Abstract
Our previously published ex vivo expansion procedure starting from cord blood CD34+ cells enables a massive expansion of total and CD34+ cells and committed progenitors without negative impact on stem cells exhibiting both short- and long-term repopulating capacity. It was upgraded to clinical scale [Macopharma HP01® medium in presence of SCF, FLT3-L (100 ng/ml each), G-SCF (10 ng/ml), and TPO (20 ng/ml)] and is in use for an ongoing clinical trial (adult allogeneic context), yielding encouraging results. In order to test the possibility to use the expanded cells in distant transplantation centers, we studied the functional stability at +4°C (usual temperature of transportation) of hematopoietic progenitors and stem cells 48 h after expansion. If the cells were washed and resuspended in 4% albumin solution (actual procedure for immediate injection), only one half of total nucleated and CD34+ cells and 30% of committed progenitors survived after 24 h. This condition has also an evident negative impact on stem cells in expansion product as demonstrated on the basis of reconstitution of NSG mice bone marrow by human CD45, CD33, CD19+ cells as well as by human committed progenitors (CFU). Surprisingly, if the cells were stored 48 h at +4°C in culture medium, very good survival of total and CD34+ cells (90 to 100%) and colony forming unit cells (CFCs; around 70%) was obtained, as well as the maintenance of stem cells (the same in vivo assay with NSG mice). These data point to the possibility of the maintenance of the full functional capacity of expanded grafts for 2 days, the time allowing for its transportation to any transplantation center worldwide.
Introduction
We have developed an ex vivo expansion procedure from cluster of differentiation 34 positive (CD34+) cells of thawed cord blood (CB), enabling simultaneous amplification of committed progenitors and maintenance of stem cells with short- and long-term repopulating capacity (5). Once “upgraded” to the clinical scale, real efficiency was proven (mean total cell expansion 400-fold, CD34+ expansion 60-fold, and that of committed progenitors 150-fold) without any loss of stem cell capacity as judged on the basis of the xenograft model (3). This technique is actually in use to produce the ex vivo expanded cells for a clinical trial named GRAPA (http://clinicaltrials.gov/ct2/show/NCT01034449) for allogeneic transplantation of adult patients suffering from myeloproliferative pathologies. The results obtained with the first seven patients are encouraging and confirm a rapid reconstitution of posttransplant hematopoiesis that seems to be long lasting (almost 2 years of follow-up so far) (11). In this clinical trial, after expansion culture, the cells are washed (in PBS/1% albumin solution), concentrated, and resuspended in 4% albumin solution to be injected within 6 h after the end of culture. There is an evident interest to enable the transportation of the expanded cells not only to local transplantation centers but also to those in distant areas. Taking into consideration the capacity of today logistics, we estimate that a 48-h lag time would be sufficient to accomplish this task.
In order to evaluate the functional stability of these expanded products, we performed in vitro and in vivo tests to assay the functional capacities of committed progenitors and stem cells after 48 h at +4°C (temperature of transportation) under two conditions: (i) the cells were resuspended in IV injectable albumin solution (actual routine) and (ii) the cells were kept at +4°C in their culture supernatant (cytokine-supplemented medium)—named “culture medium” in further text.
Materials and Methods
CD34+ CB Cells Isolation
Volume-reduced banked CB units (CB Bank of Bordeaux Transfusion Center; Etablissement Français du Sang, Aquitaine-Limousin, France) (2) were thawed in the presence of DNase (Pulmozyme Roche, Neuilly, France) and CD34+ selection made by Variomacs and Clinimacs (mean purity 94.85 ± 5%) (Miltenyi Biotech, Paris, France) as previously described (4). Samples had been rejected for storage for technical reasons such as lower volume or CD34+ cell number with respect to the minimal banking standard. Informed consent for experimental purposes (in case the blood was not suitable for banking) was systematically obtained from the mothers before collection. The experiments were performed on discarded CB units in compliance with French regulations (article R1243-49 of “Code de Santé Publique”) and with related ethical authorization granted to the laboratory.
Expansion Cultures
As previously described, 20 × 103 CD34+ cells were cultured per milliliter of HP01 medium (Macopharma, Tourcoing, France) (3) supplemented with stem cell factor (SCF; 100 ng/ml; Peprotech, Rocky Hill, NJ, USA), thrombopoietin (TPO; 20 ng/ml; Peprotech), fms-related tyrosine kinase 3-ligand (Flt-3L; 100 ng/ml; Peprotech), and granulocyte-colony stimulating factor (G-CSF; 10 ng/ml; Neupogen, Amgen SAS, Neuilly sur Seine, France), and so culture volume was dependent on the number of CD34+ cells obtained from each CB sample. This was injected into culture cell bags (VueLife 25 or 118, AFC, Gaithersburg, MD, USA) and cultured in water-saturated air (Incubators Stericult 200, Thermo Scientific, Saint Herblain, France) at 5% CO2 for 6 days. Fresh cytokine-supplemented medium was then added (four volumes of fresh medium to one volume of culture), and the culture continued until day 12. The analyses of cellular content and phenotype were performed at day 12 of culture.
Clinical grade serum-free medium (6), cytokines, bags, CD34+ selection devices, etc., were used in this study while the volume-reduced cord blood units (2) had been previously banked, and were thawed before expansion (4).
Storage of Expanded Cells at +4°C
As in the ongoing clinical trial, the expanded cells were concentrated by centrifugation, resuspended in 4% albumin pharmaceutical preparation (Vialebex, LFB, Les Ulis Courtaboeuf, France), and injected in small “sampling” bags (“Poche d'échantillonnage VSE0000A,” Macopharma, Tourcoing, France) made from the same material as routine transfer bags. The bags were stored at +4°C for 48 h before testing the functional stem and progenitor cells populations. Alternatively, the expanded cells were injected in the “sampling” bags in their culture medium and stored at +4°C for 48 h.
Cell Counts and Immunophenotypical Characterization
The cells were counted using an automatic cell counter (Cell Dyn 3000, Abbott, Rungis, France). The CD34+ cell concentrations in ex vivo expanded CB cell samples were determined as previously described (9). Briefly, trucount microbeads (Beckton Dickinson, San Jose, CA, USA) and flow cytometry (FACS Calibur, Becton Dickinson) via three-color fluorescence were used to detect and count viable CD45+/CD34+ cells.
Colony-Forming Cell (CFU) Assay
To detect the committed hematopoietic progenitors, freshly isolated CD34+ or cultured cells, cell harvested at day 12 as well as day 12 cells stored for 48 h at +4°C, were seeded in a cytokine-supplemented methylcellulose medium (Stem Alpha SA, Saint Clement les Places, France) in a Petri dish or 24-well plates (Nunc, Roskilde, Denmark), respectively, at a concentration of 250–1,000 cells/ml or a volume of 0.5 μl/250 μl, respectively (7). After 14 days of incubation at 37°C in a water-saturated atmosphere, 20% O2 and 5% CO2, the CFUs [colony-forming units-granulocyte macrophage (CFU-GM), burst-forming units-erythroid (BFU-E), and colony-forming units-mix (CFU-Mix)] (>50 cells) were enumerated using an inverted microscope (Leica, Wetzlar, Germany) and be distinguished by the red color of the hemoglobin-containing BFU-Es.
SCID-Repopulating Cells (SRC) Assay
Stem cell activity was evaluated by an in vivo assay— transplantation of human cells into immunodeficient (nonobese diabetic/severe combined immunodeficient/interleukin 2 receptor gamma-chain null; NSG) mice (central animal-keeping facility of the University of Bordeaux Segalen, France). The cell progeny of 1,000 day 0 CD34+ cells obtained after 12 days of culture, as well as their equivalents (all cells contained in the same volume of culture) after 48 h storage at +4°C (two conditions: albumin or culture medium) were injected into 6- to 10-week-old NSG mice conditioned by intraperitoneal injections of busulfan 25 mg/kg (Busilvex®, Pierre Fabre, Boulogne, France) at day -2 and day -1 with respect to the injection time point (according to the previously established protocol) (14). Positive control (mice injected with 15,000–20,000 CD34+ purified at day 0) and negative control (noninjected mice) were included in each experiment. After 8 weeks, the animals were euthanized, their femora were isolated, and the bone marrow (BM) was flushed with 1 ml of Roswell Park Memorial Institute media (RPMI) 1640 complemented with human albumin (4% final concentration). Cells were incubated with a fluorescein isothiocyanate (FITC)-coupled anti-human CD45, phycoerythrin (PE)-coupled anti-human CD19, and allophycocyanin (APC)-coupled anti-human CD33 antibodies for 20 min at 4°C in the dark. Washed cells were analyzed on a FACSCalibur to detect and quantify human chimerism. This approach provides information related to a relatively more mature SRC subpopulation, SRCCD. To avoid false-positive results due to control isotype, we used nonengrafted busulfan-conditioned mice as controls.
To detect the progenitors (CFUs) of human origin in murine bone marrow, 15 and 30 μl of femoral cell suspension (see above) were seeded per 250 μl of methylcellulose (in duplicate) (Stem Alpha SA) supplemented with 10% human plasma AB (PFC - Plasma frais congelé: Fresh frozen plasma, Aquitaine-Limousin Branch of French Blood Institute, Bordeaux, France), 25 ng/ml recombinant human interleukin-3 (rHu IL-3; Pepro Tech, London, UK), 25 ng/mL rHu granulocyte-macrophage colony stimulating factor (GM-CSF; R&D Systems Europe, Lille, France), 50 ng/ml rHu SCF (Neupogen) and 3 U/ml rHu erythropoietin (EPO; Tebu- Bio, Le Perray, France) (14). After 14 days of incubation, the human CFUs (>50 cells) were enumerated using an inverted microscope. This approach provides information related to the relatively less mature subpopulation of SRCs—SRCCFU.
Statistics
The nonparametric Wilcoxon test was used to compare values, though some of the data are represented by parametric means for clarity.
Results
The series of experiments presented here (n = 13) yielded a fold expansion for total nuclear cells (TNCs), CD34+ cells, and CFUs of 349.8 ± 219.0, 53.3 ± 19.8, and 103.6 ± 49.6, respectively (not shown). These values were similar to those obtained during the development of the clinical scale procedure (3). With respect to the culture endpoint values (day 12), one half of the total and CD34+ cells and only 30% of the committed progenitors (CFUs) persisted after 48 h in albumin solution at +4°C (Fig. 1). In the same time, and at the same temperature, virtually Analysis of NSG mice BM 7 weeks after injection of ex vivo expanded cells at the end of culture, and those stored at +4°C also revealed their better maintenance in culture medium with respect to the albumin solution, which was visible whatever phenotypic marker was used to detect human cells (CD45, CD33, CD19) in murine bone marrow (Fig. 2A.1–3). Following injection with the expansion/storage product of 1,000 day 0 CD34+ cells, in terms of the percentage of engrafted mice (0.8% threshold for CD45 and 0.23% threshold for CD33 and CD19 in this experimental series), the difference in favor of culture medium compared to albumin was evident for CD33 and CD19 (Fig. 2B.1–3). However, in terms of mean human chimerism, the culture medium ensured a twofold better preservation of SRC activity when expanded cells were stored in culture medium. These values correspond (CD45+ and CD19+ cells) to those at the end of culture (day 12) (Fig. 2C1, C3).
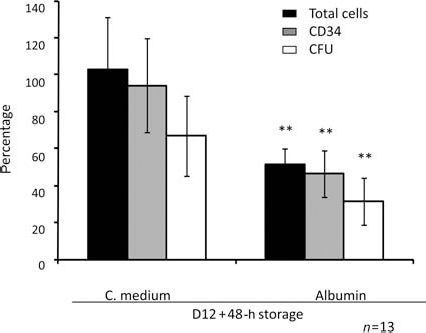
Recovery of total cells, CD34+ cells, and committed progenitors (CFUs) after 48 h at +4°C. The data represent the percentage of viable cells at 48-h storage period with respect to the culture endpoint (day 12) values. The real total cell, cluster of differentiation 34-positive (CD34+) cell, and colony forming unit (CFU) numbers/ml of culture at day 12 were 1.85 ± 0.75 × 106, 2.1 ± 0.65 × 105, and 2.0 ± 0.75 × 105, respectively. **p < 0.01, Wilcoxon test compared to equivalent medium; n = 13. all total and CD34+ cells and 70% of CFUs survived in culture medium (Fig. 1).

SRCCD—engraftment capacity of ex vivo expanded cells at culture endpoint (day 12) and after 48-h storage at +4°C. (A.1–3) analysis of human-specific CD45 (A.1), CD33 (A.2), CD19 (A.3) antigens in femoral bone marrow of nonobese diabetic/severe combined immunodeficient/interleukin 2 receptor gamma-chain null (NSG) mice 8 weeks after injection. Each point represents an individual mouse. (B.1–3) Percentage of “positive mice” with respect to human antigens, CD45 (B.1), CD33 (B.2), CD19 (B.3). (C.1–3) Mean human chimerism, CD45 (C.1), CD33 (C.2), CD19 (C.3). Mean/median numerical values are given above the bars. The limit of 0.8% was taken as threshold of positivity for CD45 and that of 0.23 % for both CD33 and CD19, as presented in A.1–3 respectively by the dotted line. B.1–3 and C.1–3 present the data derived from the analysis of A.1–3. Each mouse was injected by a culture fraction corresponding to 1,000 CD34+ cells at day 0 of culture. SRCCD, relatively more mature SCID repopulating cell (SRC) subpopulation.
The advantage of culture medium was even more obvious for the subpopulation of SRC detected on the basis of human committed progenitors grown from engrafted mice bone marrow (SRCCFU): virtually the same percentage of mice contained human CFU in their bone marrow at the end of culture (day 12) and after storage at +4°C for 48 h in culture medium (82% vs. 83%, respectively), while this percentage was only 25% for the mice that received cells stored in albumin solution (Fig. 3A). If only the positive mice were taken into account, the mean number of colonies per femur was comparable before and after the storage in culture medium and probably in albumin solution (only three positive mice for latest condition preventing reliable analysis) (Fig. 3B). The analysis of relative CFU-GM versus BFU-E proportions in these cultures revealed that day 12 differentiation ratio was maintained in culture medium after 48 h at +4°C (around 20%), while this index was twice lower in the cultures initiated with human progenitors contained in bone marrow of mice injected with expanded cells stored in albumin solution at +4°C (Fig. 3C).

SRCCFU—Engraftment capacity of ex vivo expanded cells at culture endpoint (day 12) and after 48-h storage at +4°C. Analysis of CFU of human origin grown in cultures initiated with femoral bone marrow cells of NSG mice 8 weeks after injection. (A) Total number of CFU of human origin per mouse femur. Each individual point represents one mouse. (B) Mean (±SEM) human CFU number per femur {only the mice with a detectable colony number were taken into account [>10 colonies/femur in this case, accordingly, the number of mice analyzed readable from (A)]}. (C) Relative colony-forming units-granulocyte macrophage (CFU-GM) versus burst-forming units-erythroid (BFU-E) proportions in cultures. Only mice with a detectable CFU number were taken into account. SRCCFU, relatively less mature subpopulation of SRCs.
Discussion
The most interesting results presented here concern the maintenance of hematopoietic stem cells at +4°C, showing that our culture medium is able to maintain not only total number of SRCCFU (relatively more primitive than SRCCD subpopulation) but also their proliferative and differentiation capacities. Unlike in culture medium, storage in albumin solution results in an evident decline of SRCCFU number and induces modification of their differentiation potential as revealed through CFU-GM/BFU-E ratio (Fig. 3C).
With some exceptions [for example, Moroff et al (13), who included the functional LTC-IC (long-term culture– initiating cell) test], the studies on liquid storage based their conclusions on the analysis of viable CD34+ cells and colony forming unit cell (CFC) maintenance (1,8,10,15). Since our conclusions are based not only on CD34+ cell population and committed progenitors, but also on the stem cells capable of in vivo reconstitution (SRCCD and SRCCFU), this study represents a more exaustive experimental approach to liquid storage impact on functionality of an hematopoietic graft. Furthermore, it seems to be the first study of the impact of liquid storage on ex vivo expanded hematopoietic stem and progenitor cells.
Despite the lack of data concerning expanded grafts, several studies on nonexpanded bone marrow and peripheral blood hematopoietic cells agree that the “refrigerated temperature” (2–8°C) maintains better than the room one viable CD34+ cells (1,8,10,15). Accordingly, as well as taking into consideration that the +4°C temperature condition is in use for routine liquid storage of hematopoietic cells, we focused our investigation of functional stability of expanded cells at this temperature. Our data show that IV albumin solution presently used to resuspend the cells before their injection does not have the capacity to maintain functional committed progenitors and stem cells in our expansion products for 48 h at +4°C. On the contrary, in addition to stem cells (SRCCD and SRCCFU), the culture medium exhibits a good protective action on committed progenitors as well as the overall population of CD34+ and TNCs. The data presented point to the possibility of maintainence of the expanded graft functional capacities at standard transportation temperature (+4°C) for 48 h. This period should allow for the transportation of ex vivo expanded grafts worldwide and their transplantation after washing in the distant cell therapy laboratory or transplantation center. Even if HP01 medium is fivefold more expensive than the human serum albumin, we believe that the beneficial effects justify its use. Furthermore, the expanded cord blood grafts are resuspended in low volume (200 ml), marginalizing the cost difference of the expansion/engraftment procedure. In addition, the successful clinical trials with this antioxidant-supplied (6) medium (2,12) opens up a perspective for its large-scale production and consequent decrease of the price.
The fact that culture medium exhibits a protective capacity on ex vivo expanded product confirms the biological reality of our objective: functional maintenance of expanded grafts for 2 days at 4°C.
Footnotes
Acknowledgments
This work was funded by a grant from Agence de la Biomédecine (France) – Appel d'Offres Recherche “Recherche et Greffe” 2009, by Oséo C4C grant, as well as by research funds of Aquitaine-Limousin Branch of French Blood Institute (EFS-AL). The authors are grateful to Bernard Dazey, Ph.D., Head of Cord Blood Bank of EFS-AL for providing cord blood units for this study as well as to Mrs Elisabeth Doutreloux-Volkmann for manuscript reading and correction. The authors declare no conflicts of interest.
