Abstract
Mucopolysaccharidosis type I (MPS-I; Hurler syndrome) is an inborn error of metabolism caused by lack of the functional lysosomal glycosaminoglycan (GAG)-degrading enzyme α-l-iduronidase (IDUA). Without treatment, the resulting GAG accumulation causes multisystem dysfunction and death within the first decade. Current treatments include allogeneic hematopoietic stem cell transplantation (HSCT) and enzyme replacement therapy. HSCT ameliorates clinical features and extends life but is not available to all patients, and inadequately corrects the most devastating features of the disease including mental retardation and skeletal deformities. Recent developments suggest that stem cells can be used to deliver needed enzymes to the central nervous system. To test this concept, we transplanted bone marrow-derived normal adult human MultiStem® cells into the cerebral lateral ventricles of immunodeficient MPS-I neonatal mice. Transplanted cells and human-specific DNA were detected in the hippocampal formation, striatum, and other areas of the central nervous system. Brain tissue assays revealed significant long-term decrease in GAG levels in the hippocampus and striatum. Sensorimotor testing 6 months after transplantation demonstrated significantly improved rotarod performance of transplanted mice in comparison to nontransplanted and sham-transplanted control animals. These results suggest that a single injection of MultiStem cells into the cerebral ventricles of neonatal MPS-I mice induces sustained reduction in GAG accumulation within the brain, and modest long-term improvement in sensorimotor function.
Keywords
Introduction
Mucopolysaccharidosis type I (MPS-I; Hurler syndrome) is caused by a deficiency of the lysosomal glycosaminoglycan (GAG)-degrading enzyme α-l-iduronidase (IDUA) (37). Progressive neuropsychological deterioration is one of the most devastating manifestations of this disease (32,60). Allogeneic hematopoietic stem cell transplantation (HSCT) provides IDUA, clears accumulated GAGs, and markedly improves survival (36,41,42,45,52). However, GAG clearance from the brain is incomplete since it depends on the central nervous system (CNS) penetration of small numbers of donor-derived microglia. Also, HSCT does not provide trophic/chaperone neuroprotective effects on diseased or damaged host neurons (12,56–59). Amelioration of neurocognitive and adaptive functions (communication, daily living skills, socialization, and motor function) is therefore inadequate; stabilization or slower than normal improvement occurs, largely restricted to younger patients who have better baseline cognitive function and achieve stable long-term engraftment (6,43,52). Enzyme replacement therapy (ERT) cannot improve neurological function since IDUA does not cross the blood–brain barrier (25,36). Novel cellular therapeutic approaches directed at the CNS (31) are therefore needed, both as an adjunct to allogeneic HSCT or ERT, and for patients unable to receive a HSCT. Such approaches need evaluation in animal models of MPS-I (13,20). We recently confirmed that an immunodeficient mouse model of MPS-I [non-obese diabetic/severe combined immunodeficient (NOD/SCID)/MPS-I] exhibits the biochemical and pathological features of human Hurler syndrome, including abnormalities in sensorimotor function and training (17). We and others showed that the immunodeficient background makes this model suitable for transplantation with human (xenogeneic) mesenchymal stromal cells (MSCs) (35) and gene therapy (3,13,44).
Pioneering work from Snyder and colleagues on intracranial transplantation of murine (51) and fetal human neural stem cells (NSCs) (28) in animal models of lysosomal storage diseases (LSDs) provided compelling proof of principle of the enormous potential of cellular therapy directed at the CNS. Recent studies from our group identified a different type of multipotent progenitor cell in postnatal (adult) human and mammalian bone marrow. Such cells grow without senescence (22,23,26,63,64), expand sufficiently for clinical use in humans (40), differentiate into cells representing all three embryonic layers including neuronal cells (4,21–23,26), maintain genetic stability and telomere length (8), and exhibit an epigenetic profile and differentiation potential more primitive than MSCs (1,8). We showed that normal human bone marrow-derived multipotent stem cells (similar to the MultiStem® cells used in the current study) secrete sufficient IDUA to cross-correct GAG accumulation and metabolism [correction of abnormal size (Mr 5 kDa, vs. normal size being 42 kDa) and sulfation pattern of heparan sulfate] in bone marrow-derived multipotent stem cells cultured from the marrow of patients with Hurler syndrome (19).
In vivo, we and others showed that these types of cells [rodent/human MSCs as well as multipotent adult progenitor cells (MAPCs; similar to MultiStem cells)] exert beneficial trophic/chaperone and immunomodulatory effects following transplantation in animal models of various diseases [reviewed by Auletta et al. (4)]. Human multipotent stem cells differentiate into various lineages and induce functional improvement in peripheral vascular ischemia in mice (2), and neurological improvement in rat models of cerebral ischemia (33,63) and traumatic brain injury (55). Importantly, adult human multipotent stem cells can easily be obtained from bone marrow aspirates and expanded rapidly in vitro, and are unencumbered with ethical concerns associated with human embryonic stem (ES) cells or fetal NSCs. Such cells [MultiStem®; expanded under good manufacturing practice (GMP) conditions by Athersys Inc., Cleveland, OH] are currently in phase I clinical trials in acute myocardial infarction, ischemic stroke, and allogeneic HSCT (8,33).
We hypothesized that intracerebroventricular (ICV) transplantation of MultiStem cells may provide IDUA in the brain and trophic/chaperone neuroprotective effects in MPS-I. The ICV route was used because it has the potential to (a) achieve widespread distribution of the transplanted cells, (b) provide transplanted cells in the meninges, wherein extensive GAG accumulation occurs and results in hydrocephalus in MPS-I, and (c) be clinically applicable to the treatment of patients in the future. The effect of transplantation was tested in neonatal mice since (a) the microenvironment of the brain is more conducive to stem cell survival and proliferation at an early age, and (b) neuropathology has not yet developed at this age, nor has meningeal accumulation of GAGs causing restriction of cerebrospinal fluid flow that might impair the distribution of transplanted cells. Further, the strategy used here will be more applicable towards future clinical use as it will be ideal to perform transplantation prior to the development of irreversible neuropathology and neurobehavioral deficits in patients.
This is the first report of the biochemical and functional effects of transplanting adult human bone marrow-derived multipotent (MultiStem) cells into the cerebral lateral ventricles of neonatal immunodeficient MPS-I mice.
Materials and Methods
Animals
Approval for all studies performed with animals was obtained from the Institutional Animal Care and Use Committee (IACUC) of the University of Minnesota. Immunodeficient NOD.129(B6)-PrkdcscidIduattm1Clk/J mice heterozygous for the IDUA mutation were purchased from The Jackson Laboratory (Bar Harbor, ME). Heterozygous mice were mated, producing approximately 20–25% homozygous NOD/SCID/MPS-I (IDUA-/-) mice (termed MPS-I hereafter), 50% heterozygous (IDUA+/-) mice (termed Ht), and 25% wild-type (IDUA+/+) offspring (termed WT). Mating of MPS-I males with Ht females, when successful, produced a higher proportion of MPS-I offspring. We have recently described the biochemical, phenotypic, and behavioral characteristics of this mouse model of MPS-I (3,17).
Mice were genotyped using tail clip samples, as described previously (17), according to the protocol on The Jackson Laboratory website (http://jaxmice.jax.org/strain/004083.html). The following primers were used: forward oIMR 1451 (5′-GGAACTTTGAGACTTGG AATGAACCAG-3′); WT reverse oIMR 1452 (5′-CATT GTAAATAGGGGTATCCTTGAACTC-3′); MPS reverse oIMR 1453 (5′-GGATTGGGAAGACAATAGCAGG CATGCT-3′). PCR products were resolved on a 1.5% agarose gel. Genotype assignment of individual animals was confirmed by repeating genotyping on tissue samples obtained at euthanasia.
A total of 80 mice contributed to the data included in the current article. Fourteen neonates were sacrificed at varying periods (from immediately after transplantation to 2 weeks after transplantation) for immunofluorescence staining of the brain (shown in Figs. 1 and 2). Sixty-six mice were used for the other studies and assays; these included 18 WT, 10 Ht, and 38 MPS animals; 58% were males.
ICV Transplantation of MultiStem Cells
Transplantation was performed on neonatal mice between postnatal days 3 and 6. One day prior and 1, 4, 7, and 11 days following transplantation, all neonates received an anti-natural killer (NK) cell antibody (10 μl/ g body weight IP, anti-asialo GM1, cat. #986-10001, Wako Pure Chemical Industries Ltd., Osaka, Japan) to enhance MultiStem cell survival in vivo. Previous studies have shown that mice of this immunodeficient (NOD/SCID) background can develop NK cell-mediated rejection of transplanted stem cells, which can be ameliorated by peritransplant administration of NK cell-blocking antibodies (34,48).
Human MultiStem cells were obtained in frozen aliquots from Athersys Inc. (Cleveland, OH). Immediately before transplantation, cells were thawed, centrifuged in sterile PBS to remove dimethyl sulfoxide (DMSO), counted and evaluated for viability using trypan blue exclusion, and resuspended in PBS. A midline incision was made in the disinfected skin of the scalp, which was then retracted bilaterally, exposing the external auditory orifices to facilitate placement of ear bars. The maxilla was set on a tooth bar, immobilizing the head in a Kopf head holder. The neonate's body was then placed on tin foil kept on top of a bed of ice. Stereotactic injection was performed to target the right lateral cerebral ventricle. A hole was made on the skull (coordinates: 0.0 mm anterior, 0.5 mm right lateral to bregma). Since no bony markers form at this age, orientation was achieved by visualization of the juncture of several cartilages under the surgical microscope. A Hamilton needle attached to a syringe loaded with 100,000 MultiStem cells in 3 μl PBS was introduced 1.8 mm deep from the cortex. After waiting for 1 min, the cell suspension was injected into the right lateral cerebral ventricle using a micropump (Convection Enhanced Device) at a rate of 0.5 μl/min. The needle was kept in place for another 2 min to minimize leakage and help diffusion of injected cells in ventricular system. The wound was then closed and recovery by each animal was achieved under a heating lamp. Once fully awake, the pups were returned to their parents. Genotyping was performed after weaning at 3 weeks of age. Since the genotype was not known at the time of transplantation, we randomly chose one to two neonates/litter as negative controls for injecting the 3 μl of PBS alone (sham transplantation). This also ensured that all genotypes of mice, including the MultiStem cell-transplanted and sham-transplanted mice, were litter-mates. The individuals performing the genotyping, behavioral tests, immunohistochemistry, biochemical measurements, and behavioral tests were blinded to the identity of the sham-transplanted mice; this was only disclosed at the time of data analysis.
Complications of the transplantation procedure included abscess formation on the head for which seven mice were sacrificed. Histopathological examination identified these as Staphylococcal in etiology. Two of these mice had received sham transplants (PBS injection), making it unlikely that the abscesses were related to MultiStem cells themselves or to contamination of the cell product. There were several instances of the mothers cannibalizing the transplanted neonates (three litters, and sporadic additional mice), resulting in loss of those animals for subsequent analysis. Of note, human MultiStem cells were not identified in any of the tumors (e.g., thymomas) that are known to develop in these immunodeficient mice, or teratomas that are associated with embryonic stem cells.
Immunohistochemistry
To evaluate survival and distribution of transplanted MultiStem cells, some mice were sacrificed at different intervals after transplantation. Animals were studied for histology immediately after injection and at 1, 3, 7, and 14 days after injection, or at the end of the study following behavioral testing (about 24 weeks).
For neonates 1–5 days of age, animals were anesthetized by hypothermia or with ketamine/xylazine administration for animals greater than 5 days of age, and then decapitated. Brains were then taken out and immersed in 4% ice-cold paraformaldehyde for 24 h, replaced with 30% sucrose for at least 48 h or until the brains sank to the bottom. These brains were cut on a microtome, 30 μm in thickness.
For adult mice at the end of the experiment (about 24 weeks), all mice were anesthetized with injection of ketamine/xylazine mixture, a transcardial perfusion was performed with ice-cold PBS, brains were cut in half longitudinally, right hemispheres (injection side) were immersed in 4% paraformaldehyde for 24 h of fixation, and placed in 30% sucrose until they were cut with a microtome. For biochemical studies the left hemispheres were kept on a bed of ice after removal from the skull, and with the aid of a surgical microscope, the striatum, hippocampus, cortex, cerebellum, and rest of the brain were dissected and immediately flash frozen and stored at −80°C for subsequent use in endogenous retrovirus-3 (ERV-3) real-time quantitative PCR, GAG quantification and IDUA assays.
For immunohistochemistry, brain sections were rinsed with PBS, blocked with 5% normal donkey serum (NDS) + 0.3% Triton X-100 in PBS (blocking medium) for 1 h at room temperature, then incubated with mouse anti-human nuclei (Millipore, Temecula, CA, Mab 1281, 1:100) in blocking medium at 4°C overnight, followed by a PBS wash, then incubated with AlexaFluor 488 donkey anti-mouse IgG (Invitrogen, Carlsbad, CA, A-21202, 1:500) + DAPI (Sigma, USA, D9542, 1:500) in blocking medium for 1 h at room temperature, followed by a final PBS wash. Stained sections were mounted and cover-slipped. Tissue sections were examined using a Nikon microscope (ECLIPSE E600) and images were processed using Diagnostic Instruments, Inc.'s SPOT Insight and Adobe Photoshop.
Real-Time Quantitative PCR for ERV-3
To detect and quantify small numbers of surviving human MultiStem cells that were transplanted into the mouse brain, endogenous retrovirus (ERV)-3 real-time quantitative PCR was performed on various regions of the brains of sham and transplanted mice euthanized approximately 24 weeks after transplantation. DNA was isolated from the frozen brain samples using the Qiagen DNeasy Blood and Tissue kit. Care was taken to avoid human contamination during DNA purification by using aerosol barrier pipette tips and tubes certified to be free of human DNA. Human DNA was detected by real-time quantitative PCR using primers specific for ERV-3, an endogenous retrovirus found in the human genome but not in rodents (29). The real-time PCR reaction contained 50 ng DNA with 0.4 mM of each primer (forward ERV-3 primer: 5′-atgggaagcaagggaactaat and reverse ERV-3 primer: 5′-cccagcgagcaatacagaattt) and Invitrogen's Platinum SYBR Green qPCR supermix. The following PCR profile was performed on an Applied Biosciences 7900HT Fast Real Time PCR thermocycler: 50°C for 2 min, 95°C for 10 min, and then 45 cycles of denaturation at 95°C for 15 s and annealing at 58°C for 1 min. A final dissociation step of 95°C for 15 s, 60°C for 15 s, and 95°C for 15 s concluded the amplification. A standard curve was generated using serial dilutions of human MultiStem cell DNA mixed with mouse brain DNA. PCR products were resolved on a 2% agarose gel to confirm the correct product size. Individual samples of the brains were classified as being positive if the Ct value was <40 cycles on real-time PCR, and the product from the PCR reaction was confirmed to be of the correct size on gel electrophoresis.
Measurement of GAG Levels and IDUA Activity
GAG levels and IDUA enzyme activity were determined in flash-frozen brain tissue samples from transplanted untransplanted, and sham-transplanted mice. The methods used were as described by us previoiusly (17).
GAG Content
Proteins in clarified supernatants of brain homogenates were removed by digestion with Proteinase K, and nucleic acids by DNase + RNase. GAG concentration was determined using the Blyscan Sulfated Glycosaminoglycan Assay (Accurate Chemical & Scientific Corp., Westbury, NY), and expressed as μg GAG/mg protein.
IDUA Activity
Brain samples were homogenized in 1 ml PBS, then treated with 11 μl of 10% Triton X-100 in PBS on ice for 10 min. Protein concentration in the supernatant was estimated using the Bradford method, and IDUA activity assayed according to Schuchman et al. (47). 4-Methylumbelliferyl α-l-iduronide (50 μM final conc.) was made in 0.4 M sodium formate buffer, pH 3.5, with 0.2% Triton X-100; 25 μl was added to 25 μl brain homogenate, incubated for 1 h at 37°C in the dark, then quenched with 200 μl of 0.5 M NaOH/glycine buffer, pH 10.3. Fluorescence of the supernatant was read at 365 nm excitation wavelength and 450 nm emission wavelength. Specific activity of IDUA was expressed as nmol/h/mg protein.
In addition, for measuring IDUA activity, normal human MultiStem cells and HEK293T human embryonic kidney cells were cultured in T-75 tissue culture flasks. MultiStem cells (obtained frozen from Athersys) were thawed and plated for a few days in 54% DMEM (Invitrogen) + 40% MCDB (Sigma) supplemented with 2% fetal bovine serum (FBS), penicillin/streptomycin, 1x insulin-transferrin-selenium, 100 μM ascorbic acid, 55 μM β-mercaptoethanol, and 5 ng/ml each of platelet-derived growth factor (PDGF)-BB (R&D Systems, Minneapolis, MN) and epidermal growth factor (Sigma). HEK293T were cultured in DMEM supplemented with 10% FBS, penicillin/streptomycin, 4 mM glutamine, 1 mM sodium pyruvate, and 1.5 mg/ml sodium bicarbonate. When the cells were nearly confluent, media were replaced by PBS + 0.1% BSA. After overnight incubation, IDUA activity was separately assayed in the supernatant and in the cell lysates.
RotaRod Test for Sensorimotor Function
Sensorimotor function was tested using a Rotarod machine (Model 8200 LSi Letica, PANLAB, Spain). The rotarod test measures sensorimotor function and coordination (14). We have previously validated in this mouse model (17) and in another mouse model (24) that the range of rotarod speeds used in this study (5–25 rpm) is appropriate, tests the range of sensorimotor ability, and identifies motor abnormalities. It is important to test a range of speeds, since, as shown in the present study, abnormal phenotypes of mutant animals may only be detected at higher speeds and/or consecutive days of testing.
Untransplanted/sham-transplanted WT, Ht, and MPS-I mice, as well as MultiStem cell-transplanted MPS-I mice were tested using the procedure described by us previously (17), for 5 sequential days immediately prior to sacrifice at approximately 24 weeks of age. The performance of untransplanted/sham-transplanted WT mice was equivalent to that of Ht mice; therefore, these two genotypes were grouped together as the control cohort for the rotarod test. The mean ages of the controls, untransplanted/sham-transplanted MPS-I mice, and MultiStem cell-transplanted MPS-I mice were 16 ± 0.7, 18 ± 1.6, and 22 ± 1.0 weeks, respectively. Differences in ages were not statistically significant except for the MultiStem cell-transplanted MPS-I mice being older than the control cohort (p < 0.0001). Day 1 of testing established a baseline of sensorimotor function in each animal. Improvement in balance time over 5 repetitive days of testing provided a measure of the ability of individual mice to improve their sensorimotor functioning with training.
Statistical Analysis
Data were analyzed using GraphPad Prism 5.0 software (GraphPad Software Inc., San Diego, CA). Results were expressed as mean ± SE. The significance of differences in GAG level between groups was examined using one-way ANOVA. Rotarod test results were evaluated and compared using two-way repeated measures Model I ANOVA. The subject (matching) p-values were low for all comparisons (all p < 0.002), indicating that the repeated measures design was appropriate and was effective in controlling for variability between mice. The Tukey-Kramer posttest was used with ANOVA.
Results
Distribution of Human MultiStem Cells Transplanted Into the Mouse Brain
Immediately after transplantation and at various post-transplant time periods some mice were sacrificed to determine the distribution of the MultiStem cells within the brain. MultiStem cells were identified using an antibody that recognized human cells (MAb 1281). Immediately after transplantation, MultiStem cells were observed predominantly in the cerebral lateral ventricles near the injection site, striatum, and medial septum in the 4th ventricle (Fig. 1A–F). At more distal regions from the injection site, MultiStem cells were observed near the hippocampal formation and habenula (Fig. 1G–O). One day after transplantation, transplanted cells were still found near the hippocampal formation and 3rd ventricle (Fig. 1P–X). In some instances clusters of cells appeared to be integrating with the ependymal cells (Fig. 1S–U). At this time period clusters of cells were also observed in the 4th ventricle near the cerebellum (Fig. 2A–I). These cells also appeared to be penetrating through the ependymal layer. Three days after transplantation, MultiStem cells were found integrated within the brain parenchyma dorsal to the lateral cerebral ventricle (Fig. 2J–L). By day 14 grafted cells could be observed in the habenula near the 3rd ventricle (Fig. 2M–R). In general MultiStem cells were easily detected within the brain up to 1 week posttransplantation, but their presence began to diminish thereafter.

Human cells were identified using a mouse-anti-human nucleus antibody (shown in green). DAPI counterstain (blue) was used to visualize all nucleated cells in the sections. (A–F) Localization of Multistem cells in the cerebral lateral ventricle immediately after transplantation. (A–C) Multistem cells were observed in the cerebral lateral ventricles (LV) on the side of injection immediately after transplantation and also could be found in subarachnoidal space (bold arrows). (D–F) Enlargement of boxed area from (A). CTX, cortex; ML, midline; SPT, septum; STR, striatum. Scale bars: 250 μm (B), 50 μm (E). (G–O) Localization of MultiStem cells in the cerebral ventricular system immediately after transplantation. Multistem cells were observed in the cerebral ventricle around the hippocampus on the side of injection [boxed area (J–L) in G] and midline [boxed area (M–O) in G] immediately after transplantation. (G–I) Low magnification of ventricular system surrounding hippocampus (HF), dentate gyrus (DG), and midline (ML). Scale bar: 250 μm (H). (J–O) High magnification of the corresponding areas boxed in (G), ventricle surrounding hippocampus (J–L), ventricle in the midline (M–O). Scale bar: 50 μm (K, N). (P–X) Localization of MultiStem cells adjacent to the hippocampus and in the 3rd ventricle. (P–R) Multistem cells were observed forming groups near the fornix [boxed area (S–U) in P] and in the 3rd ventricle [boxed area (V–X) in P] 24 h after transplantation. (S–U) Enlargement of boxed area S–U (fornix) from (P). (V–X) Enlargement of boxed area V–X (3rd ventricle) from (P). 3rd V, 3rd ventricle; HB, habenular nuclei, HF, hippocampus; FX, fornix. Scale bars: 250 μ (Q), 50 μm (T, W).

Human cells were identified using a mouse-anti-human nucleus antibody (shown in green). DAPI counterstain (blue) was used to visualize all nucleated cells in the sections. (A–I) Localization of MultiStem cells in the 4th ventricle. (A–C) Multistem cells were observed forming groups in the 4th ventricle near the cerebellum and brain stem 24 h after injection into the cerebral lateral ventricle. (D–F) Enlargement of boxed area (D–F) (floor of 4th ventricle) from (A). (G–I) Enlargement of boxed area (G–I) (cerebellum) from (A). 4th V, 4th ventricle; BS, brain stem; CB, cerebellum. Scale bars: 250 μm in length (B), 50 μm (E, H). (J–L) Localization of MultiStem cells in the cerebral lateral ventricles. Three days after transplantation, MultiStem cells were observed near the wall of the cerebral lateral ventricle (LV) on the side of injection. STR, striatum; SPT, septum. Scale bar: 50 μm (K). (M–R) Localization of MultiStem cells in the 3rd ventricle. (M–O) Multistem cells could be found in the 3rd ventricle near the habenula at 14 days after transplantation. (P–R) Enlargement of boxed area (P–R) (3rd ventricle and habenula area) from (M). Bold arrows in panels (P–R) indicate MultiStem cells. HB, habenula; 3rd V, 3rd ventricle; PV, paraventricular nucleus. Scale bars: 50 μm (N), 25 μm (Q).
Survival of Human MultiStem Cells in the Mouse Brain
ERV-3 is an endogenous retrovirus incorporated into the human genome but not in the genomes of rodents. Detection and enumeration of ERV-3 copies by realtime quantitative PCR therefore provides a sensitive method to detect and quantitate low levels of transplanted human cells surviving in the mouse brain (29). In control samples used to generate a standard curve (Fig. 3A), ERV-3 could consistently be detected down to a dilution of 0.0125% human MultiStem cell DNA in mouse brain DNA. Thus, this method was capable of detecting about 1 human genome (cell) in 8,000 mouse genomes (cells). Figure 3B shows one representative image of gel electrophoresis that was performed on all PCR products and appropriate controls to confirm the presence of an ERV-3 product of the correct size. The table in Figure 3C shows the frequency of detection of ERV-3 (indicative of surviving human MultiStem cells) in microdissected regions of sham- and MultiStem cell-transplanted mice, sacrificed approximately 24 weeks after transplantation. As expected, ERV-3 was not detectable in sham-transplanted mice. In contrast, ERV-3 was detected with variable frequency in the brains of MultiStem cell transplanted WT, Ht, and MPS-I mice. Overall, ERV-3 was detected somewhat more frequently in the hippocampus and striatum, compared to the cerebral cortex and cerebellum. The level of long-term (24 weeks) engraftment and survival of human cells was quite variable, but <0.1% in most samples where human DNA was quantifiable (Fig. 3D). These results suggest that small numbers of human MultiStem cells survive for at least 24 weeks following ICV transplantation into neonatal immunodeficient (NOD/SCID) mice, irrespective of the presence or absence of host IDUA. Human cells appeared to survive somewhat more often in the same regions of the brain (Fig. 3), which demonstrated significant clearance of accumulated GAGs (hippocampus and striatum) (Fig. 4). However, a direct correlation of ERV-3 with GAG clearance could not be determined since the ERV-3 PCR could not be performed on all the samples in which GAGs were measured.

Detection of human cells in the brains of transplanted mice. (A) ERV-3 real-time PCR was performed on samples of serial dilutions of human MultiStem cell DNA mixed with mouse brain DNA. ERV-3 copies were consistently detected down to a 0.0125% human DNA dilution, which corresponds to 1 human cell in 8,000 mouse cells. Ct, cycle threshold, the cycle number at which PCR product is first detected. (B) The products from the real-time PCR for ERV-3 were run on a 2% agarose gel for confirmation of product size. Lane 1: human MultiStem cell DNA; lanes 2 and 5: untransplanted mouse brain DNA; lane 3: untransplanted mouse tail DNA; lane 4: mouse brain DNA isolated immediately after transplantation of human MultiStem cells; lane 6: molecular weight markers. The arrow marks the 135-bp ERV-3 product and the asterisk marks the primer dimer band. (C) The table shows the frequency of detectable human cells (ERV-3 PCR positive) in microdissected regions of the brains of shamor MultiStem cell-transplanted mice at approximately 24 weeks of age. The numerator indicates the number of positive mice, and the denominator indicates the total number of individual animals tested. The percentage thus indicates the proportion of animals whose brains showed detectable human MultiStem cell DNA. (D) The percentage of human DNA detectable in different regions of the mouse brain at 24 weeks after transplantation of MultiStem cells was calculated using the standard curve shown in (A). WT, wild type; Ht, heterozygous; MPS, homozygous α-l-iduronidase (IDUA) knockout mice.

Reduction in GAG accumulation in the brain. GAG levels were measured in the hippocampus, striatum, cortex, cerebellum, and remaining brain of sham-transplanted (N = 3) or MultiStem cell-transplanted (N = 10) WT mice (total = 13); untransplanted (N = 9) or sham-transplanted (N = 12) MPS-I mice (total = 21); and MultiStem cell-transplanted MPS-I mice (N = 9) (MPS-I tpt.) at about 24 weeks of age. The horizontal line indicates the mean value for each group. GAG levels were significantly reduced in the hippocampus, striatum, and remaining brain of MPS-I mice transplanted with MultiStem cells compared to untransplanted/sham-transplanted MPS-I mice. GAG levels in all regions of the brains of untransplanted/sham-transplanted MPS-I mice were significantly higher than in corresponding regions of the brains of WT mice (p < 0.001 for all). Significance of differences between the indicated conditions: *p < 0.05, **p < 0.01, ***p < 0.001; NS (not significant): p > 0.05.
Reduction in GAG Accumulation in the Brains of Transplanted MPS-I Mice
GAG concentration was measured in the hippocampal formation, striatum, cerebellum, cortex, and the remainder of the brains of sham-transplanted or MultiStem cell-transplanted WT, sham-transplanted or untransplanted MPS-I mice, and MPS-I mice-transplanted with human MultiStem cells (Fig. 4). GAG levels were significantly elevated in all regions of the brains of untransplanted or sham-transplanted MPS-I mice compared to WT mice (p < 0.001 for all regions of the brain). ICV transplantation of human MultiStem cells into neonatal MPS-I mice resulted in a statistically significant long-term (at ~24 weeks) decrease of GAG levels in the hippocampus, striatum, and remaining brain. The slight reduction in GAG levels in the cortex and cerebellum did not reach statistical significance.
Long-term amelioration of GAG accumulation in the hippocampus and remaining brain was partial, since GAG levels in these regions in transplanted MPS-I mice did not completely return to WT GAG levels. Interestingly, however, GAG levels in the striatum of transplanted MPS-I mice became comparable to those of the WT animals (p > 0.05).
Iduronidase Enzyme Activity in MultiStem Cells and in the Mouse Brain
We have previously shown that human bone marrow-derived multipotent progenitor cells secrete functional IDUA that cross-corrects similar cells obtained from the bone marrow of patients with Hurler syndrome (19). We now confirm that the MultiStem cells used for the transplantation studies reported here also synthesize and secrete the enzyme. IDUA activity (9.956 ± 0.238 nmol/ h/mg protein) was present in the lysates of MultiStem cells in culture, and was detectable in the culture supernatant (0.189 ± 0.003 nmol/h/ml) of these cells harvested after overnight incubation. Enzyme activity in the lysates and supernants of MultiStem cells was substantially greater than IDUA activity in HEK293T cells (lysates: 0.228 ± 0.007 nmol/h/mg protein and culture supernatant: 0.011 ± 0.001 nmol/h/ml).
As expected, IDUA activity was undetectable in the brains of untransplanted MPS-I mice. At 24 weeks after transplantation of MPS-I mice with human MultiStem cells, IDUA activity was undetectable in the brain. The lack of IDUA activity in transplanted MPS-I mice may indicate that enzyme levels have fallen below the level of detection of the assay at 24 weeks after transplantation, consistent with the loss of most of the transplanted cells by this time.
Improvement in Sensorimotor Function
In males, the rotarod test demonstrated significant improvement in sensorimotor function (Fig. 5A–D) in MPS-I mice that received transplants of MultiStem cells.

Improvement in sensorimotor function. Mice were tested on the rotarod apparatus for 5 sequential days immediately prior to sacrifice at approximately 24 weeks of age. The time for which male mice were able to stay balanced on a rod rotating at different speeds is shown as the mean ± SE in seconds. Untransplanted/sham-transplanted WT (N = 5) and Ht (N = 6) mice (total = 11): open triangles with broken line; untransplanted/sham-transplanted MPS-I mice (N = 7): open circles; MultiStem cell transplanted MPS-I mice (N = 5): filled circles. The performance of MultiStem cell transplanted MPS-I mice became comparable to that of untransplanted/sham-transplanted WT and Ht mice at all three speeds. Significance of differences between the three groups of mice are detailed in the Results section. (A–C) Balancing time on the rotarod at 5, 15, and 25 rpm, respectively, over 5 days of testing. (D) Performance at different speeds on the fifth day of testing, demonstrating the ability of the mice to improve sensorimotor functioning upon repetitive practice.
At 5 rpm
The performance of all three groups (untransplanted/sham-transplanted WT and Ht, untransplanted/sham-transplanted MPS-I, and MultiStem cell-transplanted MPS-I mice) improved consistently and significantly on sequential days of testing (p < 0.0001), but to a comparable extent; that is, at this low speed, the performance of untransplanted/sham-transplanted MPS-I mice was not inferior to that of untransplanted/sham-transplanted WT and Ht mice and that of MultiStem cell-transplanted MPS-I mice (significance of interaction between sequential days of testing and groups of mice: p > 0.05).
At 15 rpm
The performance of all three groups of mice improved significantly on sequential days of testing (p < 0.002). There was no significant interaction between sequential days of testing and groups of mice (p > 0.05), indicating that the improvement in performance on sequential days of testing was consistently present in all three groups of mice. However, at this intermediate speed, there was a strong trend towards the performance of MultiStem cell-transplanted MPS-I mice being consistently superior to that of untransplanted/ sham-transplanted MPS-I mice (p = 0.0593).
At 25 rpm
At this high speed, the performance of MultiStem cell-transplanted MPS-I mice on sequential days of testing was significantly better than that of untransplanted/sham-transplanted MPS-I mice (significance of interaction between sequential days of testing and MultiStem cell transplanted MPS-I vs. untransplanted/sham-transplanted MPS-I mice: p = 0.0224). These differences were predominantly due to the marked impairment in performance of the untransplanted/sham-transplanted MPS-I mice compared to the other two groups (untransplanted/sham-transplanted MPS-I vs. untransplanted/sham-transplanted WT and Ht mice: p = 0.0113; untransplanted/sham-transplanted vs. MultiStem cell transplanted MPS-I mice: p = 0.0264), with mouse groups accounting for a greater percentage of the total variation than interaction or days of testing. Of note, at both 15 and 25 rpm, the performance of MultiStem cell-transplanted MPS-I mice became comparable to that of untransplanted/sham-transplanted WT and Ht mice.
Effect of Training
The response of mice to training was tested by evaluating the improvement in performance (increase in balance time) by the fifth consecutive day of testing. The results obtained in the three groups of mice tested at 25 rpm (Fig. 5C) showed that untransplanted/sham-transplanted MPS-I mice were essentially unable to improve their performance over the 5 days. In contrast, the ability of MultiStem cell-transplanted MPS-I mice to improve with training became comparable to that of the untransplanted/sham-transplanted WT and Ht mice. By the fifth day of repetitive practice on the test (Fig. 5D), the MultiStem cell-transplanted MPS-I mice were able to perform the task as well as untransplanted/sham-transplanted WT and Ht mice and remain balanced on the rotarod for comparable lengths of time at 5, 15, and 25 rpm. In contrast, the untransplanted/sham-transplanted MPS-I mice demonstrated a steep drop off in balance time at increasing speeds. The difference between the performance of MultiStem cell-transplanted MPS-I and untransplanted/ sham-transplanted MPS-I mice (Fig. 5D) was highly significant, and largely attributable to interactions between rotarod speed and the two groups of mice (significance of interaction between rotarod speed and MultiStem cell-transplanted MPS-I vs. untransplanted/sham-transplanted MPS-I mice: p = 0.0061). These results indicate that the inability of untransplanted/sham-transplanted MPS-I mice to perform at the more challenging speeds, and to improve their sensorimotor ability with training, was completely corrected following ICV transplantation of MultiStem cells.
Discussion
Since the seminal report of ICV implantation of murine NSCs in neonatal MPS-VII mice (51), several studies [reviewed by Singec et al. (50)] showed the remarkable potential of various types of murine cells, implanted directly into the brain or cerebral lateral ventricles, to engraft, migrate, acquire neural markers, and induce biochemical, pathological, and functional improvement in rodent models of MPS and other neuropathic LSDs.
Clinical application, however, will require the use of rigorously evaluated human cells (31). Towards this goal, recent studies (summarized in Table 1) examined the effect of implanting human cells into the brain or cerebral ventricles of mouse models of neuropathic LSDs including one report in MPS-IIIB, but not in MPS-I. The results of implanting human umbilical cord blood (UCB) cells, ES-like cells, MSCs, or NSCs were variable. Garbuzova-Davis et al. (16) were the first to show that a proportion of UCB mononuclear cells implanted intracerebroventricularly into 1-month-old MPS-IIIB mice survived for an extended period (7 months), migrated widely into the parenchyma, and acquired neural markers and morphology; quantitative estimates of GAG clearance and enzyme activity in the brain and evaluation of neurological functional were not provided. In a subsequent report, the authors reported that intravenous transplantation of UCB cells improved behavioral outcomes in murine MPS-IIIB (15). Croitoru-Lamoury et al. (11) reported that embryonal carcinoma (ES-like) cells or marrow-derived MSCs survived for up to 5 weeks and acquired markers of neural differentiation, but did not induce functional benefit in a mouse model of Krabbe's disease. Information on substrate/ metabolite clearance and enzyme activity was not provided. Snyder and colleagues examined the effect of implanting human NSCs: Taylor et al. (54) demonstrated that primary fetal NSCs implanted into a mouse model of Krabbe's disease survived and acquired oligodendrocytic markers. Lee et al. (28) reported that primary fetal NSCs or ES cell-derived NSCs engrafted robustly and survived for up to 22 weeks in a mouse model of Sandhoff disease. These cells migrated widely, acquired neural markers, secreted effective levels of enzyme, and induced substrate clearance and functional improvement. In contrast, using a different NSC line, Sidman et al. (49) reported that implanted NSCs were undetectable at 16 weeks, and even though there was some substrate clearance, there was no functional improvement in a mouse model of Niemann-Pick A disease. These reports indicate that variability in outcome of intracranial cell transplantation is related to the underlying disease, type of cells used, timing, and site of transplantation. The effects of intracranial implantation of human NSCs in animal models of MPS remain to be determined. These aspects are relevant to clinical advancement of intracranial cell transplantation strategies.
Studies of Direct Intracerebral/ICV Transplantation of Human Cells in Rodent Models of Lysosomal Storage Diseases
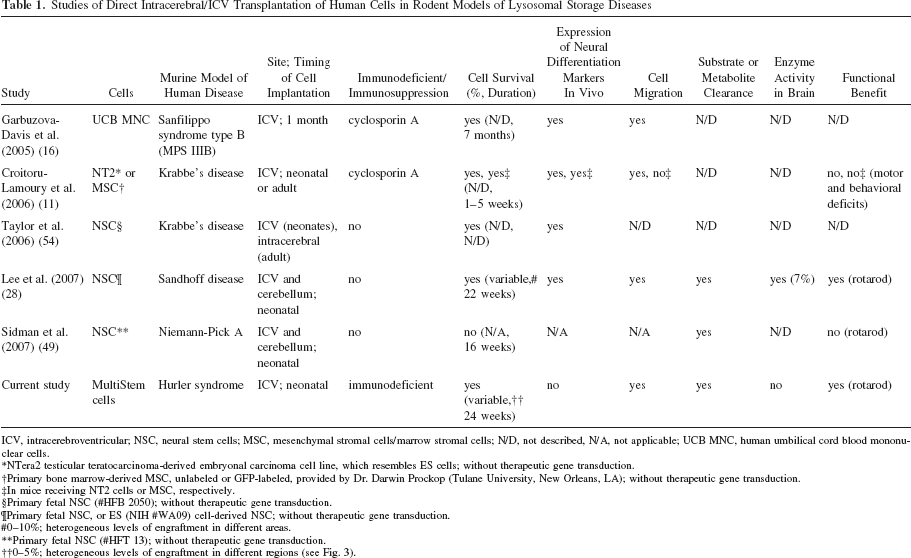
ICV, intracerebroventricular; NSC, neural stem cells; MSC, mesenchymal stromal cells/marrow stromal cells; N/D, not described, N/A, not applicable; UCB MNC, human umbilical cord blood mononuclear cells.
NTera2 testicular teratocarcinoma-derived embryonal carcinoma cell line, which resembles ES cells; without therapeutic gene transduction.
Primary bone marrow-derived MSC, unlabeled or GFP-labeled, provided by Dr. Darwin Prockop (Tulane University, New Orleans, LA); without therapeutic gene transduction.
In mice receiving NT2 cells or MSC, respectively.
Primary fetal NSC (#HFB 2050); without therapeutic gene transduction.
Primary fetal NSC, or ES (NIH #WA09) cell-derived NSC; without therapeutic gene transduction.
0–10%; heterogeneous levels of engraftment in different areas.
Primary fetal NSC (#HFT 13); without therapeutic gene transduction.
0–5%; heterogeneous levels of engraftment in different regions (see Fig. 3).
Our study is the first to examine the effect of ICV implantation of adult human MultiStem cells in a mouse model of MPS. Within a few days, MultiStem cells migrated widely into the brain parenchyma and underwent early proliferation, and even though cell survival dropped off steeply over time, implantation at the neonatal stage induced long-term improvement in substrate (GAG) levels and neurological functioning. No obvious adverse effects were identified from MultiStem cells implantation.
We found that reduced GAG levels in the hippocampus and striatum were associated with improved motor coordination and response to training. Motor coordination, measurable by the rotarod and other tests (17,62), is impaired in mouse models of MPS-I. This could be a consequence of MPS-induced dysfunction of multiple organ systems including the brain, the skeletal system, and vestibular and visual pathways. However, since functioning of the latter three systems does not change during repetitive testing, it can be inferred that serial rotarod testing accurately measures neurological performance relating to motor coordination (17). As expected, control (untransplanted) MPS-I mice demonstrated significant impairment of their ability to improve with training on serial rotarod tests. In contrast, the performance curve of MultiStem-transplanted MPS-I mice improved and approximated that of WT and Ht animals. No significant abnormality in sensorimotor functioning in MPS-I mice was discernible at the slowest speed (5 rpm), which may have been too slow to demand the animals' full attention to the task, since many mice deliberately jumped off the rod or turned around and attempted to walk backwards at 5 rpm. On the other hand, the highest speed (35 rpm) was too challenging, since none of the cohorts of mice were able to remain on the rod for a reproducible period of time.
Previous work showed that survival of adult human progenitors implanted in the rodent brain is considerably less sustained and robust than that of fetal NSCs. Azizi et al. (5) reported that survival of adult human MSCs implanted into the striatum of partially immunodeficient rats declines after 1 month. We recently reported that the majority of human MultiStem cells implanted intracerebrally into ischemic rat brains are lost within 72 h; only 1% survive for 2 months (33). The current results with MultiStem cells are consistent with these previous reports. Our results are consistent with previous reports where immunosuppression did not augment survival or functional benefit of rat or human MultiStem cells injected either into the brain or intravenously in neonatal and adult rats (33,61).
How might GAG clearance and functional improvement have been achieved without long-term survival of large numbers of transplanted cells? A consistent theme emerging from evaluation of cell therapy in various diseases is that functional improvement occurs in the absence of robust replacement of host tissue cells by differentiated transplanted cells (10,18,39,50). This apparent paradox is explained by increasing recognition of a multifaceted trophic/chaperone effect of transplanted cells in injured/dysfunctional host organs (50). Indeed, we have observed an inverse relationship between long-term survival of human MultiStem cells and trophic protection of endogenous neurons in a rat cerebral ischemia model: earlier transplantation rescued more neurons injured by acute ischemia even though fewer MultiStem cells survived in the acute ischemic brain (33).
We speculate that several complementary mechanisms of action may have contributed to the long-term functional improvement seen in our study. One possibility is that robust short-term survival and extensive early migration of transplanted MultiStem cells provides sufficient IDUA to clear GAGs and prevent/slow their usual rate of accumulation in the brains of MPS-I mice; this “resets” the quantity of GAGs in the brain to a lower level, which then results in the quantity of GAGs being lower than in control (untransplanted) animals even after several months, despite gradual resumption of GAG accumulation with the longer term decline in MultiStem cell number. By 24 weeks, few MultiStem cells survive, insufficient to provide detectable levels of IDUA. However, early clearance of GAGs [together with perhaps a trophic/chaperone effect (50) of the MultiStem cells: discussed below] provides some degree of protection from neurological damage in the critical early postnatal developmental phase (59); this may be of sufficient magnitude to achieve sustained improvement in sensorimotor function up to at least 24 weeks. A similar effect has been reported following HSCT in feline MPS-I, where reduction in GAG accumulation in the brain and improvement in mobility occurred in the absence of increase in IDUA activity in the brain (12).
Secondly, it is known that endogenous NSC proliferation is reduced in mouse models of MPS (30). The hippocampus, which plays a critical role in learning and memory, is a site of ongoing neurogenesis in the early postnatal period. It is possible that transplanted MultiStem cells that migrate into the hippocampus following ICV injection provide a trophic/chaperone effect that improves survival and/or proliferation of endogenous NSCs during the critical neonatal period. This may contribute to improvement in training ability that is maintained even after the subsequent loss of most transplanted MultiStem cells.
Thirdly, MultiStem cells may exert neuroprotective effects in MPS-I, including a trophic/chaperone effect on the survival of host postmitotic neurons, as we have shown in cerebral ischemia (63). We have also demonstrated that, following hypoxic-ischemic brain injury, transplantation of rat MultiStem cells into the hippocampus of neonatal rats improved endogenous neuronal survival and neurological functioning at 2 weeks (61,62). Even though ≤1% transplanted cells survived, transplantation was associated with significant (>20%) increase in survival of host neurons in the CA3 region of the hippocampus. Interestingly, rat MultiStem cells injected intravenously also homed to the hippocampus of these animals, suggesting that the hippocampus provides both migratory/ homing cues such as stromal-derived factor-1 (SDF-1) and a microenvironment supportive of the survival of transplanted cells. Early improvement (at 2 weeks) suggested that the effect was a result of a trophic/chaperone effect of MultiStem cells on injured host neurons and/or NSCs, rather than differentiation of MultiStem cells into neural cells. Using human MultiStem cells, we reported similar findings in an adult rat cerebral ischemia model (33). Similarly, we previously showed that human multipotent stem cells implanted around an area of cerebral ischemia in adult rats migrated, acquired neural markers (but not morphology) and reduced neurological deficits (63). Functional improvement in all these studies was most likely the result of the neuroprotective trophic/ chaperone effect (50) of implanted cells on injured host neuronal cells, without actual replacement of lost neurons by multipotent cell-derived progeny.
Finally, it is possible that MultiStem cell-induced immunomodulation (4,55) may reduce the damaging neuroinflammation that occurs in MPS (27,38). The existence and contribution of these various mechanism(s) is currently under study.
Conclusions and Future Directions
MultiStem cell transplantation merits further evaluation as a therapy for neurobehavioral abnormalities associated with MPS-I and other neuropathic disorders. While our study provides proof of principle of the potential therapeutic efficacy of these cells in MPS-I, clinical applicability will require further enhancement of cell survival, augmentation of enzyme secretion (perhaps by IDUA gene transduction), identification of the molecular and cellular mechanism(s) of action of these cells and control of their behavior in the brain, and comparative evaluation against human fetal NSCs (7,28), UCB cells (15,16), menstrual blood-derived progenitors (9,46), and other postnatal progenitors with neurogenic potential such as induced pluripotent stem (iPS) (53) and NurOwn® (18) cells. Finally, this strategy will need to be tested in the settings of HSCT or ERT that are essential for treating the systemic manifestations of MPS-I and other lysosomal storage diseases.
Footnotes
Acknowledgments
This work was supported by NIH R01-NS-48606 (P.G. and W.C.L.), the US Department of Veterans Affairs (P.G.), the Minnesota Veterans Research Institute (P.G.), the Minnesota Medical Foundation (P.G.), the Childrens Cancer Research Fund (P.G.), and NIH Grant P01-HD-32652 (W.C.L.). We thank Ms. Lan Zhao for assistance with the Rotarod experiments. R.J.D. and R.W.M. are employees of Athersys Inc., the company that holds the patent for MultiStem cells.
