Abstract
Ex vivo alloanergization of human immune cells, via allostimulation in the presence of costimulatory blockade with either a combination of anti-B7.1 and anti-B7.2 antibodies or first-generation cytotoxic T-lymphocyte antigen 4-immunoglobulin (CTLA4-Ig), induces alloantigen-specific hyporesponsiveness and expands alloantigen-specific regulatory T cells (Treg). We have successfully used this approach in the clinical setting of haploidentical hematopoietic stem cell transplantation. Recently, the in vivo use of a new second-generation CTLA4-Ig, belatacept, has shown promise in controlling alloresponses after transplantation of both human kidneys and islet cells. We therefore compared the efficiency of first- and second-generation CTLA4-Ig in alloanergizing human peripheral blood mononuclear cells (PBMCs) and investigated whether ex vivo alloanergization with belatacept could be used to engineer an alloantigen-specific immunoregulatory population of autologous cells suitable for administration to recipients of cellular or solid organ transplant recipients. Alloanergization of HLA-mismatched human PBMCs with belatacept resulted in a greater reduction in subsequent alloresponses than alloanergization with first generation CTLA4-Ig. Moreover, subsequent ex vivo re-exposure of alloanergized cells to alloantigen in the absence of belatacept resulted in a significant expansion of Tregs with enhanced alloantigen-specific suppressive function. Alloanergized PBMCs retained functional Epstein-Barr virus (EBV)-specific T-cell responses, and expanded Tregs did not suppress EBV-specific proliferation of autologous cells. These results suggest that ex vivo alloanergization with belatacept provides a platform to engineer populations of recipient Treg with specificity for donor alloantigens but without nonspecific suppressive capacity. The potential advantages of such cells for solid organ transplantation include (1) reduction of the need for nonspecific immunosuppression, (2) retention of pathogen-specific immunity, and (3) control of graft rejection, if used as an intervention.
Introduction
Recognition of donor tissue by alloreactive recipient lymphocytes contributes to rejection after cellular and solid organ transplantation whereas recognition of host tissue by alloreactive donor lymphocytes contributes to graft-versus-host disease (GvHD) after allogeneic hematopoietic stem cell transplantation (AHSCT) (37). These obstacles to successful outcome may be reduced by pharmacological immunosuppression with antiproliferatives, calcineurin inhibitors, and anti-T-cell-specific antibodies (3,35,39). Unfortunately, these approaches produce both on and off target effects in the form of nonspecific immunosuppression and toxicity (5,33,38).
Cytotoxic T-lymphocyte antigen 4 (CTLA4)-immunoglobulin (Ig) is a human fusion protein that combines the extracellular portion of CTLA4, a high-affinity receptor for B7.1 and B7.2, with the Fc domain of human IgG1 (28). CTLA4-Ig was developed to competitively inhibit the binding of B7.1 and B7.2 ligands to their counter-receptor CD28 (27). First-generation CTLA4-Ig (abatacept) inhibits T-cell immune responses and is clinically approved for patients with rheumatoid arthritis (1,22,26). To increase immunosuppressive potency, a second-generation CTLA4-Ig (belatacept, previously LEA29Y) with two amino acid substitutions resulting in higher avidity binding to B7.1 and B7.2 was developed (25). In vivo administration of belatacept in clinical trials of renal allograft transplantation has resulted in comparable patient and graft survival, superior renal function, and reduced toxicity when compared to calcineurin inhibitor-containing regimens (11,49–51). Recently, in vivo administration of belatacept has also been successfully used to reduce rejection of transplanted human islet cells in patients with type 1 diabetes (41). However, the in vivo use of costimulatory blockade may result in off-target effects. Direct blockade of CD28 costimulation is known to impair T-cell virus-specific responses in vitro (48), and use of belatacept after solid organ transplantation has been associated with a higher incidence of Epstein-Barr virus (EBV)-related posttransplant lymphoproliferative disorders (PTLD), suggesting that recipient EBV-specific immune responses are suppressed by in vivo use of belatacept (11,50).
Alloanergization ex vivo prior to transplantation to generate T-cell populations tolerant to alloantigens is an alternate approach to control T-cell alloresponses. We have previously reported that alloanergization via ex vivo allostimulation with concurrent blockade of CD28-mediated costimulation using first-generation CTLA4-Ig or a combination of anti-B7.1 and anti-B7.2 monoclonal antibodies (12,16,44) reduced the alloreactivity of human leukocyte antigen (HLA)-mismatched donor lymphocytes in the setting of HLA-mismatched AHSCT and resulted in control of alloresponses in vivo (7,10,17).
We have recently extended previous findings demonstrating that human and murine lymphocytes acquired suppressive capacity after alloanergization (21,45), showing that expansion of recipient-specific allosuppressive CD4+ regulatory T cells (Tregs) occurs in vivo after infusion of alloanergized donor T cells in the setting of AHSCT (8). Potentially, similarly expanded populations of donor-specific recipient CD4+ Tregs could provide control of alloresponses after cellular or solid organ transplantation, reducing the need for pharmacologic immunosuppression. In a murine model, infusion of T cells alloanergized to donor antigens ex vivo by allostimulation and costimulatory blockade prevented allograft rejection, demonstrating the clinical potential of the strategy to induce tolerance after solid organ transplantation (30).
We therefore hypothesized that alloanergization of recipient lymphocytes to donor alloantigens using belatacept ex vivo could be used to specifically reduce anti-donor alloresponses and generate allospecific Treg cells and that subsequent re-exposure of these alloanergized recipient cells to donor alloantigens would result in further expansion of Treg cells able to actively suppress recipient donor-specific alloresponses. Applied clinically, this strategy might reduce the need for in vivo administration of nonspecific and toxic immunosuppressive drugs after allograft transplantation. To test this hypothesis, we used an HLA-mismatched in vitro model to compare the efficiency of belatacept and abatacept as costimulatory blockade reagents to alloanergize human peripheral blood mononuclear cells (PBMCs). We went on to determine the specificity of belataceptmediated alloanergization and the subsequent generation and specificity of Tregs after re-exposure of belatacept-alloanergized cells to alloantigens.
Materials and Methods
Alloanergization of PBMCs
PBMCs were isolated from leukocyte filters from volunteer blood donors by Ficoll-Paque density-gradient centrifugation (Amersham Biosciences, Piscataway, NJ) on an institutional review board-approved protocol. Responder PBMCs (107) were alloanergized by coculture at 106/ml in complete media [RPMI, penicillin/streptomycin, 10% human AB serum, Sigma-Aldrich (SA), St. Louis, MO] in upright 25 cm2 flasks (Corning, NY) for 72 h with equal numbers of γ-irradiated (35 gray) unrelated PBMCs (first-party stimulators) in the presence of abatacept (Bristol-Myers Squibb, New York, NY, supplied by Henry Schein, Inc., Melville, NY), belatacept (a gift from Bristol-Myers Squibb, New York, NY), or humanized anti-B7.1 and anti-B7.2 monoclonal antibodies (clones h1F1 and h3D1, Wyeth, Madison, NJ) (10). Control cocultures (“allostimulated PBMCs”) were responders and irradiated stimulators without costimulatory blockade. After incubation, cocultures were washed twice to remove costimulatory blockade reagents prior to allorestimulation.
Assessment of Efficiency of Costimulatory Blockade and Alloanergization
The efficiency of costimulatory blockade (CSB) was determined by measurement of proliferation (thymidine incorporaton) in primary mixed lymphocyte reactions (MLRs), and the efficiency of alloanergization was determined by measuring residual proliferative alloresponses in secondary MLRs, as previously described (9). The percentage inhibition (PI) of alloresponses in the primary MLR was calculated as follows:
PI of alloresponses in the secondary MLR was calculated as follows (cpm, counts per minute):
Washed alloanergized PBMCs were allorestimulated with equal numbers of irradiated stimulator PBMCs for 4–5 days (without costimulatory blockade) at 106 cells/ml in upright 25-cm2 flasks.
Quantification of CD4+ Tregs
PBMCs were stained in FACS staining buffer with CD4-fluorescein isothiocyanate (FITC), CD25-allophycocyanin (APC), and CD127-phycoerythrin (PE) (all Becton Dickinson, Franklin Lakes NJ) for 20 min at 4°C, washed, and resuspended. Where appropriate, cells were fixed, permeabilized, blocked with 2% rat serum, and stained for forkhead box P3 (FOXP3)-peridinin chlorophyll protein/ Cy5.5 (PerCP-CyC5.5) (PCH101 clone, Ebioscience, San Diego, CA) as described (8). Single-color-stained cells or Compbeads (Becton Dickinson) were compensation controls. Isotype controls were used wherever possible. Events were acquired on a FACSCalibur flow cytometer (Becton Dickinson) and analyzed using FlowJo 7.0.5 software.
Immunomagnetic Cell Sorting
For purification of CD4+ Tregs, PBMCs were labeled with biotin-conjugated anti-CD8, CD19, CD123, and CD127 antibodies and with antibiotin microbeads and passed through an LD column on a QuadraMACS magnetic device (Miltenyi, Bergisch Gladbach, Germany) resulting in a CD4+ CD127lo cell fraction with a median purity above 95%. This fraction was labeled with anti-CD25 (clone 3G10) directly conjugated microbeads, passed through two sequential MS columns on an Octo-MACS, and the positive fraction eluted as described (13). The CD4+ Treg fraction contained a median 85% purity of CD4+CD25hiCD127lo cells.
Treg Suppression Assays
Purified CD4+ Tregs were added to 5×104 untreated autologous PBMCs in triplicate wells of 96-well plates. 5×104 γ-irradiated first-party stimulator PBMCs (or 0.1 ml EBV-infected human cell lysate, Microbix, Ontario, Canada) were added. Proliferation was measured after 5 days by 3H-thymidine incorporation. Percentage suppression (% sup) was calculated as
EBV-Specific Immune Responses
PBMCs (106) from HLA-A2+ donors (identified by surface expression of HLA-A2 FITC-conjugated antibody, BB7.2, Becton Dickinson) were stained for 15 min with PE-conjugated HLA-A0201-EBV BMLF-1 (GLCTLVAML) pentamer (Proimmune, Oxford), CD11a FITC, and CD8-PE-CY5 (Beckman Coulter). Propidium iodide (Becton Dickinson) was added prior to acquiring a minimum of 5×104 viable CD8+ events on a Cytomics FC500 flow cytometer (Beckman Coulter). HLA-A2neg donors were used to assess nonspecific pentamer binding. For intracellular cytokine flow cytometry, 106 PBMCs from EBV-reactive HLA A2+ healthy donors were stimulated with 1 μg of BMLF-1 peptide GLCTLVAML (GLC) or latent membrane protein (LMP) 2 CLGLLTMV (CLG) peptide (Proimmune) or 1 ml of EBV-infected cell lysate (P3H3, Microbix) for 12 h with 10 ml of CD28 (Beckman Coulter) and CD107a-PE-Cy5 antibodies (Becton Dickinson). Brefeldin A (5 μg; Sigma-Aldrich) was added after 4 h. Positive controls were stimulated with 5 μg staphylococcal enterotoxin B (Sigma-Aldrich). Combined staining for intracellular interferon-γ (IFN-γ) FITC and surface CD107a (both Becton Dickinson) was performed as previously described (10). Stimulus- specific responses were calculated by subtracting values for unstimulated PBMCs.
Transwell Plate Assays
Plasmacytoid dendritic cells (pDCs) were isolated from healthy volunteer donor PBMCs with CD304 microbeads (Miltenyi) as previously described (18). Median frequencies of lineageneg/HLA-DR+/CD123+/CD11cneg pDCs pre- and postsort were 0.3% and 85%, respectively. pDCs (7.5×104) in 200 ml complete media were incubated for 12 h in 96-well plates with/without 0.1 ml EBV lysate. After incubation, supernatant was centrifuged and loaded in triplicate into bottom wells of 96-well ChemoTx transwell polycarbonate microplates with 5-μm filter pores (Neuroprobe, Inc., Gaithersburg, MD, USA). Freshly isolated PBMCs (20,000) from the same healthy donor in 30 ml complete media were loaded into the upper wells, and microplates were incubated for 4 h. Positive and negative controls were stromal-derived factor (SDF, 0.3 ng/μl in complete media) and complete media alone in lower wells. PBMC migration was assessed by using phase contrast microscopy to count viable PBMCs in lower wells.
Statistics
Statistical analysis was performed with GraphPad Prism Version 5. Two-tailed t tests were performed throughout. Unpaired t tests did not assume equal variance, and Welch's correction was applied where appropriate. Multiple groups were compared using repeated measures ANOVA and Bonferroni's posttest correction. A value of p < 0.05 was used to reject the null hypothesis.
Results
Alloresponses of Human PBMCs Are Efficiently Reduced After Alloanergization with Belatacept
We and others have previously shown that blockade of CD28-mediated costimulation using first-generation CTLA4-Ig or anti-B7.1/2 monoclonal antibodies during alloantigen stimulation induces alloantigen-specific hyporesponsiveness in human alloreactive donor T cells (10,12,16,44). We therefore characterized the efficiency of alloanergization of human PBMCs using belatacept, abatacept, or individual anti-B7.1/2 monoclonal antibodies for costimulatory blockade during the alloanergization process.
We first compared the effect of addition of belatacept, abatacept, or individual anti-B7 antibodies to HLA-mismatched MLRs on primary alloresponses. All three costimulatory blockade reagents significantly reduced primary alloproliferative responses in stimulator–responder pairs tested (Fig. 1A). At a concentration of 10 μg/106 cells, belatacept reduced primary alloproliferative responses more effectively than abatacept, although this did not reach statistical significance (median 76% versus 66% inhibition). At a higher concentration (40 μg/106 cells) belatacept and abatacept inhibited primary alloproliferative responses to a similar degree, not significantly different to that seen with individual anti-B7.1 and -B7.2 antibodies (median 80%, 79%, and 86% inhibition, respectively) (Fig. 1B).

Primary alloresponses of human PBMCs in the absence or presence of belatacept, abatacept, or anti-B7 antibodies. Proliferation (measured by 3H-thymidine incorporation) at day +5 in human leukocyte antigen (HLA)-mismatched primary mixed lymphocyte reactions (MLRs) using human peripheral blood mononuclear cells (PBMCs) is shown in the absence (filled circles) or presence of belatacept, abatacept, or anti-B7 antibodies as indicated. (A) Results for seven different unrelated stimulator–responder pairs are shown. The addition of all costimulatory blockade reagents to the MLRs consistently and significantly reduced primary alloresponses in all pairs tested. Horizontal bars and adjacent numbers are median counts per minute (cpm). (B) Concentrations of abatacept, belatacept and anti-B7.1 antibodies expressed as μg/106 responder cells in the MLRs. The percent inhibition of primary alloresponses by addition of belatacept, abatacept, or anti-B7 antibodies to MLRs for the same seven different unrelated stimulator–responder pairs is also shown. The addition of belatacept at 10 μg/106 cells inhibited primary alloresponses significantly more than a similar concentration of abatacept. Horizontal bars and adjacent numbers represent median percent inhibition of proliferation at day +5. **p < 0.01, ***p < 0.001. NS, not significant (p > 0.05). Values of p are for repeated measures ANOVA with Bonferroni's posttest correction.
We next determined the efficiency of alloanergization using belatacept, abatacept, or individual anti-B7 antibodies by measuring residual first-party-specific alloresponses in alloanergized PBMCs. After alloanergization, cells were washed twice to remove costimulatory blockade reagents prior to restimulation with the same (first-party) allostimulators. The efficiency of alloanergization was higher with belatacept than abatacept at either lower or higher concentrations (10 or 40 μg/106 cells), with median inhibition of secondary alloresponses of 52% versus 31% (p < 0.10) and 89% versus 50% (p < 0.05). Optimal efficiency of alloanergization was seen with belatacept at 40 μg/106 cells and not significantly different to that observed with individual anti-B7 antibodies at 10 μg/106 cells (median 89% versus 96% inhibition, whereas efficiency of alloanergization with abatacept at 40 μg/106 cells remained inferior to individual anti-B7 antibodies at 10 μg/106 cells (50% vs. 96%, p < 0.01) (Fig. 2A, B).

Alloanergization of PBMCs with belatacept efficiently reduces subsequent alloresponses. (A) Secondary alloresponses (proliferation measured by 3H-thymidine incorporation at day +6 of restimulation with first-party allostimulators) are shown for washed PBMCs after alloanergization with belatacept, abatacept, or individual anti-B7 antibodies. Results are for the same seven unrelated stimulator–responder pairs shown in Figure 1. Horizontal bars and adjacent numbers are median counts per minute (cpm). Concentrations of abatacept, belatacept, and anti-B7.1 antibodies expressed as μg/106 responder cells in the MLRs. (B) The efficiency of alloanergization, expressed as percent inhibition of subsequent first-party alloresponses, is also shown. Alloanergization with belatacept at either 10 or 40 μg/106 cells resulted in better inhibition of secondary alloresponses than abatacept similar concentrations. Optimal inhibition of secondary alloresponses was seen with 40 μg/106 cells of belatacept, equivalent to that seen with an optimal concentration (10 μg/106 cells) of individual anti-B7 antibodies. Horizontal bars and adjacent numbers represent median percent inhibition of proliferation at day +6. insert: *p < 0.05, **p < 0.01, ***p < 0.001; NS, not significant; Trend, 0.05 < p < 0.10. Values of p are for repeated measures ANOVA with Bonferroni's posttest correction.
Alloanergization with Belatacept Specifically Reduces Alloresponses of Human PBMCs
As we observed maximal inhibition of residual alloresponses when belatacept was used at 40 μg/106 cells during the alloanergization process, all further experiments used this concentration. In order to determine the specificity of alloanergization using belatacept, we first confirmed the efficiency of belatacept at reducing first-party residual alloresponses in a larger number of stimulator–responder pairs (Fig. 3A, B). We then measured residual proliferative responses after third-party alloantigen or mitogen stimulation. Notably, alloanergization using belatacept did not significantly reduce subsequent proliferative responses to third-party allostimulators (Fig. 3C, D) or to mitogenic CD3 and CD28 antibodies (Fig. 3E, F) indicating that no functionally significant amount of belatacept remained after washing of alloanergized PBMCs and that the induced hyporesponsiveness was specific to first-party alloantigens used during the ex vivo alloanergization process.

Third-party alloresponses and mitogen-specific proliferation are retained after alloanergization of PBMCs with belatacept. The efficiency of alloanergization using belatacept was confirmed in a larger cohort of HLA-mismatched PBMC stimulator–responder pairs. Secondary proliferative alloresponses are shown after first-party allorestimulation of washed PBMCs that had been alloanergized with belatacept at 40 μg/106 cells. (A) Results for 22 different unrelated stimulator–responder pairs are depicted. (B) The efficiency of alloanergization, expressed as percent inhibition of subsequent first-party alloresponses, was 90%. (C–F) In contrast, proliferative alloresponses after third-party allorestimulation or stimulation with mitogenic CD3 and CD28 antibodies of washed PBMCs that had been alloanergized with belatacept at 40 μg/106 cells were not significantly reduced. Horizontal bars and adjacent numbers are median values, and p values are for paired two-tailed t tests. ns, not significant.
Repeat Exposure of Belatacept-Alloanergized PBMCs to Alloantigen Results in Expansion of Allosuppressive CD4+ Tregs
We have previously shown that expansion of allosuppressive CD4+ Tregs both in vitro and in vivo occurs when alloanergized PBMCs are re-exposed to alloantigen in the absence of costimulatory blockade (8). We therefore determined whether this phenomenon also occurred after allorestimulation of belatacept-alloanergized PBMCs. Ex vivo alloanergization using belatacept resulted in a modest increase in the proportion of CD4+ cells with a CD25+CD127lo Treg phenotype (29). In contrast, allorestimulation of washed alloanergized PBMCs resulted in a more substantial increase in the proportion of CD4+ cells with a Treg phenotype to levels up to five times greater than those seen in untreated PBMCs (Fig. 4A, B). Most of these CD4+CD25+CD127lo Tregs were also positive for FOXP3 (Fig. 4C).

Alloanergization of human PBMCs with belatacept results in the expansion of CD4+ Tregs. Alloanergization of human PBMCs by allostimulation and costimulatory blockade with belatacept resulted in a twofold increase in the proportion of CD4+ T cells with a CD25+CD127lo Treg phenotype. A further two-to threefold increase was seen upon allorestimulation of alloanergized PBMCs. Flow cytometric dot plots are shown gated on viable CD4+ cells from a single representative experiment. (A) Boxed regions depict CD25+CD127lo cells, and adjacent numbers are CD25+CD127lo cells expressed as a percentage of CD4+ cells. (B) Aggregate results using PBMC from 15 different unrelated stimulator–responder pairs are depicted. Horizontal lines are medians, boxes are 25th and 75th percentiles, and whiskers are maximum and minimum values. Values of p are for paired t tests. (C) Flow cytometry was used to determine intranuclear expression of forkhead box P3 (FOXP3) and surface expression of CD25 and CD127 in CD4+ cells within PBMCs. Approximately 2% of CD4+ cells in untreated PBMCs (top) possessed a CD25+CD127lo surface expression phenotype (left) and over 80% of these cells expressed high levels of intranuclear FOXP3 (right). In contrast, less that 0.1% of CD25negCD127pos cells expressed FOXP3 (middle). Expanded populations of CD4+ cells within allorestimulated alloanergized PBMCs (bottom) with the CD25+CD127lo surface expression phenotype (left) maintained high levels of intranuclear FOXP3 expression (right) whereas very low proportions of CD25negCD127pos cells expressed FOXP3 (middle). One representative experiment (of 6) is shown. Plots are gated on CD4+ events.
We next investigated the capacity of these CD4+ Tregs to suppress proliferative alloresponses of autologous PBMCs. CD4+ Tregs purified from untreated PBMCs inhibited alloproliferative responses of untreated autologous PBMCs only at high Treg/PBMC ratios, consistent with a polyclonal suppressive response. In contrast, CD4+ Tregs from allorestimulated alloanergized PBMCs suppressed alloresponses at lower Treg/PBMC ratios (Fig. 5), consistent with activation or enrichment of Treg populations possessing allospecificity.

Allorestimulation of belatacept-alloanergized PBMCs results in expansion of CD4+ Treg with increased allosuppressive capacity. The percentage of suppression of proliferation (measured by 3H-thymidine incorporation) of first-party PBMC-stimulated autologous responder PBMCs by CD4+ Tregs isolated from untreated alloanergized and allorestimulated alloanergized PBMCs is shown. Symbols denote the mean percent suppression of proliferation. Bars denote standard deviation. Aggregated data from nine experiments using PBMCs from different stimulator–responder pairs are shown. Values of p are for unpaired t tests comparing percent suppression with Tregs from untreated and alloanergized PBMC/ Tregs from untreated and allorestimulated alloanergized PBMCs. *p < 0.05, **p<0.01; ns, not significant.
EBV-Specific T Cells Remain Functional After Alloanergization with Belatacept
For alloanergization to be a useful strategy to specifically reduce alloresponses, the retention of potentially beneficial immunity is critical. Therefore, to further characterize the specificity of reduction of alloresponses after alloanergization with belatacept, we chose to investigate whether functional pathogen-specific immunity was retained in alloanergized cells. We chose to investigate the retention of EBV-specific immune responses as these are of great importance after solid organ transplantation. We first quantified EBV-specific CD8+ T cells with HLA A2-pentamers containing GLC peptide from the lytic BMLF-1 antigen (47). Belatacept-alloanergization of PBMCs did not significantly reduce the frequency of viable CD8+ GLC-pentamer-specific cells (Fig. 6A, B).

PBMCs alloanergized with belatacept retain functional EBV-specific CD8+ T cells. Frequencies of Epstein-Barr virus (EBV)-specific T cells within PBMC populations were measured by flow cytometry in untreated PBMCs, allostimulated PBMCs, and PBMCs alloanergized with belatacept. Flow cytometric dot plots are shown depicting frequencies of CD8+ T cells binding HLA class I-pentamers loaded with the HLA A2-restricted GLCTLVAML (GLC) peptide derived from the lytic BMLF-1 EBV antigen in one representative experiment. Plots are gated on viable cells (identified by propidium iodide exclusion). (A) Boxed regions represent CD8+ pentamer+ cells, and numbers are frequencies of such cells expressed as percentage of CD8+ cells. (B) Frequencies of CD8+ GLC-peptide pentamer+ cells in untreated, allostimulated, and alloanergized PBMCs from four different HLA A2+ donors are shown. Horizontal bars and adjacent numbers are median values, and p values are for paired two-tailed t tests. Combined intracellular cytokine cytometry for interferon-γ (IFN-γ) secretion and surface CD107a evolution after stimulation with GLC-peptide was used to measure the frequency of functional EBV-specific CD8+ T cells. (C) Flow cytometric dot plots are shown from one representative experiment. Plots are gated on CD3+CD8+ cells. Boxed regions represent IFN-γ+ CD107a + cells, and numbers are frequencies expressed as percentage of CD8+ cells. (D) Frequencies of functional CD8+ GLC-peptide-specific cells in untreated, allostimulated, and alloanergized PBMCs from three different HLA A2+ donors are shown. Intracellular cytokine cytometry for IFN-γ secretion after stimulation with CLG-peptide from the latent LMP2 EBV antigen was also used to measure the frequency of functional EBV-specific CD8+ T cells. (E) Flow cytometric dot plots are shown from one representative experiment. Plots are gated on CD3+CD8+ cells. (F) Boxed regions represent IFN-γ+ cells, and numbers are frequencies expressed as percentage of CD8+ cells. Frequencies of functional CD8+ CLG-peptide-specific cells in untreated, allostimulated, and alloanergized PBMCs from two different HLA A2+ donors are shown. Horizontal lines represent medians.
We next determined whether belatacept alloanergization affected the frequencies of functional EBV-specific T cells. EBV-specific CD8+ T cells accumulating intracellular IFN-γ and upregulating surface CD107a (a surrogate marker of degranulation) after stimulation with GLC peptide were retained after alloanergization (Fig. 6C, D). EBV-specific CD8+ T cells accumulating intracellular IFN-γ after stimulation with CLG peptide from the EBV LMP2 latent antigen were also retained after alloanergization (Fig. 6E, F) Similarly, functional EBV-specific CD4+ T cells accumulating intracellular IFN-γ after stimulation with EBV lysate were also detectable at similar frequencies to that of untreated PBMCs after alloanergization (Fig. 7).

(A) Retention of EBV-specific CD4+ T-cell responses after alloanergization with belatacept. Intracellular cytokine cytometry for IFN-γ was also used to identify EBV-specific CD4+ cells after stimulation with EBV-infected cell lysate, and dot plots are shown from one representative experiment. (B) Boxed regions represent IFN-γ+ CD4+ cells, and numbers are frequencies expressed as percentage of CD4+ cells. Frequencies of functional CD4+ EBV-specific cells in untreated, allostimulated, and alloanergized PBMCs from three different donors are shown. Horizontal lines represent medians, and p values are for paired t tests throughout. Ns, not significant.
PBMCs Maintained Capacity to Migrate in Response to EBV-Loaded pDCs After Alloanergization with Belatacept
EBV-specific CD8+ cells expressed high levels of CD11a consistent with a functional effector phenotype, and the level of expression of CD11a was maintained after alloanergization (Fig. 8A, B). However, effective memory T-cell responses also require T cells to migrate in response to soluble factors secreted by antigen presenting cells (31). Therefore, we performed transwell plate assays to determine if alloanergization affected cellular migration in response to chemotactic factors secreted by EBV-antigen-loaded pDCs. Importantly, there was no reduction in the numbers of PBMCs migrating in response to supernatant from EBV-loaded pDCs after alloanergization (Fig. 8C).
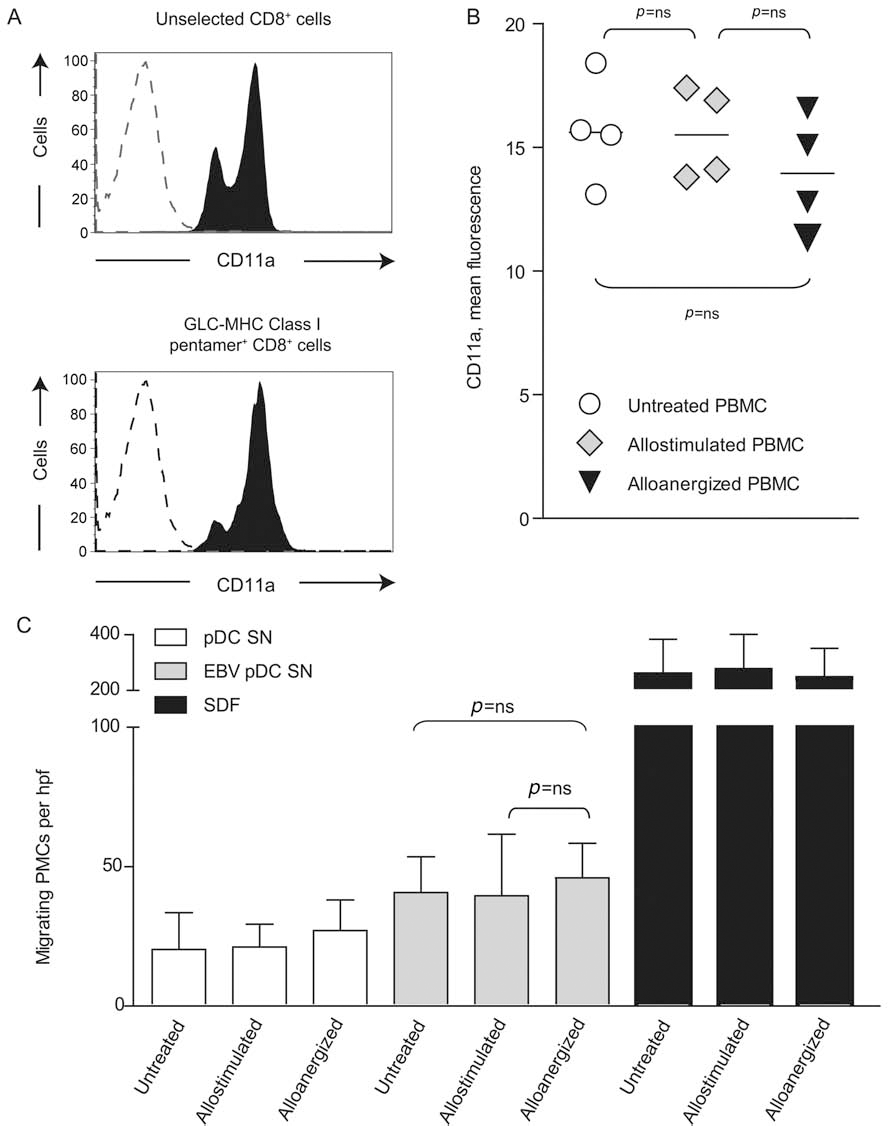
Alloanergization using belatacept did not impair expression of adhesion molecules on EBV-specific T cells or migration of PBMCs in response to EBV-loaded antigen-presenting cells. (A) Histograms depicting expression of the adhesion molecule CD11a on viable unselected (upper panel) and GLC-major histocompatibility complex (MHC) class I peptide pentamer+ CD8+ T cells (bottom) from untreated PBMCs are shown. Dotted lines represent isotype controls. GLC-peptide pentamer+ cells were almost exclusively CD11ahi. (B) There was no significant reduction in CD11a expression on GLC-peptide pentamer+ CD8+ T cells upon alloanergization of responder PBMCs in four different stimulator–responder pairs. Horizontal lines are medians, and p values are for paired t tests. Migration of untreated, allostimulated, and alloanergized PBMCs in response to soluble chemotactic factors secreted by EBV-loaded autologous plasmascytoid dendritic cells (pDCs) was assessed in transwell assays. (C) Mean (±SD) number of migrating PBMCs in response to supernatant from purified pDCs (either unstimulated or EBV-loaded) or stromal derived-factor 1 (SDF-1, positive control). Results are aggregated from three different experiments using different stimulator–responder pairs. hpf, high-power field; ns, not significant. Values of p are for paired t tests.
CD4+ Tregs Expanded After Allorestimulation of Belatacept-Alloanergized PBMCs Did Not Suppress EBV-Specific Responses
Given our finding that allorestimulation of belatacept-alloanergized PBMCs resulted in expansion of allosuppressive CD4+ Tregs, we additionally sought to determine whether these expanded populations of Tregs could suppress potentially beneficial EBV-specific T-cell responses. We compared the capacity of CD4+ Tregs purified from untreated, alloanergized, and allorestimulated alloanergized PBMCs to suppress EBV-specific proliferative responses of untreated autologous PBMCs. Tregs from untreated PBMCs suppressed EBV-specific proliferation at both high and low Treg/PBMC ratios, whereas Treg from alloanergized PBMCs were able to suppress EBV responses only at high ratios. In contrast, Tregs from allorestimulated alloanergized PBMCs did not suppress EBV responses even at high ratios (Fig. 9).

CD4+ Tregs generated by alloanergization did not suppress EBV-specific proliferative responses. The ability of CD4+ Treg purified from untreated, alloanergized, and allorestimulated alloanergized PBMCs to suppress EBV-specific proliferation of untreated autologous responder PBMCs was determined. Symbols denote the mean percent suppression of proliferation, and bars denote standard deviation. Aggregated data from three experiments using PBMCs from different stimulator–responder pairs are shown. Values of p are for unpaired t tests comparing percent suppression with Tregs from untreated and alloanergized PBMC/ Tregs from untreated and allorestimulated alloanergized PBMC. *p < 0.05, **p < 0.01; t, trend 0.05 < p < 0.10; ns, not significant.
Discussion
We have demonstrated that belatacept can be used ex vivo to alloanergize human PBMCs to HLA-mismatched alloantigens with high efficiency and specificity, without impairing potentially beneficial EBV-specific T-cell responses. Furthermore, subsequent re-exposure of alloanergized cells to alloantigen in the absence of belatacept results in expansion of allosuppressive Tregs. Thus, belatacept could be used ex vivo to generate a recipient cell population for reinfusion before, during, or after solid organ transplantation that has the capacity to selectively reduce donor-specific alloreactivity without the off-target effects associated with nonspecific immunosuppression.
Our previous studies have shown that the use of first-generation CTLA4-Ig to block costimulation during ex vivo alloanergization of human PBMCs was associated with lower efficiency of alloanergization (as determined by reduction in subsequent alloresponses) when compared to the use of the combination of anti-B7.1 and -B7.2 monoclonal antibodies during the alloanergization process (16). We now report that the use of the second-generation CTLA4-Ig belatacept was more efficacious in producing alloanergy in PBMCs in vitro than the first-generation CTLA4-Ig abatacept and had similar efficiency to that observed with the use of individual humanized anti-B7.1 and -B7.2 monoclonal antibodies. The superior inhibition of primary alloresponses we observed with belatacept at a concentration of 10 μg/106 cells compared to that seen with an equivalent concentration of abatacept suggests improved blockade of B7.1- and B7.2-mediated costimulation by belatacept during the alloanergization process and is consistent with other reports that belatacept has increased binding avidity in vitro for B7.1 and B7.2 than that possessed by first- generation CTLA4-Ig (15,25).
B7.1 and B7.2 are ligands for the T-cell counter-receptor CD28 and also the inhibitory counter-receptor CTLA4 (15). One concern with strategies that block B7.1- and B7.2-mediated signaling to human T cells is that the inhibitory signal mediated by CTLA4 may also be blocked, resulting in increased activation and proliferation of T cells. However, our data demonstrate that primary alloproliferation of PBMC when B7-mediated signaling is blocked with belatacept (or anti-B7.1 and -B7.2 monoclonal antibodies) is markedly reduced, indicating that the net result of blockade is removal of the positive signal mediated by CD28. The constitutive expression of CD28 on almost all human CD4+ T cells and most CD8+ T cells, in comparison to the induction of surface CTLA4 only upon T-cell activation (40), may underlie this observation.
B cells also mount alloresponses after solid organ transplantation, via alloantibody production, that can cause early acute humoral rejection and late transplant glomerulopathy (43). In our previous haploidentical AHSCT clinical studies, administration of ex vivo alloanergized donor cells produced using first-generation CTLA4-Ig or anti-B7 monoclonal antibodies did not result in any evidence of abnormal humoral immunity, and timely recovery of all immunoglobulin classes was observed (7). However, we have not yet directly determined the effects of alloanergization on secretion of immunoglobulins by alloreactive B cells, a question that will likely be definitively answered only by analysis of additional human clinical experiments.
Animal models have shown that infusion of activated CD4+ Tregs can prevent GvHD (20,46) and that rejection of skin grafts is associated with differential expression of genes involved in regulatory function in CD4+ T cells (52). Some studies have shown that higher FOXP3 mRNA levels or numbers of CD4+ Tregs in vivo may be associated with lower risk of harmful alloreactivity after both AHSCT and solid organ transplantation (34,36,42). Belatacept used at pharmacological doses in vivo after solid organ transplantation does not appear to result in expansion of CD4+ Tregs in the peripheral blood of patients (2,4). The absence of expanded populations of peripheral blood CD4+ Treg in these studies is consistent with ongoing belataceptmediated blockade of CD28 signaling, which is required for peripheral homeostasis and stimulates expansion of CD4+ Tregs (19). In contrast to protocols employing in vivo use of belatacept, as in this report, belatacept was removed after alloanergization and was not present during allorestimulation. Therefore, CD28 signaling was likely intact during allorestimulation after which we observed the expansion of alloantigen-specific CD4+ Tregs. These findings are of particular relevance to potential application of this strategy to solid organ transplantation. In the setting of AHSCT, all donor immune cells can be alloanergized ex vivo prior to reinfusion, whereas it is not feasible to anergize more than a fraction of the PBMCs of solid organ transplantation recipients. However, the significant expansion of allospecific Treg upon restimulation provides a mechanism by which manipulation of a relatively conservative number of cells ex vivo could be amplified in vivo to reach the desired clinical effect.
One concern relevant to all strategies to reduce unwanted immune responses is suppression of desirable immune repertoire. The impact of immunosuppressive strategies on EBV-specific immunity is of particular importance after solid organ transplantation as EBV-PTLD is more common in recipients with impaired EBV-specific T-cell immunity (32). Importantly, we demonstrate that T-cell responses specific to both latent and lytic EBV antigens are retained after ex vivo alloanergization. These findings are significant as EBV-PTLD after both solid organ transplantation and AHSCT is most commonly associated with type III latency, with expression of a wide variety of latent antigens (14), and EBV-PTLD in solid organ transplantation recipients is frequently donor-derived, stimulating a primary recipient T-cell immune response directed against lytic antigens (6). Furthermore, we demonstrate EBV-specific CD8+ T cells retained high expression of CD11a, and PBMCs retained functional capacity to migrate in response to EBV-loaded pDCs after alloanergization with belatacept. This phenotype is associated with protection against EBV-PTLD in vivo (23,24).
In order to apply this strategy effectively to control alloresponses after solid organ transplantation, practical issues relating to the generation of alloanergized PBMCs and the timing of their administration remain. Alloresponses directed against transplanted solid organs might be effectively reduced by alloanergization using irradiated PBMCs from the donor as stimulator cells. Such stimulators would be readily available from living donors prior to transplantation, thus permitting generation of alloanergized recipient cells prior to organ transplantation. In contrast, stimulator cells would only be available from cadaveric donors in the immediate peritransplant period, and thus, alloanergized recipient cells would not be available for infusion until after organ transplantation. However, the expansion of alloantigen-specific Tregs we observed after allorestimulation of alloanergized PBMCs suggests that delayed administration of alloanergized cells might nonetheless have potential to modulate anti-donor alloresponses.
In summary, we describe the successful use of the new agent belatacept to block CD28-mediated costimulatory signals during ex vivo allostimulation in order to induce alloantigen-specific hyporesponsiveness in human PBMCs. Re-exposure of alloanergized PBMCs to alloantigen in the absence of belatacept led to the expansion of a CD4+ Treg population with increased capacity to suppress specific alloresponses. Importantly, alloanergization did not reduce a broad range of potentially beneficial EBV-specific T-cell responses. Thus, this ex vivo cellular manipulation, which is simple and easily adopted, provides a platform to generate an immunoregulatory cell product with potential application to cellular or solid organ transplantation.
Footnotes
Acknowledgments
The authors would like to thank Bristol Myers Squibb for providing belatacept and Lisa L. Brennan BSN for her assistance in undertaking this work. This work was supported by NIH R21 CA137645 and U19 CA100265 with additional support from the Frank J. Hanna Jr., Carlson Family, and Pan Mass Challenge-Shea Family Funds. J.K.D. was a recipient of a New Investigator award from the American Society for Blood and Marrow Transplantation and a Clinician Scientist award from the UK Medical Research Council. Authorship was determined in accordance with according to the ICMJE criteria (http://www.icmje.org/ethical_1author.html). J.K.D. and C.M.B. contributed equally to the work, conceiving, designing and performing experiments, analyzing data, and writing the manuscript. A.V. performed experiments. L.M.N. provided critical reagents and commented critically on the manuscript. E.C.G. conceived and designed experiments and wrote the manuscript. The authors declare no conflicts of interest.



