Abstract
To improve graft preservation and consequently reduce conservation injuries, the composition of preservation solution is of outmost importance. It was demonstrated that the colloid polyethylene glycol (PEG), used in SCOT solution, has protective effects on cell membranes and immunocamouflage properties. The aim of this study was to optimize the concentration and chain length of PEG to improve pancreatic islet preservation and outcome. In a model of murine islet allotransplantation, islets were isolated with SCOT containing various concentrations of PEG 20 kDa or 35 kDa. Better islet yield (IEQ) was obtained with SCOT + PEG at 15–30 g/L versus other PEG concentrations and control CMRL-1066 + 1% BSA solution (p < 0.05). Allograft survival was better prolonged (up to 20 days) in the groups SCOT + PEG 20 kDa 10–30 g/L compared to PEG 35 kDa (less than 17.8 days) and to control solutions (less than 17.5 days). In terms of graft function recovery, the use of PEG 20 kDa 15–30 g/L induced no primary nonfunction and delayed graft function contrary to CMRL-1066 and other PEG solutions. The use of the extracellular-type solution SCOT containing PEG 20 kDa 15 g/L as colloid could be a new way to optimize graft integrity preservation and allograft outcome.
Keywords
Introduction
During the transplantation procedure, the graft is exposed to an extracorporeal conservation sequence from tissue procurement until transplantation in recipient, inducing metabolism and integrity graft disorders. Typically, the resulting conservation injury causes graft primary nonfunction (PNF) or delayed graft function (DGF), which play a major role in graft loss and development of chronic graft failure (47,50). In addition, the shortage of donors has led transplant centers to accept extended criteria donors, whose organs are more prone to graft conservation injury, resulting in increased PNF and DGF rates. This emphasizes the importance of organ preservation (25,40).
Islet transplantation provides a promising alternative to whole pancreas transplantation for type I diabetes treatment because of a reduced morbidity rate and decreased invasiveness of the surgical procedure (54). However, despite major improvements during the last years (10), islet transplants are associated with significantly lower engraftment efficiencies than whole pancreas transplants. One major reason for the poor functional islet yield obtention is that islet isolation is difficult, with important islet integrity and function disorders, variations from group to group and from pancreas to pancreas. As a consequence, many transplants are performed with a donor/recipient ratio of at least 2:1. Hence, the optimization of islet preservation/isolation method is of paramount importance (1,12,14,41,44,46,48,56).
To improve graft preservation and consequently reduce injury, the composition of preservation solution is critical (2,25,57). Several solutions, such as University of Wisconsin (UW) solution, present a high potassium content, which induces a depolarization of the cellular membrane, an adenosine triphosphate (ATP) depletion, and consequently a cellular acidosis (2,17,25,35). Current solutions, such as the current gold standard CMRL-1066 (Connaught Medical Research Laboratories-1066), are based on extracellular ionic balance (low potassium, high sodium), which is more appropriate for graft preservation (2,17,25,35).
Other elements of preservation are impermeants and colloids. Impermeants are predominantly effective at the level of cell membranes and at the interstitial compartment, while colloids are used for cell membranes and intravascular compartment (40). For long preservation times, the presence of a colloid appears important to maintain organ viability (55). However, it is important to select the right colloid. Indeed, it has been shown that HES (hydroxyethyl starch) induces side effects such as renal macrophage infiltration (31), tubular damages (11,31), renal cell vacuolization (31), and red blood cells aggregation (42,51). Other colloids, such as albumin, are used (e.g., in CMRL-1066); however, the production process is long and expensive. These elements initiated a search for other colloids, such as polyethylene-glycol (PEG) (5,7,9), an inexpensive polymer. The neutral and nontoxic PEG polymer acts as a colloid promoting cellular membrane stabilization by long-lasting hydrogen bonds, limiting cellular edema, and masking cell surface antigens (4,8,16,22,32,34,39,43,53,58,61,63). PEG binds to the membrane and polarizes H2O molecules in six to eight layers, resulting in the formation of a “water cloud” surrounding the cells (8,16).
The first study reporting the effects of PEG containing solution was from Collins et al. in 1991 (13). They introduced PEG 20 kDa (50 g/L) in a modified UW solution (Cardiosol®) to replace the HES and used it for human heart transplantation. Unexpectedly, the rate of acute rejection in recipients who received modified solution-preserved donor hearts was decreased (50% at 1 year versus 78% in all other heart transplant recipients). They suggested a direct suppressing effect of the PEG on the immune reaction. Several experimental studies confirmed the preservation efficacy and immunosuppressive property of PEG for preservation of liver (4,58) as well as for kidney (17,18,23,24,27), pancreas (63), pancreatic islets (22,59,60), small bowel (32), lung (35), and red blood cells (8,43,53).
The necessity to use a low K+ solution (“extracellular type” composition) with a nontoxic colloid such as PEG to prevent cell edema is now well established (2,25,57). Our group designed an initial preservation solution, named SCOT (Solution de Conservation des Organes et des Tissus) (17,22), with two main characteristics: (i) an extracellular K+ concentration (5 mM K+) to avoid cell membrane depolarization and ATP depletion and (ii) the presence of 30 g/L of PEG 20 kDa. The benefits of SCOT were established in models of isolated perfused pig kidney and pig kidney autotransplantation (23,26,28) where SCOT decreased ischemia/reperfusion injuries and immune cell infiltration (27). Jayle et al. in 2003 showed better vascular resistance and reduction of cellular and mitochondrial edema in a pig lung preservation model with SCOT (35). Moreover, a recent in vitro study has shown that SCOT was the most efficient commercial solution to preserve cell viability and confirmed PEG antioxidative properties (15). In our experience, the use of the initial SCOT solution (containing 30 g/L of PEG 20 kDa) as an isolation medium prolonged graft survival significantly in two models of allograft rejection, including a transgenic model in which >90% of the recipient CD8 TCR was specific for an antigen expressed on the graft islet β cells (22).
Different chain lengths (i.e., molecular weight) and concentrations of PEG can be used. Indeed, a study showed that IGL-1 solution (containing 1 g/L of PEG 35 kDa) demonstrated similar islet isolation results as the UW solution, making the PEG-containing solution a promising alternative (62). It appears that the optimal concentration and chain length of PEG remains to be determined. The goal of the present work is to complete our previous study (22,45) by testing several PEG chain lengths (20 and 35 kDa) and several concentrations of PEG in SCOT during murine pancreatic islet isolation and investigate its impact on allotransplantation.
Materials and Methods
Groups
To optimize the PEG properties in the SCOT solution (Macopharma, France), different concentrations and molecular weight (MW) of PEG (Sigma Aldrich, France) were added to SCOT and compared to control solutions: SCOT without PEG (Macopharma), UW (Bristol Meyers Squibb, France) standard preservation solution, Hank's balanced salt solution 1x (HBSS) (Gibco-BRL, France) + 0.5% bovine serum albumin (BSA) (Sigma), and CMRL-1066 (Gibco BRL) + 1% BSA currently used in islet preservation (29,37,38).
Groups consisted of allograft with islets isolated with the different solutions: SCOT without PEG, HBSS + 0.5% BSA, CMRL-1066 + 1% BSA, UW, SCOT + PEG 20 kDa at 0.5, 10, 15, 20, and 30 g/L or PEG 35 kDa 1, 5, 15, 20, 52 g/L (n = 5–8).
Osmolarity and Viscosity
Osmolarity was measured by freezing point depression, with an osmometer (Hermann Roebling Messtechnik, Berlin, Germany). Viscosity was measured at room temperature using a Cannon Fensk type viscometer 1634 (Rheotec, France). The Cannon Fenske tube measures kinematic viscosity in units of centistokes (cSt) and equates to time of flow multiplied by tube constant (size 75 according to viscosity).
Animals
Male mice were purchased from Charles River (France) and housed in a small animal facility. Mice were kept under specific pathogen-free conditions and manipulated according to European Council Directive 86/609/EEC, defining the protection guidelines of animals used for experimental and other scientific purposes. A full mismatch allograft model was used, donors were C3H (H-2Kk) mice, and recipients were Balb/c (H-2Kd) mice (6 weeks old). In all experiments, experimental groups were performed by the same operator.
Islet Isolation
Briefly, pancreas was distended in situ by injection through the biliary duct and the Wirsung duct of Hank's solution (Gibco BRL) + 0.33 mg/ml Liberase RI (Roche Diagnostics, France). The gland was then recovered and incubated in the tested solution at 37°C for 12 min without mechanical disruption (stationary digestion). Then vials were vigorously shaken, and the cell suspension was filtered through a 500-μm nylon mesh. Islets were then washed in the tested solution, purified on a discontinuous density Ficoll gradient (Amersham, France), washed in the tested solution, handpicked to obtain 100% purity, counted according to the islet equivalent (IEQ) method (49), and then cultured overnight at 37°C + 5% CO2 in CMRL-1066 + 1% BSA.
Islet Morphology
At the end of the isolation process, islets isolated with the different solutions were compared using light microscopy, evaluating islet size and integrity (dissociated or not).
Islet Transplantation
Diabetes was induced by an intraperitoneal injection of 250 mg/kg streptozotocin (STZ) (Sigma) in the recipient mice (21,22). Only recipients with two consecutive blood glucose determinations >350 mg/dl after 48 h of STZ administration were eligible. General anesthesia was induced by intraperitoneal injection of ketamine (80 mg/kg) and xylazine (4 mg/kg) (Sigma). To reach normoglycemia as soon as possible—allowing early posttransplant events assessment—1400 IEQ were grafted under the left kidney capsule as described previously (21,22). No immunosuppressive medication was administered. Grafts lost due to surgical failure were excluded.
Assessment of Graft Outcome
Blood glucose determinations were performed using a glucometer (Bayer Diagnostics, France) at 24 and 48 h after transplantation and every other day. Allograft functionality was determined by normoglycemia (<100 mg/dl), and allograft rejection was defined by two successive blood glucose >200 mg/dl. Primary nonfunction (PNF) was defined as a glycemia never returning below 200 mg/dl. Delayed graft function (DGF) was defined by the no return to normoglycemia within the first 48 h after transplantation.
Statistical Analysis
All data are expressed as means ± SEM. Statistics were performed with GraphPad and NCSS software. Statistics to islet yield data were performed using the ANOVA and Bonferroni's multiple comparison test. Statistics to allograft survival time (primary nonfunction were excluded) were performed with ANOVA and log-rank test. Statistics to graft outcome (including PNF + DGF + allograft survival time), represented by “area under the curve” (area unit), were performed with ANOVA and Dunn's test. A value of p < 0.05 was considered statistically significant.
Results
Viscosity and Osmolarity
Plasma viscosity was 1.84 cSt (centistock). UW, SCOT + PEG 35 kDa 20 g/L, and SCOT + PEG 35 kDa 52 g/L had higher viscosities (3.22, 2.49, and 7.35 cSt, respectively), while all other solutions had physiological viscosity (Fig. 1).
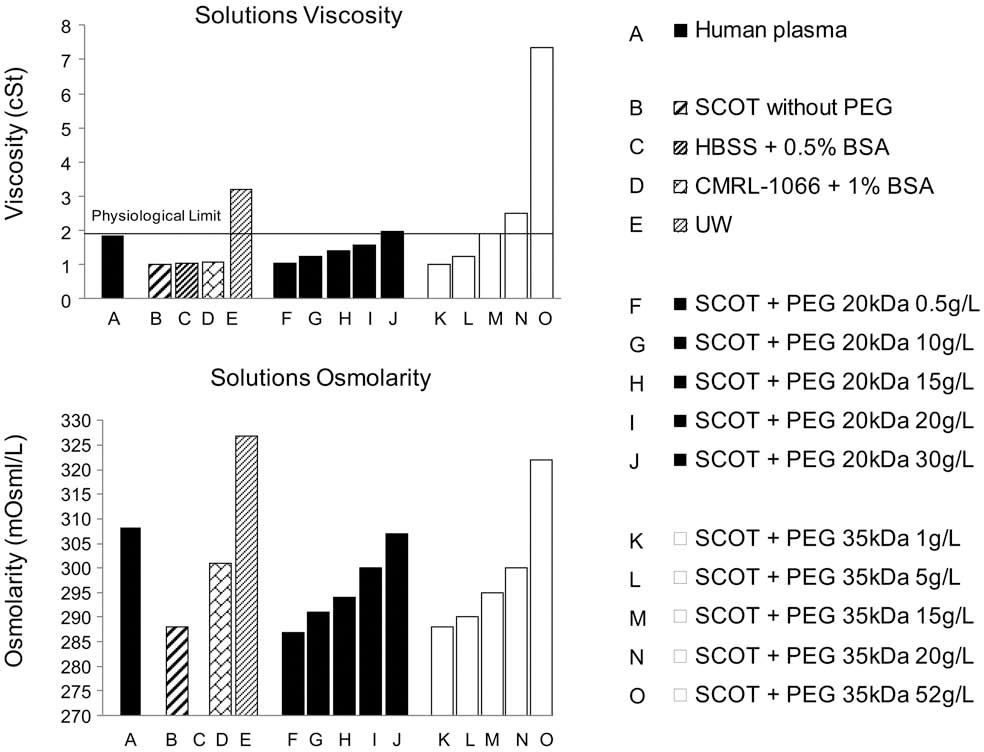
Top: Viscosity in centistoke (cSt) of solutions compared to the human plasma corresponding to the physiological limit (1.84 cSt). Bottom: Osmolarity in mOsm/L of solutions compared to the human plasma.
The osmolarity of plasma was 308 mOsm/L; normal physiologic osmolarity range is 290–320 mOsm/L. Solutions SCOT + PEG 20 and 35 kDa 10–30 g/L have an osmolarity ranging from 293 to 307 mOsm/L. UW solution (327 mOsm/L) and solution SCOT + PEG 35 kDa 52 g/L (322 mOsm/L) have osmolarity above to the acceptable range (Fig. 1). HBSS + 0.5% BSA osmolarity was 32 mOsm/L (does not appear in Fig. 1).
Islet Morphology
Microscopy studies (Fig. 2) showed that the islet morphology was better preserved (normal size and better integrity) when islets were isolated with solutions of SCOT containing PEG and CMRL-1066 compared to other solutions: UW, HBSS + 0.5% BSA, and SCOT without PEG, where islets were more dissociated.

Islet morphology at the end of the isolation process with the different solutions. Original magnification: 40x. The majority of islets were dissociated after isolation with control solutions (SCOT without PEG, HBSS + 0.5% BSA, and UW), compared to the initial integrity islet preserved with the use of CMRL-1066 + 1% BSA and SCOT + PEG solutions. Scale bars: 100 μm, included at the bottom right of each picture.
Islet Yield
Islet yields (IEQ/pancreas), calculated at the end of the isolation process, are represented in Figure 3. In association with the islet morphology, the islet yield was significantly improved with SCOT + PEG >5 g/L (up to 527 IEQ/pancreas) compared to controls solutions (p < 0.001): UW (371 ± 11 IEQ/pancreas), SCOT without PEG (432 ± 19 IEQ/pancreas), HBSS + 0.5% BSA (448 ± 9 IEQ/pancreas), and CMRL-1066 + 1% BSA (494 ± 24 IEQ/pancreas). Islet yield was significantly increased with the use of SCOT + PEG 20 kDa at 15, 20 and 30 g/L compared to SCOT + PEG 35 kDa at 1 and 52 g/L and to SCOT + PEG 20 kDa at 0.5 g/L (p < 0.01).
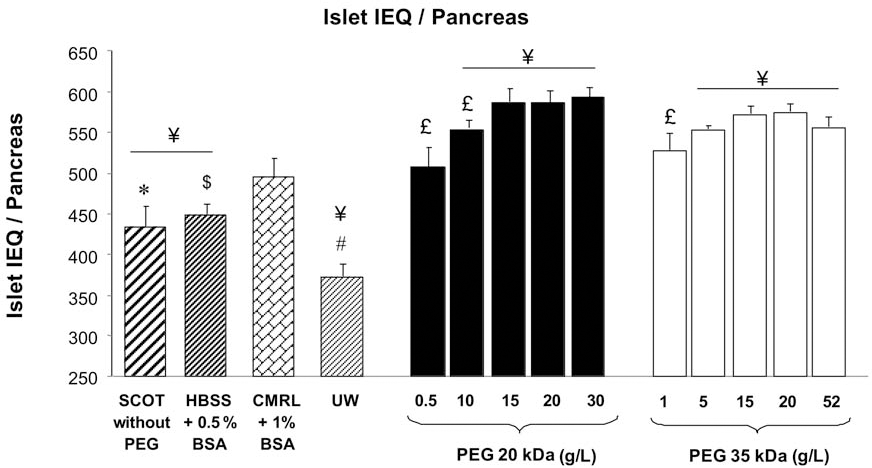
Islet yield (IEQ/pancreas) obtained at the end of the isolation process with different solutions. Results are expressed as mean ± SEM (n = 7). ANOVA and Bonferroni's multiple comparison test (significant values of different PEG concentration in a same chain length group are represented by £p < 0.05; significant values of groups vs. CMRL-1066 + 1% BSA are represented by ¥p < 0.001; UW vs. other groups is represented by #p < 0.001; SCOT without PEG vs. other groups is represented by *p < 0.001; and HBSS – 0.5% BSA vs. other groups is represented by $p < 0.001).
Allograft Outcome
Allograft outcome, including PNF, DGF, and allograft survival time, are represented in Table 1 (grafts lost due to surgical failure were excluded). No PNF was observed in the groups SCOT + PEG 20 and 35 kDa 5–30 g/L. In contrast, the rate of PNF was 14% in the CMRL-1066 + 1% BSA, 17% in HBSS + 0.5% BSA, 25% in SCOT without PEG, and 28% in UW group. Regarding the risk of DGF, SCOT + PEG 20 kDa > 10 g/L led to rapid function recovery compared to a DGF rate ranging from 14% to 42% in controls and SCOT + PEG 35 kDa groups.
Allograft Outcome Representation

Statistics to allograft survival time (primary nonfunction were excluded) were performed with ANOVA and log-rank test (significant values of p < 0.05 represented by * to SCOT without PEG, $ to HBSS + 0.5% BSA, £ to CMRL-1066 + 1% BSA, or # to UW; p < 0.01 represented by **, $$, or ##; p < 0.001 represented by ***, $$$, or ###). PNF, primary nonfunction; DGF, delayed graft function, in the different groups solutions used.
Longer graft survivals were obtained with SCOT + PEG 20 kDa ≥ 10 g/L, resulting in a mean survival of up to 20 days, better than CMRL-1066 + 1% BSA (17.5 ± 1 days, £p < 0.05), UW solution (17.2 ± 0.4 days, #p < 0.05), SCOT solution without PEG (14 ± 0.9 days, **p < 0.01), and HBSS + 0.5% BSA solution (14 ± 0.7 days, $$p < 0.01) (Table 1). Graft survivals appeared improved when islets were isolated with SCOT + PEG 20 kDa at 10–30 g/L compared to SCOT + PEG 35 kDa at any concentration (Table 1).
Percentages of normoglycemic mice per groups from allotransplantation at day 0 to rejection are represented in Figure 4 (grafts lost due to surgical failure were excluded). Area under the curve data showed a significantly better allograft outcome (function recovery and allograft survival) in the SCOT + PEG 20 kDa > 10 g/L compared to CMRL-1066+1% BSA, to SCOT without PEG, to HBSS + 0.5% BSA, and to UW groups (p < 0.05). Allograft outcome of SCOT+PEG 35 kDa was not significantly different to the control CMRL-1066+1% BSA group.

Percentages of normoglycemic mice per groups after allotransplantation per day (n = 5–8, grafts lost due to surgical failure were excluded). Statistics to graft outcome (including PNF + DGF + allograft survival time), represented by area under the curve (area unit), were performed with ANOVA and Dunn's test (significant values p < 0.05 are represented by * vs. SCOT without PEG, $ vs. HBSS, # vs. UW, and £ vs. CMRL-1066).
Discussion
The goal of this study was to optimize chain lengths and concentrations of the PEG colloid to improve islet preservation and allograft outcome. Currently, two preservation solutions containing PEG are used in clinical settings: SCOT solution containing (for the initial solution) 30 g/L of PEG 20 kDa (30) and IGL-1 solution containing 1 g/L of PEG 35 kDa (62). In the current study, we compared PEG 35 kDa 1 g/L to 20 kDa 0.5 g/L of a similar molarity than IGL-1 and groups PEG 20 kDa 30 g/L to 35 kDa 52 g/L of a similar molarity than SCOT. Intermediary groups with PEG concentration between 5 and 20 g/L were added to obtain better determination of chain length effect.
Itasaka and Bradley, in 2002, suggested that small PEG molecular weights are not optimal for transplantation (10). In fact, Itasaka reported that only PEG 20 kDa, compared to 8 kDa, appears to significantly increase the survival of the small bowel allograft (32). Bradley showed that only PEG 20 kDa induced normal in vivo survival of “PEGylated” red blood cells in mice at immunoprotective concentrations (up to 2 mM) compared to PEG 5 kDa (8). A previous study tried to elucidate the biophysical effects of PEG polymer size, density, and linker chemistry on immunocamouflage propriety and found that the PEG 20 kDa can camouflage cellular membrane antigens to 13.8 nm compared to 6 nm with the use of a PEG 5 kDa (8). For these reasons and in accordance with our previous study (45), we compared only intermediate PEG molecular weights 20 and 35 kDa in our model of mouse islet pancreatic transplantation.
This islet model is interesting for the study of the effect of preservation solution, as it presents the opportunity to evaluate the preservation effect with regards to islet morphology and islet yield after the isolation process. Indeed, in the clinic, the functional islet yield obtained at the end of the isolation process remains a problem (1,6,48), and the preservation quality determines a successful islet transplantation (6,36). Furthermore, evaluation of graft outcome in a diabetic recipient mouse is based on a simple glycemia control for the judgement of the graft insulin secretion functionality. Use of islet pancreatic tissue as graft is a perfect model at the junction of whole organ and cell transplantation.
In order to determine which solutions might correspond to physiological conditions, viscosity and osmolarity of the different solutions tested were determined. It is recommended in the clinic to use a “blood-like” preservation solution. Blood viscosity, principally determined by the colloid presence, is 1.84 cSt. In order to limit intracellular or extracellular water movement, solution osmolarity must be between 290 and 320 mOsm/L. Our results showed that the solutions SCOT + PEG 20 or 35 kDa between 0.5 and 30 g/L have physiological viscosity and osmolarity.
Pancreatic islet transplantation could be a precious therapy for type I diabetes (54); however, the processes of isolation and preservation culture disturb islet function and integrity (56). In order to remediate this, one of the major goals is to improve the islet yield (islet integrity) after islet isolation. Importantly, herein, we show that SCOT + PEG at 15–30 g/L significantly improved the IEQ (up to 527 IEQ/pancreas), while the current gold standard CMRL-1066 does not permit a better islet yield (494 ± 24 IEQ/pancreas) as the SCOT without PEG (432 ± 19 IEQ/ pancreas). These last data show the importance of the colloid presence in a preservation solution to improve the tissue integrity.
In the current study, too low or too high PEG concentration resulted in a suboptimal allograft function recovery (14–28% of PNF) in contrast of no PNF observed in PEG 20 and 35 kDa used between 5 and 30 g/L. Regarding the risk of DGF, PEG 20 kDa > 10 g/L led to immediate function recovery compared to a DGF rate ranging from 14% to 42% in PEG 35 kDa groups. This significant graft recovery is linked to the use of PEG colloid since the use of SCOT without PEG resulted in significantly more PNF (25%) and DGF (42%). Hence, the right choice of colloid size and concentration can provide superior protection to the graft, increasing quality above the level of the standard solution CMRL-1066 (14% PNF and 28% DGF).
Our results showed that use of PEG 20 kDa at 10–30 g/L significantly prolonged islet allograft survival up to 20 days compared to PEG 35 kDa (p < 0.05), SCOT without PEG (p < 0.01), and CMRL-1066 (p < 0.05 vs. PEG 20 kDa 30 g/L). This significant prolongation of the allograft survival time is linked to the use of PEG as the use of SCOT without PEG resulted in lower allograft survival (14 ± 1.7 days, p < 0.01).
Allograft outcome data, determined as percentages of normoglycemic mice per groups from allotransplantation at day 0 to rejection, showed significantly better outcome with the use of SCOT + PEG 20 kDa > 10 g/L compared to CMRL-1066 group and to other control groups (p < 0.05). In turn, allograft outcome with use of SCOT + PEG 35 kDa were not significantly better to CMRL-1066 and other control groups.
In summary, our results showed that better graft function recovery and allograft survival are associated with a better cellular preservation, according to the islet yield and to the graft outcome curve. This significant improvement in graft outcome is associated with the use of preservation solution containing PEG as colloid, compared to no colloid control solution (SCOT without PEG) and to BSA or HES colloid. Indeed, SCOT containing PEG 20 kDa at 15–30 g/L significantly increased islet yield and graft function recovery with no PNF, no DGF, and significantly prolonged the allograft survival time. These results are in accordance with a study by Hubert et al., reporting that better organ conservation was associated with graft survival prolongation and better metabolic control after islet transplantation (30).
Current practice is to collect the pancreas with one solution such as UW, Celsior, or histidine-tryptophanketoglutarate (HTK) (3,19,33) and CMRL-1066 for isolation. As SCOT + PEG 20 kDa 15 g/L is compatible with other abdominal organs like kidney and liver (52) due to its better circulation in the organs vascular network, it presents an interesting alternative to the standard method. Use of SCOT + PEG 20 kDa 15 g/L from pancreas procurement to islet isolation would simplify this process and improve both quality and yield.
Perico et al. explained, in 2004, that the preservation modalities affect the graft functionality and the recipient immune system reaction, thus influencing graft outcome (47). If the graft is badly preserved, there are increased injuries (necroses, cellular integrity damages, inflammation, etc.), strongly activating the immune system at transplantation (47), a phenomenon called “Danger Signal” (20). This acute, short-lasting situation has deleterious long-term effects. A better graft integrity preservation, hence a likely transient reduction of antigen presentation and thus decreased inflammation through the use of SCOT with PEG 20 kDa 15–30 g/L during preservation, could reduce the “Danger Signal” to improve the graft outcome and decrease the long-term consequences of recipient immune reactivity.
Footnotes
Acknowledgments
We thank I Rodde-Astier (Macopharma, France) for providing the SCOT solution; L. Lagorce, J. Decocq, M. Jacquard, S. Tillet, and I. Khalifeh, for technical help; and Bayer Diagnostics and Roche France for kindly providing some of the reagents used. We particularly thank Dr. N. Chatauret for precious help in the preparation of this manuscript. This work was supported by grants from MacoPharma France, Association Française des Diabétiques (AFD), Agence de BioMedecine (ABM), Fondation pour la Recherche Medicale (FRM), and Institut National de la Santé et de la Recherche Médicale (INSERM). The authors declare no conflicts of interest.
