoc comparisons indicated was significantly greater than the volumes of either the Full/metal/CPu group or the Full/glass/CPu group. The grafts of the Full/glass/SN group were of intermediate size and did not differ significantly from the grafts in the Part/metal/CPu group but were significantly bigger than grafts in the Full/metal/CPu and the Full/glass/CPu groups.
VMAT staining of selected sections across intrastriatally and intranigrally grafted groups showed robust colabeling with TH (Fig. 5), indicating that these TH+ neurons were functional neurons as VMAT is needed in DA transmission.

Confocal photomicrographs of VMAT and TH double-labeled sections. The top row displays images of intrastriatal grafts (STR) under 20x magnification. The middle row shows images of intranigral grafts (SN) under 20x magnification. The bottom row discloses a higher magnification (63x) of the boxed area in the middle row highlighting a neuron that expresses both TH and VMAT. Nuclear staining of all cells is presented in the first column (DAPI), and the overlay of all three staining is shown in the last column (MERGE). Scale bars: 150 μm (top two rows) and 50 μm (bottom row).
Axonal Projections
The human-specific neurofilament marker hNF70 was used to identify axonal growth of grafted neurons into the host tissue (Fig. 6). Animals of the Part/metal/CPu group exhibited projections throughout the entire striatum, the corpus callosum and the globus pallidus (Fig. 6A). In caudal direction, fibers were detected in the ventral pallidum, in the lateral globus pallidus, in the medial forebrain bundle, and in the stria terminalis. The most caudal fibers were identified in the MFB and in the lateral hypothalamic area (anterior–posterior −4.8 mm, measured from bregma). No graft-derived projections could be observed in the substantia nigra. In the Full/metal/CPu and Full/glass/CPu groups limited axonal projections were observed crossing the graft-host border (Fig. 6B, C), and no hNF70+ fibers were seen in other brain regions. In the Full/glass/SN group graft-derived hNF70+ fibers were exclusively confined to the substantia nigra and the needle tract. There was no outgrowth of fibers in rostral direction along the nigrostriatal pathway (Fig. 6D).
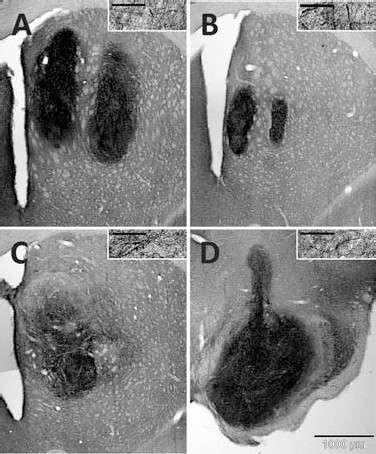
Photomicrographs of hNF70-stained sections. (A) Intrastriatal graft (Part/metal/CPu). (B) Intrastriatal graft (Full/metal/CPu). (C) Intrastriatal graft (Full/glass/CPu). (D) Intranigral graft (Full/glass/SN). Each case is illustrated at low magnification. Scale bars: 1,000 μm (A–D). The insets display hNF70-positive fibers under higher magnification. Scale bars: 50 μm (A–D).
Discussion
In the present study, we investigated graft survival, graft integration, and functional recovery in intrastriatal and intranigral hVM-derived xenografts. The cell suspensions were prepared either as a full (single cell) suspension or as a partial (tissue pieces) suspension, and they were implanted either by means of a glass capillary or by means of a metal cannula. The results confirm that fetal hVM-derived cells are able to survive for 14 weeks and functionally integrate into the immunosuppressed rodent recipient. We could demonstrate a good cell survival and integration as well as high graft volume in the Part/metal/CPu group. Worst graft survival was observed in the groups with an intrastriatally injected full suspension (Full/metal/CPu and Full/glass/CPu). The intranigrally injected group (Full/glass/SN) developed an intermediate graft survival with a significant reduction of rotational bias in the apomorphine- but not in the amphetamine-induced rotation. Intrastriatal grafts generally reduced the rotational bias after apomorphine and amphetamine injection; the Part/metal/CPu group, however, not only reduced the amphetamine-induced rotational bias but also led to overcompensation to the contralateral side 14 weeks posttransplantation. Graft-derived axonal projections in this group were found in higher numbers outside the graft core and were detected at distances farthest away from the graft than projections of grafts derived from single cell suspensions at either sites, the striatum and substantia nigra (Full/metal/CPu, Full/glass/CPu, Full/glass/SN).
Intrastriatal Grafts: Tissue Pieces Versus Single Cell Suspension
Previous grafting studies using metal cannulae (rat donor into rat host) have demonstrated a better graft survival of partial (tissue pieces) suspensions for intrastriatal grafts than survival of full (single cell) suspensions (3,33,35). The detrimental effects of the vigorous trituration process during the preparation of a full (single cell) suspension were given as a potential cause for this effect. The authors speculated that, on rat embryonic day 14 (E14), the neuronal processes have already built such dense network that any mechanical damage of the neuronal connections injures the neurons and decreases the survival of the graft. Interestingly, by replacing the metal cannula with a fine glass capillary to reduce the implantation trauma, graft survival of a full (single cell) suspension compared to the partial (tissue pieces) suspension implanted by metal cannula was significantly better in rat allografts (35). The smaller impact on the host brain tissue by the glass capillary seemed to have outweighed the negative effects of preparing a full (single cell) suspension. Interestingly, in another study using primate tissue, Redmond et al. (38) reported no differences between single cells and tissue pieces in parkinsonian monkeys in terms of behavioral recovery and graft integration; the tissue pieces, though, elicited an increased glial response. The authors conclude that single cells might still be the preparation of choice as they are easier in use, less immunoreactive, and closer to clinical application considering that stem cell-derived DA (single) cells (and not ethically challenging fetal cells) will be used for clinical trials in the long-term future.
In contrast to these studies using rodent or primate donor tissue, the present study demonstrates better graft survival of hVM-derived cells in the group transplanted with tissue pieces injected by means of a metal cannula (Part/metal/CPu) than in the group transplanted with a full (single cell) suspension injected by means of a glass capillary (Full/glass/CPu). There seems to be a fundamental difference between human and rodent cells. A possible explanation could be differing needs of the human and rat fetal tissue during their early developmental stages [7- to 9-week-old human embryos are in the first trimester of pregnancy, which roughly compares to E14 in rats, based on gene expression (30)]. Creating a “intrinsic microenvironment” with growth factors from intact human glial cells, keeping the fetal neuronal network and efficient vascularization within the xenograft seem to be crucial for graft survival of the immature human neurons and is most likely to be achieved by the use of tissue pieces. The use of tissue pieces, however, requires a metal cannula for infusion because the tissue pieces are too big for a narrow glass capillary. Hence, the benefit of the intact microenvironment within the donor tissue pieces seems to outweigh the host tissue-damaging effects of the metal cannula. Furthermore, we observed that human full (single cell) suspensions were stickier and more adherent compared to rat full (single cell) suspensions (personal experience); as a consequence, the human full (single cell) suspension had to be frequently resuspended during the surgery session to avoid blocking of the tip of the glass capillary by cell aggregates. This increased mechanical trituration process might have contributed to a reduced graft survival during surgery, although cell viability was not affected immediately posttransplantation.
This effect was independent of the tissue quality as all animals within each group showed the same graft survival pattern, although they were grafted during different surgery sessions. This is interesting as donor tissue varied in age (7–9 weeks) between cell preparations; that is, the dissected VMs were derived from different stages of development. Hence, animals from different groups were transplanted during each surgery session using the same cells. This schedule was chosen, first, because of the limited availability of fetal tissue in general, second, because this procedure most closely resembles a situation during potential clinical trials where the availability of human tissue counts and less the exact donor age, and third and most importantly, this method allowed us to overcome differences in cell viability, embryo age, and cell preparation between groups.
Fetal hVM tissue pieces (Part/metal/CPu) not only integrated best into the host striata but also showed best functional recovery as seen by a reversed rotational bias after amphetamine injection. This is in line with a series of publications by Strömberg et al. presenting evidence for functional integration of hVM-derived grafts (using tissue pieces): apomorphine-induced rotation could be reversed; DA receptor levels were partially normalized; grafts developed normal vascularization; and functional synapses between host and donor were formed (41,42,44). Interestingly and in contrast to a previous study using rodent tissue (31,33), the intrastriatal injection of a full (single cell) suspension caused only a reduction of amphetamine-induced rotational bias (and no overcompensation), indicating a worse functional integration of the full (single cell) suspension into the host tissue than the partial (tissue pieces) suspension. These results correlate with best graft survival as seen in highest numbers of TH+ cells as well as greatest graft volumes in the Part/metal/CPu group.
Intranigral Grafts
Behavioral recovery following the intranigral transplantation of hVM in the 6-OHDA rat model of PD has not been described previously. It has been reported in rodent transplantation studies that about 1,000–1,300 intranigrally grafted TH+ neurons seem to be required to achieve a 50% reduction of rotational bias after dopamine agonist injection (31). In this study, we found similar results: a reduction in apomorphine-induced rotational asymmetry greater than 50% was observed in most rats with >1,000 surviving TH+ cells, whereas rats with <1,000 surviving TH+ neurons developed a reduction of rotational bias that was less than 50%. However, we did not find a significant correlation between the numbers of surviving intranigrally grafted TH+ cells and the degree of rotational bias. Therefore, we argue that a threshold value has to be reached to show a significant compensation and that there is no close correlation of cell number and degree of functional compensation for values below or above this number. Nevertheless, it is remarkable to which extent the intranigral hVM-derived grafts reduced apomorphine-induced turning behavior (-54%) considering the fact that the striatum remained denervated and received no DA input from the transplant. It has been reported that the behavioral response to DA agonists in 6-OHDA lesioned rats is partially induced by nigral mechanisms (22,26,40). Hence, it has been assumed that intranigral grafts are able to restore dendritic DA release sufficient for a reversal of the lesion-induced DA receptor supersensitivity within the substantia nigra (31,52). The importance of the dendritically released dopamine in the substantia nigra of the healthy animal has been pointed out in several reports and should be reconsidered in neurorestoration (12,29,39).
In line with some previous reports, amphetamine-induced turning behavior was not affected by homotopic transplantation of human DAergic neurons (Full/glass/SN group) (31). Amphetamine works presynaptically and facilitates DA release into the synaptic cleft (54); hence, its actions are mainly on the nonlesioned healthy hemisphere due to the lack of DA on the lesioned side. Therefore, in case of intranigral grafts, the ipsilateral striatum remains DA depleted, and the DA imbalance in the striatum continues leaving amphetamine-induced rotation unaffected.
In contrast to studies using mouse donor and (adult) mouse recipients (19,46), we did not observe any long-distance outgrowth from the intranigral grafts towards striatal target areas (as observed in TH and hNF staining) that would have helped to counterbalance striatal DA levels. It is noteworthy that a functional reconstruction of the nigrostriatal pathway accompanied with significant changes in the amphetamine-induced rotation asymmetry following intranigral transplantation of VM-derived cells has been reported in neonatal rats; their brains present a milieu that permits neuronal growth and migration (4,32). The adult brain, however, can be considered to provide a less favorable environment for outgrowth over long distances (17), and reconstructing the nigrostriatal projections from an intranigral graft may be more challenging. Thompson et al. (46) and Gaillard et al. (19), on the other hand, reported the opposite and provided evidence that standard intranigral mouse VM-derived graft form targeted functional long-distance connections. One reason for these contradictive reports is that the more recent studies could track grafted neurons and fibers in more detail due to GFP-overexpressing donor material. Furthermore, the authors used mouse models, whereas other studies with no nigrostriatal reconstruction used rat models. Here, we used human cells in a xenografting paradigm, which obviously does not provide optimal growth conditions and thereby might be the reason why we could not see long-distance growth either. However, other studies using porcine or human tissue report that long-distance axonal growth along the nigrostriatal pathway is possible (14,21,49). Isacson and Deacon summarize in their 1997 review (20) that cells derived from animals with protracted development (such as human or pig) have a relatively longer time window of development (than their host, i.e., the rat), which may enable axonal growth to reach distant targets during graft maturation. Yet, the reconstruction of DA pathways remains incomplete, and it seems that axonal growth of xenogenic neurons is equally as impeded as axonal growth in syngenic transplantation paradigms, indicating that both human or porcine cells recognize inhibitory signals from the (rodent) host.
TH+ cell survival and graft volume in the SN revealed intermediated graft integration into the host nigra, which, interestingly, was still better than in the other two full (single cell) suspension groups that were ectopically injected into the striatum. This might be due to the fact that the SN forms a homotopic environment where, although targeted long-distance growth could not be observed in our case (see above), hVM-derived (future nigral) cells still recognize their homotopic placement cross species and growth conditions for DA cells seem superior to striatal coordinates. The benefits of homotopic graft placement appear to even outweigh the disadvantageous preparation of a full (single cell) suspension, which caused poor results in the striatum.
Graft-Derived Neuronal Projections
In our human-to-rat study we observed dense and long neuronal projections: the Part/metal/CPu group displayed axonal growth from the injection site to caudal brain regions (5–9 mm) compared to 2–4 mm outgrowth typically seen in studies with rodent donor tissue. Surprisingly, we observed extended growth only in this group (Part/metal/CPu group) and not in any of the other groups using full (single cell) suspensions. Hence, cell preparation of human VM-derived tissue (vigorous trituration in case of the full single cell suspension) seemed to act negatively on top of the mostly inhibitory environment in the adult host preventing extensive fiber outgrowth and graft integration. Nevertheless, previous studies have demonstrated that human primary fetal tissue transplanted into the rat brain in Huntington's disease or PD models can send out extensive projections (23,45,50,51). Wictorin et al. (50) reported that transplantation of hVM, dispersed mechanically into a crude cell suspension, which compares best to our partial (tissue pieces) suspension, can send axonal projections from the implantation sites along the nigrostriatal pathway to the ventral striatum and to the olfactory bulb. Wictorin and colleagues used a different PD rat model though where rats received an additional injection of ibotenic acid into the rostral mesencephalon, shortly before the fetal cell implantation. The authors concluded that the extended long-distance growth of human tissue may be explained by the relatively larger size of the human brain requiring a longer inhibition-insensitive growth phase or that the human cells do not recognize the rodent type of inhibitory molecules. On the contrary, Thompson et al. (46) and Gaillard et al. (19) showed that the lesioned host brain is not as hostile and inhibitory towards newly grafted cells and fiber outgrowth as always thought. In fact, in mouse models of PD including mouse donors for transplantation, the authors observed long-distance outgrowth along the nigrostriatal pathway including functional improvements as observed in drug-induced rotation. This information is supported by a clinical safety trial carried out by the Mendez group in Canada (29): two patients with PD received intrastriatal and intranigral grafts simultaneously. Remarkable improvement of motor scores and quality of life were reported; postmortem analyses revealed great graft survival and an almost complete reinnervation of the host putamen. Whether this impressive rewiring of the nigrostriatal pathway was due to the striatal or nigral grafts could not be determined, behavioral improvement and degree of reinnervation, however, had been exceptionally good. These results and the results from the present study may reopen the discussion of the best transplantation site reconsidering the SN for clinical trials. From our results, we suggest that the potential for long distance axonal growth of hVM cells is not dependent of the number of surviving cells but depends at least partially on cell preparation and the grafting technique. More detailed analyses are necessary though to explain the encapsulated-like nigral grafts and the outgrowth from grafted tissue pieces investigating the effects of, for example, extracellular matrix molecules and neurotrophic factors on neurorestoration.
Conclusions
Intrastriatal transplantation of hVM-derived partial (tissue pieces) suspension compared to transplantation of a full (single cell) suspension results in better graft survival, reverses amphetamine-induced rotational bias to a higher degree, and supports better graft integration into the host tissue (long distance axonal growth). The transplantation of hVM-derived cells into the rodent substantia nigra results in an intermediate graft survival. Intranigral grafts showed no long distance axonal growth in contrast to intrastriatal graft which—particularly in the Part/metal/CPu group— invaded the host tissue with their neuronal processes far into caudal areas. Interestingly, the greater surgical trauma caused by the use of a wider metal cannula (compared to the impact of a glass capillary) is outweighed by better graft survival and integration of a partial (tissue pieces) suspension compared to a full (single cells) suspension.
Further studies are needed to investigate graft–host interactions in the context of behavioral (motor/cognitive) recovery and how donor age influences grafting success of the tissue pieces suspension.
Footnotes
Acknowledgments
The authors would like to thank Dr. Máté D. Dobrossy for his help with histological analyses. Furthermore, the authors would like to acknowledge Johanna Wessolleck and Marlene Löffler for excellent technical assistance. This study was supported by the Deutsche Forschungsgemeinschaft and the Bundesministerium fuer Bildung und Forschung (01GW0730) to G.N. The Medical Research Council UK and the European Commission [EC contract #222918 (REPLACES) FP7-Thematic priority HEALTH] kindly funded to A.K. and S.B.D.'s work. S.B.D. and G.N. would like to acknowledge shared funding from the TransEuro consortium [European Commission under the 7th Framework Programme-HEALTH Collaborative Project TransEuro (EC contract #242003)]. The authors declare no conflicts of interest.
