Abstract
Although islet transplantation can achieve insulin independence in patients with type 1 diabetes, sufficient number of islets derived from two or more donors is usually required to achieve normoglycemia. Activated neutrophils and neutrophil elastase (NE), which is released from these neutrophils, can directly cause injury in islet grafts. We hypothesized that inhibition of NE improves islet isolation and islet allograft survival. We tested our hypothesis by examining the effects of modified ET-Kyoto solution supplemented with sivelestat, a NE inhibitor (S-Kyoto solution), on islet yield and viability in islet isolation and the effect of intraperitoneally injected sivelestat on islet graft survival in a mouse allotransplant model. NE and proinflammatory cytokines such as tumor necrosis factor (TNF)-α and interleukin (IL)-6 increased markedly at the end of warm digestion during islet isolation and exhibited direct cytotoxic activity against the islets causing their apoptosis. The use of S-Kyoto solution significantly improved islet yield and viability. Furthermore, treatment with sivelestat resulted in significant prolongation of islet allograft survival in recipient mice. Furthermore, serum levels of IL-6 and TNF-α at 1 and 2 weeks posttransplantation were significantly higher in islet recipients than before transplantation. Our results indicated that NE released from activated neutrophils negatively affects islet survival and that its suppression both in vitro and in vivo improved islet yield and prolonged islet graft survival. The results suggest that inhibition of NE activity could be potentially useful in islet transplantation for patients with type 1 diabetes mellitus.
Introduction
Since the reporting of the Edmonton protocol, islet transplantation has become one of the treatment options for patients with type 1 diabetes mellitus (8, 41, 43–48). Islet transplantation is a minimal invasive approach for β-cell replacement compared with pancreas transplantation (18, 44, 47). However, a sufficient number of islets derived from two or more donor pancreas are usually required to achieve insulin independence, since a substantial number of transplanted islets fail to engraft into the recipient liver for a variety of reasons such as apoptosis, inflammation, and ischemia (1, 3, 27, 29, 37, 41, 42, 47, 52, 57–59). Furthermore, research into islet transplantation has been hindered by the inability to isolate a sufficient number of islets from a single donor pancreas (4, 15, 16, 45, 47). Thus, there is a need for novel strategies that increase islet yield, maintain high islet quality, and protect transplanted islet grafts.
Indeed, the islet isolation procedure itself can lead to tissue destruction and activation of cellular and non-cellular components of the pancreas, including resident neutrophils, macrophages, and T cells, which probably play an important role in impairment of islet survival (1, 4, 31, 37, 42). In the present study, we focused on the role of neutrophils, in particular neutrophil elastase (NE), against islets during islet isolation. The NE is a 29-kDa (kilodalton) glycoprotein chymotrypsin-like serine protease stored in azurophil granules in its inactive form until it is released after neutrophil exposure to inflammatory stimuli (17, 49, 55). Once released, NE is fully active, and the excessive release of NE degrades elastin, collagens, laminins, and other extracellular matrix components, thereby leading to subsequent tissue damage through endothelial cell injury (12, 17, 49, 54, 55).
Sivelestat (ONO-5046) is a low molecular weight synthetic specific and competitive inhibitor of NE activity (12, 17, 21, 30, 49, 50, 54, 55, 60). This agent has been employed clinically in Japan and shown to attenuate acute lung injury associated with systemic inflammation response, which is sometimes seen after infection, surgical intervention, traumatic, or burn injury (11, 49, 50, 60). In addition, sivelestat exhibits potent cytoprotective properties in animal models of liver and lung transplantation (30, 54), hepatectomy (17, 21), and ischemia/reperfusion injury (17, 54, 55).
The objectives of the present study were to determine whether the addition of sivelestat to the islet isolation solution improves islet yield and viability. We also investigated the cytoprotective effects of sivelestat in islet recipients. The results suggested that NE inhibition using sivelestat is an attractive new therapeutic option in islet isolation and transplantation and could have a significant impact on patients with type 1 diabetes by allowing successful one donor to one recipient.
Materials and Methods
Drugs and Reagents
Sivelestat (ONO-5046) is a newly synthesized agent known to selectively inhibit NE. Sivelestat was a generous gift from Ono Pharmaceutical Co., Ltd. (Osaka, Japan). A stock solution was prepared by dissolving 200 mg of sivelestat at room temperature in 20 ml of phosphate-buffered saline (PBS) with 24.5 mg of sodium carbonate and stored at 4°C until use.
Preservation Solutions
University of Wisconsin (UW, Bristol-Myers Squibb Company, Princeton, NJ) and extracellular-type trehalosecontaining Kyoto (ET-Kyoto; Otsuka Pharmaceutical, Tokyo, Japan) solution were prepared. Stock solutions of 20 μM sivelestat in UW and ET-Kyoto were prepared as S-UW, S-Kyoto, each. Sivelestat did not change the density of islets or other tissue components of the pancreas.
Mice
Male C57BL/6J mice and Balb c/A mice, 10–12 weeks old, weighting 20–30 g, were purchased from CLEA Japan, Inc. (Tokyo). All experiments were approved by the international animal care and use committee (IACUC) of Osaka University Medical School.
Islet Isolation and Assessment
Briefly, after clamping the distal common bile duct under anesthesia, the common bile duct was cannulated. Then the pancreatic tissue was distended by using 3 ml of isolation solution containing 1 mg/ml of collagenase VIII (Sigma-Aldrich; USA). The distended pancreas was excised and incubated in 37°C warm shaker for 15 min. The digested pancreas was washed with appropriate isolation solution three times by centrifugation (270 × g, 2 min, 4°C), then purified with a discontinuous density gradient (1.111, 1.104, 1.097, 1.072 g/ml) in isolation solution containing iodixianol (Optiplep®, Axis-Shild, Oslo, Norway). The purified islets were collected and cultured with Roswell Park Memorial Institute (RPMI) 1640 medium (Sigma-Aldrich) supplemented with 10% fetal bovine serum (FBS) (Sigma-Aldrich), 100 U/ml penicillin, 100 μg/ml streptomycin, and 0.1 mM nonessential amino acids (Invitrogen, Carlsbad, CA) under 5% CO2 atmosphere at 37°C (26).
To evaluate the isolated islets, islet count, islet equivalents (IEQ), distribution of islet size, and islet purity were determined as described previously (35, 40). Islet yield and distribution of islet size were determined by measuring islets after dithizone staining (Wako, Osaka) using VH analyzer (Keyence, Osaka). The purification recovery rate was defined as the percentage of IEQ recovered after purification compared to the IEQ before purification (34). Islet purity was assessed by four independent investigators.
In Vitro Cytotoxicity Assay
The cytotoxic activity of NE against isolated islets was assessed by the lactate dehydrogenase (LDH) assay kit (Roche Applied Science, Mannheim, Germany). Briefly, the harvested islets were plated at 30 islets/well in 96-well round plate. NE (Calbiochem, San Diego, CA) was then added to the wells at various concentrations, and the plates were incubated for 24 h at 37°C. Next, 100 μl of the culture supernatant was transferred into the wells of 96-well flat plate. The reaction mixture was added to each well, and then absorbance was measured at 490 nm. Moreover, to examine the cytoprotective effects of sivelestat on NE cytotoxicity, 2, 20, or 200 μM of sivelestat was simultaneously added to the NE-containing wells and the plates were incubated for 24 h at 37°C followed by measurement of absorbance at 490 nm.
Staining for Naphthol AS-D Chloroacetate Esterase
To assess the accumulation of activated neutrophils in the pancreas during islet isolation, the tissue was stained by naphthol AS-D chloroacetate esterase (Sigma-Aldrich) as described previously (14, 17). Briefly, the pancreas specimens obtained before and at the end of warm digestion were fixed in 10% formalin and embedded in paraffin. Tissue sections (2-μm thick) were stained with naphthol AS-D chloroacetate esterase. In addition, the specimens were counterstained with hematoxylin. Activated neutrophils were positively stained red-brown and counted under the microscope at a magnification of 100x.
Neutrophil Elastase Activity Assay
NE enzyme activity was measured in the supernatant at each step of islet isolation, including before warm digestion, at the end of warm digestion, and after purification, using the method described previously (11, 12, 17, 55, 59). For this purpose a 20-μl sample was incubated with 1 mM of N-methoxysuccinyl-Ala-Ala-Pro-Val-p-nitroanilide (p-NA) (Sigma-Aldrich), which is a highly specific synthetic substrate for NE, in 0.1 M Tris–HCl buffer (pH 8.0) containing 0.5 M NaCl at 37°C for 24 h. The incubated samples were plated onto a 96-well plate, and then absorbance was measured at 405 nm to detect free p-NA.
Assessment of Apoptosis of Isolated Islets
Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining was performed to detect apoptotic cells during islet isolation, using Apop Tag® Peroxidase In Situ Apoptosis Detection Kit (Chemicon International, Temecula, CA) as described previously (53–55). The negative control was prepared by omission of the terminal transferase. Positive control was generated by treatment with deoxyribonuclease (DNase) I. Peroxidase activity was visualized with diaminobenzidine (DAB) substrate, which yielded a brown oxidation product, and 0.5% methyl green was used for counter-staining. TUNEL-positive cells were counted under the microscope at 400x magnification.
Scanning Electron Microscopy (SEM)
Morphological analysis of the isolated islets was carried out by SEM as described previously (31). The isolated islets were fixed with 2.5% PBS (0.1 M, pH 7.4) glutaraldehyde (Tokyo Chemical Industry, Tokyo) solution at 4°C for 24 h. After washing, they were postfixed with 1% OsO4/PBS for 2 h at 4°C. Subsequently, the fixed islets were incubated in 1% tannin acid solution at 4°C overnight and then dehydrated. They were transferred to isoamyl acetate and dried in a critical point dryer using liquid CO2. They were mounted on the stage and observed under a scanning electron microscope (S-800, Hitachi, Tokyo). The morphology was assessed by four independent investigators.
Islet Viability Assay
The viability of isolated islets was assessed by tetramethyl rhodamine ethyl ester (TMRE; Molecular Probes, Eugene, OR) assay and 7-aminoactinomycin D (7-AAD; Molecular Probes) assay, as described previously (9). TMRE is an indicator of the mitochondria membrane potential (MMP) and used as a marker for live cells, while 7-AAD is used as a marker for dead cells or apoptotic cells. Briefly, the isolated islets were cultured for 24 h and then incubated in 1 ml of TrypLE Express (Invitrogen) for 15 min at 37°C to prepare single islet cells and then dispersed. The single islet cells were incubated with 100 ng/ml of TMRE for 30 min at 4°C. Subsequently, the fluorescence intensity of TMRE was analyzed with a FACS Calibur flow cytometer (BD Immunocytometry, San Jose, CA). In a similar fashion, the isolated islets were dispersed into single cells and then incubated with 1 μg/ml of 7-AAD. Subsequently, the fluorescence intensity of 7-AAD was analyzed with a FACS Calibur flow cytometer.
To further determine the islets viability, the colorimetric methyl tetrazolium salt (MTS) assay was performed as described previously (56). The viability of freshly isolated islets or cultured islets over either 24 or 48 h was evaluated by monitoring metabolic activity with MTS assay using the cell Titer 96 Aqueous One reagent (Promega, Madison, WI). The colorimetric reagent was added to each well and incubated for 2 h, and the absorbance values were read at 490 nm.
Glucose-Stimulated Insulin Release In Vitro
To evaluate the in vitro insulin function of islets, static glucose change was measured as described previously (33, 35). Twenty islets were cultured overnight at 37°C and then preincubated in low-glucose culture medium (2.8 mM glucose) for 60 min at 37°C. After preincubation, the islets were incubated in low-glucose culture medium (2.8 mM glucose) at 37°C for 60 min. Subsequently, the supernatant of the culture medium was collected, and the islets were incubated in high-glucose culture medium (20 mM glucose) at 37°C for 60 min. Similarly, the supernatant was collected, and insulin concentration was measured by the mouse-insulin enzyme-linked immunosorbent assay (ELISA) kit (Mercodia, Uppsala, Sweden). Glucose-stimulated insulin concentration was expressed as the stimulation index (SI), calculated as the ratio of insulin released during exposure to high glucose over the insulin released during low-glucose incubation.
Islet Transplant Experiments
The recipient 10- to 12-week-old male Balb c/A mice were divided at random into two experimental groups (Fig. 6A) (n = 5/group) to receive allogeneic islets isolated from C57BL/6J mice by the use of either isolation solution (i.e., ET-Kyoto and S-Kyoto solutions). The recipient mice were rendered diabetic by a single injection of streptozotocin (STZ) (Nacalai tesque, Kyoto, Japan) at a dose of 180 mg/kg intraperitoneally. Hyperglycemia was defined as glucose level of >400 mg/dl measured twice consecutively after STZ injection. Then 500 of freshly isolated islets were transplanted under the kidney capsule. After transplantation, nonfasting blood glucose level was monitored using samples from tail blood by Glutest PRO (Sanwa kagaku, Nagoya, Japan). Normoglycemia after transplantation was defined as two consecutive blood glucose levels below 200 mg/dl. Islet rejection after transplantation was defined when two consecutive blood glucose levels exceeded 200 mg/dl. At 1 week posttransplantation, the engrafted kidneys were excised to assess the survival of islet grafts by immunohistochemistry. Moreover, the beneficial effects of monotherapy with intraperitoneal sivelestat in recipient mice were assessed. Sivelestat was administered at 100 mg/kg/day for 1 day before transplantation and every day until 14 days after transplantation, as described previously (32). The recipient Balb c/A mice treated with sivelestat were divided at random into two experimental groups (n = 5/group) to receive allogeneic islets isolated from C57BL/6J mice using ET-Kyoto or S-Kyoto solution.
Intraperitoneal Glucose Tolerance Test (IPGTT)
The IPGTT was performed at 1 week posttransplantation, using the method described previously (2, 9, 58). Mice (n = 3/group) were fasted overnight and then injected intraperitoneally (IP) with 2 g glucose in saline/kg body weight. Untreated diabetic mice and nondiabetic wild-type mice were transplanted with saline as a control. Blood glucose levels were measured before injection and at 15, 60, and 120 min after injection.
Immunohistochemical Analysis
Immunohistochemical analysis was performed using the method described previously (36). The engrafted kidneys were excised, fixed in formalin, and embedded in paraffin. Tissue sections (2-μm thick) were placed in 0.3% H2O2/methanol to quench endogenous peroxidase activity and incubated with 5% bovine serum albumin (BSA)-PBS to block nonspecific reaction. The slides were incubated with rabbit anti-insulin polyclonal antibody (pAb, Santa Cruz Biotechnology, CA) to detect the transplanted islets. The sections were incubated with horseradish peroxidase (HRP)-conjugated secondary antibody (Bethyl Laboratories, Inc., Montgomery, TX) and then immunostaining was visualized with 0.02% DAB (Sigma-Aldrich) as the chromogen. After washing, the sections were counterstained with hematoxylin. Control tissue sections were prepared in a similar fashion, except no primary antibody was used.
Measurement of Proinflammatory Cytokines
The supernatant was collected after each step of islet isolation, including before warm digestion, at the end of warm digestion and after purification. Moreover, serum samples were collected from islet recipients at day 1 before transplantation and days 4, 7, 14, 21, 28 after transplantation. These samples were frozen immediately at −80°C until analysis. Proinflammatory cytokine (IL-2, IL-4, IL-6, IL-10, IL-17A, IFN-γ, TNF-α) levels in these samples were measured using the BD™ Cytometric Bead Array Mouse Th1/Th2/Th17 CBA kit (BD Biosciences, San Jose, CA) and analyzed on a FACS Calibur Flow cytometer (BD Immunocytometry).
Statistical Analysis
Values were expressed as mean ± SD. Differences between groups were examined for statistical significance using the two-tailed unpaired Student's t test or one-way analysis of the variance (ANOVA) followed by Bonferroni's post hoc test when multiple comparisons were made. A value of p < 0.05 denoted the presence of significant difference.
Results
Direct Cytotoxicity of Neutrophil Elastase and Optimum Effective Concentration of Sivelestat
To assess the direct cytotoxicity of NE against isolated islets, LDH assay was performed at various concentrations of this enzyme. NE caused 43–48% killing at both 5 and 10 μg/ml. To prevent this killing, sivelestat was added to the culture medium at 2, 20, or 200 μM. The cytotoxicity induced by 10 μg/ml of NE could not be inhibited by sivelestat (Fig. 1A). However, the cytotoxicity induced by less than 5 μg/ml of NE was significantly abrogated by either 20 or 200 μM of sivelestat, but not by 2 μM of sivelestat (Fig. 1A).

In vitro cytotoxicity assay of neutrophil elastase against islets and optimum concentration of sivelestat in ET-Kyoto solution. (A) The cytotoxicity of neutrophil elastase against islets and the inhibitory effects of sivelestat were assessed by lactate dehydrogenase (LDH) assay. Data are mean ± SD of three independent experiments; *p < 0.05, **p < 0.01. (B) Islet yields under various concentrations of sivelestat. Data are mean ± SD of three independent experiments; *p < 0.05.
To elect the optimum concentration of sivelestat for islet isolation, islet isolation was performed by the addition of various concentrations of sivelestat to the isolation solution (Fig. 1B). Islet yields using a high concentration of sivelestat (200 μM or 2 mM) were significantly lower than that with 20 μM of sivelestat. In contrast, no significant improvement in islet yield was observed by 2 μM of sivelestat isolation compared with that of the control group (ET-Kyoto isolation). These findings correlated with those from LDH assay, and thus, the following experiments were performed using 20 μM of sivelestat.
Activation of Neutrophils in Pancreas During Collagenase Digestion
Naphthol AS-D chloroacetate esterase staining was performed to demonstrate whether resident neutrophils in the pancreas tissue are activated by collagenase digestion. Many neutrophils were activated (red-brown staining) by collagenase digestion in islet isolation using both UW and ET-Kyoto solutions (Fig. 2A), but their number (cells/field) was significantly reduced by the addition of 20 μM of sivelestat in ET-Kyoto solution (UW: 10.2 ± 1.1, S-UW: 6.8 ± 0.4, ET-Kyoto: 9.2 ± 1.3, S-Kyoto: 3.6 ± 0.5) (UW, ET-Kyoto vs. S-Kyoto: p < 0.001, S-UW vs. S-Kyoto: p < 0.01) (Fig. 2B). These results suggest a marked increase in enzyme activity by collagenase digestion in all types of isolation solutions (Fig. 2C). However, the enzyme activity was significantly suppressed in S-Kyoto islet isolation than those in other isolations (UW, S-UW, and ET-Kyoto isolation).
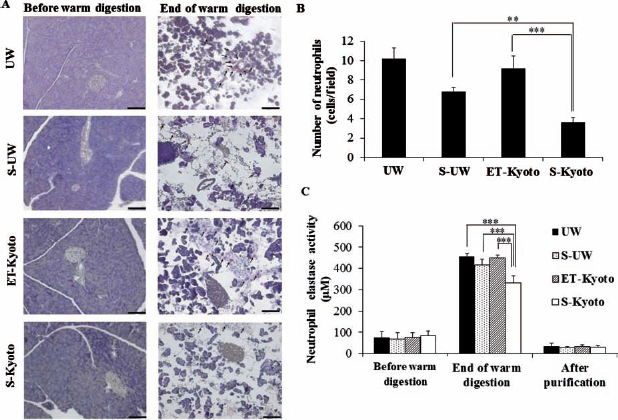
Accumulation of activated neutrophils in the pancreas and neutrophil elastase activity during islet isolation. (A) Representative histology of the pancreas before and at the end of warm digestion. Activated neutrophils are stained red-brown (black arrow) by naphthol AS-D chloroacetate esterase. Scale bars: 100 μm. (B) Number of activated neutrophils per field (original magnification: 100x) was counted under microscopy. Data are mean ± SD of five sections; **p < 0.01, ***p < 0.001. (C) Neutrophil elastase activity in the supernatant during islet isolation measured at various time points of islet isolation (before warm digestion, at the end of warm digestion, after purification). Data are mean ± SD of five independent samples; ***p < 0.001. UW, University of Wisconsin solution.
Sivelestat Suppresses Apoptosis in Islets After Warm Digestion During Islet Isolation
Many TUNEL-positive cells were identified in the islets at the end of warm digestion (Fig. 3A), whereas no such cells were detected before digestion (data not shown). Quantitative analysis indicated a significantly lower number of TUNEL-positive cells within the islets of the S-Kyoto group (6.6 ± 1.5) compared with those of UW, S-UW, and ET-Kyoto groups (23.1 ± 3.9, 20.8 ± 5.0, and 15.3 ± 2.6, respectively) (p < 0.001, each) (Fig. 3B).
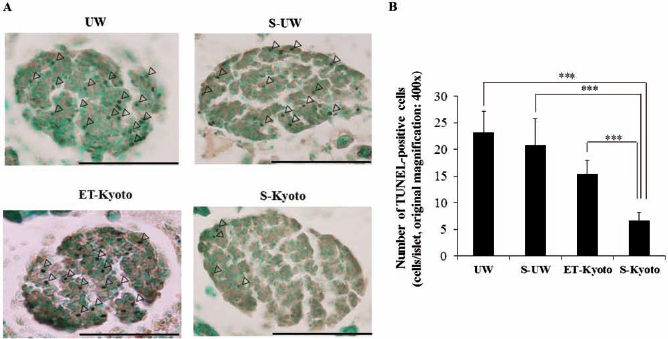
Assessment of apoptosis during islet isolation by TUNEL staining. (A) Representative histological sections of the islets at the end of warm digestion. Note the brown terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL)-stained apoptotic cells (arrowhead). Scale bars: 100 μm. (B) The density of TUNEL-positive cells (cells/islet) was determined under magnification of 400x. All experiments were done using each independent mouse. One mouse was used in each experiment (n = 1). Data are mean ± SD of five independent sections; ***p < 0.001.
Improvement of Islet Yield Elicited by Islet Isolation with S-Kyoto Solution
To investigate the beneficial effects of sivelestat in islet isolation, islet isolations were performed using either UW or ET-Kyoto solution, with or without 20 μM of sivelestat. Islet yield, isolation index, and islet purity after purification were significantly improved by S-Kyoto islet isolation (Table 1). The recovery rate of purification was also significantly improved by S-Kyoto islet isolation, compared to those by other isolation solutions (UW: 55.8 ± 10.4%, S-UW: 56.6 ± 12.7%, ET-Kyoto: 56.6 ± 4.6%, S-Kyoto: 76.0 ± 5.2%). Based on the results of UW isolation groups (UW, S-UW), no beneficial effects of sivelestat were elicited under these islet isolation parameters. With regard to islet size distribution, the percentage of large islets (>150 nm in diameter) in the S-Kyoto group was markedly higher than that in other isolation groups (Fig. 4A).

Results of islet isolation according to the type of isolation solution. (A) Size distribution for each diameter (50–150, 150–250, 250–350, 350– μm) of isolated islets assessed by using each isolation solution. All experiments were done using each independent mouse. One mouse was used in each experiment (n = 1). Data are the mean percentage from five independent experiments. (B) Representative microscopic images of the islets immediately after isolation. The morphology of isolated islets was observed by scanning electron microscopy. Scale bars: 100 μm.
Results of Islet Isolation According to the Isolation Solution

Data are expressed as mean ± SD of five independent experiments.
p < 0.05,
p < 0.01,
p < 0.001, compared with S-Kyoto isolation solution. Islet count and purity were measured by the VH analyzer software. Isolation index was calculated as the ratio of IEQ to islet count. The recovery rate of purification (%) = IEQ after purification / IEQ before purification x 100. IEQ, islet equivalents; UW, University of Wisconsin.
To further examine the morphological changes in isolated islets, SEM was employed. As shown in Figure 4B, islets obtained from the UW groups (UW, S-UW) were poor, based on the irregular islet surface and detection of islet damage during the isolation procedure. Similarly, islets obtained from ET-Kyoto isolation had irregular shape. On the other hand, islets isolated by S-Kyoto solution were well preserved, with round and smooth surface.
Improvement of Islet Viability Elicited by Islet Isolation with S-Kyoto Solution
Next, we evaluated islet viability using fluorescence labeling with TMRE and 7-AAD (Fig. 5). In the UW groups (UW, S-UW), no significant difference was noted in islet viability assay using TMRE and 7-AAD between with or without sivelestat [UW (n = 5) vs. S-UW (n = 5) (60.0 ± 5.6% vs. 63.3 ± 4.4%, not significant)]. However, the percentage of dead cells, represented by 7-AAD-positive cells, was significantly reduced in S-UW isolation group [UW (n = 5) vs. S-UW (n = 5), 25.8 ± 5.3% vs. 17.2 ± 1.3%, p < 0.05]. In contrast, the viability of islets isolated with S-Kyoto solution was significantly better relative to that of ET-Kyoto isolation [ET-Kyoto (n = 5) vs. S-Kyoto (n = 5), 67.0 ± 1.2% vs. 75.4 ± 2.0%, p < 0.001]. Moreover, the percentage of dead cells in the S-Kyoto group was also lower compared with that in ET-Kyoto isolation [ET-Kyoto (n = 5) vs. S-Kyoto (n = 5), 18.6 ± 2.2% vs. 11.6 ± 2.4%, p < 0.01]. Similar findings of islet viability were noted by MTS assay (Fig. 6A). The highest viability of fresh islets was noted with S-Kyoto solution [ET-Kyoto (n = 5) vs. S-Kyoto (n = 5), 0.172 ± 0.013 vs. 0.206 ± 0.026, p < 0.05] (Fig. 6A). With regard to the in vitro culture after islet isolation, although islet viability in each group decreased gradually, that of the S-Kyoto group was well preserved compared with other isolation groups (Fig. 6B).

In vitro viability assay of isolated islets by TMRE and 7-AAD. (A) Representative flow cytometry analysis by tetramethyl rhodamine ethyl ester (TMRE) assay. (B) Representative flow cytometry analysis by 7-aminoactinomycin D (7-AAD). Percentage data represent percentages of cells with high fluorescence.

In vitro viability assay of isolated islets by MTS. (A) Viability of fresh islets (30 islets) was assessed using MTS assay. Data are mean ± SD of five independent experiments; *p < 0.05. (B) Isolated islets (30 islets) were cultured for 0, 1, 2 days, and their viabilities were assessed by MTS assay. Data are mean ± SD of five independent experiments; *p < 0.05,**p < 0.01versus fresh islets.
Improvement of Islet Function Elicited by Islet Isolation with S-Kyoto Solution
Next, we evaluated insulin function of islets using static glucose change. Although no significant difference was observed in insulin concentration under low glucose (2.8 mM) using each solution, insulin concentration under high glucose (20 mM) and the SI improved significantly by S-Kyoto islet isolation in comparison with those of other isolation solutions (Table 2). Based on the results of UW isolation groups (UW, S-UW), no beneficial effects of sivelestat were elicited as determined by the SI.
Insulin Concentration Under Low (2.8 mM) and High (20 mM) Glucose and Stimulation Index According to the Isolation Solution Used in the Present Study
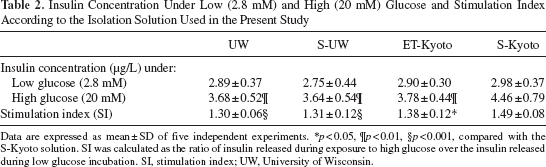
Data are expressed as mean ± SD of five independent experiments.
p < 0.05,
p < 0.01,
p < 0.001, compared with the S-Kyoto solution. SI was calculated as the ratio of insulin released during exposure to high glucose over the insulin released during low glucose incubation. SI, stimulation index; UW, University of Wisconsin.
Prolongation of Islet Graft Survival in S-Kyoto Group and in In Vivo Experiments
Transplantation of 500 allogeneic islets of the ET-Kyoto group was associated with graft survival of 7.4 ± 1.1 days (Fig. 7A, Table 3). A significant prolongation of islet graft survival was noted after transplantation of the S-Kyoto group, compared with islet grafts of the ET-Kyoto group (11.2 ± 1.3 days, p < 0.05). Importantly, islet transplantation coupled with 15-day intraperitoneal administration of 2 mg/day sivelestat elicited significant prolongation of graft survival, compared with both ET-Kyoto and S-Kyoto groups, respectively (Fig. 7A, Table 3). Next, NE activity in sera of recipient mice was measured. NE activity increased gradually after islet transplantation in the S-Kyoto group. However, sivelestat significantly suppressed the enzyme activity measured at day 28 after transplantation (Fig. 7B). These results suggest that prolongation of islet graft survival in the sivelestat IP group is due to inhibition of NE enzyme activity and the anti-inflammatory properties of sivelestat.

Results of in vivo transplant experiments. Islets were isolated using one of the isolation solutions (ET-Kyoto, S-Kyoto), then immediately transplanted (500 allogeneic islets) under the kidney capsule of diabetic mice. Sivelestat was injected intraperitoneally at 2 mg/day at 1 day before transplantation and every day until posttransplantation day 14. (A) Blood glucose levels of recipient mice transplanted with the islets. Data are mean ± SD of five independent mice. (B) Serum neutrophil elastase activity of recipient mice was measured at day 1 before transplantation and days 4, 7, 14, 21, 28 posttransplantation. Data are mean ± SD of three independent samples; *p < 0.05, versus S-Kyoto. (C) IPGTT of recipient mice was performed at posttransplantation day 7. Blood glucose was measured in each group (nondiabetic wild-type mice, untreated diabetic mice, recipient mice transplanted with islets isolated by the use of ET-Kyoto, S-Kyoto, and S-Kyoto with sivelestat) before injection and at 15, 60, 120 min after injection. Data are mean ± SD of three independent mice. (D) Immunohistochemical analysis of insulin in a representative mouse kidney engrafted with islets was performed at posttransplantation day 7. Scale bars: 100 μm.
Survival of Islet Allografts in Mice With Streptozotocin-Induced Diabetes

Data are mean ± SD of five independent experiments.
p < 0.01,
p < 0.001, compared with the ET-Kyoto isolation solution without intraperitoneal injection of sivelestat.
p < 0.001, compared with the S-Kyoto isolation solution without intraperitoneal injection of sivelestat.
To examine islet graft function in vivo, IPGTT was performed at day 7 after transplantation. In nondiabetic wild-type mice, injection of glucose-induced hyperglycemia with the peak blood glucose level recorded at 15 min, but the level returned to normal at 60–120 min after the injection (Fig. 7C). A similar pattern was observed in mice transplanted with S-Kyoto and S-Kyoto and treated with sivelestat. On the other hand, blood glucose levels of mice transplanted with ET-Kyoto group and untreated diabetic mice were significantly higher than those of the control wild-type mice before injection and at 15, 60, and 120 min after injection of glucose (Fig. 7C). Immunohistochemical staining for insulin confirmed that islet transplantation with S-Kyoto and S-Kyoto with sivelestat prolonged graft survival. Small islet grafts with many infiltrating inflammatory cells were detected at day 7 after transplantation in the ET-Kyoto group, whereas insulin-positive islet grafts with well preserved islet structure were found in the S-Kyoto and S-Kyoto with sivelestat groups (Fig. 7D).
Sivelestat Suppresses Proinflammatory Cytokines After Warm Digestion During Islet Isolation
We measured the levels of proinflammatory cytokines (IL-2, IL-4, IL-6, IL-10, IL-17A, IFN-γ, TNF-α) in the isolation solution at each step of islet isolation, including before warm digestion, at the end of warm digestion, and after purification. The levels at the end of warm digestion were markedly higher than before warm digestion (Fig. 8). On the other hand, IL-6 and TNF-α levels were significantly lower at the end of warm digestion in S-Kyoto solution compared with other isolation solutions such as UW and ET-Kyoto (Fig. 8).

Assessment of proinflammatory cytokines in isolation solution during islet isolation. The levels of proinflammatory cytokines [interleukin (IL)-2, IL-4, IL-6, IL-10, IL-17A, interferon (IFN)-γ, tumor necrosis factor (TNF)-α] in the isolation solution were measured during islet isolation (before warm digestion, at the end of warm digestion, after purification). Data are mean ± SD of four independent experiments; *p < 0.05.
Sivelestat Suppresses Proinflammatory Cytokines in Serum of Islet Recipients After Transplantation
The serum levels of IL-6 and TNF-α in islet recipients were significantly higher at 7 and 14 posttransplantation days than before transplantation (day -1), whereas no significant increases were noted in the levels of other cytokines except IL-6 and TNF-α (Fig. 9). The serum levels of IL-6 and TNF-α were significantly lower in the sivelestat intraperitoneal (IP) group compared with nonsivelestat IP group at 7 and 14 posttransplantation days (Fig. 9).

Assessment of proinflammatory cytokines in sera of islet mouse recipients after islet transplantation (ITx). The levels of proinflammatory cytokines (IL-2, IL-4, IL-6, IL-10, IL-17A, IFN-γ, TNF-α) in the sera of islet recipients were measured at pretransplantation day 1 and posttransplantation days 4, 7, 14, 21, and 28. Data are mean ± SD of three independent samples; **p < 0.01, versus nonsivelestat IP group, #p < 0.05 and ##p < 0.01, versus before transplantation (day -1).
Discussion
Islet transplantation is currently one of the most attractive strategies for the treatment of type 1 diabetes. One important key to achieving successful insulin independence after islet transplantation is acquiring a sufficient donor islet mass (1, 10). The number and quality of islets recovered from isolation are influenced by several factors (10). Tissue damage of donor pancreas starts to occur as early as the onset of brain death associated with hypotension, peripheral vasoconstriction, tissue ischemia, and release of stress hormones and inflammatory mediators (10, 20, 24, 38). Moreover, at the time of procurement, donor pancreas is exposed to warm ischemia after donor cross-clamping and cold ischemia storage in preservation solution, such as UW, ET-Kyoto solution (23, 40). Further more, during islet isolation, warm digestion, trauma, and hypoxia may cause cell damage in isolated islets similar to ischemia/reperfusion injury (IRI) of other transplant organs (10). Therefore, we need to design an efficient isolation method to reduce cell damage toward donor pancreata.
The major finding of the present study was identification of the crucial role of NE in islet isolation and transplantation. First, we showed a marked increase in NE activity during islet isolation, especially at the end of warm digestion by collagenase (Fig. 2C), and that NE was cytotoxic against isolated islets (Fig. 1A). Second, the addition of sivelestat to the isolation solution during islet isolation inhibited NE activity (Fig. 2C) and significantly improved islet yields, islet viability, and insulin function of isolated islets (Tables 1 and 2). Furthermore, we also showed that treatment of recipient animals with sivelestat significantly prolonged the survival of insulin-secreting islet allografts (Fig. 7A, Table 3).
In clinical liver transplantation, IRI, an exogenous antigen-independent inflammatory event, remains a major problem, because IRI causes early transplant graft failure and can lead to a higher incidence of both acute and chronic rejections (6, 54, 55). The mechanisms underlying liver IRI involve leukocyte accumulation and activation of neutrophils, Kupffer cells (macrophages), and T cells, secretion of proinflammatory cytokines and chemokines, complement activation, and activation of vascular cell adhesion molecules (49, 51, 53--55). Recent reports also indicated that the cross-talk interaction between NE and toll-like receptor (TLR) 4 promotes liver damage and that NE inhibitors, such as sivelestat, ameliorated the hepatocellular damage by reducing the expression of proinflammatory cytokines, chemokines, and TLR4 (24, 54, 55).
In islet transplantation, proinflammatory cytokines such as TNF-α and IL-1β induce β-cell apoptosis (1, 3, 27). These proinflammatory cytokines are produced by acinar and ductal cells (10). Moreover, in isolated islets, resident macrophages and monocytes produce cytokines (3, 10, 25). During islet isolation, the pancreatic tissues are warmed up to 37°C for collagenase digestion (10). As shown in Figure 2, many neutrophils were activated and released NE at the end of warm digestion. The released NE caused injury to membrane components of macrophages, acinar cells, and islets. Activated macrophages and acinar cells produce proinflammatory cytokines, including TNF-α and IL-6 (3). In fact, significantly high levels of TNF-α and IL-6 were found in isolation solution at the end of warm digestion compared with those before warm digestion (Fig. 8). Moreover, NE may also serve as a putative endogenous TLR4 ligand, causing TLR4 upregulation on macrophages and islets during the isolation process (5, 39, 54, 55). Such excessive expression of TNF-α and TLR 4 affects the surrounding islets causing their apoptosis (7, 19). Indeed, to detect apoptotic cells during islet isolation, TUNEL staining was performed before and at the end of warm digestion. Many TUNEL-positive cells were detected in UW solution at the end of warm digestion, whereas no such cells were detected in pancreatic tissue before warm digestion (Fig. 3A). What is the mechanism of NE-induced islet injury? Sivelestat effectively prevented the cross-talk between NE and inflammation responses, including the expression of proinflammatory cytokines (TNF-α, IL-6) and TLR4, resulting in a significant improvement in islet yields, islet viability, and insulin function in islet isolation with S-Kyoto solution. Furthermore, analysis of serum cytokine production profile (IL-2, IL-4, IL-6, IL-10, IL-17A, IFN-γ, TNF-α) in islet recipient mice showed significant increase in IL-6 and TNF-α production in serum at 1 and 2 weeks after islet transplantation, compared with those before islet transplantation (day -1), whereas no such increase was observed in the production of other cytokines before/ after islet transplantation (Fig. 9). As shown in Figure 7B, NE activity increased gradually after islet transplantation. However, sivelestat significantly suppressed NE activity and the increase in IL-6 and TNF-α production in mice (Figs. 7B and 9). Taken together, local inflammatory reaction, resulting in functional loss of islet grafts occurred after transplantation and treatment with sivelestat inhibited such inflammatory reactions, as evidenced by reduced IL-6 and TNF-α production, and resulted in prolongation of islet allograft survival.
Trypsin released from pancreatic acinar cells directly destroys islets (35). Previous studies demonstrated that inhibition of trypsin by pefabloc or ulinastatin during pancreas digestion improved islet yield and reduced the fraction of embedded islets (22, 35). These findings suggest that trypsin may degrade pancreatic ductules, resulting in reduced delivery of collagenase solution (35). However, the molecular weight of the new recombinant NE inhibitor sivelestat is much lower than α1 protease inhibitors and appears to exert its cytoprotective effect in the microenvironment between neutrophil and pancreatic tissues (12, 30). In this study, we examined the effects of the addition of sivelestat to ET-Kyoto solution. ET-Kyoto solution has a high-sodium/low-potassium composition with comparatively low viscosity (35). Therefore, it allows sufficient organ flushing after harvesting the pancreatic tissues (28, 35). Furthermore, we also examined the effects of the addition of sivelestat to the UW solution. The results suggested that sivelestat also provided cytoprotection when added to the UW solution. This conclusion was based on the finding that the decrease in the number of activated neutrophils and neutrophil elastase activity in the S-UW group tended to be larger than in the UW group (Fig. 2B, C), albeit statistically insignificant. The islets isolated from the UW solution are easily damaged by mechanical stress due to the high viscosity of the UW solution (13). Indeed, in our experiment, membrane shear damage had a negative impact on the isolated islets of the UW group during the isolation process, as shown in Figure 4B. This was probably due to one or more of the following reasons: (1) higher viscosity of the UW solution relative to that of the ET-Kyoto solution, (2) the high percentage of activated neutrophils present in the pancreatic tissue, and (3) the higher level of neutrophil elastase released in the UW than in ET-Kyoto. We speculate that the balance between the amount of released neutrophil elastase and its inhibition by collagenase digestion tilts towards excess elastase activity when sivelestat is added to the UW solution. We also assessed the synergistic effects of ulinastatin and sivelestat on improvement of islet yield and islet viability. However, no additive effects were observed in comparison with S-Kyoto solution alone and S-Kyoto solution with ulinastatin (data not shown).
As shown in Figure 4B, SEM showed well-preserved islets that were isolated by S-Kyoto solution, as evident by their round and smooth surface. NE is reported to increase the permeability of vascular endothelial cells, a process known to be involved in tissue edema (30). Indeed, during islet isolation, inhibition of NE activity by sivelestat may maintain cell membrane stability and permeability of endothelial cells in isolated islets, preventing tissue edema and leading to improvement of islet yield and insulin function of isolated islets.
In conclusion, we succeeded in isolating large numbers of islets using a new preservation solution, S-Kyoto solution, and in significant prolongation of islet graft survival in recipient mice treated with sivelestat. Our results emphasize the role of NE in the pathophysiology of islet damage during islet isolation and after transplantation. NE contributes to the accumulation of neutrophils and secretion of proinflammatory mediators during the isolation procedure and after islet transplantation. Therefore, treatment with NE inhibitors is potentially suitable for better harvest of transplantable islets and long-term islet allograft survival, allowing successful management of diabetes with islets from a single donor. We plan to assess in the near future the beneficial effects of S-Kyoto solution and monotherapy with sivelestat in human islet transplantation.
Footnotes
Acknowledgments
The authors thank Dr. F. G. Issa (www.word-medex.com.au) for the careful reading and editing of this manuscript, ONO Pharmaceutical Co. for kindly providing sivelestat, and Otsuka Pharmaceutical Co. for generously providing ET-Kyoto solution. The authors declare no conflict of interest.
