Abstract
Cell replacement therapies have yielded variable and short-lived benefits in Huntington's disease (HD) patients. This suboptimal outcome is likely due to the fact that graft survival is compromised long term because grafts are subjected to a host's microglial inflammatory response, to a lack of adequate trophic support, and possibly to cortical excitotoxicity. However, graft demise may also relate to more straightforward issues such as cell preparation methodology (solid grafts vs. cell suspension). Indeed, we recently reported that solid grafts are poorly revascularized in HD patients transplanted 9 and 12 years previously. To evaluate whether methodological issues relating to cell preparation may have an impact on graft viability, we implanted green fluorescent protein (GFP+) single-cell suspensions of fetal striatal neuronal cells into the striatum of YAC128 HD mice. Postmortem evaluation yielded comparable graft survival in YAC128 mice and their wild-type littermates (noncarrier) at 1 and 3 months posttransplantation. Additionally, the degrees of graft revascularization in the YAC128 and noncarrier mice were similar, with both capillaries and large-caliber vessels observable within the grafted tissue. Furthermore, GFP+ cells interacted well with host blood vessels, indicating integration of the donor cells within the recipient brain. These observations, combined with our recent report of poor revascularization of solid grafts in the HD-transplanted patients, suggest that the success of cell transplantation can be improved by optimizing methodological aspects relating to cell preparation.
Introduction
Huntington's disease (HD) is an autosomal dominant neurodegenerative disorder of the central nervous system (CNS) that is defined by a cytosine–adenine–guanine (CAG; glutamine codon) expansion in exon 1 of the huntingtin gene leading to the production of the mutant huntingtin (mHtt) protein. There is no disease-modifying treatment for HD, and current clinical interventions are limited to symptom management (7). The development of new therapeutic strategies is thus actively pursued, and a number of open-label trials have already been conducted to test the feasibility and efficacy of cell replacement approaches in the context of HD. These trials were predicated on the grounds that the major pathology in HD involves the striatum, and thus replacing it with unaffected fetal striatal tissue would be beneficial, as shown experimentally (11,21,34). However, transplanted HD patients have demonstrated only marginal and short-lived clinical improvements (7–9).
The reasons for these suboptimal clinical outcomes are not entirely clear, but in a unique series of postmortem analyses of the brains of HD-transplanted patients who have come to autopsy, we have observed that graft survival is compromised long term. The mechanisms underlying this poor graft survival may be numerous, including an important microglial inflammatory response, the lack of adequate trophic support, as well as graft demise via cortical excitotoxicity (7,8,16). However, more easily modifiable technical issues relating to cell preparation, for example, the use of solid versus single-cell suspension approaches, have shown diverse outcomes in the revascularization of transplanted neural tissue, a critical factor in long-term graft integration and survival (16). For example, solid grafts show delayed neovascularization, as observed in the parenchyma of both wild-type mice (6,24) and 6-hydroxydopamine (6-OHDA)- lesioned rats (27). Despite the fact that blood vessels of the recipient brain are mitotically active, they rarely penetrate the donor tissue (6,24). However, cells placed in an enriched vascular environment, such as the choroid plexus, survive better than those implanted into the parenchyma (39).
We have similarly reported significant vasculature disparities between the host brain and solid tissue grafts in HD-transplanted patients (9). In these cases, solid tissue grafts had a vascular network that was much less developed than that of the host brain. While capillaries were observable within the transplants, they were not as frequently encountered as in the host, and large-caliber blood vessels were almost entirely absent in the transplanted tissue, another important difference with the host brain (9). We suspect that the compromised revasculature of the solid striatal tissue transplants in HD patients may have further contributed to poor graft survival long term. Based on these observations (7,9), we undertook this in vivo study in the murine yeast artificial chromosome containing 128 CAG repeats (YAC128) model of HD to specifically address whether single-cell suspension would favor graft survival and vascularization in the HD diseased brain.
Materials and Methods
Cell Preparation
Pregnant hemizygous female mice genetically engineered to express the green fluorescent protein (GFP)+/-[C57BL/6-Tg(ACTB-EGFP)1Osb/J] (30) (kindly provided by Dr. Serge Rivest from Laval University, Québec, QC, Canada) were subjected to a sublethal dose of a ketamine (100 mg/ml)/xylazine (1 mg/ml) (Vetalar, Bioniche, Belleville, ON, Canada/Rompun, Bayer, Toronto, ON, Canada) mix at embryonic day 13.5 (E13.5). Fetuses were retrieved and placed in sterile ice-cold solution of 20 mM glucose (Sigma, Oakville, ON, Canada) in Hanks' balanced salt solution (HBSS) 1× (Invitrogen, Burlington, ON, Canada). The GFP+ phenotype of each embryo was confirmed using ultraviolet (UV) lamp illumination. The use of GFP+ tissue allowed for the easy tracking of transplanted cells within the host brain. The lateral ganglionic eminence (LGE), which largely yields fetal cells committed to a striatal phenotype, was dissected and kept in HBSS– glucose solution on ice. The HBSS solution was subsequently replaced by a solution of 0.2% trypsin (Sigma) and 0.5 mM ethylenediaminetetraacetic acid (EDTA) (Sigma) in HBSS–glucose and incubated for 20 min in a 37°C water bath to allow enzymatic digestion of the tissue; 0.5 ml of 0.2% DNase (Sigma) in HBSS–glucose was added, followed by three additional washes of 0.2% DNase. The solution was triturated using a series of fire-polished Pasteur pipettes of decreasing diameters in order to obtain a single-, or near single-, cell suspension. Cell viability was assessed using the trypan blue (Sigma) exclusion assay. The cell suspension was diluted to obtain a concentration of approximately 100,000 cells/μl (4,19) (Fig. 1A).

Comparable graft survival in YAC128 and noncarrier mice. (A) Eight-month-old YAC128 and noncarriers (NC) were sacrificed 1 and 3 months following transplantation of E13.5 green fluorescent protein, positive (GFP+) fetal striatal cell suspensions. (B) Body weight was monitored for the entire duration of the study (11 months), and neither the YAC128 nor the NC mice suffered weight loss following the transplantation procedure. In fact, the YAC128 mice demonstrated a greater weight gain than the control animals (p < 0.005), as previously reported in these animals (34). (C) Representative examples of GFP+ grafts, as observed throughout the rostrocaudal extent of the striatum in YAC128 mice and NCs at 1 and 3 months posttransplantation. The asterisk next to the schematic drawing of the striatum indicates the location of the transplant for which the photomicrograph has been provided. (D–G) Plots of the total number of GFP+ surviving cells (D), graft area [both total graft area (E) and area of the largest graft identified in a single animal (F)] as well as the total graft volume (G). None of these analyses yielded a significant difference in the extent of graft survival in YAC128 and NC mice at either 1 or 3 months posttransplantation, indicating that single-cell suspensions favor graft viability and that the Huntington's disease (HD)-like diseased environment of the YAC128 mouse model does not impact on graft viability at these time points. Scale bar: (C) 100 μm. Abbreviations: CC, corpus callosum; LGE, lateral ganglionic eminence; LV, lateral ventricle; NC, noncarrier; WGE, whole ganglionic eminence; 1m and 3m, 1 month and 3 months.
Animal Model and Transplantation Procedure
A total of 36 transgenic HD male mice of the YAC128 line and 36 noncarrier (NC) littermates were purchased from Jackson Laboratories (Bar Harbor, ME, USA) at 2 months of age. They were housed one per cage under standard conditions with free access to food and water, randomized, and handled by one investigator. Genotype was confirmed for all mice in the study. Mice were transplanted with GFP+ cell suspensions at 8 months of age— the time at which they are reported to be symptomatic (42)—and were sacrificed at 1 or 3 months posttransplantation (total of n = 18 mice per experimental group).
To perform intracranial cell implantation, mice were anesthetized with 1.5% isoflurane (Abbott, Saint-Laurent, CA, USA) in 100% oxygen at a rate of 2 ml/min and placed in a stereotaxic frame (Kopf Instruments, Tujunga, CA, USA). They received bilateral striatal injection of 1 μl of the cell suspension (100,000 cells/μl) per side over a 2-min period using a Hamilton syringe (Hamilton, Reno, NV, USA) and a 26-gauge needle (ThermoFisher Scientific, Ottawa, ON, Canada). The following striatal coordinates were used: 1) AP +0.5 mm, ML −1.8 mm, and DV −3.5 mm; 2) AP +0.5 mm, ML +1.8 mm, and DV −3.5 mm (32). At the completion of cell inoculation, the needle was left in position for 3 min and then slowly withdrawn. The cranial skin was closed using silk 4.0 sutures (Ethicon, Norwalk, CT, USA). Mice were removed from the stereotactic apparatus and put back in their home cage for recovery. They received 0.5 ml/day of NaCl 0.9% (Hospira, Montreal, QC, Canada) for rehydration and 0.1 ml/12 h of buprenorphine (Vetergesic, CDMV, Québec, QC, Canada) to reduce pain on the day of surgery and for 3 subsequent days. All mice were monitored daily for weight loss and general health. All experiments were performed in accordance with the Canadian Guide for the Care and Use of Laboratory Animals, and all procedures were approved by the Institutional Policy of the Centre de Recherche du CHU de Québec (CHUQ).
Postmortem Histological Evaluation
At the completion of the experimental protocol, mice were sacrificed by intracardiac perfusion of saline (0.9%) followed by 4% paraformaldehyde (PFA; Sigma), pH 7.4. Brains were collected and postfixed in the same PFA solution for 6 h and transferred to 20% sucrose (Sigma) made in phosphate-buffered saline (PBS; Sigma) 0.1 M, pH 7.4, for cryoprotection. Coronal sections of 25 μm thickness were collected using a freezing microtome (Leica Microsystem, Montreal, QC, Canada), placed in antifreeze solution (sodium phosphate monobasic 0.2 M, pH 7.3; sodium phosphate dibasic 0.2 M, pH 7.3; ethylene glycol 30%; and glycerol 20%; all Sigma), and stored at −20°C until use.
Immunofluorescence
Sections were washed three times in potassium phosphate-buffered saline (KPBS; Sigma) 0.1 M, pH 7.4. The sections stained for GFP were directly incubated in a blocking solution after the initial washes, while sections double stained for GFP and laminin were incubated with proteinase K (Roche Diagnostic, Laval, CA, USA) diluted in a 0.1 M Tris (Sigma) and 0.5 M EDTA (Sigma) buffer diluted in water, at 37°C for 20 min. Sections were cooled down at room temperature (RT), further washed in KPBS, and preincubated in a blocking solution containing 0.1% bovine serum albumin (BSA; Bioshop, Burlington, ON, Canada), 0.04% Triton X-100 (Sigma), 4% normal goat serum (NGS; Wisent Inc., St-Jean-Baptiste de Rouville, QC, Canada) in KPBS for 30 min. Sections were next washed with KPBS and incubated overnight at 4°C in a blocking solution containing the primary antibody anti-GFP (mouse anti-GFP; Invitrogen, 1:1,000) alone or in combination with anti-laminin (rabbit anti-laminin; DAKO, Burlington, ON, Canada; 1:1,000). After KPBS washes (3 × 10 min), sections were incubated with appropriate secondary antibodies in a blocking solution for 90 min at RT. After an additional three washes, sections were incubated in KPBS containing 0.022% 4′,6-diamidino-2-phenylindole (DAPI, 2 mg/ml; Molecular Probes, Eugene, OR, USA), washed and mounted on Superfrost slides (Fisher Scientific, Ottawa, ON, Canada), coverslipped with Fluoromount-G (SouthernBiotech, Birmingham, AL, USA), and sealed with nail polish. Photomicrographs were acquired using Simple PCI version 5.0 (Hamamatsu, Sewickley, PA, USA) software linked to a Nikon eclipse 90i microscope (Nikon Instruments, Toronto, ON, Canada). Confocal laser scanning microscopy was performed using an Olympus FV500 confocal laser scanning microscope (Olympus America Inc., Melville, N Y, USA). Images were acquired by sequential scanning and optimized by a two-frame Kalman filter and analyzed using acquisition software from Olympus (Fluoview SV500 imaging software 4.3, Olympus America Inc.) and ImageJ (NIH, Bethesda, MD, USA). All images were prepared for illustration using Adobe Photoshop CS5 and Adobe Illustrator CS5 (San Francisco, CA, USA).
Assessment of Graft Survival
All volumetric quantifications were performed by three-dimensional (3D) graft reconstruction using GFP immunofluorescence-stained striatal sections. The contours of the grafts, as well as the striatum, were delineated using the Tracing Contours option in StereoInvestigator [Microbrightfield (MBF) Bioscience, Williston, VT, USA] operated on a E800 Nikon microscope (Nikon Instruments, Toronto, ON, Canada), imported into the software NeuroExplorer (Microbrightfield), and aligned for 3D reconstructions. After delineating the grafts at low magnification (4× objective), GFP+ cells were further stereologically counted using the optical fractionator method at higher magnification (20× objective). In cases of graft misplacement (observed outside the anatomical borders of the striatum), the mouse was discarded from further analysis.
Blood Vessel Measurement and Quantification
Diameter measurements as well as quantification of the number of laminin+ blood vessels were performed using 20× magnification images collected with the software Simple PCI version 5.0 linked to a Nikon eclipse 90i microscope. The diameters of laminin+ vessels were measured from 40 sampled vessels and are expressed as mean ± SEM. (9). The number of blood vessels was calculated in each sampled field of equal area using the standardized method of ImageJ (NIH) for particle analysis (9). The results are reported as a number of blood vessels per field of view and expressed as mean ± SEM.
Statistical Analysis
A two-way ANOVA was used to compare both graft viability and vasculature in YAC128 and NC mice at 1 and 3 months posttransplantation. For the total number of GFP+ cells and total graft area analyses, a different variance was estimated for each of the four groups in order to meet the model assumptions. Step-down Bonferroni correction was used for multiple comparison tests, and the level of statistical significance was set at p < 0.05. Data are expressed as group mean ± SEM. Statistical analyses were performed using the MIXED procedure of SAS (version 9.2, SAS, Cary, NC, USA) and PRISM 4 (Graphpad Software, San Diego, CA, USA).
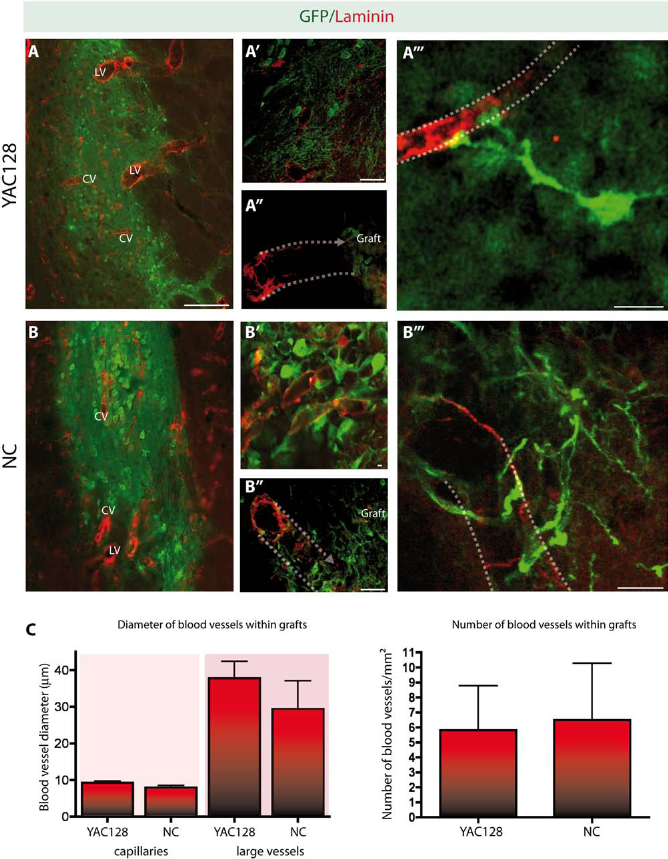
Comparable graft vascularization in YAC128 and noncarrier littermates. (A–B) Single-cell suspension grafts showed similar patterns of vascularization in both the YAC128 and noncarrier (NC) mice. Host blood vessels, identifiable as laminin+ (red) but GFP– (green) blood vessels, were as frequently encountered in the transplants of the YAC128 as in the NC mice. The vascularization pattern was similar regardless of the time points evaluated. Both capillaries (diameter size ranging between 6 μm and 13 μm) and large-caliber blood vessels (ranging between 20 μm and 45 μm) were observed within the transplanted tissue (A–B). (A′ and A″ and B′ and B″) Confocal images of GFP negative host blood vessels revascularizing the grafts. Dotted lines have been added to indicate the direction of the blood vessels. (A′″ and B′″) Representative images of interactions observed between GFP+ grafted cells and host blood vessels. (C) Quantification of blood vessel diameters, as well as the total number of blood vessels encountered within the grafted tissue of YAC128 and NC mice, revealed similar patterns in each group of animals. Note that the detection of GFP+-grafted cells was further enhanced using GFP immunofluorescence staining (green), while blood vessels were identified using an antibody against the basement membrane protein laminin (red). Scale bars: (A, B) 100 μm, (A′–B″) 25 μm, (A′″) 10 μm, (B′″) 12.5 μm. Abbreviations: CV, capillary vessels; LV, large-caliber vessels; NC, noncarrier.
Results
Cell Suspension Grafts Survive Well in the YAC128 HD Mouse Model
All animals in this study were closely monitored throughout the 11-month period of the experimental protocol. YAC128 and NC mice, transplanted at 8 months of age, recovered well from the grafting procedure as evidenced by progressive weight gain and the absence of any health-related issues (Fig. 1B). Furthermore, the weight gain observed in the YAC128 mice was significantly greater than in the NC controls (p < 0.05). Similar weight gains have previously been reported in YAC128 mice and have been suggested to be due to levels of full-length Htt and plasma insulin-like growth factor (IGF-1) (33) (Fig. 1B). At postmortem, GFP-enhanced immunofluorescence staining was performed to confirm graft location. In both YAC128 and noncarriers, GFP+ cell suspension grafts could be identified throughout the rostrocaudal extent of the striatum. A series of quantitative analyses were subsequently performed to determine the extent of graft survival, which included calculation of the total number of surviving GFP+-transplanted cells (Fig. 1D), the total graft area (Fig. 1E), the area of the largest transplant found in a single animal (Fig. 1F), and the total graft volume (Fig. 1G). None of these quantifications revealed a significant difference between survival of grafted cells, surface areas, or graft volumes at the time points evaluated, further suggesting that the evolution of pathology, observed in the YAC128 mice between 8 and 11 months of age, does not significantly impact on the survival of cell suspension grafts.
Cell Suspension Grafts Are Well Vascularized in the YAC128 HD Mouse Model
We further evaluated whether the methodological transplantation approach of cell suspension grafts would yield adequate vascularization of transplanted cells by the host. Grafts were stained with GFP and blood vessels with laminin, a marker of the basement membrane. Confocal imaging microscopy revealed similar patterns of vascularization of grafted tissue in YAC128 mice and noncarriers, at the two time points studied (Fig. 2A, B, and C). In the YAC128 and NC mice, cell suspension grafts were clearly vascularized by both capillaries and larger-caliber blood vessels (Fig. 2C). Importantly, the absence of colocalization of laminin and GFP suggests that the vascular network of the transplanted tissue derived exclusively from the host (Fig. 2A, A′, A″ and B, B′, B″). Confocal microscopy further revealed interactions between grafted cells and blood vessels of the host in both YAC128 and noncarrier mice (Fig. 2A′″ and B′″).
Discussion
This study was designed to specifically address whether single-cell suspension methodology favored graft survival and vascularization in the context of HD-like pathology. We transplanted dissociated E13.5 LGE cells derived from GFP+ embryos into 8-month-old YAC128 and noncarrier mice and performed postmortem analyses at 1 and 3 months following grafting. In this short communication, we report that similar graft viability and vascularization patterns were observable in both the YAC128 and noncarrier mice. Contrary to our recent observations of poor vascularization of solid fetal striatal allografts in HD patients (9), single-cell suspensions in YAC128 and NC mice were characterized by a blood network composed of both capillaries and large-caliber blood vessels. Furthermore, grafts survived well at 1 and 3 months posttransplantation despite the pathological environment characterizing the YAC128 brain. These results suggest that methodological aspects relating to cell preparation may impact graft survival and therefore the outcome of cell replacement therapy.
Studies investigating the efficacy of neural cell transplantation in transgenic animal models of HD are scarce. We have itemized only six publications, which have reported on the survival and efficacy of cell transplantation in in vivo HD transgenic mouse models (see Table 1 for summary). These studies were performed in R6/2 (transgene insertion 62; exon 1 with 150 CAG repeats) (12,14,26,28), YAC128 (20), and N171-82Q (transgene insertion 81; first 171 aa and 82 CAG repeats) (37) mice and evaluated the efficacy of a variety of cell types including embryonic neuronal cells (12), adipose stem cells (18), mesenchymal stem cells (28,37), and more recently human neuronal stem cells (14) (Table 1). Overall, these studies demonstrated that transplanted cells increased striatal volume and endogenous cell proliferation, likely via heightened trophic support. Our findings are very similar to those reported in the R6/2 mice by Dunnett et al. (12). In their study, the authors demonstrated that intracerebral engraftment of striatal fetal cell suspension in the R6/2 mouse, which expresses the truncated mHtt gene, yields similar survival to that seen in control animals (12). However, their effects on motor behavior are modest and not observable on disease progression. For the purpose of our study, we chose to utilize the HD YAC128 murine model, which was generated to express the full-length mHtt gene (36). The YAC128 model recapitulates several HD-like features including behavioral and cognitive deficits, selective striatal cell loss, as well as nuclear aggregates, the characteristic neuropathological hallmarks of the disease (36,41). Compared to the R6/2 mouse, the YAC 128 presents a more progressive development of pathological features, providing a longer time frame to investigate graft viability and events such as graft vascularization (10,36). We observed similar graft viability in the YAC 128 transgenic mice and control littermates at both 1 and 3 months posttransplantation, as previously reported in the R6/2 model (12). The percentage of grafted surviving cells ranged between 7% and 8%, which is also similar to previous reports (38,40).
Summary of Transplantation Studies Performed in Transgenic Animal Models of Huntington's Disease

FGF-2, fibroblast growth factor 2; hASCs, human adipose-derived stem cells; hBM-MSCs, human bone marrow-derived mesenchymal stem cells; HD, Huntington's disease; LGE, lateral ganglionic eminence; NGF, nerve growth factor; QA, quinolinic acid; VEGF, vascular endothelial growth factor; vWF, Von Willebrand factor.
In a recent publication, we have shown limited revascularization of solid tissue grafts in HD patients a decade posttransplantation (9). The transplants were vascularized by capillaries but not larger blood vessels. Similar results were observed 18 months postsurgery in a Parkinson's disease individual who also received a solid fetal tissue transplant (22). Adequate vascular development between the host and donor tissue is essential to graft viability and prevention of ischemic damage (24,29) and can have significant implications on overall functional and structural graft integration (43) (Table 2). Transplants that are rapidly revascularized by the host have indeed a higher survival rate (24,29). However, graft reperfusion can be affected by various factors such as graft size (31) and techniques of cell preparation (cell suspension vs. solid tissue grafts) (5). Solid tissue grafts transplanted directly into the parenchyma have a less rich vascular environment and so have poorer survival and functionality. For example, solid tissue transplants implanted into 6-OHDA-lesioned striata require a longer time to develop functional neuronal contacts from the grafted neurons and generate motor improvements when compared to cell suspension grafts (3). Generally, solid grafts are more slowly revascularized. At 24 h posttransplantation, there is no integration between the host and the solid tissue transplant vasculature, as observed in piebald Virol Glaxo (PVG) rats transplanted with hippocampal tissue (25). One week postsurgery, marginal vessels start to penetrate the transplant, and only by 1 month following the implantation are the grafts revascularized and depict a vascular organization similar to the host (25,35). Human neuronal tissue engrafted into the kainic acid-lesioned striatum of rats has demonstrated even greater delayed revascularization. In this particular study, the presence of capillaries was detected only 2 months posttransplantation and revascularization at 3 months postsurgery (19). However, endothelial cells originating from the grafts interacted with the host to form blood vessel chimeras and functional revascularization (2,29,43).
Cell Transplantation Studies Addressing Vascularization of Grafted Tissue in Animal Models and Human Cases

6-OHDA, 6-hydroxydopamine; α-SMA, a smooth muscle actin; AKR, aldo keto reductase inbred mouse strain; BBB, blood-brain barrier; E, embryonic day; GLUTI, glucose transporter 1; NA, not available; P0, day of birth; P1, postnatal day 1; PC 12, pheochromocytoma of the rat adrenal medulla; PD, Parkinson's disease; PVG, Piebald Virol Glaxo inbred rat strain; vWF+, Von Willebrand factor.
The connection between host and graft blood networks predominantly takes place at the border of the transplanted tissue and rarely infiltrates the core of the grafts, despite the fact that host blood vessels are mitotically active (24,27). The method of cell suspension was in fact developed to improve survival and integration of the transplanted cells (3). However, mechanical trituration, required to prepare cell suspension grafts, compromises the intrinsic vasculature of the dissected tissue (2). Contrary to solid grafts, which maintain their own angioarchitecture but are characterized by sparse blood vessels from the donor (1,2), cell suspension revascularizes more rapidly and relies exclusively on the host vasculature and angiogenic process (2,5). One study has demonstrated that nimodipine, a pharmacological approach to promote angiogenesis, can ameliorate the vascular supply and survival of neural grafts in 6-OHDA-lesioned mice (15), demonstrating the importance of proper graft revascularization and the possibility to pharmacologically ameliorate the outcome of graft vasculature.
Our findings, combined with our recent observations in transplanted HD patients, support the idea that graft survival may be improved by optimizing methodological aspects of tissue preparation and vascularization.
Footnotes
Acknowledgment
This work was supported by Huntington Society Canada and the International Organization of Glutaric Acidemia (IOGA) to Francesca Cicchetti. Giulia Cisbani was supported by a Bourse d'excellence du Centre thématique de recherche en neurosciences (Université Laval). The authors wish to thank Mr. Gilles Chabot for artwork. The authors declare no conflicts of interest.
