Abstract
Transplantation of bone marrow (BM) CD34+ cells, an endothelial/hematopoietic progenitor-enriched cell population, has shown therapeutic efficiency in the treatment of ischemic diseases enhancing neovascularization. However, the number of CD34+ cells obtained from bone marrow is not sufficient for routine clinical application. To overcome this issue, we developed a more efficient and clinically applicable CD34+ cell expansion method. Seven-day ex vivo expansion culture of BM CD34+ cells with a cocktail of five growth factors containing VEGF, SCF, IL-6, Flt-3 ligand, and TPO resulted in reproducible more than 20-fold increase in cell number. The favorable effect of the local transplantation of culture expanded (cEx)-BM CD34+ cells on rat unhealing fractures was equivalent or higher than that of nonexpanded (fresh) BM CD34+ cells exhibiting sufficient therapeutic outcome with frequent vasculogenic/osteogenic differentiation of transplanted cEx-BM CD34+ cells and fresh BM CD34+ cells as well as intrinsic enhancement of angiogenesis/osteogenesis at the treated fracture sites. Specifically, cEx-BM CD34+ cell treatment demonstrated the best blood flow recovery at fracture sites compared with the nonexpanded BM CD34+ cells. In vitro, cEx-BM CD34+ cells showed higher colony/tube-forming capacity than nonexpanded BM CD34+ cells. Both cells demonstrated differentiation potential into osteoblasts. Since fresh BM CD34+ cells can be easily collected from fracture sites at the time of primary operation and stored for future use, autologous cEx-BM CD34+ cell transplantation would be not only a simple but also a promising therapeutic strategy for unhealing fractures in the field of orthopedic trauma surgery.
Introduction
Although most fractures can be healed with bridging callus formation by conservative or surgical treatments, 5–10% of fractures do not achieve union and culminate in delayed union or persistent nonunion (9, 19, 46, 61), causing serious problems for the patient's quality of life. Therefore, establishment of a novel therapeutic strategy for nonunion healing is clinically warranted. Deficient bone formation after fracture is mainly caused by the impairment of neovascularization (9, 45, 46, 48, 61). The majority of tissues require appropriate vascularization and blood supply to support proper function. Without them, cells suffer from hypoxia, lack of nutrients, accumulation of waste products, and disruption of biomechanical signaling pathways, affecting tissue homeostasis and preventing tissue regeneration (17). Aiming for the induction of neovascularization at sites of fracture, cell therapy may provide a novel therapeutic strategy to modify and improve the fracture healing process and prevent nonunion.
Since Asahara et al. reported that circulating CD34+ cells isolated from adult human peripheral blood (PB) contained endothelial progenitor cells (EPCs) and contributed to vasculogenesis (3), their role in angiogenesis and neovascularization has been studied extensively in vitro and in vivo (25, 31, 33, 62, 64, 67). Currently clinical studies of autologous EPC transplantation are ongoing, aiming for the development of a safe and effective treatment for nonoption patients with severe ischemic diseases (26, 29, 32, 53, 59, 68, 72). With cooperation between neovascularization and osteogenesis having been reported to be a key event in fracture healing (1, 4, 6, 9, 41), stem/progenitor cell-based therapies are gaining interest in the field of skeletal biology and orthopedic surgery. Several reports have also suggested that EPC-rich cell populations can promote osteogenic differentiation (8, 14, 15, 39, 69) in addition to its well-studied vasculogenic differentiation potential. Encouraged by the accumulating evidence for a role of EPCs in bone homeostasis and repair, our group performed a series of studies focusing on the effect of CD34+ cell transplantation on fracture repair (48, 50, 52). First, we demonstrated that the systemic infusion of human PB CD34+ cells contributed to fracture repair via vasculogenesis and osteogenesis in immunodeficient rats with unhealing fractures (48). Subsequently, it was revealed that local transplantation of human granulocyte-colony stimulating factor-mobilized PB (GM-PB) CD34+ cells also contributed to fracture healing in a dose-dependent manner (52). Based on these preclinical achievements, we have recently also started a phaseI/II clinical trial of local transplantation of autologous GM-PB CD34+ cells in patients with nonunion fracture, demonstrating a case in which fracture healing was accelerated radiographically and symptomatically following cell therapy (37).
Recently, not only human PB CD34+ cells but also BM CD34+ cell transplantation for hindlimb ischemia or myocardial ischemia has shown therapeutic efficiency in enhancing ischemic neovascularization (5, 40). Chen et al. (8) identified osteoblast (OB) precursor cells in human BM CD34+ cells. Moreover, a subpopulation of CD34+ cells (less than 5%) from adult human BM expressing the mesenchymal marker STRO-1 has been shown to differentiate into fibroblasts, adipocytes, smooth muscle cells, and macrophages (65). These findings strongly suggest the therapeutic feasibility of BM-derived CD34+ cells not only for vasculogenesis but also for osteogenesis. Especially, in trauma surgery, the use of BM CD34+ cells that can be easily collected at the fracture site in a primary operation is more convenient than hPB CD34+ cells as a cell source. However, since EPCs represent less than 1% of all BM cells and less than 0.01% of PB mononuclear cells (MNCs) (66), the number of CD34+ cells obtained regardless of their origin, BM or PB, is not sufficient for routine clinical applications. For instance, results from preclinical studies have suggested that more than 10 L of autologous PB will be required to collect a sufficient number of EPCs to induce angiogenesis in one patient (28). In addition, aging and cardiovascular risk factors will also make it difficult to obtain a high enough number of EPCs for cell therapy (12, 23). Access to a functional and adequate amount of EPCs appears thus to be a critical factor for effective cell therapy, with ex vivo expansion of these cells offering a way to overcome this initial and important hurdle.
Therefore, we explored strategies to expand EPCs efficiently while avoiding rapid cellular senescence. Since proliferative senescence is frequently observed in hematopoietic stem cells (HSCs) during ex vivo suspension culture (34, 57), we addressed and tried to overcome this issue by utilizing a recently developed and optimized serum-free nonadhesive culture condition for the ex vivo expansion of stem/progenitor cells, shown to result in an increase of cell number up to 20- to 50-fold within 1 week (H. Masuda et al., manuscript in preparation). In the present study, we have examined the in vitro and in vivo properties of culture expanded (cEx)-BM CD34+ cells and tested the hypothesis that local transplantation of cEx-BM EPCs exhibits a similar or higher potent therapeutic effect for fracture healing when compared with freshly isolated nonexpanded BM CD34+ cells.
Materials and Methods
Preparation of Human Cells
We purchased BM-CD34+ cells isolated from healthy men (21 years old, African-Americans) from Lonza (Basel, Valais, Switzerland) and used them for all subsequent experiments.
Ex Vivo BM CD34+ Cell Expansion
Ten thousand BM CD34+ cells in 500 μl of medium were plated into each well of a 24-well tissue culture dish (Primaria™, BD Falcon), cultured in Stem Span medium (STEM CELLS Veritas, Japan) without serum, and supplemented with five growth factors, 50 ng/ml vascular endothelial growth factor (VEGF; R&D Systems, Minneapolis, MN, USA), 20 ng/ml interleukin-6 (IL-6; R&D Systems), 100 ng/ml stem cell factor (SCF; Kirin), 20 ng/ml thrombopoietin (TPO; Wako), 100 ng/ml Fms-related tyrosin kinase-3 (Flt-3) ligand (Wako), and antibiotics. Cultures were maintained at 37°C with 5% CO2 in a humidified incubator. After 7 days in culture, the cells were harvested by washing with phosphate-buffered saline (PBS) and used for flow cytometry analysis. One-week culture-expanded BM CD34+ cells (cEx-BM CD34+ cells) and BM CD34+ cells were used for additional experiments as described below.
Flow Cytometry Analysis
We analyzed BM CD34+ cells and cEx-BM CD34+ cells to assess their content of CD34+ cells and checked also for the expression of other hematopoietic and mesenchymal lineage markers via fluorescence-activated cell sorting (FACS) using a FACSCalibur™ flow cytometer (BD Biosciences, San Diego, CA, USA) and CellQuest software (Becton, Dickinson and Company, Mountain View, CA, USA) as described previously (48).
Cell Proliferation Assay of cEx-BM CD34+ Cells and Fresh BM CD34+ Cells
The proliferation activity of cEx-BM CD34+ cells and BM CD34+ cells was examined using a Cell Counting Kit-8 (Dojindo Laboratories, Inc., Kumamoto, Japan) according to the manufacturer's instructions. Briefly, both cells were seeded onto 96-well culture plates at a density of 5 × 103 cells per well and cultured in DMEM with 10% fetal bovine serum (FBS) for 48 h at 37°C in 20% O2/5% CO2. Optical density was measured using a plate reader at 490-nm wavelength. Both groups were studied at least in triplicate.
Relative Telomere Measurement with Real-Time Quantitative PCR
Genomic DNA was purified with the standard procedure described in PureLink Genomic DNA Purification Kit (Invitrogen, Carlsbad, CA, USA). The eluent was concentrated with ethanol precipitation, and genomic DNA was dissolved in 20 ml of Tris–EDTA (TE) buffer (pH 8.0).
Relative telomere length was analyzed by using the technique as described previously (7, 10). Real-time qPCR mixture included MESA GREEN qPCR MaterMix Plus (Eurogentec, Seraing, Belgium) and 10 pmol of forward and reverse primers. One microliter of DNA diluted in pure water at 100-fold dilution was used in the qPCR reaction. PCRs were performed on the 7900 Sequence Detection System (Applied Biosystems, Foster City, CA, USA) into each reaction well of a 384-well plate. The PCR thermal conditions consisted of an activation of Meteor Taq polymerase at 95°C for 10 min followed by a total of 40 cycles at 95°C for 5 s, 54°C for 2 min for telomere repeat sequence (T) or followed by a total 40 cycles at 95°C for 5 s, 58°C for 1 min for a reference single copy gene, 36B4 (S) and a positive control gene for validation of 36B4, β-globin. The primer sequences (written 5′→3′) were as follows: tel 1, GTTTTTGAGGGTGAGGGTGAGGGTGAGGGTGA GGGT; tel 2, TCCCGACTATCCCTATCCCTATCCCTAT CCCTATCC-CTA; 36B4u, CAGCAAGTGGGAAGGTGT AATCC; 36B4d, CCCATTCTATCATCAACGGGTACAA.
The relative ratio of telomere repeat sequence (T) versus single copy gene (S), referred to as T/S ratio, was calculated to determine the relative telomere length of pre- and postexpanded BM-CD34 cells. It was confirmed for correct amplification of 36B4 in all experimental DNAs that the range of relative ratio (36B4/β-globin) was 0.95–1.05 (data not shown).
EPC Colony-Forming Assay of cEx-BM CD34+ Cells and Fresh BM CD34+ Cells
The EPC colony-forming assay established in our laboratory was performed as reported previously (38, 47). The number of EPC colonies was assessed after culture of fresh BM CD34+ cells and cEx-BM CD34+ cells in a six-well plate for 16 days in methyl cellulose-containing medium M3236 (Stem Cell Technologies, Vancouver, BC, Canada) with 20 ng/ml SCF (Kirin, Tokyo, Japan), 50 ng/ml VEGF (R&D Systems), 20 ng/ml interleukin-3 (Kirin), and 50 ng/ml basic fibroblast growth factor (bFGF) (Wako, Osaka, Japan). Two types of attaching cell colonies made of small EPCs and large EPCs were counted.
In Vitro Tube Formation Assay
To compare the functional activity between BM CD34+ cells and cEx-BM CD34+ cells (n = 3), tube formation assay was performed. In vitro formation of cord-like structures was assessed using a cell culture system with growth factor-reduced Matrigel (BD Bioscience, Bedford, MA, USA). Fifty microliters of Matrigel basement membrane matrix diluted with vascular cell-based medium (ATCC) without supplement (1:1 dilution) was pipetted into each well of a 96-well plate and allowed to solidify for 20 min in a 5% CO2 incubator at 37°C. To evaluate incorporation of both types of CD34+ cells into cord-like structures, cells were labeled with fluorescent carbocyanine 1,1′-dioctadecyl-I to 3,3,3′,3′-tetramethylindocarbocyanine perchlorate (DiI) dye (Molecular Probes), with each 1,000 DiI-labeled CD34+ cells and 12,500 human umbilical vein endothelial cells (HUVECs) being mixed in 50 μl of endothelial basal medium-2 (EBM-2) supplemented with 1% FBS and seeded onto growth factor-reduced Matrigel-coated 96-well plates. Twenty-four hours and 7 days in culture, morphological changes were examined with a phase-contrast microscope. Digital microphotographs were taken from four randomly selected fields (original magnification: 40x) per subject. The number of intact tube formations was assessed by measuring the total length and number of incorporated DiI+ cells in the fields with the aid of ImageJ (http://rsbweb.nih.gov/ij/).
Induction of Osteogenic Differentiation In Vitro
To induce mesenchymal stem cells from BM CD34+ cells similar to a previously described method using BM MNCs (69), we seeded 1 × 105 BM CD34+ and cEx-BM CD34+ cells in six-well plates with α-minimum essential medium (α-MEM) (Gibco-BRL, Tokyo, Japan) supplemented with 10% FBS (Vitromex, Vilshofen, Germany), 2 mM L-glutamine, (Gibco-BRL), and 10% conditioned medium (CM) during the first 7 days of culture. Cells were then cultured in α-MEM supplemented with 10% FBS and 2 mM L-glutamine for the next 2 weeks changing the medium twice a week. To induce osteogenic differentiation, 100,000 of each pretreated CD34+ cell population were plated in six-well plates and cultured for 3 weeks under specific osteogenic conditions in α-MEM supplemented with 10% FBS, 2 mM L-glutamine, 60 μM ascorbic acid (Sigma-Aldrich, St. Louis, MO, USA), 10 mM β-glycerophosphate (Sigma-Aldrich), and 0.1 μM dexamethasone (Sigma-Aldrich). Cells were also cultured with α-MEM supplemented with 10% FBS and 2 mM l-glutamine as a negative control.
Animal Model of Unhealing Bone Fracture
Female athymic nude rats (F344/N Jcl rnu/rnu, CLEA Japan, Inc., Tokyo, Japan) aged 8–12 weeks and weighing 150–170 g were used in this study. All surgical procedures were performed under anesthesia and normal sterile conditions. As described previously, a reproducible model of unhealing bone fracture was created in the femur of nude rat by cauterizing the periosteum on each side of the fracture according to the modification of the original methods (16, 35), which led to nonunion 8 weeks after fracture creation. All animal procedures were performed in accordance with the Japanese Physiological Society Guidelines for the Care and Use of Laboratory Animals, and the study protocol was approved by the Ethical Committee of the RIKEN Center for Developmental Biology.
Cell Transplantation
Following FACS analysis, the CD34+ cell numbers were adjusted to the effective doses as determined in a previous study (52), with one cEx-BM CD34 group, two BM CD34 groups, and one PBS control group. Immediately after creation of the unhealing fractures, rats received local administration of the following materials mixed in atelocollagen gel (KOKEN, Tokyo, Japan), which was used as a bioabsorbable scaffold to retain the cells in the transplantation site (24, 27): (A) cEx-BM CD34+ cells (cEx-BMCD34 group, 1 × 104 generated from about 5 × 102 BM CD34+ cells); (B) high-dose BM CD34+ cells [BMCD34 (Hi) group, 1 × 104]; (C) low-dose BM CD34+ cells [BMCD34 (Lo) group, 1 × 103] with all cells suspended in 100 μl of PBS; or (D) the same volume of PBS alone (n = 23 in each). The contralateral intact femurs were used as controls for histological and functional analyses.
Tissue Harvesting
Rats were euthanized with an overdose of ketamine and xylazine for biomechanical and histological analyses in the indicated time course described below. The femurs were directly frozen for biomechanical analysis, embedded in OCT compound (Sakura Finetek Japan) for histological analysis, and quickly frozen in liquid nitrogen and stored at −80°C for conventional or real-time RT-PCR analysis. Three rats were randomly selected and euthanized in each group at weeks 1, 2, and 4 after surgery. The remaining rats were sacrificed at week 8. Bilateral femurs were harvested and quickly treated for the above-mentioned analyses as described previously (29, 49).
RT-PCR and Quantitative Real-Time RT-PCR Analyses
Total RNA was isolated from granulation tissues and callus tissues present at the perifracture sites 1 and 2 weeks after transplantation using TRIzol kit (Life Technologies, Gaithersburg, MD, USA) according to the manufacturer's instructions. RNA isolated at week 1 was analyzed to detect human-specific cell markers and the expression of cytokines in the recipient fracture sites. First-strand cDNA was synthesized using a RNA LA PCR Kit Ver1.1 (TAKARA BIO, Inc., Shiga, Japan) and amplified by Taq DNA polymerase “Advantage-GC cDNA PCR Kit” (Clontech, Mountain View, CA, USA) and “AmpliTaq Gold DNA polymerase” (Applied Biosystems). PCR oligonucleotide primers are listed in Table 1. PCR was performed using a PCR thermocycler (MJ Research, Waltham, MA, USA) under the following conditions: 35 cycles of 30-s initial denaturation at 94°C, annealing at 56°C for 1 min, and 30 s of extension at 72°C. Human umbilical vein endothelial cells and normal human osteoblasts (hOBs) (Cambrex, East Rutherford, NJ, USA) were used as positive controls for human-specific endothelial and bone-related genes, respectively.
Specific Primers for RT-PCR Amplifications
hVE-cad, human vascular endothelial cadherin; hCD31, human CD31; hOC, human osteocalcin; hCol1A1, human collagen 1 α1; hGAPDH, human glyceraldehyde-3-phosphate dehydrogenase; rGAPDH, rat glyceraldehyde-3-phosphate dehydrogenase; bp, base pair.
For quantitative real-time RT-PCR, after the first-strand cDNA was synthesized using an RNA LA PCR Kit Ver1.1 (TAKARA BIO, Inc., Shiga, Japan), the converted cDNA samples (2 μl) were amplified in triplicate by real-time PCR (ABI PRISM 7700, Applied Biosystems) in a final volume of 20 μl using SYBR Green Master Mix reagent (Applied Biosystems) with the gene-specific primers listed in Table 2. Melting curve analysis was performed with Dissociation Curves Software (Applied Biosystems). Results were obtained using sequence detection software (ABI PRISM 7700), and the mean cycle threshold (Ct) values were used to calculate gene expression with normalization to human or rat glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
Specific Primers for Real-Time RT-PCR Amplifications

rVEGF, rat vascular endothelial growth factor; rANG-1, rat angiopoietin-1; rBMP-2, rat bone morphogenetic protein-2; rBMP-4, rat bone morphogenetic protein-4; hVE-cad, human vascular endothelial cadherin; hCD31, human CD31; hOC, human osteocalcin; hCol1A1, human collagen 1 α1; rGAPDH, rat glyceraldehyde-3-phosphate dehydrogenase; hGAPDH, human glyceraldehyde-3-phosphate dehydrogenase; bp, base pair.
Immunohistochemical Staining
To detect differentiation of transplanted human cells in the rat perifracture sites and to evaluate capillary density and osteoblast (OB) density, immunohistochemistry was performed with the following human- or rat-specific antibodies: human nuclear antigen (hNA) (1:50 dilution, Chemicon, Billerica, MA, USA) as a sensitive marker for all transplanted human cells, biotinylated Ulex europaeus agglutinin 1 (UEA-1) (1:100 dilution; Vector Laboratories, Burlingame, CA, USA) as a sensitive marker for human endothelial cells (hECs), fluorescein-labeled isolectin B4 (FITC-conjugated ILB4) (1:100; Vector Laboratories) as a sensitive marker for rat ECs (rEC), anti-human osteocalcin (hOC) antibody (1:50; Biomedical Technologies, Inc., Stoughton, MA, USA) as a marker for human OBs, and anti-rat osteocalcin (v-19) (rOC) (1:50; Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) as a marker for rat OBs (rOB). The secondary antibodies used for each immunostaining were as follows: Alexa Fluor 594-conjugated goat anti-muse IgG1 (1:500; Invitrogen) for hNA staining, Alexa Fluor 594-conjugated streptavidin (1:200; Invitrogen) for UEA-1 staining, Alexa Fluor 594-conjugated goat anti-rabbit IgG1 (1:200; Molecular Probes, Invitrogen Japan K.K., Tokyo, Japan) for hOC, and Alexa Fluor 594-conjugated rabbit anti-goat IgG1 (Molecular Probes) for rOC. 4′,6-Diamino-2-phenylindole (DAPI) solution (1:500; Sigma-Aldrich) was applied for 5 min of nuclear staining.
Capillary and OB density was morphometrically evaluated as average values in four randomly selected soft tissue fields in the perifracture sites under a fluorescence microscope. OBs were recognized as lining or floating cells positive for OC on the new bone surface. All morphometric studies were performed by two blinded examiners.
Physiological Assessment of Tissue Perfusion
Laser Doppler perfusion imaging (LDPI) system (43, 71) (Moor Instrument, Wilmington, DE, USA) was used to measure serial blood flow in both hindlimbs at 0, 1, 2, and 3 weeks postfracture according to the manufacturer's instruction. The ratio of fractured/intact contralateral blood flow was calculated to evaluate the serial blood flow recovery after surgery. The measurement was performed under anesthesia with animals supine and both limbs fully extended. At first, we palpated the femoral fracture site under the skin and pasted a small stainless steel marker on the skin above the fracture site. The steel marker was scanned as a defect spot in the LDPI image, and we recognized it as the center of the region of interest (ROI). The steel marker was removed and scanned again. Then, we set ROI squares on the second scanned image referring to the first image with the marker's defect spot. The blood flow recovery following fracture was evaluated as a ratio of mean flux within the ROI in the fractured hindlimb to the same-size ROI in the contralateral intact hindlimb.
Radiographic Assessment of Fracture Healing
Radiographs of fractured hindlimbs were serially taken at weeks 0, 2, 4, and 8 following creation of fractures under anesthesia with the animals' supine and both limbs fully extended. Fracture union was identified by the presence of a bridging callus on two cortices. Radiographs in each animal were examined by three blinded observers. For quantification of callus/trabecular bone formation, micro-computed tomography (CT) imaging analysis was performed 8 weeks after fracture creation using LaTheta (Aloka, Co., Ltd., Tokyo, Japan). To evaluate the fracture healing process, relative callus areas around fracture sites in scanned radiographs at week 8 were quantified using LaTheta. Bone densities in the callus areas were calculated with CT intensity in scanned images by single-energy X-ray absorptiometry method and averaged using LaTheta. To recognize fracture healing more clearly, three-dimensional (3D) CT images were reconstructed using Volume Graphics GmbH Studio MAX software (Heidelberg, Germany).
Histological Assessment of Fracture Healing
Histological evaluations were performed with toluidine blue staining to address the process of endochondral ossification at weeks 1, 2, 4, and 8. The degree of fracture healing was evaluated using a 5-point scale (grades 0–4) proposed by Allen et al. (2), Samples were sectioned at 6-μm thickness on slides followed by fixation with 4% paraformaldehyde at 4°C for 5 min.
Biomechanical Analysis of Fracture Union
Three rats in each treatment group were used for biomechanical evaluation at week 8 postfracture. Biomechanical evaluation was performed at Kureha Special Laboratory (KSL, Tokyo, Japan). Fractured femurs and contralateral nonfractured femurs were prepared, and intramedullar fixation pins were removed prior to the bending test. The standardized 3-point bending test was performed using a load torsion and bending tester (MZ-500S, Maruto Instruments, Co., Ltd., Tokyo, Japan). The bending force was applied with cross-head at a speed of 2 mm/min until rupture occurred. The extrinsic stiffness (N/mm) and the ultimate stress (N) were interpreted and calculated from the load deflection curve. The relative ratio of fractured (right) femur to nonfractured (left) femur was calculated in each group and averaged.
Statistical Analysis
The results were statistically analyzed using a software package (GraphPad Prism™, MDF Software, Inc.). All values were expressed as mean ± SEM. Unpaired t tests were performed for comparison between two groups. Multiple comparisons among groups were made using one-way analysis of variance (ANOVA) followed by post hoc testing with Tukey's procedure. The comparison of radiological results was performed with a chi-square test. A value of p < 0.05 was considered to denote statistical significance.
Results
Characterization of cEx-BM CD34+ Cells and Fresh BM CD34+ Cells
The total cell number of BM CD34+ cells was increased 22.7 ± 7.8 times after culture expansion within 1 week. FACS analysis demonstrated 99.3% and 60.9% positivity of CD34 expression in BM CD34+ cells and cEx-BM CD34+ cells, respectively (Fig. 1A). cEx-BM CD34+ cells were positive for CD31 (93.3 ± 0.2%), CD133 (54.8 ± 0.4%), CD44 (98.0 ± 0.1%), CD90 (14.2 ± 0.3%), CD105 (15.8 ± 0.3%), and CD166 (13.8 ± 1.3%) and negative for c-Kit (0.7 ± 0.07%) (Fig. 1B–H). Based on the results, we determined that 104 cEx-BM CD34+ cells contained around 6 × 103 CD34+ cells, which was shown to be an effective dose of CD34+ cells in a previous study (49, 52).

Characterization of cEx-BM CD34+ and BM CD34+ cells used in this study. (A) About 60% of the culture-expanded bone marrow (cEx-BM) CD34 cells are positive for CD34 after 1 week of ex vivo culture. (B–H) Fractions of cells positive for CD31 (B), CD133 (C), c-Kit (D), CD44 (E), CD90 (F), CD105 (G), and CD166 (H) before and after expansion. (I) Proliferation assay of cEx-BM CD34+ cells and BM CD34+ cells. The proliferation activity is expressed as optical density (490 nm). **p < 0.001. (J) Relative telomere length of cEx-BM CD34+ cells and BM CD34+ cells analyzed by real-time RT-PCR.
cEx-BM CD34+ Cells Show High Ability of Proliferation and Maintain Telomere Length
The proliferation activities of cEx-BM CD34+ cells and BM CD34+ cells were expressed as a mean optical density value at a wavelength of 490 nm. cEx-BM CD34+ cells show significantly high proliferation activities compared with fresh BMCD34+ cells (cEx-BM CD34, 0.2020 ± 0.0108; BM CD34, 0.1000 ± 0.0016; p < 0.001 for cEx-BM CD34 vs. BM CD34) (Fig. 1I). These results suggest that culture expansion increases proliferation activity. The mean telomere lengths were not significantly different between cEx-BM CD34+ cells and BM CD34+ cells (cEx-BM CD34, 1.056 ± 0.01947; BM CD34, 1.038 ± 0.02754; p = NS for cEx-BM CD34 vs. BM CD34) (Fig. 1J). These findings suggest that cEx-BM CD34+ cells maintained their telomere length, that is, cell senescence was not promoted even in the specific culture conditions for expansion.
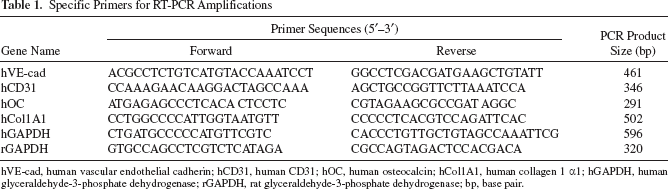
BM-Derived CD34+ Cells Show High Capacity of Colony/Tube Formation In Vitro
To evaluate the function of cEx-BM CD34+ cells and BM CD34+ cells as EPCs in vitro, both types of cells were analyzed by EPC colony-forming assay. After 16 days in culture with methylcellulose-based medium, the average number of EPC colonies per dish of cEx-BM CD34+ cells was significantly greater than that of BM CD34+ cells (small colony: cEx-BM CD34, 19.14 ± 1.65; BM CD34, 12.71 ± 1.89; p < 0.001 for cEx-BM CD34 vs. BM CD34; large colony: cEx-BM CD34, 7.00 ± 0.69; BM CD34, 4.71 ± 0.68; p < 0.05 for cEx-BM CD34 vs. BM CD34) (Fig. 2A, B). Each EPC colony-forming unit showed a differentiation hierarchy from small EPC to large EPC colonies, indicating a primitive EPC stage with high proliferative activity and a definitive EPC stage with vasculogenic properties, respectively. These findings suggest that culture expansion increases the function of BM CD34+ cells as EPCs in vitro.
EPC colony and tube formation assay. (A) Representative figures of a large colony and a small colony. (B) Average number of endothelial progenitor cell (EPC) colonies per dish of cEx-BM CD34+ cells was significantly greater than of BM CD34+ cells. *p < 0.05, **p < 0.001. (C–E) Representative immunofluorescence tube formation image of human umbilical vein endothelial cell (HUVEC)/ CD34+ cells after 7 days of coculture. The number of tube formations in cEx-BM CD34+ cells was significantly greater compared with BM CD34+ cells or HUVEC only after 7 days of coculture. Arrow, DiI-labeled CD34+ cells. (C) Addition of cEx-BM CD34+ cells. (D) Addition of BM CD34+ cells. (E) Control HUVEC only (original magnification: 4x). (F) Measured total tube length after 7 days of coculture. (G) Measured DiI-positive cell number in tubes after 7 days of coculture. *p < 0.05, **p < 0.001. HPF, high-power field.
Tube formation assay was performed to examine the functional activity of each EPC population on Matrigel. Either DiI-labeled cEx-BM CD34+ cells or fresh BM CD34+ cells were cocultured with HUVECs in Matrigelbased medium, and tube formation was observed for several days. After 24 h in culture, tube-like structures were observed in all groups, and there was no significant difference among the groups. After 7 days in culture, cEx-BM CD34/HUVEC demonstrated the highest tubulogenic activity, showing more than twofold increase compared to BM CD34/HUVEC. In the HUVEC-only group, no or very few tubes could be found by 7 days after culture (Fig. 2C–E) [7 days: total tube length: cEx-BM CD34/HUVEC, 6,700 ± 162.6; BM CD34/ HUVEC, 3,256 ± 175.9; HUVEC only, 1,174 ± 356.0 μm (high-power field, HPF); p < 0.001 for cEx-BM CD34 vs. HUVEC only, p < 0.05 for cEx-BM CD34 vs. BM CD34 and BM CD34 vs. HUVEC only; incorporated DiI-positive cell numbers: cEx-BM CD34/HUVEC, 19.2 ± 2.03; BM CD34/HUVEC, 15.7 ± 3.11; HUVEC only, 0.00 ± 0.00/mm; p < 0.001 for cEx-BM CD34 vs. HUVEC only and BM CD34 vs. HUVEC only, p < 0.05 for cEx-BM CD34 vs. BM CD34] (Fig. 2F, G). These results suggest that EPCs from cEx-BM CD34+ cells have a likely higher functional endothelial activity. These in vitro results further indicate that cEx-BM CD34+ cells exhibit EC-like characteristics.
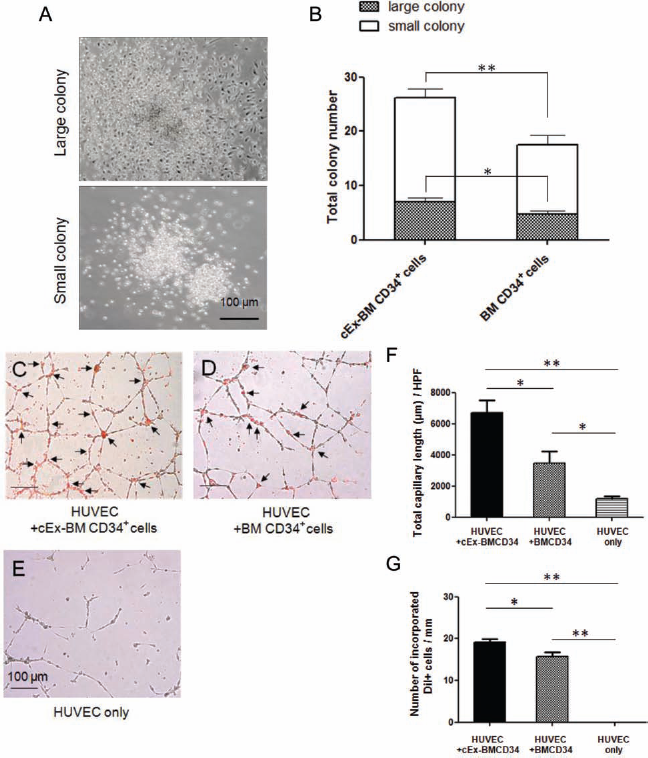
Both cEx-BM CD34+ Cells and BM CD34+ Cells Show High Capacity for Osteogenic Differentiation In Vitro
During primary culture aiming for mesenchymal stem cell induction, a part of the cEx-BM CD34+ cells and BM CD34+ cells obtained fibroblast-like spindle shapes (Fig. 3A, B). After 2 weeks in primary culture, adherent cells were observed both in cEx-BM CD34+ cells and in BM CD34+ cells and proliferated quickly to form colonies (Fig. 3C, D). Treatment for 3 weeks with specific conditions for osteogenic induction resulted in a morphological transformation of the cells from a long and spindle-like shape into a cuboidal one. In contrast, no transformation was observed in the negative control group (data not shown). Following osteogenic induction, remarkable matrix mineralizations were confirmed by Alizarin Red staining for calcium detection (Fig. 3E, F). For quantification, the proportion of Alizarin Red-positive area to total area was measured by computerized image software (Adobe Photoshop Elements 10, Adobe Systems, Inc., CA, USA). Each 1-mm2 area (10 areas in both groups) was randomly selected. There was no significant difference between the two groups (Fig. 3G). In addition, mRNAs of hOC and hCol1A1 were highly expressed in both cEx-BM CD34+ cells and BM CD34+ cells after osteogenic induction (osteogenesis-induced cells) compared with the negative control (nonosteogenic induction) (Fig. 3H). Differentiated human OBs were identified as human OC-positive cells in both cEx-BMCD34 and BMCD34 osteogenesis-induced cells, with no obvious positivity being observed in the noninduction cells by immunofluorescence staining (Fig. 3I–K). These results indicate that human cEx-BM CD34+ cells and BM CD34+ cells are capable of differentiating into OBs under specific culture conditions.
Osteogenic differentiation of cEx-BM CD34+ cells and BM CD34+ cells in vitro. (A–D) Morphology of cEx-BM CD34+ and BM CD34+ cells, which were cultured in α-minimal essential medium (α-MEM) supplemented with 10% fetal bovine serum (FBS), 2 mM l-glutamine, and 10% conditioned medium (CM) during the first 7 days and in the same medium without CM for the following 1 week, characterized by a fibroblast-like spindle shape (original magnification: 200x) (A, B). These spindle-shaped cells proliferated quickly forming colonies (original magnification: 40x) (C, D). (E, F) In wells with osteogenesis-inducing conditions, matrix mineralization was clearly demonstrated by Alizarin Red staining, indicating the presence of calcium. In contrast, no mineralization was observed under noninducing conditions (original magnification: 40x). Each upper right panel shows the entire image of the well. (G) Quantification of Alizarin Red staining. The Alizarin Red-positive area (red) and total area in the field were measured, and the percentage of Alizarin Red-positive area to total area was calculated and averaged in cEx-BM CD34+ cells versus BM CD34+ cells. NS, not significant. (H) mRNAs of human osteocalcin (hOC) and human collagen 1 α1 (hCol1A1) were markedly expressed in osteogenesis-induced cells (Os+) but not in noninduced cells (NI). (I–K) Immunofluorescence staining of human osteocalcin-positive cells (original magnification: 40x). Scale bars: 100 μm. GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
cEx-BM CD34+ Cells Exhibit Potent Therapeutic Potential in Fracture Healing
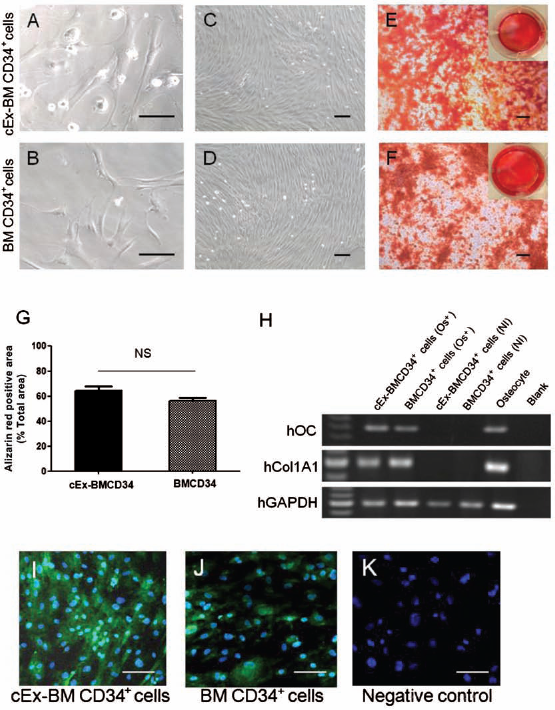
To evaluate fracture repair morphologically, radio-graphic and histological examinations were performed. Fractures were united radiographically with bridging callus formation in 90% (9 of 10) of the cEx-BMCD34 group, in 70% (7 of 10) of the BMCD34 (Hi) group, and in 20% (2 of 10) of the BMCD34 (Lo) group at week 8. In contrast, fracture sites in all animals receiving PBS (control group) showed no bridging callus formation and finally fell into nonunion at week 8. The time course of union rate in each group is described in Figure 4A. Morphological and functional fracture healing in each group was further evaluated by micro-CT 8 weeks after surgery. The results of micro-CT exhibited striking trabecular bone formation in the cEx-BMCD34 group and BMCD34 (Hi) group compared to the BMCD34 (Lo) group or PBS group (Fig. 4B).
Radiographical and histological assessment of fracture healing following cell transplantation. (A) Representative radiographs of fracture sites at weeks 2, 4, and 8 in each group (n = 10 in each group and at each time point). Arrowheads show rat femur fracture sites. (B) Representative micro-computed tomography (CT) images of fracture sites in each group at week 8 (top) and 3D reconstruction images at week 8 (bottom). Arrowheads show rat femur fracture sites. (C, D) Micro-CT analysis of fracture healing. Callus volume and bone density were quantified in the micro-CT images and averaged. *p < 0.05, **p < 0.001. (E) Histological evaluation of endochondral ossification by toluidine blue staining (original magnification: 40x). White dotted, curved line marks the edge of the callus formation. Arrowheads show rat femur fracture sites. (F) The extent of fracture healing assessed by Allen's classification was significantly higher in the cEx-BMCD34 and BMCD34 (Hi) groups than in the other groups throughout the time course of fracture healing (n = 3 in each group and at each time point). *p < 0.05, **p < 0.001.
Quantitative analysis for bone formation was performed using micro-CT images and expressed as callus volume and bone density. Both parameters exhibited significantly greater values in the cEx-BMCD34 and BM CD34 (Hi) groups than in the two other groups [callus volume: cEx-BMCD34, 4.833 ± 0.241; BMCD34 (Hi), 4.543 ± 0.334; BMCD34 (Lo), 1.893 ± 0.442; PBS, 1.537 ± 0.321 mm3; p < 0.001 for cEx-BMCD34 vs. PBS group and BMCD34 (Hi) vs. PBS group; p < 0.05 for cEx-BMCD34 vs. BMCD34 (Lo) and BMCD34 (Hi) vs. BMCD34 (Lo) group, and; bone density: cEx-BMCD34, 694.1 ± 20.31; BMCD34 (Hi), 594.2 ± 34.53; BMCD34 (Lo), 442.7 ± 19.99; PBS, 407.1 ± 22.02; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group and BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group; p < 0.05 for cEx-BMCD34 vs. BMCD34 (Hi) group] (Fig. 4C, D).
Fracture healing was also histologically evaluated by toluidine blue staining. In animals receiving cEx-BM CD34+ cells, enhanced endochondral ossification and newly formed trabecular bone were observed at week 2, bridging callus formation at week 4, and finally fractures were united completely at week 8. In animals of the BMCD34 (Lo) and PBS groups, although callus formation was observed at week 4, and the fracture gaps were not filled with bridging callus at week 8. Moreover, in the PBS group, the callus formation was absorbed at week 8, and all fractures resulted in pseudoarthritis (Fig. 4E). The degree of fracture healing assessed by Allen's classification (2) was significantly higher in the cEx-BMCD34 group compared to the other groups at all time points of fracture healing [2 weeks: p < 0.05 for cEx-BMCD34 vs. PBS group; 4 weeks: p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group and BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group; 8 weeks: p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group and BMCD34 (Hi) vs. PBS group; p < 0.05 for BMCD34 (Hi) vs. BMCD34 (Lo) group] (Fig. 4F). These results indicate that a nonhealing femoral fracture created by periosteal cauterization can be repaired by local administration of cEx-BM CD34+ cells or high-dose BM CD34+ cells.
cEx-BM CD34+ Cells Promote Tissue Regeneration, Differentiating Into Endothelial and Osteoblastic Lineages in Sites of Fracture
Transplanted cells (cEx-BM CD34+ and BM CD34+) present in fracture sites were characterized by immunofluorescence staining. We found that transplanted human cells were located at perifracture site 1 week after transplantation in the cEx-BMCD34 and BMCD34 (Hi) and (Lo) groups but not in the PBS group (Fig. 5A–H). To histologically validate the phenomenon of human cell-derived vasculogenesis, histochemical staining for UEA-1, a human-specific EC marker, was performed with tissue samples obtained 1 week after local cell transplantation. Differentiated human ECs in the vasculature of the perifracture area were detected as UEA-1-positive cells in the cEx-BMCD34 and BMCD34 (Hi) and (Lo) groups but not in the PBS group (Fig. 5I–L). We performed similar staining using samples obtained 2, 4, and 8 weeks after surgery but could not detect UEA-1-positive cells in all groups. To identify osteogenic differentiation of cEx-BM CD34+ cells and BM CD34+ cells, immunohistochemical staining for human-specific OC, an OB marker, was performed with tissue samples obtained 1 week after cell transplantation. Differentiated human OBs derived from cEx-BM CD34+ cells and high- and low-dose BM CD34+ cells were detected as hOC-positive cells in the perifracture area, while hOC-positive cells were not identified in the PBS group (Fig. 5M–P). We could not detect hOC-positive cells in samples obtained 2, 4, and 8 weeks after surgery in all groups. These results indicate that cEx-BM CD34+ cells and BM CD34+ cells can differentiate into both EC and OB lineages in a fracture-induced environment at the initial phase of fracture repair process.

Fracture site vasculogenesis and osteogenesis derived from cEx-BM CD34+ and BM CD34+ cells. (A–H) White dotted lines mark the edge of the callus formation. Arrowheads show rat femur fracture sites. (A–D) Yellow closed square in toluidine blue staining (original magnification: 2x) is the region of interest (ROI) observed by fluorescence human nuclear antigen (hNA) staining. *Point observed by fluorescence vascular staining. **Point observed by fluorescence osteoblast staining. (E–H) Representative transplanted human cells staining with hNA (red) and DAPI (blue) using tissue samples in perifracture sites collected at week 1 in all groups (original magnification: 4x in E–H). (I–L) Representative double immunostaining for Ulex europaeus agglutinin 1 (UEA-1) (red) and isolectin B4 (green) using tissue samples of the fracture site at week 1 (original magnification: 400x in I–L). Differentiated human ECs in the vasculature of the perifracture area were detected as UEA-1-positive cells (red) in the cEx-BMCD34 (I) and BMCD34+ (Hi) and (Lo) groups (J, K) but not in the PBS group (L). Transplanted human cells differentiated to mature endothelial cells (ECs; arrows) at the site of neovascularization. (M–P) Representative immunostaining for hOC (red) using tissue samples of the fracture site at week 1 (original magnification: 100x in M–P). Differentiated human osteoblasts (OBs) derived from cEx-BMCD34+ cells (M) and high-dose (N) and low-dose (O) BM CD34+ cells were detected as hOC-positive cells (arrows, red) in the perifracture area, while hOC-positive cells were not identified in the PBS group (P).
Next, we confirmed the differentiation of transplanted cEx-BM CD34+ cells into human endothelial and osteoblastic lineage cells at the fracture sites by RT-PCR analysis. The mRNA upregulation of human-specific EC markers, vascular endothelial cadherin (hVE-cad) and CD31 (hCD31), and human bone-related markers, osteocalcin (hOC) and collagen 1 α1 (hCol1A1), as well as hGAPDH, was confirmed in cEx-BM CD34+ cell and BM CD34+ cell transplanted fracture tissue samples collected 1 week after surgery (Fig. 6A, B). No human-specific genes were detected in the PBS group.

Human CD34+ cell-derived vasculogenesis and osteogenesis at fracture site. (A, B) RT-PCR analysis of tissue RNAs isolated from the perifracture sites demonstrated the expression of human-specific EC markers [human vascular endothelial cadherin (hVE-cad) and hCD31] and human-specific bone-related markers (hOC and hCol1A1) in animals treated with cEx-BM CD34+ cells and high- and low-dose BM CD34+ cells, but not in animals receiving PBS. Cultured HUVECs and cultured hOBs were used as positive controls (Posi). Nega, negative control (no RNA applied). (C–F) Quantitative real-time RT-PCR analysis for human-specific cytokines at week 1 (n = 4 in each group). Relative mRNA expression levels of human-specific VE-cad (C), CD31 (D), OC (E), and Col1A1 (F) to hGAPDH. *p < 0.05, **p < 0.001.
The mRNA expression level of hVE-cad was significantly greater in the cEx-BMCD34 group than in the other groups [cEx-BMCD34, 4791 ± 554.4; BMCD34 (Hi), 2532 ± 257.0; BMCD34 (Lo), 1739 ± 367.8; PBS, 6.733 ± 4.583; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Hi) or BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. PBS group, BMCD34 (Lo) vs. PBS group, and p < 0.05 for BMCD34 (Hi) vs. BMCD34 (Lo) group] (Fig. 6C). The mRNA expression level of hCD31 was greater in animals in the cEx-BMCD34 and BMCD34 (Hi) groups than that in the other groups [cEx-BMCD34, 3937 ± 324.4; BMCD34 (Hi), 2494 ± 256.7; BMCD34 (Lo), 340.5 ± 270.1; PBS, 19.33 ± 26.24; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Hi) or BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group] (Fig. 6D). The mRNA expression level of hOC was also significantly greater in the cEx-BMCD34 and BMCD34 (Hi) groups than in the BMCD34 (Lo) and PBS groups [cEx-BMCD34, 2658 ± 418.7; BMCD34 (Hi), 2555 ± 223.1; BMCD34 (Lo), 998.2 ± 152.0; PBS, 10.72 ± 4.376; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group, BMCD34 (Lo) vs. PBS group] (Fig. 6E). The mRNA expression level of hCol1A1 was greater in the BMCD34 (Hi) and cEx-BMCD34 groups than in the other groups [cEx-BMCD34, 636.7 ± 129.9; BMCD34 (Hi), 724.7 ± 63.30; BMCD34 (Lo), 127.3 ± 15.48; PBS, 2.713 ± 3.478; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group] (Fig. 6F). These results suggest that cEx-BM CD34+ cells could promote fracture healing via not only paracrine effects by upregulation of endogenous proangiogenic/-osteogenic cytokines but also differentiation into both EC and OB lineages in the fracture sites at early stages of fracture healing.
Enhancement of Intrinsic Angiogenesis and Osteogenesis by Local cEx-BM CD34+ Cell Transplantation
Enhanced angiogenesis and osteogenesis by the paracrine effect of the transplanted cells on the recipients' tissues were confirmed by immunostaining for rat-specific markers (Fig. 7). Histochemical vascular staining with isolectin B4, a rat-specific marker for EC, using tissue samples collected 2 weeks postfracture, demonstrated enhancement of intrinsic neovascularization around the endochondral ossification area (Fig. 7A) in animals treated with cEx-BM CD34+ cells and BM CD34+ cells (Fig. 7D). Capillary density was significantly greater in the cEx-BMCD34 and BMCD34 (Hi) groups compared with the other groups [cEx-BMCD34, 87.75 ± 8.18; BMCD34 (Hi), 71.50 ± 8.18; BMCD34 (Lo), 20.4 ± 5.13; PBS, 15.17 ± 5.27/mm2; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group, and p < 0.05 for BMCD34 (Hi) vs. BMCD34 (Lo) group] (Fig. 7B).
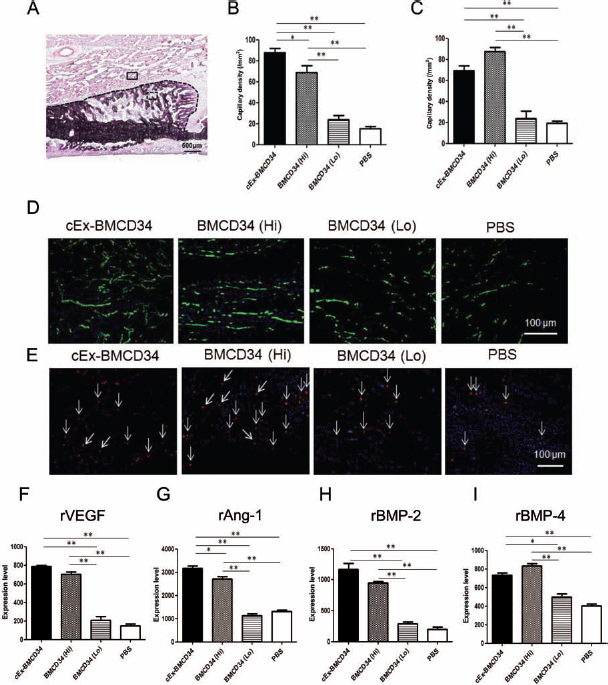
Enhancement of angiogenesis and osteogenesis by recipient cells following cEx-BM CD34+ and BMCD34+ cell transplantation. (A) Closed square in toluidine blue staining (x2) is the region of interest (ROI) observed by fluorescence vascular staining. Dotted square is the ROI observed by fluorescence osteoblast staining. Black dotted, curved line marks the edge of the callus formation. (B) Angiogenesis assessed by capillary density at week 2 was significantly greater in the cEx-BMCD34 group compared to the other groups (n = 5 in each group). *p < 0.05, **p < 0.001. (C) OB density was significantly greater in the cEx-BMCD34 and BM CD34+ (Hi) groups than in the other groups (n = 5 in each group). **p < 0.001. (D) Representative vascular staining with isolectin B4 (green) and DAPI (blue) using tissue samples at perifracture sites collected at week 2 in all groups (original magnification: 100x). (E) Representative osteoblast staining with anti-rat osteocalcin (arrows, red) and DAPI (blue) using tissue samples at perifracture sites collected at week 2 in all groups (original magnification: 100x). (F–I) Gene expression of intrinsic cytokines for angiogenesis and osteogenesis at week 2 (n = 4 in each group). Relative expression levels of rat-specific vascular endothelial growth factor (VEGF) (F), angiopoietin-1 (Ang-1) (G), bone morphogenetic protein-2 (BMP-2) (H), and BMP-4 (I) to rGAPDH. The expression level of rVEGF and rAng-1 was significantly greater in animals of the cEx-BMCD34 and BMCD34 (Hi) groups compared with the other groups (F, G). The expression levels of rBMP-2 and rBMP-4 were also significantly greater in animals of the cEx-BMCD34 and BMCD34 groups compared with the BMCD34 (Lo) and PBS groups (H, I). *p < 0.05, **p < 0.001.
OB staining with anti-rat OC on tissue samples collected 2 weeks postfracture revealed augmentation of intrinsic osteogenesis in the area of new bone formation (Fig. 7A) in animals treated with high doses of BM CD34+ cells (Fig. 7E). OB density was significantly greater in the BMCD34 (Hi) and cEx-BMCD34 groups than in the other groups [cEx-BMCD34, 69.40 ± 4.23; BMCD34 (Hi), 87.5 ± 3.71; BMCD34 (Lo), 23.75 ± 7.19; PBS, 19.40 ± 2.64/mm2; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS groups] (Fig. 7C).
To explore the underlying mechanism by which intrinsic osteogenesis and angiogenesis were enhanced following cEx-BM CD34+ cells transplantation therapy, we assessed the mRNA expression of pro-angiogenic and pro-osteogenic cytokines in perifracture sites 2 weeks after surgery by real-time RT-PCR, quantifying the expression of rat vascular endothelial growth factor (rVEGF), rat angiopoietin 1 (rAng1), rat bone morphogenetic protein 2 (rBMP-2), and rat bone morphogenetic protein 4 (rBMP-4) around the fracture sites. The relative mRNA expression level of rVEGF to rGAPDH was significantly greater in the cEx-BMCD34 and BMCD34 (Hi) groups compared with the other groups; however, there was no significant difference between the BMCD34 (Lo) and PBS groups [cEx-BMCD34, 788.8 ± 8.45; BMCD34 (Hi), 705.3 ± 20.84; BMCD34 (Lo), 207.3 ± 21.27; PBS, 149.9 ± 16.80; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group] (Fig. 7F). The relative mRNA expression level of rAng-1 was greater in animals of the cEx-BMCD34 group compared with the other groups [cEx-BMCD34, 3176 ± 164.3; BMCD34 (Hi), 2721 ± 143.3; BMCD34 (Lo), 1131 ± 126.6; PBS, 1311 ± 101.8; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group, and p < 0.05 for cEx-BMCD34 vs. BMCD34 (Hi) group] (Fig. 7G). The relative mRNA expression level of rBMP-2 was also significantly greater in the cEx-BMCD34 and BMCD34 (Hi) groups compared with the BMCD34 (Lo) and PBS groups [cEx-BMCD34, 1168.0 ± 96.15; BMCD34 (Hi), 948.3 ± 27.94; BMCD34 (Lo), 288.5 ± 34.79; PBS, 200.0 ± 40.02; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group] (Fig. 7H). The relative mRNA expression level of rBMP-4 was greater in the BMCD34 (Hi) and cEx-BMCD34 groups compared with the other groups [cEx-BMCD34, 734.2 ± 24.33; BMCD34 (Hi), 834.1 ± 25.59; BMCD34 (Lo), 496.4 ± 38.77; PBS, 405.7 ± 19.57; p < 0.001 for cEx-BMCD34 vs. PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group, and p < 0.05 for cEx-BMCD34 vs. BMCD34 (Lo) group] (Fig. 7I).
These results suggest that cEx-BM CD34+ cells enhance, perhaps paracrinely, not only intrinsic angiogenesis but also osteogenesis in fracture sites via upregulation of pro-angiogenic and pro-osteogenic cytokines, leading to rapid fracture healing with blood perfusion recovery. The regenerative potential of cEx-BM CD34+ cells appeared to be stronger than that of BM CD34+ cells.
cEx-BM CD34+ Cell Transplantation Promotes Blood Flow Recovery in Sites of Fracture
To evaluate blood flow recovery via neovascularization at fracture sites, LDPI was serially examined after fracture generation (Fig. 8A). In all groups, severe reduction of blood flow was observed 1 h after fracture. There was no significant difference in the blood flow ratio of fractured to intact hindlimbs 1 h after fracture creation among all groups. The blood perfusion ratios were significantly higher in the cEx-BMCD34 group than in the other groups at week 1 [cEx-BMCD34, 1.520 ± 0.043; BMCD34 (Hi), 1.412 ± 0.017; BMCD34 (Lo), 1.224 ± 0.013; PBS, 1.190 ± 0.042/mm2]; a similar trend continued until 2 weeks after surgery [cEx-BMCD34, 1.672 ± 0.060; BMCD34 (Hi), 1.457 ± 0.06; BMCD34 (Lo), 1.382 ± 0.052; PBS, 1.359 ± 0.020/mm2]. At week 3, the blood flow ratio exhibited no significant difference among all groups (Fig. 8B). These results indicate that local transplantation of cEx-BM CD34+ cells contributes to early improvement of tissue perfusion in sites of fracture.
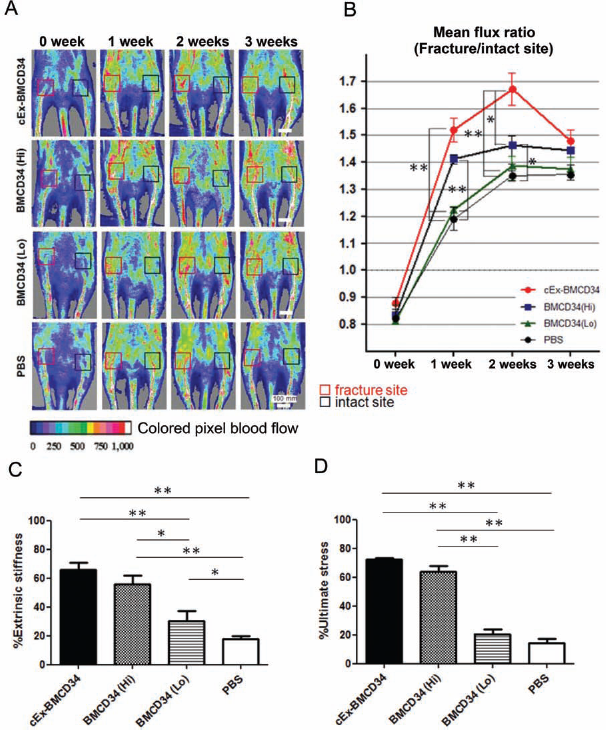
(A) Representative laser Doppler perfusion imaging (LDPI) at weeks 0 (1 h after fracture), 1, 2, and 3 are shown. In these digital color-coded images, maximum perfusion values are indicated in white to red, medium values in green to yellow, and lowest values in dark blue. The blood flow within the fracture site (red square) and that within the intact contralateral site (black square) were evaluated as mean flux, and the ratio of the mean flux in the fractured site to that in the contralateral site (mean flux ratio) was calculated. (B) Quantitative analyses of local blood perfusion. The mean flux ratio at week 1 was significantly higher in animals of the cEx-BMCD34 group compared to the other groups at week 2, the ratio was significantly higher in the cEx-BMCD34 group compared with the Lo and PBS group, as well as in BMCD34 (Hi) compared to the PBS group. *p < 0.05, **p < 0.001. (C, D) Functional recovery after fracture is assessed by biomechanical 3-point bending test at week 8. Percentage of each parameter (extrinsic stiffness and ultimate stress) indicating the ratio of each value in the fracture site to the contralateral intact femur was significantly superior in the cEx-BMCD34 and BMCD34 (Hi) groups over the BMCD34 (Lo) and PBS groups (n = 3 in each group and each parameter). *p < 0.05, **p < 0.001. (C) Percent extrinsic stiffness. (D) Percent ultimate stress.
cEx-BM CD34+ Cell Transplantation Leads to Functional Bone Healing After Fractures
To confirm the functional recovery of the fractured bones, biomechanical evaluation by a 3-point bending test was performed at week 8 in all groups (n = 3 in each group). The specimen length was similar in the cEx-BMCD34 group (30.2 ± 0.5 mm), BMCD34 (Hi) group (31.2 ± 0.7 mm), BMCD34 (Lo) group (30.7 ± 0.6 mm), and PBS group (31.1 ± 1.4 mm). The percentage ratios of extrinsic stiffness and ultimate stress in the fractured femur versus contralateral intact femur were significantly superior in the cEx-BMCD34 and BMCD34+ (Hi) groups over the BMCD34 (Lo) and PBS groups [percent extrinsic stiffness: cEx-BMCD34, 65.80 ± 5.153; BMCD34 (Hi), 55.69 ± 5.986; BMCD34 (Lo), 24.06 ± 2.846; PBS, 17.90 ± 1.789%; p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. PBS group, and p < 0.05 for BMCD34 (Hi) vs. BMCD34 (Lo) group; percent ultimate stress: cEx-BMCD34, 72.24 ± 1.263; BMCD34 (Hi), 64.06 ± 3.794; BMCD34 (Lo), 20.56 ± 3.195; PBS, 14.25 ± 3.047%: p < 0.001 for cEx-BMCD34 vs. BMCD34 (Lo) or PBS group, BMCD34 (Hi) vs. BMCD34 (Lo) or PBS group] (Fig. 8C, D). These findings indicate that incomplete union created by periosteum cauterization is functionally repaired by cEx-BM CD34+ cell transplantation in nude rats.
Discussion
The current study revealed a therapeutic potential of cEx-BM CD34+ cells when transplanted together with a bioabsorbable scaffold into fracture sites of a rat unhealing fracture model. Moreover, our data indicated several advantages of using cEx-BM CD34+ cells for fracture repair, including their promotional effect on neovascularization, their multidifferentiation capacity into endothelial and osteoblastic lineages, and their easy accessibility as a potential tool for cell therapy. These characteristics of cEx-BM CD34+ cells may thus lead to the development of promising novel therapeutic strategies applicable to the clinical setting, specifically in the field of trauma surgery.
Our 7-day culture expansion technique increased around 23 times the initial number of BM CD34+ cells used in this study. Although we observed a certain reduction of CD34 positivity through the culture expansion, around 60% of the cells remained positive for CD34. Before expansion, BM CD34+ cells expressed predominantly CD34, CD31, CD133, and CD44. The cells reached confluence after 1 week of culture and displayed a typical EC phenotype including enhanced CD31 expression. CD34+ EPCs adopted a more mature endothelial phenotype during their in vitro culture and expansion, which was consistent with a previous study (11, 13, 63). It was reported that stem cells experience a long replicative history during their expansion and are thus subjected to damage from intracellular and extracellular insults (42). However, previous reports showed that CD34+ cells had telomere stabilization correlated with maintenance of high levels of telomere activity (18) and showed genetic stability of CD34+ cells by using Wright–Giemsa staining (70). In this study, we also showed that cEx-BM CD34+ cells maintained their telomere length. Moreover, we demonstrated that cEx-BM CD34+ cells exhibited high ability of proliferation and had a higher capacity of colony and tube formation than fresh (nonexpanded) BM CD34+ cells in vitro (Fig. 2). O et al. (55) also showed that culture-expanded cord blood-derived CD34+ cells exhibited more endothelial activities and better ability for blood flow recovery following ischemic injury than freshly isolated CD34+ cells. Moreover, we could also demonstrate that some of the cEx-BM CD34+ cells and BM CD34+ cells have the potential to differentiate into OBs after 3 weeks in culture (Fig. 3). Although these results are supported by other studies (8, 44), the therapeutic potential of cEx-BM CD34+ cells and BM CD34+ cells for fracture has not yet been well explored, and we have now demonstrated it in the present study.
In the presented study, the favorable effects of cEx-BM CD34+ cells on nonunion fractures were evidenced by a series of assessments of fracture healing, including biomechanical tests and micro-CT image analysis. To confirm whether the effect is specific for cEx-BM CD34+ cells or not, we compared the therapeutic potential of cEx-BM CD34+ cells with that of high- and low-dose nonexpanded BM CD34+ cells and demonstrated the superiority of cEx-BM CD34+ cells to the same number of BM CD34+ cells. In addition, it is notable that the healing ratio in the cEx-BMCD34 group and the BMCD34 (Hi) group in the present study is higher than that reported in a previous study (52), in which 50% of animals received local transplantation of 1 × 104 of GM-PB CD34+ cells. In present study, 90% of animals transplanted with the same number of cEx-BM CD34+ cells and 70% of animals transplanted with the same number of fresh BM CD34+ cells were shown to achieve fracture union. These results might be related to the different intrinsic properties of BM CD34+ cells, possibly due to their more immature nature when compared with PB-derived CD34+ cells, or their distinct proliferative advantages of higher cell cycle rate and longer telomere length. Indeed, Koutna et al. demonstrated that CD133+ populations from BM showed better proliferation activity and a higher distribution of primitive progenitors than those from other sources (36).
The results of immunohistochemistry and RT-PCR analyses for human-specific marker expression in the fracture sites indicate that one of the mechanisms underlying the angiogenic and osteogenic effects of the cEx-BM CD34+ cells and BM CD34+ cells may be direct differentiation into endothelial and osteoblastic lineages at the initial phase of fracture repair process. Immunofluorescence staining and quantitative RT-PCR analysis of rat-specific EC and OB markers revealed, on the other hand, also a significant enhancement of intrinsic angiogenesis and osteogenesis in the cEx-BMCD34 and BMCD34 groups. Interestingly, real-time RT-PCR analysis revealed that not only the rat-specific angiogenesis-related markers rVEGF and rAng-1 but also the rat-specific bone formation-related markers rBMP-2 and rBMP-4 were upregulated in the perifracture sites in the rats that received cEx-BM CD34+ and BM CD34+ cells, indicating a possible paracrine mechanism underlying enhanced intrinsic angio-osteogenesis. Such an indirect effect of the transplanted cEx-BM or BM CD34+ cells on the resident tissue cells might be also critical for fracture repair. Our unpublished cDNA microarray data (data not shown) suggest that human BM CD34+ cells possibly secreting certain growth factors (GFs) [e.g., hANG-1, human platelet-derived growth factor (PDGF)-B, or hVEGF] into sites of fracture, as a part of the proposed albeit still hypothetical scenario awaiting experimental verification, could thus “directly” upregulate the expression and increase the secretion of endogenous GFs or play a synergistic role with the GFs. These GFs were reported to induce and upregulate the expression of Ang1 (22, 54), VEGF (20, 21, 51), and BMP-2/4 (30, 56, 58, 60). The observed increase in the transcript levels of the osteogenic and angiogenic factors could be also due to a secondary effect of BM CD34+ cell transplantation, characterized by intrinsic gene expression changes in the recipient tissue concomitant with or triggered by increased osteogenesis, enhanced neovascularization, and blood flow recovery. Therefore, it is speculated that expression of the endogenous angiogenic cytokines rVEGF/rAng-1 and the osteogenic cytokines rBMP-2/rBMP-4 was upregulated by transplanted CD34+ cells in fracture sites.
Despite the promising initial results obtained with cEx-BM CD34+ cells for unhealing fracture repair, there are several issues that need to be solved before we can use cEx-BM CD34+ cells in clinical applications. Firstly, the safety of ex vivo expanded BM cells, specifically their tumor formation potential following cell transplantation, remains to be determined. Secondly, there are persistent concerns that ex vivo expanded CD34+ cells may lose their stem cell characteristics and their ability to contribute meaningfully to long-term engraftment. Indeed, the percentage of cells expressing surface markers such as CD34 and CD133 declines after expansion in culture, suggesting that the cells undergo a certain degree of differentiation. Nevertheless, our experiments clearly indicate that the therapeutic efficacy of cEx-BM CD34+ cells is equivalent or even higher than that of freshly isolated BM CD34+ cells.
Conclusion
Our 7-day culture expansion technique allowed us to increase the initial number of BM CD34+ cells around 23 times, maintaining 60% purity of CD34 positivity. Moreover, the culture expanded BM CD34+ cells exhibited striking therapeutic efficacy for the treatment of unhealing fractures, promoting neovascularization and osteogenesis in fracture sites even with the same number of freshly isolated BM CD34+ cells. These results suggest that this novel cell therapeutic approach may require only a small amount of bone marrow to be initially collected, avoiding complicated procedures such as multiple G-CSF administration and time-consuming apheresis of PB in the clinical setting. Autologous culture expanded BM CD34+ cell transplantation therapy would be a simple but powerful therapeutic strategy for nonunion fracture treatment.
Footnotes
Acknowledgments
We would like to thank Janina Tubby for her editing assistance in preparing this manuscript. The authors declare no conflict of interest.
