Abstract
The aim of the study was to determine the long-term effect of transplantation of adipose-derived stromal cells (ADSCs) in a preclinical model of ischemia/reperfusion (I/R). I/R was induced in 28 Goettingen minipigs by 120 min of coronary artery occlusion followed by reperfusion. Nine days later, surviving animals were allocated to receive transendocardial injection of a mean of 213.6 ± 41.78 million green fluorescent protein (GFP)-expressing ADSCs (n = 7) or culture medium as control (n = 9). Heart function, cell engraftment, and histological analysis were performed 3 months after transplantation. Transplantation of ADSCs induced a statistically significant long-lasting (3 months) improvement in cardiac function and geometry in comparison with control animals. Functional improvement was associated with an increase in angiogenesis and vasculogenesis and a positive effect on heart remodeling with a decrease in fibrosis and cardiac hypertrophy in animals treated with ADSCs. Despite the lack of cell engraftment after 3 months, ADSC transplantation induced changes in the ratio between MMP/TIMP. Our results indicate that transplantation of ADSCs, despite the lack of long-term significant cell engraftment, increases vessel density and prevents adverse remodeling in a clinically relevant model of myocardial infarction, strongly suggesting a paracrine-mediated effect. ADSCs thus constitute an attractive candidate for the treatment of myocardial infarction.
Introduction
Over the last decade, the use of multiple types of stem cells for cardiovascular diseases has extensively been tested in a broad range of experimental settings from in vitro assays to clinical trials (22, 26). Although the desired goal of tissue regeneration has not really been achieved, most studies have suggested that transplantation of stem cells is associated with a modest beneficial effect on cardiac function. The limited cell engraftment and differentiation of transplanted cells into cardiomyocytes (4, 15) point toward a paracrine mechanism of action in which transplanted cells are able to secrete or induce host secretion of certain molecules with therapeutic capacity (11).
Among the various cell types, adipose-derived stem cells (ADSCs) represent an attractive source of stem cells for cardiovascular repair (17, 18). ADSCs are endowed with a remarkable differentiation potential as well as the capacity to release significant amounts of growth factors involved in healing and tissue regeneration (21, 24, 25, 28). Moreover, adipose tissue stands out for being easy to harvest and having high-efficiency stem cell culture. In spite of these facts, only a limited number of studies have been performed using ADSCs in models of cardiac disease (17, 18) and particularly in only two reports have adipose-derived stem cells been applied to large animal models of myocardial infarction (MI) (29, 32).
With few exceptions, studies either in rodents or in large animals have employed protocols that cannot be translated directly into the human setting owing to, for instance, the need for the prolonged culture of stem cells required to obtain enough cells for transplantation in an acute myocardial infarct model (32). In the current study, we compared the potential of ADSCs to prevent cardiac derangement and to contribute to improving cardiac function in a swine model of ischemia/reperfusion. ADSCs were obtained at the time of cardiac ischemia and injected within a clinically meaningful time frame after a short culture without multiple passages.
Materials and Methods
Animal Procedures and Study Design
Adult male and female Goettingen minipigs (average weight, 60–80 kg) procured from our breeding center were maintained in the animal facilities of CIFA (GLP accredited center at the University of Navarra, Spain). All the studies were performed in accordance with the US National Institutes of Health guidelines, the Declaration of Helsinki, and approved by the Ethics Committee for Animal Experimentation from the University of Navarra. In each procedure, animals were premedicated with a combination of 15 mg/kg ketamine (Imalgene 1000, MERIAL) and 2 mg/kg azaperon (Stresnil, Esteve Veterinaria). After sedation, animals received 5 mg/kg of etomidate (Hypnomidate, Janssen-CILAG) and 0.03 mg/ kg cisatracurium besylate (Nimbex, GlaxoSmithKline) IV and were endotracheally intubated and mechanically ventilated with supplementary oxygen. During surgery, anesthesia was maintained with a combination of 3% isofluorane (ISOFLO, Abbott) and 0.01 mg/kg/h fentanyl (Fentanest, Kern Pharma) administered by continuous infusion, and electrocardiogram (ECG), heart rate, saturation of peripheral oxygen (SpO2), end tidal carbon dioxide (EtCO2), and rectal temperature were controlled with a multiparameter monitor. At the end of the procedure and after extubation, all animals received the nonsteroidal analgesic ketoprofen (Ketofen 10%, MERIAL) 3 mg/ kg/24 h IM for 3 days as well as the antibiotic amoxicillin (Clamoxyl L.A., Pfizer) 7 mg/kg/24 h for 5 days.
Myocardial infarction was induced as previously described (10) in 28 pigs. Briefly, a 7-Fr introducer sheath was placed by dissection in the left carotid artery and 1.5 mg/kg heparin (Heparina 1%, Rovi) was intravenously infused. A 7-Fr coronary artery-guiding catheter was placed within the ostium under fluoroscopic guidance using a mobile C-arm (Powermobil, Siemens), and myocardial infarction was produced by occluding the left anterior descending coronary artery with a balloon catheter 2.5 just below the second diagonal branch. Temporary and complete occlusion was performed by balloon dilatation (8 atm) and maintained for 120 min, followed by reperfusion, as demonstrated by coronary angiography and ST segment elevation and reversion in the electrocardiogram. Lidocaine (B. Braun) 2 mg/kg IV and advanced life support were used when needed. Finally, the delivery catheter was removed, the carotid artery was ligated, and the cut down site was sutured. After induction of myocardial infarction, adipose tissue was collected after cleaning and disinfection of the right abdominal zone. An incision was performed in the skin of approximately 20 cm in length, which allowed us to dissect and obtain between 90 and 120 g of abdominal subcutaneous fat.
Nine days after ischemia/reperfusion (I/R), surviving animals (n = 20) were randomized to receive medium or ADSCs. Single-cell suspension was verified by microscopy to avoid the possible arrhythmogenicity of cell clumps. A percutaneous access through dissection of the femoral artery was performed. Treatment was intramyocardially delivered under simultaneous fluoroscopic and echocardiography (HP SONOS 4500) guidance using the Myocath® catheter (Bioheart, FL, USA) (Fig. 1). Multiple injections (average of 20 per time point of 0.25–0.5 ml per injection) were performed in each animal in and around the infarct area.

MyoCath® catheter (Bioheart, FL, USA). The device includes a double input, which permits cell injection as well as connection to the washing system to avoid the obstruction of the needle. The MyoCath provides for multiple injections to a predetermined needle insertion depth with a single core needle of 25-gauge diameter that can be advanced and retracted from the tip of the catheter.
Cell Culture and Characterization
Adipose tissue biopsies and serum were obtained from pigs at the time of I/R. Samples were processed as previously described (24) with minor modifications. Briefly, fat was carefully minced and digested in 2 mg/ml collagenase type I (Invitrogen, Barcelona, Spain) for 45 min at 37°C. After serial filtering through 100 μm and 40 μm mesh (BD, Madrid, Spain), cells were suspended in erythrocyte lysing solution (155 mM NH4Cl, 10 mM KHCO3, 0.1 mM EDTA) for 5 min, centrifuged, and counted. The resulting stromal vascular fraction (SVF) was plated in a 10-tray cell culture factory (Thermo, Barcelona, Spain) in minimum essential medium Eagle α modification (α-MEM; Invitrogen) supplemented with 10% fetal calf serum (Biochrom, Berlin, Germany) and 1% antibiotics (Invitrogen). Approximately 2 million cells/g of adipose tissue were obtained. After 2 days, cells were washed in PBS (Invitrogen) and infected as described below. Forty-eight hours prior to cell injection, medium was replaced with α-MEM supplemented with antibiotics and 10% autologous serum to avoid unwanted immune reactions. Cells were characterized by fluorescence-activated cell sorting (FACS) analysis using antibodies against swine leukocyte antigen (SLA)-DR, CD29, CD31, and CD90 (all from BD) before plating and before injection.
Lentiviral Production and ADSC Infection
Lentiviruses were produced in 293T cells by cotransfection of four plasmids as previously described (7). Briefly, the packaging plasmids MDL.RRE and pRSV.REV and the VSV-G envelope protein expression plasmids were mixed with the PPT.CMV plasmid [which encodes green fluorescent protein (GFP) under the control of the constitutively expressed cytomegalovirus (CMV) promoter], and 293T cells were transiently transfected using calcium chloride. Medium was changed every 24 h. After 72 and 96 h posttransfection, supernatants were collected, filtered through 0.45 μm, concentrated by ultracentrifugation at 50,000 × g for 120 min, and stored at −80°C. Two days after ADSC transplantation, one viral production (from forty 100-mm dishes) was resuspended in 150 ml of basal medium, added to the culture factory, and incubated for 1 h at 37°C with gentle agitation. Finally, 550 ml of complete medium was added.
Assessment of Ventricular Function and Arrhythmias
Animals under general anesthesia were placed in the left lateral decubitus position, and transthoracic two-dimensional echocardiography was performed using a Sonos 4500 ultrasound system (Philips) and a 4-MHz linear array transducer. Left ventricular remodeling was assessed by measuring end-systolic and end-diastolic volumes and diameters according to the American Society of Echocardiography and adjusted for animal weight at the time of analysis. Assessment of diastolic function was performed by mitral filling pulse Doppler and mitral annulus tissue Doppler. Left ventricular ejection fraction (LVEF) was determined in parasternal short axis (19) due to the unreliability of the four-camera approach in swine (12, 33). Echocardiogram was performed at baseline (before MI), before the implantation procedure, and at the time of sacrifice (3 months posttransplantation) by two investigators blinded to the type of treatment. Major arrhythmic events (ventricular tachycardia and ventricular fibrillation) during procedures were registered in all groups.
Histological Processing and Immune Staining
After sacrifice with pentobarbital and a saturated solution of potassium chloride, the heart was excised and fixed in formalin and paraffin embedded for histological analysis. The organ was transverse-cut in 2- to 3-mm-thick sections, and location of MI was visually assessed. Sampling of tissues consisted of scar surrounded by a ring of viable myocardium. After paraffin embedding, 5-μm sections were stained with hematoxylin–eosin and Gallego Tricromic for qualitative assessment and with the appropriate antibodies. These included anti-GFP (Invitrogen), antimatrix metalloproteinase 2 (MMP2), tissue inhibitor of metalloproteinase 1 (TIMP1), and TIMP2 (Abcam, Cambridge, UK). EnVision™ horseradish peroxidase (HRP)-conjugated system (Dako, Barcelona, Spain) was used as secondary reagent and diaminobenzidine (Dako) as chromogen.
Vasculogenesis (arterioles and arteries) was evaluated in paraffin-embedded sections by measuring the area occupied by smooth muscle-covered vessels in the infarct border using an anti-α-smooth muscle actin antibody coupled to cyanine 3 (Cy3; Sigma, Madrid, Spain). Angiogenesis was assessed by counting the density of small vessels (capillaries/mm2) stained with Bandeiraea simplicifolia lectin-I (BSL-I, Sigma) and using Vectastain® (Vector, Barcelona, Spain) as secondary reagent and diaminobenzidine as chromogen. Infarct size was quantified in transverse macroscopic sections using the image analyzing software NIS-Elements (Nikon). The degree of fibrosis was determined by quantification of collagen deposition stained by Sirius Red staining. Briefly, sections were deparaffinized and immersed in 0.1% Fast Red (Sigma) in a saturated solution of picric acid, for 90 min, differentiated for 2 min in HCl (Sigma) 0.01 N, dehydrated, and mounted in DPX. Measurement of cardiomyocyte (CM) hypertrophy was performed on laminin-stained (antilaminin, Sigma) sections. High-power field images were acquired, and CM cross-sectional area was quantified using Image J software. A minimum of 120 CM per heart was analyzed. A minimum of 40 sections per animal were analyzed for each morphological measurement. All microphotographs were obtained on a Nikon Eclipse E800 microscope and analyzed with a computerized system (Axiovision 4.6, Zeiss, Germany). Sampling and measuring were performed in all cases by an investigator blinded to the treatment of individual animals.
PCR Analysis
DNA was extracted from paraffin-embedded tissue. Fifty to 80 slides per heart (periinfarct region) were deparaffinized and hydrated. Tissue was carefully separated from glass, and DNA was extracted with DNAeasy mini kit (Quiagen, Venlo, The Netherlands), following the manufacturer's instructions. GFP expression was analyzed by PCR (GFP-upstream primer: 5′-GCACCATCTTCTTCAAG GAC-3′; GFP-downstream primer: 5′-ACTTGTACAGCT CGTCCATG-3′). Amplification conditions were as follows: 95°C for 2 min followed by 40 cycles consisting on 94°C for 30 s, 59°C for 30 s, and 72°C for 30 s, plus an extension phase of 72°C for 10 min. All samples were also amplified for glyceraldehyde 3-phosphate dehydrogenase (GAPDH-upstream primer: 5′-ACCTGCCGCCTGGAGAAACC-3′; GAPDH-downstream primer: 5′-GACCATGAGGTCCA CCACCCTG-3′) as control for the presence of amplified DNA.
Serum Zymography
Protein in serum samples was quantified by the Bradford method (Thermo). One hundred micrograms of total protein was diluted in saline and mixed with equal amounts of sodium dodecyl sulfate (SDS) sample buffer (Novex, Carlsbad, CA, USA) and electrophoresed on 10% SDS-polyacrylamide gels (Novex) containing 1 mg/ml gelatin as the protease substrate. Following electrophoresis, gels were placed in 2.7% TritonX-100 for 1 h to remove SDS, and then incubated for 20 h at 37°C in developing buffer (50 mmol/L Tris base, 40 mmol/L HCl, 200 mmol/L NaCl, 5 mmol/L CaCl2, and 0.2% Brij 35; Novex). After incubation, gels were stained with gel code blue stain reagent (Pierce) for 1 h followed by destaining. Quantification was performed using QuantityOne™ software.
Statistical Analysis
All data are expressed as mean ± SD. Comparisons were performed using the paired or unpaired t test. In case of nonnormal distribution (demonstrated by Shapiro–Wilk and Komogorov–Smirnov tests), Wilcoxon or Mann–Whitney U test was used. Statistical analysis was performed with the SPSS 17.0 software, and differences were considered statistically significant when p < 0.05.
Results
Phenotypic Characterization and Transplantation of ADSCs
Flow cytometry analysis showed fresh SVF cells to be a heterogeneous population with low expression of the endothelial cell marker CD31 and higher CD90 and CD29 expression consistent with stromal cells (Fig. 2A). After 9 days in culture, a more homogenous population of cells with no expression of SLA-DR or CD31 was obtained (Fig. 2B). Animals received a mean of 213.6 ± 41.78 millions of ADSCs. GFP expression measured by flow cytometry and immunocytochemistry was consistently above 50% of the transplanted cells (Fig. 2C).

Fluorescent activated cell sorting (FACS) analysis of adipose-derived stromal cells (ADSCs). Cells were labeled with antibodies against swine leukocyte antigen (SLA)-DR, cluster of differentiation 29 (CD29), CD31, and CD90 (red line) or isotype controls (dashed lines), at the time of harvesting (A) and after 9 days in culture (B). (C) Green fluorescent protein (GFP) expression measured by immunocytochemistry (ICC) and FACS. A representative experiment is shown.
Transplantation of ADSCs Improves Cardiac Function in a Preclinical Model of Ischemia/Reperfusion
Cardiac function was assessed by echocardiography at baseline before MI, at day 9 before cell transplant, and at 3 months after transplant. Ischemia/reperfusion induced a statistically significant decrease in LVEF in comparison with baseline values, which was similar between ADSC-treated animals and controls (baseline LVEF was 76 ± 4% in the control group and 76.66 ± 3.98% in ADSC-treated animals; day +9 LVEF was 47.77 ± 8.45% in the control group and 46.69 ± 5.62% in ADSC-treated animals; p = 0.0001 between baseline and day +9). Reperfusion alone was associated with an improvement in LVEF from 47.77 ± 8.45% to 55.79 ± 6.39%; p = 0.026 3 months after transplantation (Fig. 3A), consistent with early reperfusion-induced preservation of cardiac function (34). However, when ADSCs were injected, transplanted animals showed a greater increase on cardiac contractility (from 46.69 ± 5.62% to 64.73 ± 5.14% at 3 months; p = 0.0001) (Fig. 3A). The LVEF at 3 months and the increase in LVEF were significantly greater in animals treated with ADSC in comparison with the control group (LVEF at 3 months: Control: 55.79 ± 6.39%; ADSC: 64.73 ± 5.14%; p = 0.027; Increase in LVEF: Control: 8.02 ± 2.94; ADSC: 18.04 ± 2.16; p = 0.021) (Fig. 3A, B). ADSC transplant was also associated with a statistically significant prevention of ventricle geometry worsening, as indicated by the significant decrease in end-systolic diameter (ESD, from 3.27 ± 0.71 cm before transplant to 2.69 ± 0.45 cm at 3 months; p = 0.029) and both end-diastolic and end-systolic volumes (EDV, from 87.06 ± 10.34 ml to 64.67 ± 4.64 ml at 3 months; p = 0.038; ESV, from 46.44 ± 8.03 ml to 25.58 ± 4.76 ml at 3 months; p = 0.028) (Fig. 3D–F), thus suggesting an effect on the remodeling process. Arrhythmias were registered at the time of ischemia induction (Control: n = 3; ADSC: n = 1) and transplantation (Control: n = 1; ADSC: n = 1) due to surgical procedures. No pericardial effusions or other safety issues were found during the duration of the study.
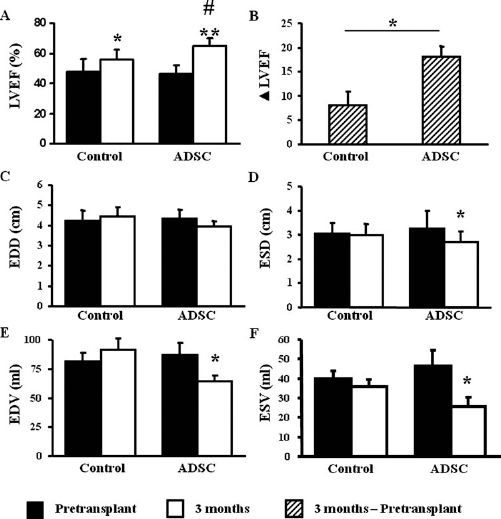
Transplantation of ADSCs induces an improvement in cardiac function and geometry. Cardiac function was measured by echocardiography before transplant and 3 months after transplant in the control (n = 9) and ADSC-treated (n = 7) animals. (A) Left ventricular ejection fraction (LVEF). (B) ΔLVEF between control and ADSC-treated animals. (C) End diastolic diameter (EDD). (D) End systolic diameter (ESD). (E) End diastolic volume (EDV). (F) End systolic volume (ESV). Results are shown as mean ± standard deviation (SD). *p < 0.05; **p < 0.01 between pretransplant and 3 months after ADSC or medium transplantation; #p < 0.05 between ADSC of medium-injected groups.
Transplantation of ADSCs Promotes Angiogenesis and Vasculogenesis and Inhibits Cardiac Remodeling in Infarcted Hearts
To evaluate whether changes in cardiac performance could be related to cell-derived effects upon tissue architecture, we measured infarct collagen content (fibrosis), vessel density (capillaries and arteries/arterioles), and myocyte hypertrophy in the border zone of the infarct. Although no significant difference in infarct size (Control: 11.14%; ADSC: 9.6%; p = 0.31), fibrosis was significantly reduced in ADSC-treated animals (Control: 57.79 ± 5.18%; ADSC: 47.84 ± 7.13%; p = 0.007) (Fig. 4A), while tissue vascularization was significantly improved in animals treated with ADSCs as indicated by an increase in the area occupied by smooth muscle-covered vessels (Control: 2.54 ± 1.08%; ADSC: 5.44 ± 2.28%; p = 0.037) (Fig. 4B) and in the capillary density (Control: 153.28 ± 29.3 vessels/high power field (HPF); ADSC: 235.22 ± 53.28 vessels/HPF; p = 0.003) (Fig. 4C). Finally, CM-hypertrophy was decreased in the ADSC-transplanted animals (Control: 460.27 ± 80.47 μm2; ADSC: 206.38 ± 32.66 μm2; p = 0.0001) (Fig. 4D).

ADSC transplant is associated with an increase in vasculogenesis and a decrease in cardiac fibrosis. Five-micromolar sections obtained from the infarct border zone of animals sacrificed at 3 months after transplant were analyzed. Fibrotic index (A), capillary (B), and arterioles/arteries (C) densities and cardiomyocyte (CM) hypertrophy (D) were determined by quantification of Sirius Red collagen staining/high power field (HPF), α-smooth muscle actin (SMA)-positive arterial vessels/mm2, Bandeiraea simplicifolia lectin-I (BSL-I) staining, and laminin-positive CM staining, respectively, in animals treated with media or ADSCs. Results represent the mean ± SD. A representative image is shown for each analysis. *p < 0.05; **p < 0.01.
Due to the multipotentiality of ADSCs, transplantation could be associated with differentiation of ADSCs into unwanted tissues such as adipose cells or other mesoderm-derived tissues (1). However, no evidence of ectopic adipose, bone, or cartilage tissue was observed in any of the treated animals (Fig. 5A). Three months after medium or cell transplantation, only minor foci of inflammatory cells were found in the myocardium, with no differences between ADSC and control groups (Fig. 5A). However, no trace of GFP-expressing cells could be found either by immunohistology or by PCR (Fig. 5B).

Transplantation of ADSCs is not associated with ectopic tissue formation and long term engraftment. (A) No differences in amounts of adipocytes, microcalcifications, or inflammatory infiltrates were found between control and ADSC-treated animals. (B) ADSC engraftment was not found 3 months after transplant either by immune-staining or PCR analysis for GFP.
We next sought to examine whether the improvement in cardiac function and remodeling could be related to changes in the composition of matrix metalloproteinases (MMPs) or their inhibitors (TIMPs) induced by transplantation of ADSCs (Fig. 6). In this case, both the border zone and the noninfarcted area were analyzed. Although we found no significant differences at 3 months between control and ADSC-treated animals in the expression of MMP2, the most abundant metalloproteinases in the porcine heart (6) (Fig. 6A–D), expression of TIMP1 and 2 was downregulated in CM in the noninfarcted area in the control group (Fig. 6E–F, I–J) in comparison with animals treated with ADSC. Furthermore, an increased number of TIMP1- and 2-positive vessels were found in the infarcted border zone of ADSC-transplanted animals (Fig. 6G–H, K–L). A decrease in the activity of MMP2 was found by serum zymography (Fig. 6M). As the proteinase activity depends on the balance between MMPs and TIMPs, these results suggest that ADSCs induced a decrease in the net activity of MMP2, thus limiting the adverse remodeling of the heart.

Transplantation of ADSC is associated with changes in the matrix metalloproteinases (MMP)/tissue inhibitors of matrix metalloproteinases (TIMP) balance. Immunohistochemistry using antibodies against MMP2 (A–D), TIMP1 (E–H), and TIMP2 (I–L) was performed in 5-μm sections obtained from the noninfarcted and the infarct border zone of animals sacrificed at 3 months after transplantation of ADSCs or media control. No differences in the expression of MMP2 were found between ADSC or control animals (pigs) in the noninfarcted myocardium (A, B) or in the border zone (C, D). Expression of TIMP1 and 2 was downregulated in CM (arrowheads in E and I), and an increase in TIMP1- or 2-expressing vessels was found in the border zone of ADSC-treated animals (asterisks in G, H, K, and L). Serum MMP2 activity by gelatin zymography was also diminished in ADSC-treated pigs (M). Results are shown as mean ± SD. *p < 0.05.
Discussion
Despite the impressive results obtained with stem cell therapy in animal models of myocardial infarction (27), transfer to the bedside has not fulfilled expectations and only modest results comparable to those of conventional treatments have been obtained. Some of the lessons learnt from previous research have recently been highlighted (20) and indicate that an important reason for discrepancies in the results resides in the divergences between animal models and the human disease these models try to mirror. For instance, in the acute MI setting, the use of culture cells (such as ADSCs or MSCs) would not be feasible unless allogeneic or previously stored cells were to be used. This is the case with the only two studies using adipose cells for the treatment of acute and subacute MI in large animal models (29, 32) in which cells were cultured several weeks before transplant. An additional limitation of both studies was the short-term follow-up of 30 and 21 days, respectively, which prevents conclusions regarding the long-term effect of cells from being drawn, an important issue considering that clinical trials have shown that the effect of stem cell transplantation can be transitory, as was the case with the bone marrow transfer to enhance ST elevation infarct regeneration (BOOST) trial (31). In our study, pigs were transplanted 9 days after harvesting ADSCs, a setting that optimally balances the time necessary to grow the cells with the decrease in the inflammatory reaction post-MI (3, 34), providing evidence for the improvement of cardiac function in a model that better resembles the human setting. The value of our model is also supported by the fact that the control group also experienced an improvement in cardiac function consistent with the reperfusion effect. In addition, timing and volume of adipose tissue extraction would be feasible in a human setting. Unlike previous studies (29, 32), we were unable to detect ADSCs at 3 months despite the functional effect observed.
Transplantation of ADSCs had an effect on two of the main detrimental factors related to the disease: the lack of sufficient blood supply and the late remodeling. In this regard, ADSCs have consistently been shown to contribute to cardiac regeneration by the release of the proangiogenic factors vascular endothelial growth factor and hepatocyte growth factor, a potential that is enhanced under hypoxia conditions as is the case in ischemia (13, 28).
Adverse remodeling is one of the major players in the transition from MI to heart failure (8). In our model, transplantation of ADSCs induced a decrease in collagen content. The lack of differences in MMP2 expression suggests that the net effect of collagen decrease was not due to an increased digestion but rather to prevention of collagen deposition. The decrease in TIMP1 and 2 in animals treated with ADSCs could contribute to decreased collagen deposition. In fact, the MMPs/ TIMPs system is tightly regulated in the myocardium, and the misbalance could result in severe organ damage (3, 14). Thus, ADSCs may be contributing to the correction of the imbalance that happens after MI, thus preventing ventricular dilatation (16, 30), which in turn could lead to a less stressed ventricle and reduced cardiomyocyte hypertrophy.
The lack of meaningful long-term engraftment remains a major caveat in cell therapy, which has been observed uniformly for every type of cell that has been used in animal models (2). Several mechanisms have been implicated in the cell attrition observed after transplantation, such as cell apoptosis due to the hypoxic milieu or immune rejection associated with GFP (2). Different means to improve engraftment have been tested including genetic manipulations (23). Nevertheless, improvement in engraftment remains a moving target in the field. Based on the results from a number of studies including ours, an increase in engraftment would be expected to improve the functional results. Simple strategies, such as repeated injections of cells, are associated with a modest improvement in function and engraftment but do not seem to be a definitive solution (9).
The results of two randomized placebo-controlled clinical trials with adipose-derived stem cells that have recently been completed, APOLLO (3D adipose-derived stem cell transplantation in the treatment of patients with an acute ST elevation myocardial infarction) and PRECISE (3D adipose-derived stem cells in the treatment of patients with nonrevascularizable ischemic myocardium), should provide important information regarding the usefulness of this therapy and possibly reinforce its applicability. In this sense, the present report sheds light on the behavior of ADSC therapy in a clinically relevant model of reperfused MI, showing that transplantation of ADSCs induces a significant benefit in cardiac contractility. This effect is related to the prevention of cardiac remodeling, an increase in damaged tissue perfusion, and a decrease in myocyte hypertrophy.
Limitations of the Study
Besides the lack of long-term engraftment of the cells, other limitations of our study deserve some comment: despite being a preclinical model, the induction of I/R in a healthy young animal does not reflect the human disease where most of the patients develop an acute ischemic event in the context of a preexistent disease. Thus, the observed functional improvement might not be a reflection of the result in humans. Several studies have demonstrated that sick patients may have “sick” stem cells less capable of providing a functional benefit (5). In that sense, the use of autologous cells could also be considered a limitation. Lastly, the follow-up of our study does not allow determining whether the functional benefit will be maintained for a long term. New studies with longer follow-up are warranted.
Footnotes
Acknowledgments
The authors would like to thank Beatriz Pelacho for her critical reading of this work. This work was supported in part by Ministerio de Ciencia e Innovacion: ISCIII PI070474, CP09/00333, and RETIC RD06/0014, PLE2009-0116, and FEDER funds from Comunidad de Trabajo de los Pirineos (CTP), European Union Framework Project VII (INELPY), Caja de Ahorros de Navarra (Programa Tu Eliges: Tu Decides), and the “UTE project CIMA”. The authors declare no conflict of interest.
