Abstract
As of October 1, 2007, 25 North American medical institutions and one European islet transplant center reported detailed information to the Registry on 315 allograft recipients, of which 285 were islet alone (IA) and 30 were islet after kidney (IAK). Of the 114 IA recipients expected at 4 years after their last infusion, 12% were insulin independent, 16% were insulin dependent with detectable C-peptide, 40% had no detectable C-peptide, and 32% had missing C-peptide data or were lost to follow-up. Of the IA recipients, 72% achieved insulin independence at least once over 3 years and multiple infusions. Factors associated with achievement of insulin independence included islet size >1.0 expressed as IEQs per islet number [hazard ratio (HR) = 1.5,
Background and Purpose
The Collaborative Islet Transplant Registry (CITR) accumulates and compiles data from completed and ongoing human islet transplantation studies conducted since 1999. Each year the Registry provides a comprehensive overview of the cumulative data. The present report highlights results on recipients of one or more islet cell transplants from 1999 through 2006, with detailed data reported to the Registry as of September 30, 2007, from 25 North American centers and one European center, whose participation in the CITR began in 2006.
The CITR data to date comprise single-arm Phase I/II trials. These trials were designed to investigate factors associated with islet transplant success, measured through improvement in glycemic control and/or reversal of insulin dependency, achievement of long-term islet function, and assessment of risks related to the infusion procedure or the immunosuppressive medication. Each islet transplant center publishes the results of their studies and provides information regarding their open and recruiting protocols through their own means and/or at the National Library of Medicine's website (www.clinicaltrials.gov).
Patients and Methods
Patients
All recipients of allogeneic islet-alone (IA), islet-after-kidney (IAK) transplants, and autologous transplants, since 1999, are eligible for inclusion in CITR. The vast majority (>95%) of CITR allograft recipients had type 1 diabetes mellitus (T1DM) for more than 5 years, were between 18 and 65 years of age, and had poor diabetes control, hypoglycemia unawareness, and severe hypoglycemia.
Data Collection
Data reported to the Registry are abstracted from medical information that is routinely collected by investigators for scientific evaluations and for reports to various agencies and entities that regulate trials according to the requirements of the respective country. Data collection utilizes the Coordinating Center's (The EMMES Corporation, Rockville, MD, USA) web-based data entry and management. For each islet/β-cell infusion, information is collected on the pancreas donor(s), islet processing and testing of all pancreata used for the infusion procedure, and recipient status during follow-up. Donor and islet processing data are also obtained through data sharing agreements with the United Network for Organ Sharing (UNOS) and the Administrative and Bioinformatics Coordinating Center (ABCC) of the Islet Cell Resource Centers (ICR).
Follow-up data are abstracted at day 30, month 6 and month 12 post-first islet infusion for four main indicators: severe hypoglycemic episodes, glycated hemoglobin (A1C or HbA1c, measured as percent of total hemoglobin) values, fasting blood glucose and C-peptide, and daily insulin status. Detailed follow-up data are abstracted at month 6, month 12, and annually postinfusion. At each new infusion, a new follow-up schedule is established to abstract data at 6-month and annual anniversaries of the most recent infusion. Event-driven data include reportable adverse events, islet graft dysfunction and failure, other major medical events, and loss to follow-up. All grade 3, 4, and 5 adverse events, according to the Common Terminology Criteria for Adverse Events (CTCAE) of the National Cancer Institute, and all serious adverse events (regardless of grade) are reported to CITR. Site transplant investigators determine the relationship between the adverse event and immunosuppressive therapy and/or islet infusion. A copy of the CITR data collection forms is available at the CITR Website (www.citregistry.org).
All islet recipients provided written informed consent for their data to be reported to the Registry. The data are monitored by the CITR Coordinating Center for quality assurance. The database was closed for this analysis on October 1, 2007.
Missing Data
Increasing levels of missing data accrue with longer follow-up times. Patients lost to follow-up (two or more expected consecutive annual visits not completed) are analyzed in two ways: 1) data after loss to follow-up are censored, namely, considered missing at random or unrelated to any outcome (“best-case scenario”), or, alternatively, 2) persons lost to follow-up are imputed to have experienced complete islet failure due to a presumed discontinuation of their immunosuppression regimen (“worst-case scenario”). The truth lies somewhere in between. Study resources are being focused on capturing as much of the missing data as possible while the Registry is still active to obtain the best estimates of these important outcomes.
Outcomes
Primary outcomes are: achievement of insulin independence; maintenance/loss of insulin independence; hypoglycemia status; A1C level; C-peptide level; islet graft dysfunction or failure; or combinations of these.
Secondary outcomes are: diagnosis or progression of chronic complications of diabetes (nephropathy, neuropathy, and retinopathy among others); variations of kidney and liver function; lipid and blood pressure stasis and other concomitant medications; and adverse events.
Statistical Analysis
Results of continuous variables are expressed as mean ± SD or median (interquartile range) and categorical variables as number of cases (%). The primary outcomes (achievement of insulin independence, loss of insulin independence, graft dysfunction and failure, or combinations of these) were analyzed as time-to-event (Kaplan-Meier) estimates or frequency distributions of categorical factors. Analysis of the effect of various factors on the primary outcomes has begun and will continue as the registry grows and the data are more completely reported. Explanatory factors included preinfusion recipient, donor, procurement and final product characteristics, immunosuppressive agents, as well as time-dependent factors such as reinfusion and the occurrence of other events and subsequent interventions, which present competing risks. Analyses are presented for events occurring after first infusion up to reinfusion, current follow-up, or complete graft loss, as well as analyses on outcomes after the recipient's last infusion regardless of the total number of infusions the recipient has received. Values of
Results
Islet Allograft Transplantation Activity (1999–2007)
Pooling the reported data from the 30 registered North American and one European centers, the CITR as of September 30, 2007 comprised 315 allograft recipients (285 IA and 30 IAK) with detailed data reported, who received 617 infusion procedures derived from a total of 674 donors. Ninety of the recipients (26%) received a single islet infusion, 153 (49%) received two, 67 (21%) received three, and five (1.6%) received a total of four islet infusions. The Registry is also collecting information on islet autograft transplantation. The cumulative number of allograft transplants in the Registry according to date of first infusion is shown in Figure 1. Allogeneic islet transplantation experienced a substantial slow-down during 2007 compared with previous years. The remainder of this report is limited to the recipients of islet alone allogeneic islet transplants.

Cumulative number of allogeneic (
Recipient Characteristics
The mean age of the 285 allograft recipients at the time of first infusion was 44 ± 8.4 years and the duration of diabetes was 29 ± 10 years. The majority of the recipients were female (~64%), the mean weight was 66 ± 10 kg and the body mass index (BMI) was 23.6 ± 3.4 kg/m2. More detailed clinical characteristics of allogeneic recipients (IA and IAK) are shown in Table 1.
Islet Allograft Recipient Characteristics

Values are mean ± SD or
Data available on 283 subjects.
Percent of total hemoglobin.
Most of the patients from IA protocols were receiving intensive insulin treatment, meaning insulin pump or three or more insulin injections per day, long before the procedure (~91% of recipients for 18.7 ± 1.0 years), and the mean daily insulin requirements was 37 ± 14 units. Even receiving the best available treatment only 19% of the recipients had a A1C <6.5%, 0.7% were free of hypoglycemic episodes, while 78% had severe hypoglycemia in the previous year and about 20% had employment problems resulting from the disease.
Donor Information
Donor characteristics are summarized in Table 2. Mean donor age was 43 ± 13 years, BMI was 29.3 ± 171 kg/m2, and 58% of the donors were male. Regarding the cause of death, 53% had a cerebrovascular event or stroke while 29% experienced a head trauma. The mean time from cross clamp to pancreas recovery was 39 ± 20 min and mean cold ischemia time was 7.3 ± 3 h. In 64% of the pancreas procurement procedures, the surgical team was not related to or affiliated with the processing/transplant team. However, 91% of the pancreas processing procedures took place at the same institution as the islet transplant center. University of Wisconsin (UW) solution, two layer (UW + PFC), and UW followed by two layer were the most common (88%) methods used for pancreas preservation.
Donor Characteristics (

Liberase HI was the collagenase type used during most islet processing (94%) followed by Thermolysin/Liberase combinations (4%). All of the pancreata processed used a density gradient for islet purification. When islet culture was used, defined as ≥6 h in a specially prepared nutrient medium, the mean culture time was 30 ± 17 h.
Islet Characteristics and Islets Infused
Islet final product characteristics of the 674 preparations infused in the IA recipients are shown in Table 3. These variables were related to donor age and BMI, as well as recovery and cold ischemic time; these relationships deserve more in-depth analysis, especially in correcting correlated factors to primary outcomes.
Islet Preparation Characteristics (

Characteristics of infused islets are shown in Table 4. Allograft recipients received a 825,112 ± 369,916 total islet equivalents (IEQs), or 12,700 ± 5,900 IEQs/kg of body weight over all their infusions. Mean islet size infused, as estimated by the ratio of IEQs to total particles, was 1.6 ± 0.41 (assuming ratio 1 = islet size of 150 μg).
Characteristics of Islets Over All Infusions per Recipient: IA Recipients With Available Data

Immunosuppression Therapy
The majority of the IA recipients received a daclizumab, sirolimus, and/or tacrolimus immunosuppression regimen at the time of first infusion (1). Daclizumab was used for induction alone in 76% of IA first infusions, and in combination with other T-cell antibodies in another 6% of first infusions; 18% received etanercept at one or more infusions. Anti-thymocyte globulin was given alone or in combination in 11% of first infusions.
Graft Function
After the first infusion, increasing proportions of IA recipients were reinfused: 12% by day 30, 37% by day 75, 54% by month 6, and 64% by year 1 (Fig. 2, top). The proportion of insulin-independent recipients without reinfusion remained fairly constant (9–13%) throughout the first year (Fig. 2, top) and 4–14% retained detectable C-peptide with insulin dependence but without reinfusion in the same period (Fig. 2, top, 1-year time point).
Of the IA recipients, 90% were expected to have 1-year post-first infusion data at the time of analysis (Fig. 2, center). Twenty-four percent were insulin independent, 16% were insulin dependent with detectable C-peptide, 5% had no detectable C-peptide, and 60% either had missing C-peptide data (expected but not yet reported) or were lost to follow-up.
Analyzed from last infusion (Fig. 2, bottom), the percentage of insulin-independent IA recipients declined steadily from 40% at month 6 to 12% at year 4. A stable 18–22% retained graft function with exogenous insulin over the 4 years. The proportion with loss of islet function (reported graft failure or no detectable C-peptide or lost to follow-up with imputed graft failure) increased steadily from 20% at month 6 to 70% at year 3 post-last infusion. These trends of increasing prevalence of graft loss and decreasing prevalence of insulin independence over time post-last infusion prevail regardless of the total number of infusions given, although the outcomes improve with more infusions per recipient (9).
Focusing only on insulin independence status, defined as 2 weeks or longer of no exogenous insulin with good glycemic control by capillary glucose measurements, both available from daily diaries, the prevalence of insulin independence from last infusion rose to 60% at month 6 then declined to 28% at year 3 post-last infusion (Fig. 3). Two or three infusions boost the prevalence of insulin independence in the first year to a peak of 65%, with a subsequent decline to levels that are comparable to those with a single infusion.
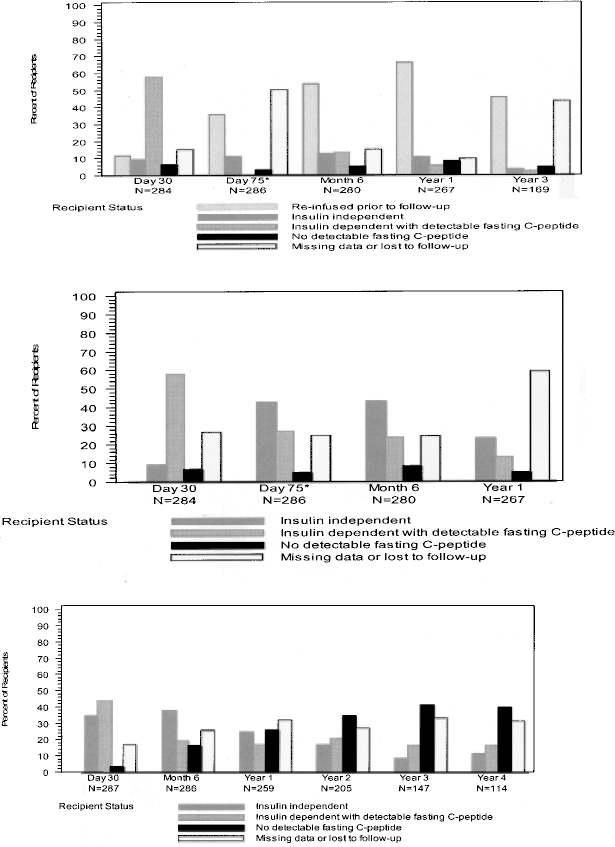
Insulin status and detectable fasting C-peptide; islet alone recipients. (Top) Post-first infusion accounting for reinfusion. (Center) Post-first infusion regardless of reinfusion. (Bottom) Post-last infusion. C-peptide data not available at day 75 post-first infusion.

Insulin independence post-last infusion, by total number of infusions received, IA recipients.
In IA recipients, the cumulative incidence of achieving insulin independence in the first year post-first infusion was 56% (censored at last follow-up regardless of the total number of infusions given), and over the second year this increased to 67%; and similar rates were observed for IAK recipients (Fig. 4, top). The greater the number of infusions per recipient, the higher the rate of achieving insulin independence (9). The proportion of recipients attaining insulin independence quickly after each reinfusion is substantially higher for second infusion and slightly higher post-third infusion than for first infusion.
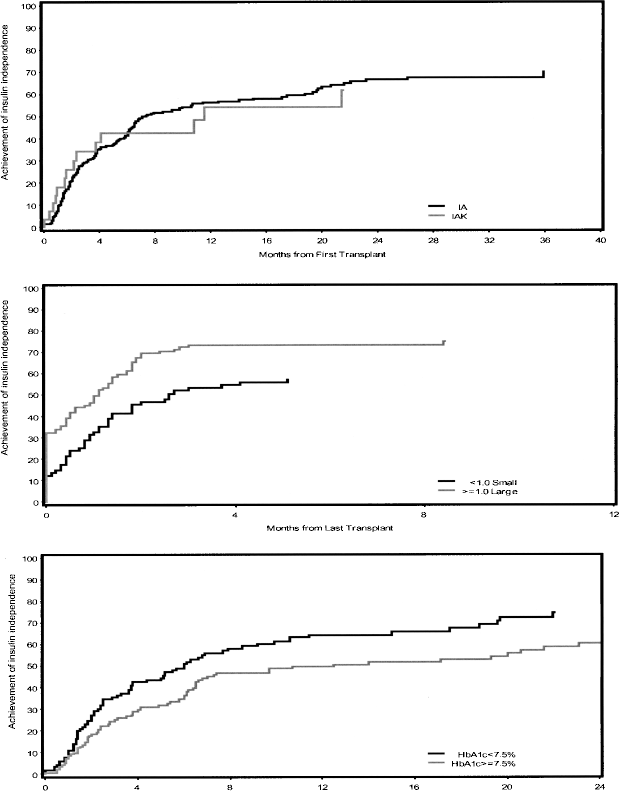
Achievement of insulin independence after first islet infusion, censored at last follow-up, regardless of reinfusion or graft loss (primary nonfunction and those with less than 30 days follow-up excluded). (Top) IA (dark line): 151 events/262 recipients; IAK (light line): 14 events/28 recipients; event is last instance of attaining insulin independence for 2 weeks or longer, time is from first infusion. (Center) IA only, computed islet size >1.0 (light line) versus <1.0 (dark line), log-rank = 3.17,
Over time there was a decrease in the sustainability of insulin independence (Fig. 5, top): of all recipients who ever achieved insulin independence, only 71% retained this status 1 year after achieving it and this decreased to 47% at 2 years. C-peptide levels substantially increased (Fig. 6, top) while fasting plasma glucose and A1C levels fell (Fig. 6, bottom), after islet transplantation. These trends were seen both overall and by total number of infusions. Those who achieved insulin independence after the first or second infusion were more likely to reachieve it and more likely to retain it after their last infusion. They were also less likely to lose graft function (9). However, a gradual loss of benefit in both variables was observed with longer follow-up.

(Top) Persistence of insulin independence, IA recipients achieving insulin independence, censored at last follow-up. (Bottom) Persistence of islet graft function in IA recipients achieving insulin independence (solid line) versus those never achieving insulin independence (dashed line).

(Top) C-peptide (mg/dl) at scheduled follow-up post-last infusion, IA recipients. (Bottom) HbA1c (%) at scheduled follow-up post-last infusion, IA recipients. Follow-up: preinfusion 1, preinfusion 2, preinfusion 3, then post-last infusion.
Variables Associated with Islet Transplant Success
Multivariate Cox regression models were used to investigate the effect of preinfusion factors on primary outcomes of islet transplantation. Factors associated with achieving insulin independence included larger islet size ratio (≥1.0) (Fig. 4, center), lower A1C (Fig. 4, bottom), donor without insulin treatment in hospital, induction with daclizumab, and lower cold ischemia time (Table 5).
Factors of Insulin Independence Post-Last Infusion, Censored at Last Follow-Up (95 Events/153 Recipients With Data)
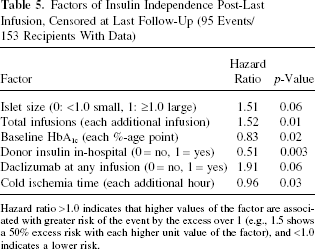
Hazard ratio >1.0 indicates that higher values of the factor are associated with greater risk of the event by the excess over 1 (e.g., 1.5 shows a 50% excess risk with each higher unit value of the factor), and <1.0 indicates a lower risk.
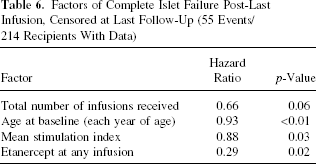
Factors protective against complete islet failure included more infusions given, older age (which was strongly correlated with longer diabetes duration and years of intensive insulin therapy), higher stimulation index of the graft(s), and no etanercept at induction (Table 6).
Factors of Complete Islet Failure Post-Last Infusion, Censored at Last Follow-Up (55 Events/214 Recipients With Data)
Hypoglycemia
A striking continuous decrease in the prevalence of severe hypoglycemic events was observed after islet transplantation. Severe hypoglycemia prevalence was reduced from 78–83% preinfusion to less than 5% throughout the first year post-last infusion, and to an estimated 18% (adjusted for missing data) at 3 years post-last infusion (Fig. 7). Hypoglycemia awareness was also markedly improved and was eroded only by loss of graft function or loss to follow-up (9). All recipients that experienced a severe hypoglycemic event during follow-up were on insulin at the time of the event.

Severe hypoglycemia at scheduled follow-up after last infusion, IA recipients.
Adverse Events
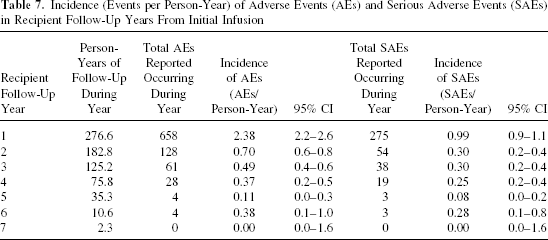
Sixty-five percent of the IA recipients experienced at least one reportable (CTCAE Grade 3–5 or serious) adverse event (AE) in the first year after initial infusion and 41% experienced one or more serious adverse (SAE) events in this same period. Of the 574 AEs reported in the first year after first infusion, 32.5% were judged by the local investigator as being at least possibly related to the immunosuppression therapy and 28.4% to the infusion procedure. Of the 211 SAEs reported in the first year after first infusion, 26.5% were related to the immunosuppression therapy and 45.5% were related to the islet infusion procedure.
Overall, a total of 337 SAEs were reported. Approximately 91% resolved with no residual effects. Most of the reported SAEs were categorized as specified threshold laboratory results (22%), gastrointestinal disorders (19%), and blood and lymphatic system disorders (15%) as classified by the MedDRA classifications system.
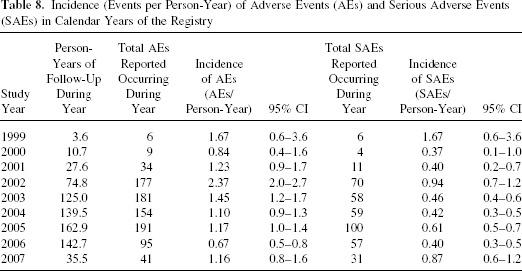
The incidence of both AEs and SAEs peaked in the first year post-first infusion and then dropped to substantially lower levels (Table 7). Considering that 2002 was the first year of follow-up for the majority of IA recipients reported to the CITR, the first-year effect artificially raises the apparent incidence rates for the years 2002–2003 (Table 8).
Incidence (Events per Person-Year) of Adverse Events (AEs) and Serious Adverse Events (SAEs) in Recipient Follow-Up Years From Initial Infusion
Incidence (Events per Person-Year) of Adverse Events (AEs) and Serious Adverse Events (SAEs) in Calendar Years of the Registry
Increments in laboratory results two times or greater than the upper limit of normal were subject of CITR reporting. Only a small subset of recipients have shown liver tests abnormalities [alanine aminotransferase (ALT) 4%, aspartate aminotransferase (AST) 4%, alkaline phosphatase 6%, and total bilirubin 1%, and nine (4.5%) recipients had an elevation in triglycerides.
There were 28 reports in 13% of IA patients of increment in serum creatinine greater than 0.5 mg/dl above patients' baseline level. When the estimated glomerular filtration rate, calculated by the Modification of Diet in Renal Disease (MDRD) formula (4), was used to evaluate the trends of kidney function, a decline was observed over time in both IA (
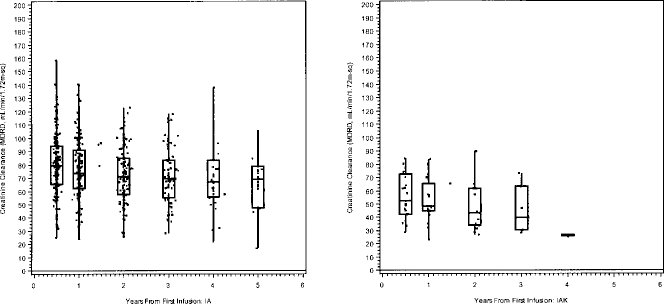
Creatinine clearance from first infusion, IA (left) and IAK (right). Individual results (points) and box plots showing median, interquartile range, and minimum/maximum.
The percent of subjects using medication at 1-year post-last infusion compared to preinfusion increased somewhat for antihypertensives (48% vs. 40%) and doubled for lipid lowering agents (61% vs. 31).
Neoplasms have been diagnosed in 10 allograft recipients, of which 8 were proved to be malignancies, meaning an incidence of 2.5% over the reported period of follow-up (1999–2006). Five were skin cancers, comprising four cases of squamous cell carcinoma, three of which were treated and resolved with no need for discontinuation of immunosuppression, and one with no further information; and one case of basal cell carcinoma, occurring after discontinuation of immunosuppression due to islet graft failure, treated and resolved with no residual effects. Two recipients had papillary thyroid cancer, diagnosed at 2 and 21 months post-first infusion, respectively, and treated without immunosuppression discontinuation. One subject had breast cancer, diagnosed 22 months post-first infusion and treated without discontinuation of immunosuppressive therapy. However, nodal regional metastases were found 1 month later leading to immunosuppression discontinuation.
There were five reports of death to the CITR up to the report cut-off. One recipient died due to viral meningitis, 3 years after second islet infusion and classified by the local CITR investigator as possibly related to the immunosuppressive therapy. The others were classified with no relation with the islet transplant: drug intoxication (methadone and diphenhydramine) 70 days after third infusion; a stroke more than 2 years post-second infusion; a death due to unknown causes (discovered in obituaries) more than 4 years after the recipient's second infusion; and a suicide.
Discussion
The CITR likely represents the most comprehensive collection of the human islet transplantation experience since 1999. The main result of the CITR analysis is the confirmation of previous reports from individual islet transplant centers experience, but with enhanced power, because this sample represents almost the totality of islet recipients in North America, plus the contribution of one European center. Thus, it represents the strongest evidence of islet transplantation benefits: stabilization of glucose metabolism, sustained decrease in severe hypoglycemic episodes, restoration of symptoms awareness, and normalization of A1C.
Moreover it provides accurate description of AEs experienced by islet recipients, relying on site-visit data monitoring. Most AE were related to known effects of the immunosuppressive regimens and were resolved without sequelae, thus confirming the overall relative safety of the procedure and therapy. The decrease in glomerular filtration rate posttransplantation suggested by some authors (5–7), but not confirmed by others (1, 3) was observed in the collective CITR experience. However, this decline could be solely a reflection of the clinical course of kidney disease in patients with long-term T1DM, because comparable controls were not available for either CITR or most of the individual cohort analysis. In fact, the two centers reporting stable kidney function after islet transplantation based their results on comparisons with controls (patients in the waiting list for islet transplantation in one case and with pretransplant own patients trends in another) (1, 3). Another possible explanation for these differences is the individualized clinical management for concomitant risk factors for kidney disease offered in different centers (3). A not unexpected number of neoplasms occurred, namely the reported skin and breast cancers, though it is impossible to rule out a relation with immunosuppressive therapy.
Another contribution of CITR was the possibility to analyze factors associated with better islet graft function and survival. As expected, higher chances of insulin independence were observed in subjects receiving higher number of islet infusions and with islet preparations with shorter cold ischemia time. Donor need of insulin treatment in-hospital negatively affected insulin independence achievement, reflecting lower β-cell secretion capacity in an in vivo model of increased insulin demands. As well, higher A1C recipient values pretransplant had a negative impact on insulin independence. Plausible explanations for this finding could be either an acute glucose-induced damage to transplanted islets or a higher pretransplant A1C could be a marker of recipient lower insulin sensitivity since baseline. Surprising, larger islet size was associated with better islet function, which has been previously reported in autograft recipients (8) but contrasts with in vitro reports (2). Protocols with daclizumab or etarnecept during induction had higher rates of insulin independence and lower rate of total function lost, respectively, which endorse the current approaches.
The CITR report definitely has proven the concept raised by experimental studies: pancreatic islets infused intraportally can engraft in the human liver, detect blood glucose levels, and release insulin accordingly. Insulin independence is achieved in most recipients, but long-term graft failure has been consistently observed. Therefore, clinical islet transplantation can be considered a successful procedure if more realistic end-points—such as glucose stabilization and severe hypoglycemia prevention, instead of insulin independence—are taken into account. Even though this issue is still a source of debate, the aggregate experience from the present results of the Registry confirms the inarguably positive impact of islet transplantation on metabolic control.
The main limitation of a full interpretation of the present results is the level of missing data, especially in later years of follow-up. In order to solve it, the CITR is increasing efforts to reduce the amount of missing data. The lack of a control group is a limitation that affects many other results in T1D.
Conclusions
From 1999 through 2006, the chief clinical benefits of human islet transplantation included a remarkable reduction in the occurrence of severe hypoglycemia and a success rate of 67% in achievement of insulin independence, which often persists for 2 years or more. These results are quite consistent throughout the 8 years of follow-up included in the Registry. The Registry is growing large enough to begin investigating factors predictive of and/or associated with primary outcomes, the results of which will undoubtedly guide researchers in designing more effective and safer protocols, leading the field to overcome most of the steps required to be approved for use in the clinical setting.
CITR and Contributors
The Collaborative Islet Transplant Registry (CITR) is sponsored by the National Institute of Diabetes & Digestive & Kidney Diseases under contract number N01-DK-6-2868 and by a supplementary grant from the Juvenile Diabetes Research Foundation International. Additional data are made available through cooperative agreements with participating transplant centers; the Islet Cell Resource Consortium Administrative and Bioinformatics Coordinating Center, City of Hope Medical Center; and the United Network for Organ Sharing. Reprints and additional information may be requested via email to
The following institutions and individuals contributed to the reporting and/or analysis of the data included in this report.
