Abstract
Mesenchymal stem cells (MSCs) exhibit immunosuppressive capabilities, which have evoked interest in their application as cell therapy in transplant patients. So far it has been unclear whether allogeneic MSCs and host regulatory T-cells (Tregs) functionally influence each other. We investigated the interaction between both cell types using perirenal adipose tissue-derived MSCs (ASCs) from kidney donors and Tregs from blood bank donors or kidney recipients 6 months after transplantation. The immunomodulatory capacity of ASCs was not prejudiced by both Tregs from healthy donors and Tregs from graft recipients, indicating that ASCs were not targeted by the inhibitory effects of Tregs and vice versa. In addition, Tregs supported ASC function, as they did not alter the secretion of IFN-γ by immune cells and hence contributed to ASC activation and efficiency. ASCs exerted their suppressive role by expressing IDO, reducing levels of TNF-α, and by inducing the production of IL-10 in effector cells and Tregs. In conclusion, this study presents evidence that donor ASCs and acceptor Tregs do not impair each other's function and therefore encourages the use of MSC therapy for the prevention of graft rejection in solid organ transplantation.
Introduction
Increasing investigational efforts are undertaken to employ cell therapy for the advancement of graft survival in the field of transplantation. Mesenchymal stem cells (MSCs) are explantable from various sites including adipose tissue, bone marrow, and amniotic fluid (2, 25, 46, 59) and easily expandable. MSCs have the capacity to differentiate into multiple cell lineages such as osteoblasts, adipocytes, chondrocytes, and myocytes (14, 46). It has been demonstrated that MSCs mediate the repair of bone and cartilage (1, 28, 49, 58), the improvement of cardiac muscle and kidney function (11, 29, 33, 35), and the restoration of lung function (30). The main characteristic of MSCs relevant to transplantation is their potent in vitro immunosuppressive effect on immune cell activation and proliferation (17, 34); MSCs exhibit this feature upon stimulation by inflammatory conditions (12). Soluble factors associated with the inhibitory effect of MSCs are transforming growth factor-β (TGF-β), hepatocyte growth factor (HGF), interleukin-10 (IL-10), prostaglandins, human leukocyte antigen G5 (HLA-G5), and nitric oxide (NO) (6, 16, 17, 21, 26, 42, 51). In addition, MSCs create a local immunosuppressive microenvironment by production of indoleamine 2,3-dioxygenase (IDO), leading to tryptophan catabolism. Utilizing these mechanisms, MSCs promote heart and skin allograft survival in animal models (5, 48). Further, there is clinical evidence that MSCs alleviate graft-versus-host disease (GVHD) after hematopoietic stem cell transplantation (36–37). Currently ongoing clinical studies aim to confirm the feasibility and safety of MSC therapy in solid organ transplantation (27, 45).
Regulatory T-cells (Tregs), defined by the expression of cluster of differentiation 4 (CD4), CD25, and forkhead box P3 (FOXP3) and the lack of CD127, possess immunomodulatory capacities. The influential role of Tregs on maintaining tolerance was observed in patients suffering from immunodysregulation polyendocrinopathy enteropathy X-linked syndrome (IPEX); these patients lack functioning Tregs due to a point mutation in the Foxp3 gene (4, 7, 32). Tregs have been reported to control tissue injury and allo-reactivity (8, 23, 31, 41, 57) by functional inhibition of multiple cell types including CD4+ helper T-cells, CD8+ cytotoxic T-cells, and antigen presenting cells (55). In clinical studies, ex vivo expanded Tregs diminished the symptoms and reduced the incidence of GVHD (9, 56). Main factors involved in Treg function are the anti-inflammatory cytokines IL-10 and TGF-β (20, 22, 54). In the transplantation setting, some immunosuppressants were identified to be permissive for Treg function including corticosteroids, anti-CD3 antibodies, anti-thymocyte globulin (ATG), anti-CD52 antibodies, and mammalian target of rapamycin (mTOR) inhibitors (39), whereas others, such as the calcineurin inhibitors cyclosporine A and tacrolimus, were found to negatively influence the development and the immunosuppressive activity of Tregs (15). With MSCs emerging as a prospective therapy to prevent graft rejection after transplantation due to their potent inhibitory influence on effector T-cells, the effect of MSCs on host Tregs remains to be investigated. Conversely, it is still unknown whether Tregs, as a consequence of their immunosuppressive capacities, inhibit the activation of MSCs and consequently MSCs' repressive features; similar effects have been reported for other immunosuppressive agents such as rapamycin, tacrolimus or cyclosporine A (18, 24).
The present study investigated the impact of interaction between kidney donor perirenal adipose tissue-derived MSCs (ASCs) and Tregs, derived from either transplant recipients or healthy donors, on the suppression of allo-reactivity.
Materials and Methods
Origin of Adipose Tissue
Perirenal adipose tissue was surgically removed from living kidney donors (n = 17; mean age 51.5 years, range 38.1–63.2 years; 10 female, 7 male) and collected in minimum essential medium Eagle a modification (MEM-α) (Sigma-Aldrich, St. Louis, MO, USA) supplemented with 2 mM l-glutamine (Lonza, Verviers, Belgium), 1% penicillin/streptomycin solution (P/S; 100 IU/ml penicillin, 100 IU/ml streptomycin; Lonza). Samples were obtained with written informed consent as approved by the medical ethical committee of the Erasmus University Medical Center Rotterdam (protocol no. MEC-2006-190).
Isolation and Culture of Human ASCs From Perirenal Adipose Tissue
Perirenal adipose tissue was mechanically disrupted with a scalpel. The tissue was washed twice with 1x Dulbecco's phosphate-buffered saline (PBS; PAA Laboratories, Pasching, Austria). Subsequently, the tissue was enzymatically digested with sterile 0.5 mg/ml collagenase type IV (Life Technologies, Paisley, UK) in Roswell Park Memorial Institute (RPMI) 1,640 medium with GlutaMAX™-I (Life Technologies) and 1% P/S for 30 min at 37°C. ASC culture medium consisting of MEM-α with 2 mM l-glutamine, 1% P/S, and 15% fetal bovine serum (FBS; Lonza) was added, and the digested tissue was centrifuged at 1,200 × g for 10 min. The cell pellet was resuspended in ASC culture medium and filtered through a 70-μm cell strainer (BD Biosciences, San Jose, CA, USA). Finally, cells were transferred into a 175-cm2 cell culture flask (Greiner Bio-One, Alphen a/d Rijn, the Netherlands) and expanded in a humidified atmosphere with 5% CO2 at 37°C. Nonadherent cells were removed after 3–4 days. Culture medium was refreshed twice weekly. Cells were removed from culture flasks at subconfluency using 0.05% trypsin-EDTA (Life Technologies) and reseeded at 1,000 cells/cm2 to ensure optimal proliferation. ASCs were used for experiments between passages 2 and 6. ASCs from these passages did not differ in their ability to differentiate or to exert their immunosuppressive functions.
Immunophenotyping of ASCs
Adherent ASCs were removed from culture flasks following incubation with 0.05% trypsin-EDTA at 37°C and washed twice with FACSFlow (BD Biosciences). Cells were incubated with monoclonal antibodies (mAb) against CD14-phycoerythrin (PE), HLA-ABC-PE (all Serotec, Düsseldorf, Germany), CD90-allophycocyanin (APC), CD105-fluorescein isothiocyanate (FITC; all R&D Systems, Abingdon, UK), CD34-APC, CD45-FITC, CD166-PE, and HLA-DR-APC-cyanine 7 (Cy7) (all BD Biosciences) at room temperature and protected from light for 30 min. Subsequently, samples were washed twice with FACSFlow, and immunophenotypical analyses of ACSs were performed using the BD FACSCanto II flow cytometer, BD FACSDiva software (BD Biosciences), and FlowJo software (Tree Star, Ashland, OR, USA).
Differentiation Assays
Adipogenic Differentiation
Adipogenic differentiation was induced by culturing confluent ASCs in MEM-α supplemented with 2 mM l-glutamine, 1% P/S, 15% heat-inactivated FBS (FBS-HI), 50 μg/ml l-ascorbic acid, 500 μM 3-isobutyl-1-methylxanthine (IBMX; Sigma-Aldrich), 60 μM indomethacin (Sigma-Aldrich), and 10 nM dexamethasone (Sigma-Aldrich) for 14 days in a humidified atmosphere with 5% CO2 at 37°C. Lipid-filled vesicles were detected by Oil Red O staining. Cells were washed with PBS, fixed with 60% isopropanol for 1 min, and incubated with filtered 0.3% Oil Red O (Sigma-Aldrich; in 60% isopropanol) for 30 min. Following three washes with PBS, the cells were photographed.
Osteogenic Differentiation
Osteogenic differentiation was induced by culturing confluent ASCs in MEM-α supplemented with 2 mM l-glutamine, 1% P/S, 15% FBS-HI, 5 mM β-glycerophosphate (Sigma-Aldrich), 50 μg/ml l-ascorbic acid (Sigma-Aldrich), and 10 nM dexamethasone for 21 days in a humidified atmosphere with 5% CO2 at 37°C. The deposition of calcified nodules was identified using von Kossa staining. Cells were washed with PBS and fixed with cold 4% paraformaldehyde for 5 min. Following an additional wash step with PBS and two wash steps with distilled water, cells were incubated with 1% silver nitrate (in water) on a light box until blackening occurred. Cells were washed three times with water, incubated with 2.5% sodium thiosulfate (in water) for 5 min, washed again twice with water, and photographed.
Isolation of Peripheral Blood Mononuclear Cells
Buffy coats from healthy blood donors were obtained from Sanquin Blood Bank, Rotterdam, the Netherlands. Peripheral blood samples from living kidney donors were collected prior to the donation procedure and from kidney recipients 6 months after transplantation. Kidney transplant patients received a triple immunosuppressive therapy consisting of tacrolimus (Astellas Pharma Europe B.V., Leiderdorp, the Netherlands), mycophenolate mofetil (MMF; Novartis Pharma B.V., Arnhem, the Netherlands), and prednisolone (Sigma-Aldrich). Peripheral blood mononuclear cells (PBMCs) were isolated from buffy coats or heparinized peripheral blood samples by density gradient centrifugation using Ficoll-Paque PLUS (density 1.077 g/ml; GE Healthcare, Uppsala, Sweden). Cells were washed with RPMI 1640 supplemented with 1% P/S and frozen in RPMI 1640 + GlutaMAX-I with 1% P/S, 10% human serum (Sanquin), and 10% dimethyl sulfoxide (DMSO; Merck, Hohenbrunn, Germany). PBMCs were stored at −150°C until further use.
Isolation of Tregs and Effector Cells From PBMCs
PBMCs were thawed and washed twice with RPMI 1640 supplemented with 20% FBS-HI, 1% P/S, and 0.01 mg/ml DNase I (Roche Diagnostics, Mannheim, Germany). PBMCs were resuspended in PBMC culture medium (PCM) consisting of MEM-α supplemented with 2 mM l-glutamine, 1% P/S, and 10% heat-inactivated human serum. The CD25bright cells (Tregs) were isolated by means of CD25 MicroBeads II (Miltenyi Biotec, Bergisch Gladbach, Germany). Cells and beads were incubated for 15 min at 4°C in autoMACS rinsing solution (Miltenyi Biotec) supplemented with 0.5% bovine serum albumin (BSA; Sigma-Aldrich). Cells were centrifuged for 10 min at 2,000 rpm, resuspended in buffer, and subjected to a positive selection (Posseld program) on the autoMACS Pro Separator (Miltenyi Biotec). The residual fraction (CD25-/dim cells; effector cell fraction) and the Tregs were washed and resuspended in PCM for functional assays. Cell fraction purity was determined by flow cytometry using mAb against CD3-AmCyan, CD4-Pacific Blue, CD25-PE-Cy7 (epitope B), CD127-PE (all BD Biosciences), and FOXP3-APC (eBioscience, San Diego, CA, USA). The characterization of both fractions from a representative separation example is depicted in Figure 1. Intracellular FOXP3 staining was carried out according to the manufacturer's instructions using the anti-human FOXP3 staining set APC (eBioscience). Flow cytometric analyses were performed using the BD FACSCanto II flow cytometer and BD FACSDiva software.

Characterization of Tregs and CD25-/dim effector cells. The CD25bright cells (Tregs) and CD25-/dim effector cells were isolated from peripheral blood mononuclear cells (PBMCs) by incubation with CD25 MicroBeads II followed by a positive selection. Both cell fractions were characterized using flow cytometry. (A) CD25bright fraction: The CD25bright cells are gated within the CD3+CD4+ cell population. The percentage CD127-FOXP3+ cells within the CD4+CD25bright T-cells are shown. (B) CD25-/dim fraction: The CD25-/dim cells are gated within the CD3+CD4+ cell population. The percentage CD127+FOXP3- cells within the CD4+CD25-/dim T-cells are shown. FOXP3, forkhead box P3.
Mixed Lymphocyte Reaction and Suppression Assay
Mixed lymphocyte reactions (MLRs) consisted of 5×104 CD25-/dim effector cells (residual fraction from Treg isolation) stimulated with 5 × 104 γ-irradiated (40 Gy) allogeneic PBMCs in PCM in round-bottomed 96-well plates (Nunc, Roskilde, Denmark). Effector–stimulator cell combinations were chosen based on a minimum of four HLA mismatches. In suppression assays, the immunomodulatory capacities of various concentrations of ASCs and Tregs on the MLRs were determined. Following an 8-h incubation period, [3H]thymidine incorporation (0.25 μCi/well; PerkinElmer, Groningen, the Netherlands) was measured on day 7 using the Wallac 1450 MicroBeta TriLux Liquid Scintillation Counter and Luminometer (PerkinElmer). Proliferation capacity of CD25-/dim effector cells was determined by stimulating 5 × 104 cells with 1 μg/ml phytohemagglutinin-L (PHA; Roche Diagnostics, Mannheim, Germany). After 3 days, proliferation was measured by [3H]thymidine incorporation. When MLRs were performed in microtiter plates with different well sizes, the number of cells was adjusted accordingly. When applicable, 50 μl cell culture supernatant was harvested prior to the addition of [3H]thymidine and frozen until further use at −80°C.
PKH labeling and Flow Cytometric Analysis
In order to study the effects of ASCs and Tregs on the proliferative capacities of various lymphocyte subsets, CD25-/dim effector cells were labeled using the PKH67 Green Fluorescent Cell Linker Kit (Sigma-Aldrich). PKH67 dilution due to proliferation was measured by flow cytometry. Tregs and allogeneic stimulator PBMCs were discriminated from the effector cell population by labeling with PKH26 (PKH26 Red Fluorescent Cell Linker Kit, Sigma-Aldrich) or PKH26/67 double-labeling, respectively. Labeling was performed prior to the MLR set-up and according to the manufacturer's instructions. After 7 days, cells were harvested and washed twice with FACSFlow. Cells were stained with mAb against CD3-AmCyan, CD4-peridinin chlorophyll protein (PerCP), CD8-APC-Cy7, CD25-PE-Cy7 (all BD Biosciences), and CD45RO-Pacific Blue (BioLegend, San Diego, CA, USA). Following a 30-min incubation period at room temperature and protected from light, cells were washed twice and subjected to flow cytometric analysis using the BD FACSCanto II flow cytometer and BD FACSDiva software.
mRNA Expression Analysis
ASCs were cultured alone or cocultured with MLRs (1:5, ASCs/effector cells) for 7 days. In cocultures, MLRs were separated from ASCs by cell culture inserts with permeable membrane supports (0.4-μm pore size; Greiner Bio-One). ASCs were harvested and washed twice with PBS-diethylpyrocarbonate (DEPC; Sigma-Aldrich). Cells were either handled immediately or snap-frozen in liquid nitrogen and stored at −80°C. Total RNA was purified using the High Pure RNA Isolation Kit (Roche Diagnostics) according to the manufacturer's instructions. Complementary DNA (cDNA) was synthesized by reverse transcription using random primers. Quantitative real-time PCR (qPCR) was performed using 500 ng cDNA, the StepOnePlus™ Real-Time PCR System, TaqMan Universal PCR Master Mix, and the assay-on-demand primer/probes for indoleamine 2,3-dioxygenase (INDO; Hs00158027.m1), interferon-γ (IFN-γ; Hs00174143.m1), interleukin-6 (IL-6; Hs00174131.m1), IL-10 (Hs0017 4086.m1), transforming growth factor-β (TGF-β; Hs 00171257.m1), and tumor necrosis factor-α (TNF-α; Hs99999043.m1) (all Applied Biosystems, Foster City, CA, USA). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA served as endogenous control for normalization (Hs99999905.m1; Applied Biosystems). Intra- and interassay variations were determined by negative controls and positive reference samples. Changes in expression of the target genes relative to the GAPDH gene were quantified using the comparative CT method (38). Fold changes of less than 3 were considered insignificant.
Cytometric Bead Array (CBA) and ELISA
Cell culture supernatants were obtained from suppression assays using Tregs (1:10; Tregs/effector cells), ASCs (1:40; ASCs/effector cells), or Tregs and ASCs in combination after a 7-day incubation period. Secreted IL-10, IFN-γ, and TNF-α were measured in cell culture supernatants by means of the BD Cytometric Bead Array Human Th1/Th2/Th17 Cytokine Kit (BD Biosciences). Secreted IL-6 was determined by ELISA (U-CyTech biosciences, Utrecht, the Netherlands). Both methods were carried out according to the manufacturer's instructions.
Statistical Analysis
Statistical analyses were performed by means of one-way analysis of variance (ANOVA), Bonferroni multiple comparison test, and (un)paired t tests using GraphPad Prism 5 software (GraphPad Software, San Diego, CA, USA). A value of p < 0.05 was considered statistically significant. One-tailed p values are stated.
Results
Characterization of ASCs
ASCs were isolated from perirenal adipose tissue of living kidney donors. The cells adhered to the plastic surface of culture flasks and had a spindle-shaped morphology. Immunophenotyping of ASCs by flow cytometry demonstrated that the cells expressed low levels of CD34 and the cell surface markers CD90, CD105, CD166, and HLA-ABC. ASCs did not express CD14, the leucocyte marker CD45, or HLA-DR (Fig. 2A). The culture of undifferentiated ASCs under adipogenic conditions for 14 days induced the development of lipid-filled vesicles. These vesicles, characteristic for cells of adipogenic lineage, were detected by Oil Red O staining (Fig. 2B). Osteogenic differentiation of ASCs for 21 days resulted in the deposition of calcified nodules, which were blackened by von Kossa staining (Fig. 2B). The ability of the isolated cells to induce these morphological changes confirmed that they were ASCs.

Characterization of adipose tissue derived mesenchymal stem cells (ASCs). (A) Immunophenotyping of ASCs for the cell surface proteins CD14, CD34, CD45, CD90, CD105, CD166, HLA-ABC, and human leukocyte antigen (HLA)-DR. Open histograms show nonspecific staining for the indicated marker; solid histograms show specific staining for indicated marker. (B) Microscopic images of undifferentiated ASCs, and ASCs differentiated into adipocytes and osteocytes. Adipogenic differentiation (14 days) induced the development of lipid-filled vesicles; these were detected by Oil Red O staining. Osteogenic differentiation (21 days) induced the deposition of calcified nodules that were blackened by von Kossa staining. Scale bars: 200 μm.
ASCs and Tregs Dose-Dependently Inhibit the Proliferation of CD25–/dim Effector Cells
The effect of ASCs and Tregs on the proliferation of CD25-/dim effector cells was examined by means of [3H]thymidine incorporation. Stimulation of CD25-/dim effector cells with γ-irradiated allogeneic PBMCs for 7 days resulted in a strong proliferative activity of these cells. Coculture with third-party ASCs (Fig. 3A) or autologous Tregs (Fig. 3B) suppressed the effector cell proliferation in a dose-dependent manner (both p < 0.0001). ASCs significantly reduced the proliferation from a concentration of 1:160 (ASCs/effector cells). Tregs significantly suppressed the proliferative capacity of CD25-/dim effector cells from a concentration of 1:10 (Tregs/effector cells).

Effect of ASCs or Tregs on the proliferation of allo-activated CD25-/dim lymphocytes. Mixed lymphocyte reactions (MLRs) (white bars) consisted of CD25-/dim effector cells stimulated with γ-irradiated allogeneic PBMCs (both derived from healthy individuals) with a minimum of four HLA mismatches. MLRs were performed in the presence of various concentrations of ASCs (A; n = 6, mean ± SEM, one-way ANOVA: p < 0.0001) or Tregs (B; n = 5, mean ± SEM, one-way ANOVA: p < 0.0001) for 7 days. Effector cell proliferation was determined by [3H]thymidine incorporation. Bonferroni multiple comparison test was used to compare conditions with MLRs. *p < 0.05, **p < 0.01, ***p < 0.001.
ASCs and Tregs Do Not Impair Each Other's Immunosuppressive Functions
The influence of ASCs and Tregs on each other's T-cell-suppressive capacities was analyzed by measurement of [3H]thymidine incorporation. At first, the effect of Tregs on the suppressive capacity of various ASCs concentrations was examined. The addition of Tregs 1:10 (Tregs/effector cells) permitted the immunomodulatory function of ASCs (Fig. 4A). In the reverse set-up, it was analyzed whether ASCs (1:40; ASCs/effector cells) had an effect on the inhibition of responder cell proliferation by diverse Treg concentrations; ASCs did not hamper Treg function (Fig. 4B).

Effect of ASCs and Tregs on the proliferation of allo-activated CD25-/dim lymphocytes. MLRs (white bar) consisted of CD25-/dim effector cells stimulated with γ-irradiated allogeneic PBMCs (both derived from healthy individuals). Effector cell proliferation was determined by [3H]thymidine incorporation. (A) Varying ASC concentrations were added to MLRs in the absence (black bars; n = 6; mean ± SEM) and in the presence of a constant Treg concentration (1:10; hatched bars; n = 5; mean ± SEM). (B) Various Treg concentrations were added to MLRs in the absence (gray bars; n = 5; mean ± SEM) and in the presence of a constant ASC concentration (1:40; hatched bars; n = 3; mean ± SEM). Paired t test, *p < 0.05, **p < 0.01.
ASCs and Tregs From Kidney Donor–Recipient Pairs Have Immunosuppressive Capacities
In order to examine ASC–Treg interaction in a transplantation setting, it was investigated whether ASCs derived from kidney donors and Tregs obtained from the respective graft recipient 6 months posttransplantation inhibited recipient effector cells in a similar way as observed for healthy controls. The effect of both cell types was examined by means of MLRs and [3H]thymidine incorporation. ASCs (1:40, ASCs/effector cells) and Tregs (1:10, Tregs/effector cells) inhibited the proliferation of allo-activated CD25-/dim effector cells by 42% and 48%, respectively (Fig. 5). The combination of ASCs and Tregs did not negatively influence the T-cell suppressive capacity of each cell type. Therefore, ASC–Treg interaction in a transplantation setting is comparable to a setting using healthy controls.

Effect of ASCs or Tregs on the proliferation of allo-activated CD25-/dim lymphocytes using cells from kidney donor–recipient pairs. MLRs (white bar) consisted of recipient-derived CD25-/dim effector cells stimulated with γ-irradiated allogeneic PBMCs from the corresponding donor. ASCs were obtained from the kidney donor (n = 7; mean age 54.9 years, range 43–63.2 years; 5 male, 2 female). Tregs and CD25-/dim effector cells were isolated from PBMCs of the respective graft recipient (n = 7; mean age 51.7 years, range 18.6–75.7 years; 6 male, 1 female) at 6 months posttransplantation. Effector cell proliferation was determined by [3H]thymidine incorporation. The condition MLR was used as reference for statistical analyses. Mean ± SEM, paired t test, *p < 0.05.
ASCs and Tregs Inhibit the Proliferative Capacities of Various Lymphocyte Subsets
The effect of ASCs and Tregs on the proliferation of different lymphocyte subsets within the CD25-/dim effector cell population was investigated by PKH dilution assay. The results of a representative experiment are shown in Figure 6; a summary of a series of experiments is presented in Table 1. High proliferative activities (≥74%) (Table 1) were observed for total CD4+, total CD8+, CD4+ memory (CD45RO+), and CD8+ memory (CD45RO+) T-cells following stimulation with γ-irradiated allogeneic PBMCs for 7 days in the absence of ASCs and Tregs. Individually, ASCs (1:40, ASCs/effector cells) as well as Tregs (1:10, Tregs/effector cells) abrogated the proliferative capacity of all analyzed lymphocyte subsets. In addition, the presence of Tregs (1:10) to ASCs did not diminish the suppressive effects achieved by ASCs alone. Further, in comparison to the CD45RO+ subpopulations, the CD45RO- subsets were found to be more susceptible to the suppressive capacities of ASCs and Tregs.
Effect of Adipose Tissue-Derived Mesenchymal Stem Cells (ASCs) and Tregs on the Proliferation of Allo-Activated CD25-/dim Lymphocytes

Mean ± SEM.
n = 6.
n = 3.
p < 0.05 versus mixed lymphocyte reaction (MLR); paired t test.

Effect of ASCs or Tregs on the proliferative capacities of various lymphocyte subsets. MLRs consisted of CD25-/dim effector cells stimulated with γ-irradiated allogeneic PBMCs (both derived from healthy individuals). CD25-/dim effector cells were labeled with PKH67. PKH67 dilution due to proliferation was measured by flow cytometry on day 7. For discrimination purposes Tregs (1:10; when applicable) and allogeneic stimulator PBMCs were labeled with PKH26. Cell concentrations stated refer to the ratio of indicated cells to effector cells. The percentages indicate the fraction of proliferating cells of the respective lymphocyte subset. A representative experiment is shown. Data of multiple experiments are shown in Table 1.
Immunosuppressive ASCs Overexpress INDO
In order to examine which genes were involved in the immunosuppressive effect of ASCs, the gene expression profile of ASCs cultured in the absence or in the presence of MLRs was assessed by qPCR after 7 days. MLRs were separated from ASCs by cell culture inserts with permeable membrane supports. Changes in expression of the target genes relative to the GAPDH reference gene were quantified using the comparative CT method. When compared to untreated ASCs, cocultured ASCs expressed substantially more INDO (120,000-fold change) (Fig. 7), a gene coding for an enzyme involved in the inhibition of lymphocyte proliferation. Gene expression of the anti-inflammatory cytokine TGF-β was downregulated by 5.7-fold, whereas the expression of the proinflammatory cytokine IL-6 was upregulated by 15.5-fold. Fold changes of less than 3 were considered insignificant. Therefore, changes in gene expression as a result of ASC stimulation were not observed for the anti-inflammatory cytokine IL-10 nor for the proinflammatory cytokine TNF-α. IFN-γ expression by ASCs was not detectable.

Gene expression of ASCs cocultured with MLRs in comparison to ASCs cultured alone. ASCs were cultured alone or in coculture with MLRs for 7 days. MLRs were separated from ASCs by cell culture inserts with permeable membrane supports. Quantitative real-time PCR (qPCR) was performed using 500 ng cDNA. Changes in expression of the target genes relative to the glyceraldehyde 3-phosphate dehydrogenase (GAPDH) gene were quantified using the comparative CT method (n = 5; mean ± SEM). Fold changes in gene expression between ASCs and ASCs cocultured with MLRs are shown. *Significant fold changes (≥3). n.d., not detectable; IFN, interferon; TNF, tumor necrosis factor; IL, interleukin; TGF, transforming growth factor; INDO, indoleamine 2,3-dioxygenase.
ASCs Contribute to an Anti-inflammatory Microenvironment
The effect of ASCs and Tregs on the generation of an immunosuppressive microenvironment was examined using cytometric bead array technology. Concentrations of secreted cytokines were analyzed in cell culture supernatants after coculture of MLRs with ASCs (1:40, ASCs/effector cells) and/or Tregs (1:10, Tregs/effector cells). The proliferation data corresponding to the analyzed supernatants are shown in Figure 8A. IL-10 is not expressed by ASCs. However, ASCs mediated an elevated production of IL-10 by the effector cell population (31 pg/ml) (Fig. 8B). Although no increase in IL-10 secretion was observed when Tregs alone were added, in the presence of both Tregs and ASCs IL-10 levels increased by 41 pg/ml. The addition of Tregs, ASCs, or both cell types in combination to MLRs did not lead to significant changes of IFN-γ concentrations (Fig. 8C). Further, ASCs mediated a reduction of TNF-α secretion into the culture medium (Fig. 8D); Tregs did not have the capacity to decrease levels of TNF-α. In addition, unstimulated ASCs secreted IL-6. In the presence of ASCs and allo-activated effector cells, a strong increase in IL-6 production was observed when compared to the MLR condition (Fig. 8E). Tregs had no effect on the IL-6 production by the immune cells. However, Tregs influenced the amount of secreted IL-6 when ASCs were present; the IL-6 concentration was reduced by 135 ng/ml.
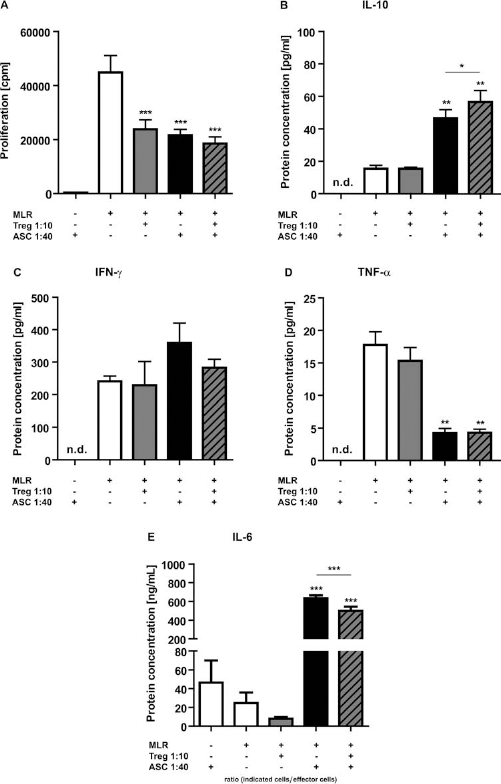
Determination of cytokine concentrations secreted into cell culture supernatants. (A) MLRs consisted of CD25-/dim effector cells stimulated with γ-irradiated allogeneic PBMCs (both derived from healthy individuals). Proliferative capacity of effector cells following the addition of various concentrations of ASCs and/or Tregs was analyzed by [3H]thymidine incorporation on day 7. The secretion of IL-10 (B), IFN-γ (C), and TNF-α (D) into the cell culture supernatant was determined by cytometric bead array (CBA). Concentrations of IL-6 (E) were determined by ELISA. Unless indicated differently, the MLR condition was used as reference for statistical analyses (n = 4; mean ± SEM). Paired t test, *p < 0.05, **p < 0.01, ***p < 0.001. n.d., not detectable.
Discussion
As cellular therapy presents itself as an emerging alternative to current drug-based immunosuppressive regimens in the field of transplantation, it becomes essential not only to investigate the potential of this new treatment to suppress effector cells but also to evaluate its influence on existing immunomodulatory mechanisms operating in the cell therapy recipient. Focusing on the interactions between donor and acceptor immunomodulatory cells following treatment, the present study provides evidence that kidney donor perirenal adipose tissue-derived MSCs (ASCs) and Tregs, originating from healthy donors, do not impair each other's suppressive effect on allo-reactivity. Further, this study confirmed earlier reports that Tregs isolated from kidney recipients 6 months after transplantation maintained their suppressive capacities despite being exposed to high-dose medication with strong effects on T-cell activation and proliferation (52). Importantly, these Tregs were found to permit the inhibitory effect of donor ASCs. Vice versa, ASCs did not abolish the suppressive effect of Tregs despite their potent inhibitory influence on effector T-cells. ASCs and Tregs inhibited the same lymphocyte subsets within the CD25-/dim effector cell population. However, in comparison to nonmemory cells, CD4+CD45RO+ and CD8+CD45RO+ memory cells were less efficiently suppressed by ASCs and Tregs, possibly due to the highly activated nature of these cells. The limited effectiveness of ASCs and Tregs in altering the proliferation of memory T-cells is not unique. Similar observations were made for other clinically available immunosuppressive agents (43). MSCs exert their immunosuppressive function on effector cells by secreting anti-inflammatory factors and inducing the reduction of proinflammatory factors. A possible explanation for the absence of repressive influences between ASCs and Tregs may be that both cell types exercise their immunosuppressive abilities by orchestrating non-interfering mechanisms. A mechanism employed by MSCs to create a local immunosuppressive microenvironment is tryptophan starvation. This process is mediated by INDO, a gene coding for the tryptophan catabolizing enzyme indoleamine 2,3-dioxygenase (IDO). We and other research groups observed that activated MSCs produce increased concentrations of IDO, which contribute to the inhibition of various effector cell populations (40, 53). Yet, we found that elevated levels of IDO did not have a negative impact on Treg function. Low levels of tryptophan and the resulting presence of kynurenine have also been reported to induce and amplify the suppressive functions of Tregs in the periphery (19). Furthermore, we found that ASCs exerted their inhibitory capacities by mediating the secretion of IL-10 by immune cells, an anti-inflammatory cytokine that MSCs do not produce themselves. IL-10 is also one of the main molecules implicated in the functional activity of Tregs (22). As Tregs not only react upon but also produce IL-10, we were surprised not to find increased levels of the anti-inflammatory cytokine in the supernatant during the Treg-mediated suppression of allo-activated effector cells. The lack of an IL-10 increase can be explained by autocrine IL-10 signaling on Tregs and therefore consumption of IL-10. However, when ASCs and Tregs in combination inhibited allo-reactivity, we found increased concentrations of IL-10 when compared to ASC-mediated suppression. This indicates that ASCs boost the IL-10 production of Tregs. In addition, we observed that not Tregs but only ASCs were able to reduce concentrations of the proinflammatory cytokine TNF-α. On the other hand, both cell types were not able to decrease IFN-γ concentrations. Partly, this is in contrast to the finding by Park et al. (44) that Tregs diminish the production of IFN-γ by CD4+ and CD8+ T-cells in an autoimmune setting. Although not expressed by MSCs, IFN-γ is required for their activation and therefore crucial for the immunosuppressive function of MSCs (34). Thus, the availability of IFN-γ in the presence of Tregs allows optimal functionality of MSCs. It has been shown that IFN-γ-stimulated MSCs prevent GVHD mortality in patients receiving bone marrow transplants (47). In contrast to the effect of ASCs on TNF-α and IFN-γ, we found that activated ASCs secreted levels of IL-6, demonstrating the duality of ASC immunomodulation. However, IL-6 has been reported to play a role in the induction of regulatory T-cells (13), suggesting that ASCs indirectly promote immunosuppression via this pathway. In addition, MSCs also employ nonsoluble factors to achieve their immunosuppressive effect. Upon activation, MSCs show increased expression of the ligand of the costimulatory molecule programmed death-1 (PD-L1) and the intercellular adhesion molecule-1 (ICAM-1) (3, 50). ICAM-1 facilitates the binding of lymphocytes to MSCs and thus promotes MSC-mediated immunosuppression. Binding of PD-L1 to its receptor PD-1 results in the inhibition of IL-2 production leading to a reduction of target cell proliferation (10). In summary, MSCs and Tregs appear to utilize both distinct and partially overlapping mechanisms to alter immune responses.
The present study demonstrates that donor ASCs and recipient Tregs do not impair each other's function. This finding clearly encourages the use of MSC therapy for the prevention of graft rejection in solid organ transplantation. MSCs, in contrast to some currently applied immunosuppressive interventions, preserve the function of Tregs and therefore potentially mediate the establishment of an advanced immunosuppressive environment in transplant patients without inflicting the known side effects observed for T-cell inhibitory drugs.
Footnotes
Acknowledgments
The authors thank Dr. Nicolle H. R. Litjens for support with flow cytometric analysis. The authors declare no conflict of interest.
