Abstract
Islet transplantation has shown great success in the treatment of type 1 diabetes since the Edmonton protocol was established. However, it still has two major problems to overcome: the lack of organ donors and the side effects of immunosuppression. Encapsulated islets have emerged as a potential option for islet transplantation because it can, at least partly, overcome these two problems. Wistar rat islets suspended in 3% polyvinyl alcohol (PVA) hydrogel were frozen-thawed to make macroencapsulated islets (MEIs). The recovery rate, insulin content, and morphological change in culture medium with/without fresh human plasma (FHP) were measured in MEIs and free islets in vitro. In vivo, MEIs of either Wistar or Lewis rats were transplanted into the peritoneal cavity of streptozotocin (STZ)-induced diabetic Lewis rats and nonfasting blood glucose (NFBG), body weight, and histological evaluations were processed. FHP destroyed rat free islets but did not affect the islet morphology, islet recovery rate, or insulin content of rat MEIs. The transplantation of MEIs decreased the NFBG level and prevented body weight loss without a significant difference between the donor strains. Insulin-positive islets were observed in PVA MEIs 24 weeks after allotransplantation. These results suggest that PVA MEIs may be used as a cure for type 1 diabetes.
Introduction
Type 1 diabetes is an autoimmune disease that results from the permanent destruction of insulin-producing β-cells of the pancreas (14,34). Exogenous insulin injection to replace the missing hormone is utilized to treat type 1 diabetes; however, hypoglycemia is unavoidable (15) and diabetic chronic complications may lead to morbidity and mortality (4).
Islet transplantation could replace the exogenous insulin therapy, because β-cells within islets can secret insulin. Successful clinical islet transplantation had been utilized to treat patients with type 1 diabetes since the Edmonton protocol was established in 2000 (21,27,28). However, two major obstacles, namely the lack of organ donors and the side effects of immunosuppression, prevent its success rate (10,12). Encapsulated islets could be considered to be a therapeutic strategy for islet transplantation, because they can, at least partly, overcome these two problems.
Encapsulated islets are donor islets enclosed in a semipermeable membrane that prevents high-weight molecules and immune cells from coming in contact with the islets, thereby protecting the donor islets from the attack of host immune system. On the other hand, low-weight molecules, such as oxygen, glucose, some nutrients, and insulin, etc., are allowed to exchange freely; therefore, islets inside can receive the sufficient oxygen and nutrients to maintain their function and insulin secreted from islets can be released freely (18,30). Because of the properties of the encapsulated islets, encapsulated islet transplantation can be performed without any immunosuppressive treatments and a variety of cells can be used as donor cells, such as allografts (30), xenografts (8,31,32), and some insulin-producing cells derived from various cell sources (35). Therefore, two major obstacles for islet transplantation could be solved easily.
A variety of biocompatible materials have been utilized as semipermeable membranes to make encapsulated islets. This study focused on a polyvinyl alcohol (PVA) hydrogel because of its lower protein binding tendency, higher water content, and higher elasticity (6). Previous studies investigated its function in vitro and in vivo, such as in xenotransplantation, diabetic nephropathy, and islet preservation (17,18,23,24). However, the effective period for immunoisolation and the discordant immunoisolation effect of PVA macroencapsulated islets (MEIs) have not yet been evaluated.
This study describes a successful allotransplantation (Wistar rat cells into Lewis rat; W-L) without any immunosuppressive treatments and an excellent effect of discordant immunoisolation from fresh human plasma (FHP) using PVA MEIs.
Materials and Methods
Animals
Nine- to 10-week-old male Wistar and Lewis rats were purchased from Shimizu Laboratory Supplies Co. Ltd. (Kyoto, Japan). Approval of this experiment was obtained from the Animal Care Committee of Institute for Frontier Medical Sciences, Kyoto University, and the animals were treated according to the experimental protocols under its regulations.
Islet Isolation
Wistar or Lewis rat islets were isolated as described previously (16). Briefly, the pancreas was digested by type XI collagenase (Sigma-Aldrich, Inc., St. Louis, MO, USA) and the islets were separated by dextran (Amersham Biosciences, Sweden) gradient. Separated islets were purified by handpicking and cultured in RPMI-1640 medium with 10% heat-inactivated fetal bovine serum (FBS) and 1% antibiotics solution overnight.
Protocol of PVA MEI Preparation
The protocol was originally developed by Qi et al. (17). Briefly, PVA solution was made up of 3% PVA (molecular weight: 387200, saponification degree: 99.8 mol %), 10% 10 times concentrated Euro-Collins (EC) solution, 10% heat-inactivated FBS, 5% dimethyl sulfoxide (DMSO), and 1% antibiotics. The rat islets pretreated with Cell Banker (Nippon Zenyaku Kogyo Co., Fukushima, Japan) were suspended in 200 μl PVA solution and this mixture was then sandwiched between two pieces of polyethylene terephthalate (PET) mesh for reinforcement, and then further sandwiched between two pieces of glass plates to control its thickness of 1 mm. After this molding process, MEIs were frozen in a computer-controlled program freezer (TAIYO Nippon Sanso Corporation, Tokyo, Japan), the freeing program was initiated at 4°C and the cooling rate was set to −3°C/min until the surface temperature of MEIs reached −10°C, and then was changed to −1°C/min until the surface temperature of MEIs reached −30°C. The MEIs were then immediately moved to −80°C ultra low freezer (SANYO Electric Co., Ltd) and stored for 24 h to form a crystallized gel. This crystallized gel was thawed rapidly in 37°C RPMI-1640 medium for a few seconds followed by immersing it in 4°C University of Wisconsin (UW) solutions for 24 h. Finally the MEIs were used in this study after overnight culture in RPMI-1640 (37°C, 5% CO2, 95% air).
Culture with FHP
FHP was collected from a healthy adult male with AB blood type, after obtaining informed consent. Fifty Wistar rat islets were enclosed into MEIs (size: 20 × 15 × 1 mm), and cultured in CMRL-1066 medium supplemented with 10% FHP, 20% FHP, or 10% heat-inactivated FBS (37°C, 5% CO2, 95% air). As a control, an equal number of free Wistar rat islets were also cultured in the same conditions. Morphological changes of MEIs and free islets were observed under microscopy after a 24- and 48-h culture. The number of islets in MEIs or number of free islets was counted after a 1-, 12-, 24-, and 48-h culture, respectively. The islet recovery rate was defined as the ratio of the number of islets after culture to number of islets before culture. Following a 48-h culture, MEIs and free islets were treated with HCl-ethanol solution, and extracted insulin was measured using an ELISA kit (Shibayagi Co. Ltd. Gunma, Japan).
Islet Transplantation
Type 1 diabetes mellitus (DM) models were induced in Lewis rats by a single injection of streptozotocin (STZ; Sigma, St. Louis, MO, USA; 55 mg/kg body weight in citrate buffer, pH 4.5, IV) 7 days before the islet transplantation. Rats with nonfasting blood glucose (NFBG) level >450 mg/dl served as the recipients. Islets of three donor rats were transplanted to a Lewis rat recipient. Recipients were randomly divided into five groups of five rats each as described in Table 1. MEIs (size: 30 × 25 × 1 mm) were placed into the peritoneal cavity and free islets were then transplanted into the renal subcapsular space of the recipients. The NFBG and body weight were observed until 24 weeks after transplantation.
Groups for Islet Transplantation (n = 5)
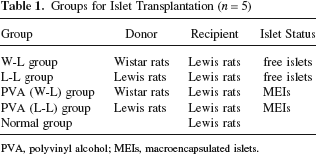
PVA, polyvinyl alcohol; MEIs, macroencapsulated islets.
Histological Assessments
MEIs were retrieved from the sacrificed recipient rats 24 weeks after transplantation for histological assessments. The samples were fixed in 4% paraformaldehyde solution for 24 h, followed by immersion in 70% ethanol at 4°C for 24–48 h. Next, the samples were embedded in paraffin and 5-μm-thick sequential sections were made. The sections were stained for insulin as described previously (29). Briefly, glass slides were washed in PBS (six times, 5 min) and mounted with 1% rabbit normal serum in PBS for 30 min. Subsequently, primary antibody (anti-insulin guinea pig polyclonal antibody; DakoCytomation) was applied overnight at 4°C. They were incubated with peroxidase-conjugated rabbit anti-guinea pig immunoglobulins (second antibody; Dako Cytomation) diluted to 1:100 in PBS for 30 min. After washing in PBS (six times, 5 min), coloring reaction was carried out.
Statistical Analysis
All of the results were expressed as the mean ± SEM. Significant differences between the groups were tested by Student-Newman-Keuls' test. Statistical significance was defined as p < 0.05.
Results
Morphological Changes
After a 24-h culture, the destruction of islets was observed in the free islets group, when the islets were cultured with 10% or 20% FHP. The destruction of islets became more obvious after a 48-h culture; the islets lost their normal morphologies and some fragments of islets were observed in the free islets group. In contrast, this phenomenon was not seen in the PVA MEIs group, even when islets were cultured with 10% or 20% FHP. No significant morphological changes of the islets were observed in either group when the islets were cultured with 10% heat-inactivated FBS (Fig. 1).

Morphological changes of the islets after a 24-h (A) and 48-h culture (B). Original magnification: 40x. Scale bars: 100 μm. FHP, fresh human plasma; FBS, fetal bovine serum; PVA MEIs, polyvinyl alcohol macroencapsulated islets.
Islet Recovery Rates
Islet recovery rates were calculated after a 1-, 12-, 24-, and 48-h culture, respectively. The free islets showed lower recovery rates in the 10% or 20% FHP group in comparison to the 10% heat-inactivated FBS group, especially after a 24-h (p < 0.05) and a 48-h culture (p < 0.01) (Fig. 2A). However, PVA MEIs showed no significant differences among the three groups at any time point (Fig. 2B).

Islet recovery rates of free islets (A, n = 4) and PVA MEIs (B, n = 6) cultured with 10% (circle), 20% FHP (square), and 10% FBS (triangle). *p < 0.05, **p < 0.01 (vs. 10% FBS group).
Insulin Content
The insulin levels were measured after a 48-h culture. The islets in the PVA MEIs group showed significantly higher insulin contents than those in the free islets group when islets were cultured with 10% or 20% FHP, respectively (p < 0.05). However, no significant difference was observed between the two groups when the islets were cultured with 10% heat-inactivated FBS (Fig. 3).

Insulin contents of free islets (white bars, n = 4) and PVA MEIs (black bars, n = 6) after 48-h culture with 10% FHP, 20% FHP, and 10% FBS. *p < 0.05.
Blood Glucose and Body Weight
The mean NFBG level of the recipients before transplantation was 574 ± 8 mg/dl. The average number of transplanted islets in the four transplantation groups was 1940 ± 39, without significant differences among the four groups (data not shown). As a positive control, the Lewis rat cells transplanted into a Lewis rat (L-L) group showed a lower NFBG and its level was maintained at about 200 mg/dl until 24 weeks after transplantation. In contrast, the Wistar-Lewis (W-L) group, as a negative control, maintained a lower NFBG for only 1 week after islet transplantation, then the NFBG was rapidly increased and the recipients became hyperglycemic again. Although NFBG in PVA (W-L) group did not reach the normal level in the first 6 weeks after islet transplantation (p < 0.05 vs. normal group; p < 0.05 vs. L-L group), it showed a lower NFBG value in comparison to the W-L group (p < 0.05). However, its NFBG significantly increased from the sixth week after transplantation in comparison to the initial stage (p < 0.05). No significant difference of NFBG was observed between the PVA (W-L) group and PVA (L-L) group. In contrast, a significantly higher NFBG was observed in the PVA (L-L) group in comparison to the L-L group (p < 0.05) (Fig. 4A).

Changes of nonfasting blood glucose (NFBG) after transplantation (A). After islet transplantation, a lower NFBG value was observed in the PVA group (Wistar cells into Lewis rat; W-L) in comparison to the W-L group (p < 0.05). However, NFBG in the PVA (W-L) group was increased after a 6-week transplantation. There was no significant difference between the PVA group (W-L) and PVA group (L-L), and a significantly higher NFBG was observed in the PVA group (L-L) in comparison to the L-L group (p < 0.05). Changes in body weight after transplantation (B). Recipients in the W-L group did not show a significant increase of body weight throughout the study (p > 0.05), but the recipients in the other four groups showed a significant increase of body weight in a time-dependent manner (p < 0.05).
The normal and L-L groups showed significantly higher body weight than the other three groups (p < 0.05). The recipients in the W-L group did not show a significant increase of body weight throughout the study (p > 0.05), but the recipients in the other four groups including the PVA group (W-L) and PVA group (L-L) showed a significant increase of body weight in a time-dependent manner (p < 0.05) (Fig. 4B).
Histological Findings
Dithizone staining was performed before transplantation to confirm the survival status of PVA MEIs. PVA MEIs maintained their normal shapes and intact structures and all the islets were positively stained (Fig. 5A). The transplanted PVA MEIs were retrieved 24 weeks after transplantation for insulin immunostaining. The islets were positive for insulin staining even in the PVA group (W-L); however, their normal appearances were somewhat changed (Fig. 5B).
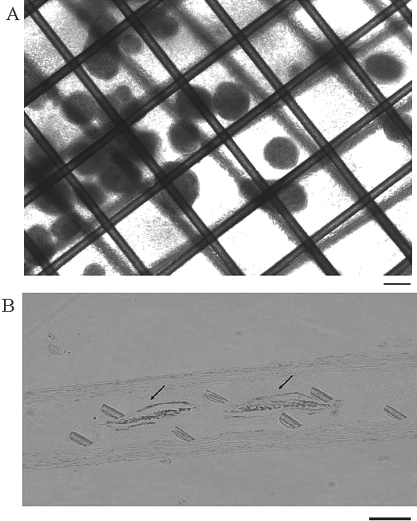
Dithizone staining before PVA MEI transplantation (A). Original magnification: 10x. Insulin immunostaining of the PVA group (W-L) 24 weeks after transplantation (B). Islets within PVA MEIs (arrows) were positive for insulin staining; however, their normal appearances were somewhat changed. Original magnification: 100x. Scale bars: 100 μm.
Discussion
The rat islets were encapsulated using PVA hydrogel and used for experiments in vitro and in vivo. The results demonstrated that the PVA MEIs could protect islets against the discordant immune attack by humoral factors, and transplantation of PVA macroencapsulated Wistar rat islets could reverse hyperglycemia and prevent loss of body weight in STZ-diabetic Lewis rats.
An immuoisolation material must be harmless to transplanted grafts and recipients, and have selective permeability for different kinds of molecules. Demerlis et al. (6) proved the nontoxicity of PVA and Burczak et al. (5) investigated its permeability in their studies. Several investigations of PVA MEIs have been conducted in this laboratory (9,11,17,25,26); in particular, Qi et al. (18) recently evaluated the function of PVA MEIs in islet cryopreservation. Sakata et al. (23,24) found that PVA MEIs could ameliorate diabetic renal dysfunction. These findings indicated the efficacy of PVA MEIs in type 1 DM therapy.
Hyperacute rejection is a complement-mediated response that occurs in a discordant immune response, and causes transplanted xenografts to lose their functions within a short time. Badet et al. (1) and Bennet et al. (2,3) reported the influence of hyperacute rejection in islet transplantation. Preventing contact between a transplanted xenograft and complement should inhibit the hyperacute rejection. The current study used FHP as a source of immune attack to confirm the immunoisolation effect of PVA macroencapsulated Wistar rat islets in a discordant immune rejection. The free islets of the FHP groups showed a lower recovery rate in comparison to the 10% heat-inactivated FBS group (Fig. 2A), but PVA MEIs showed no significant differences between the FBS and FHP groups (Fig. 2B). The morphological changes (Fig. 1) and insulin content (Fig. 3) demonstrated that PVA MEIs could protect islets and maintain their function when cultured with FHP. Therefore, these findings demonstrated the immunoprotection effect of PVA MEIs from the discordant humoral factors.
A previous study (17) in mice using the same PVA MEIs showed excellent results with xenotransplantation and maintained normoglycemia for 1 month. However, the duration of the effect was not evaluated. Therefore, one of the aims of this study was to extend the period of observation and determine the duration of the effect of PVA MEIs after allotransplantation. The NFBG was maintained at a relatively lower level for the initial 6 weeks following transplantation, followed by an increase in the PVA group (W-L) (Fig. 4A), suggesting that the immuoisolation effect of PVA MEIs might become ineffective 6 weeks after allotransplantation, although the statistical analysis did not show a significant difference between the PVA (W-L) and PVA (L-L) groups. The exact reasons for the reduction of graft function are not clear. However, the following explanations may be possible. 1) Breakage of whole or parts of MEIs might allow islets to be attacked by the host immune system. Therefore, the islets would lose their functions and the recipients would become hyperglycemic again. Omer et al. suggested that capsulated islets in larger animals (rats) have more breakage than in smaller animals (mice), probably due to the higher abdominal pressure from the visceral weight, activity, and peristaltic forces (7,16). However, no obvious breakages was observed in this study when MEIs were retrieved 24 weeks later; suggesting that PET mesh-reinforced MEIs is robust to such physical forces. 2) Fibrotic tissue due to foreign body reaction. Yang et al. suggested that fibrotic tissue on the surface of encapsulated islets could envelope the graft and block the diffusion of some life-essential sub-stances, therefore leading to dysfunction of islets (33). However, no thick fibrotic tissue was observed on the surface of retrieved PVA MEIs, although a thin layer of mesenchymal cells was attached (Fig. 5B). 3) Ischemic necrosis of islets. Small molecules including oxygen and some nutrients are allowed to exchange freely through the PVA semipermeable membrane (data not shown) only depending on passive diffusion to maintain the normal metabolism and function of islets inside. The thickness of this device was 1 mm and the islets inside were not evenly but randomly distributed. These two factors (i.e., thickness of the gel and uneven distribution of islets) may cause hypoxia in some parts of islets inside, therefore leading to ischemic necrosis of MEIs.
At the same time, the NFBG not only in the PVA group (W-L) but also in the PVA group (L-L) of iso-transplanted MEIs failed to reach the normal level after transplantation with a significant difference from the free islets isotransplantation group (L-L group; p < 0.05) (Fig. 4A). No immune rejection should occur with isotransplantation. Therefore, even though the exact reasons for higher NFBG in the PVA group (L-L) are unclear, this difference could be attributed to some factors associated with the encapsulation. Sheet-type MEIs were produced by using a freezing–thawing technique. The most important problem with the cryopreservation of islets is the reduced number and function in comparison to fresh islets because of cryodamage (13,20). The islets were pretreated with Cell Banker (cell cryopreservative medium), then suspended in the PVA solution that was supplemented with 5% DMSO as cryoprotectants in a previous study (18) and in the present study. Furthermore, Rajotte et al. (19) suggested that slow cooling and rapid thawing could protect islets better during cryopreservation. Therefore, unlike the previous study, the currently study used a computer-controlled program freezer to reduce the cryodamage to islets by making the cooling rate slower. Although several trials have been attempted to reduce the cryodamage of islets during cryopreservation, a considerable loss of islets number and function is unavoidable and probably led to the decreased MEIs function in this study.
Although the objective of clinical islet transplantation is to make type 1 DM patients insulin independent, this is difficult to achieve in practice. The Edmonton group found that only 10% patients remained insulin independent at 5-year postislet transplantation (22). Therefore, at the present stage, the practical aim of therapy in islet transplantation is stabilization of the blood glucose level, which leads to remarkable improvement of the disease. Although transplantation of PVA MEIs did not normalize the NFBG of recipients, the NFBG remained within a relatively lower range and prevented body weight loss, suggesting that PVA MEIs can, at least, attenuate severe hyperglycemia without immunosuppression. Considering the current state of clinical islet transplantation and the survival of beta cells in PVA MEIs 24 weeks after transplantation (Fig. 5B), PVA MEIs seems to be a potential and hopeful alternative to islet transplantation.
This study confirmed the immunoisolation effect of PVA hydrogel on discordant immune rejection and allotransplantation. PVA hydrogel could protect rat islets against FHP in vitro. In vivo, transplanted PVA Wistar rat MEIs decreased the STZ-induced diabetic Lewis rat NFBG level and prevented body weight loss. In conclusion, this study showed the immunoprotective effect of PVA MEIs against discordant xenoimmunoreaction in vitro and lasting but gradually subsiding effect of PVA MEIs transplantation on severe DM in rats. PVA MEIs seem to be a potential and hopeful alternative to islet transplantation, although further improvements are obviously necessary.
Footnotes
Acknowledgments
This research was partially supported by the Ministry of Education, Science, Sports and Culture, Grant-in-Aid for Scientific Research (B), (No. 18390364) 2006–2007 and Pancreas Research Foundation of Japan. The authors thank Dr. Yoshinobu Toda for his support in immune-staining. The authors declare no conflict of interest.
