Abstract
The major endpoints for clinical islet transplantation for type 1 diabetes are insulin independence and reduction of hypoglycemic episodes. Both endpoints are influenced by patients' and physicians' preferences regarding the use of exogenous insulin. Therefore, development of an objective endpoint for assessing clinical islet transplantation is desirable. HOMA-beta score is useful in assessing functional β-cell mass. However, this score uses blood insulin levels that are influenced by exogenous insulin injection and therefore is not suitable for patients who receive exogenous insulin. For assessing functional β-cell mass for type 1 diabetic patients after islet transplantation, we created the Secretory Unit of Islet Transplant Objects (SUITO) index using fasting C-peptide and fasting glucose. The formula of the SUITO index is fasting C-peptide (ng/ml)/[fasting blood glucose – 63 (mg/dl)] x 1500. We demonstrated that, within 1 month of islet transplantation, an average SUITO index of >26 was an excellent predictor of achieving insulin independence. In addition, daily SUITO index scores correlated with a reduction of insulin dose and adversely correlated with blood glucose levels during an intravenous glucose tolerance test. Other important endpoints, reduction of hypoglycemic episodes and quality of life, also correlated with the SUITO index. Thus, the SUITO index is excellent for assessing important endpoints (insulin independence, reduction of hypoglycemia, improved quality of life) after allogeneic islet transplantation.
Introduction
Islet transplantation is a promising treatment for type 1 diabetes (11,26,27). Current major endpoints of islet transplantation are insulin independence and elimination of severe hypoglycemic episodes (11,26,27). Insulin independence is influenced by patient and physician preferences. Some patients and physicians prefer to use insulin more frequently to reduce hemoglobin A1c, and others prefer to minimize insulin use to avoid hypoglycemia. Severe hypoglycemic episodes are defined as those involving a third party's help and the patient's hypoglycemic unawareness. The frequency of third-party help depends on the patient's social circumstances, and hypoglycemic unawareness depends on how frequently patients measure blood glucose levels. Therefore, both insulin independence and elimination of severe hypoglycemia are subjective. An objective method to evaluate clinical outcomes of islet transplantation is necessary. It is obvious that islet graft function directly correlates with clinical outcomes of islet transplantation.
Matthews et al. established homeostasis model assessment (HOMA) for both insulin resistance (HOMA-R) and deficient β-cell function (HOMA-beta) (21). HOMA-beta is useful in assessing functional islet mass. However, HOMA-beta includes the blood concentration of insulin and therefore cannot evaluate islet function for patients who receive exogenous insulin. The recipients of islet transplantation are type 1 diabetes injected with exogenous insulin; hence, HOMA-beta is not applicable. To solve this issue, we introduced the Secretory Unit of Islet Transplant Objects (SUITO) index, which can be calculated using blood glucose and C-peptide levels (20). Of note, SUITO consists of SUI and TO, which means pancreas and island or islet in Japanese; therefore, SUITO index means pancreatic islet index. In this review article, we describe the development and value of the SUITO index for assessing clinical islet transplantation.
Development of the Suito Index
Problems with Using the HOMA-Beta Score for Islet Transplant Recipients
Steady-state basal plasma glucose and insulin concentrations are determined by their interaction in a feedback loop. A computer model has been used to predict the homeostatic concentrations that arise from varying degrees of β-cell deficiency and insulin resistance. Comparison of a patient's fasting values with the model's predictions allows a quantitative assessment of the contributions of insulin resistance and deficient β-cell function to fasting hyperglycemia (homeostasis model assessment, HOMA) (21). Insulin resistance (HOMA-R) is calculated as follows: fasting blood insulin level (μU/ml) x fasting blood glucose level (mg/dl)/405. A HOMA-R score of <1.6 is normal, and a score of >2.5 is considered insulin resistance. β-Cell function (HOMA-beta) is calculated as follows: fasting blood insulin level (μU/ml)/[fasting blood glucose level (mg/dl) - 63] × 360. A HOMA-beta score <40 is considered decreased β-cell function.
Candidates for islet transplant have unique characteristics. First, they lack insulin secretory ability. Therefore, both their HOMA-R and HOMA-beta scores are zero. While insulin secretory ability is zero, insulin resistance is not necessarily zero. In fact, patients who are insulin resistant cannot be candidates for islet transplantation. Since the patients have no insulin secretory ability, insulin resistance is diagnosed by a high body mass index (>30 kg/m2) and daily insulin dose (>1.0 IU/kg body weight) (26).
After islet transplantation, the transplanted islets begin secreting insulin. However, exogenous insulin injection is continued to enhance engraftment of transplanted islets for several months. Even after achieving insulin independence, the patients are encouraged to use insulin injections when their blood glucose is high to protect transplanted islets. Therefore, even after islet transplantation, the HOMA-beta score cannot reflect insulin secretory ability from islets. Thus, we thought a new index using blood C-peptide levels would be useful in assessing the function of transplanted islets.
Formula for SUITO Index
After allogeneic islet transplantation, we and others noticed that the blood levels of C-peptide correlated with those of glucose (5,20,22,35) (Fig. 1). In addition, the ratio between C-peptide and glucose seemed to be associated with islet graft function (Fig. 1). This suggested that transplanted functional islets secrete insulin based on blood glucose levels, and when the number of transplanted functional islets is high, the total amount of secreted insulin is also high. Based on HOMA-beta (21) and data from normal healthy persons (35), we created the SUITO index to reflect functional mass (20). The formula is as follows: SUITO index = fasting C-peptide level (ng/ml)/[fasting blood glucose level (mg/dl) - 63] x 1500.

Relationship between fasting blood glucose levels and fasting C-peptide levels before and after islet transplantation. The angle of the regression line reflects functional islet mass.
The SUITO index is 100 for normal healthy persons and 0 for type 1 diabetic patients with no ability to secrete insulin (Fig. 1). After islet transplantation, patients who achieve insulin independence secrete more C-peptide than patients who do not achieve insulin independence at the same glucose levels, resulting in a high SUITO index (Fig. 1).
Suito Index to Evaluate Clinical Islet Transplantation
Prediction of Insulin Independence by Average SUITO Index Within 1 Month of Islet Transplantation
One of the endpoints of islet transplantation for the treatment of type 1 diabetic patients is insulin independence (11,26,27). We hypothesized that functional islet mass during the first month after islet transplantation as assessed by the SUITO index could predict future insulin independence. C-peptide is released from broken islets up to 1 day after islet infusion (4), so C-peptide levels do not reflect functional islets in that period. Therefore, we do not use C-peptide levels for the SUITO index at day 1 and 2 after islet transplantation.
We calculated the average SUITO index of eight patients after islet transplantation using pancreata from non-heart-beating donors between postoperative days 3 and 30 (20). When the average SUITO index was >26.0, the patients were insulin independent. This suggested that >26% of functional β-cell mass is required for insulin independence. We also calculated the SUITO index between postoperative days 3 and 30 for our first successful living-donor islet transplant patient (20). The average SUITO index of this patient was 40.7, and the patient was insulin independent.
Recently, we calculated the average SUITO index between postoperative days 3 and 30 for patients who received islet transplants from pancreata from brain-dead donors (19). In this study, we used both the Edmonton-type protocol and our own protocol (Baylor protocol). Under the Edmonton-type protocol, three type 1 diabetic patients received islet transplantation twice. After the first islet transplantation, the average SUITO indices during the first month were 10–25 in all three cases. None of the three patients achieved insulin independence. After the second islet transplantation, the average SUITO indices during the first month posttransplantation were >26.0 in all three patients, and all three achieved insulin independence. Under the Baylor protocol, three patients received single-donor islet transplantation. All patients became insulin independent, and the average SUITO indices during the first month posttransplantation were >26.0.
Thus, based on the analysis of the SUITO index after allogeneic islet transplantation using pancreata from non-heart-beating donors (17), a living donor (16,18), and brain-dead donors (12,13,19), a SUITO index >26 is associated with insulin independence.
Correlation Between SUITO Index and Daily Insulin Dose
We examined the correlation between daily SUITO index and reduction of insulin dose after islet transplantation (12,14). Since the current standard protocols use intensive insulin therapy during the first month after islet transplantation, there was no correlation between the daily SUITO index and insulin reduction rate during the first month after islet transplantation. However, after 2 months, the daily SUITO index strongly correlated with the daily insulin dose (14). When the cutoff value of the daily SUITO index was set at 26 for insulin independence, the positive predictive value for a reduced insulin dose was 84.1%, and the negative predictive value was 89.4%. Therefore, the daily SUITO index is also useful for assessing functional islet mass.
We have a unique case in which a very high total dose of islets (>20,000 IEq/kg body weight) was transplanted after two islet transplantations (15). In this case, the average SUITO index within 1 month after the second islet transplantation was 48.5, and the daily SUITO index on postoperative day 1006 was 44.6. This patient has been insulin independent for more than 3 years. Thus, maintaining a high SUITO index with a transplantation of high islet yield might be one strategy to achieve long-term insulin independence.
Correlation Between SUITO Index and Intravenous Glucose Tolerance Test
The intravenous glucose tolerance test (IVGTT) is a standard evaluation method after islet transplantation (25). However, this method requires bolus glucose injections and frequent blood glucose measurements. We examined whether the SUITO index, which is much simpler than IVGTT, correlated with the results of IVGTT (30). Fifteen blood samples from each of six islet transplant recipients were examined before and after 3, 5, 10, 20, and 30 min of injection of 0.5 g/kg 50% dextrose. Glucose-level areas under the receiver–operating characteristic curve (AUC) during IVGTT were assessed based on the SUITO index. The AUC was significantly lower in cases with a SUITO index >10 than in cases with a SUITO index <10. In addition, when the SUITO index was <10, glucose levels at baseline, 10, 20, and 30 min were significantly higher than when the SUITO index was >10. Hence, a SUITO index <10 is associated with impaired islet function assessed by IVGTT.
Rickels et al. demonstrated that acute insulin response to glucose and arginine could predict β-cell secretory capacity in human islet transplantation (23). In addition, glucose disappearance rate (K-value) during IVGTT (9) was also used to assess β-cell function after islet transplantation (1,3,34). In our IVGTT, we only measured blood glucose for assessing acute response, and then we examined the correlation between the K-value and the SUITO index. The K-value significantly correlated with the SUITO index (Spearman r = 0.571, p = 0.01) (Fig. 2A). We also assessed β-cell secretory capacity using the difference between the basal C-peptide levels before IVGTT and stimulated C-peptide levels at the end of IVGTT. The β-cell secretory capacity also significantly correlated with the SUITO index (Spearman r = 0.581, p = 0.01) (Fig. 2B). These findings suggest that the SUITO index can be a surrogate marker for IVGTT to assess the β-cell secretory capacity.

The glucose disappearance rate and β-cell secretory capacity in IVGTT. The glucose disappearance rates (K-values) were assessed with SUITO index (A). The β-cell secretory capacity was evaluated by C-peptide responses calculated by the stimulated C-peptide levels minus the basal C-peptide levels in IVGTT (B). Both K-values and C-peptide responses were significantly correlated with the SUITO index (Spearman r = 0.571, p = 0.01 and r = 0.581, p = 0.01, respectively). The solid line shows a linear regression. IVGTT, intravenous glucose tolerance test; SUITO index, secretory unit of islet transplant objects index.
Correlation Between SUITO Index and Hypoglycemic Episodes
To assess the relationship between the frequency of hypoglycemic episodes and the SUITO index, data were examined from 310 clinical evaluations of 11 islet recipients (32). Hypoglycemia was defined as a self-monitoring blood glucose (SMBG) level <3.8 mmol/L. Severe hypoglycemia was defined as an SMBG level <2.2 mmol/L or the presence of hypoglycemic unawareness. Cases were categorized into three groups based on their SUITO index: low (<10), middle (10–25), and high (≥ 26). The frequency of hypoglycemia in the high-SUITO group was significantly lower than in the other groups, including pretransplantation. The frequency of total severe hypoglycemia was significantly lower in the low-SUITO group compared to the pretransplant status and further decreased in the middle- and high-SUITO groups. Therefore, even the low-SUITO group benefited in the reduction of severe hypoglycemia.
Correlation Between SUITO Index and Quality of Life
Quality of life (QOL) is another important endpoint for type 1 diabetic patients after islet transplantation (6,7). To elucidate the correlation between the SUITO index and QOL, we used the 36-item Short-Form Health Survey (SF-36), Diabetes Form 2.1, and the Insulin Treatment Satisfaction Questionnaire (ITSQ) before transplant and 3, 6, 12, 18, and 24 months posttransplant (29). When the SUITO index was >10, an item of physical health in the SF-36 related to glycemic control and the total score in the ITSQ were significantly improved compared with the pretransplant status. In addition, when patients' SUITO index was >10, items related to physical health, energy, and emotions in the SF-36, items related to symptoms and social interactions in Diabetes Form 2.1, and items related to inconvenience of regimen, glycemic control, and total score in the ITSQ were significantly higher compared with those who had a SUITO index <10. Thus, a SUITO index >10 was associated with improvement of QOL after islet transplantation. Of note, when the SUITO index was <10, almost all items were worse than in the pretransplant status. Since the main purpose of islet transplantation is improving QOL, it is important to maintain a SUITO index of at least 10.
Other Applications of the Suito Index
SUITO Index for Autologous Islet Transplantation
Autologous islet transplantation after total pancreatectomy is a useful treatment for chronic pancreatitis with severe abdominal pain (10,24,28,31,33). Islets are isolated from the removed pancreas, and isolated islets are immediately infused into the same patient (10). To assess the correlation between the SUITO index and metabolic outcomes of autologous islet transplantation, we analyzed 27 of our patients who received autologous islet transplantation. Cases were divided into an insulin-independent group (N = 12) and an insulin-dependent group (N = 15). The average SUITO index within the first month after transplantation, except for day 1 and 2, was calculated in each case and was significantly higher in the insulin-independent group (24.6 ± 3.4 vs. 14.9 ± 2.0; p < 0.02). Thus, the average SUITO index within 1 month seems to be an excellent predictor of insulin independence after total pancreatectomy followed by autologous islet transplantation.
SUITO Index for other Species
Recently, Igarashi et al. performed islet xenotransplantation, transplanting pig islets into streptozotocin-induced diabetic nonhuman primates (8). They analyzed functional β-cell mass based on the SUITO index. The SUITO index did not correlate with the β-cell mass. The normal blood glucose of nonhuman primates is very low compared with that in humans (2); therefore, fasting blood glucose minus 63 is not a suitable formula for nonhuman primates. The authors created a new score using fasting blood glucose, daily glycosuria, postprandial glycosuria, polydipsia, and polyuria with excellent results. However, this score requires both blood and urine samples and frequent measurement. A modified SUITO index for nonhuman primates should provide a simple and reliable assessment method.
Future Directions
We have shown the usefulness of the SUITO index for predicting insulin independence, assessing hypoglycemic episodes, and correlating with IVGTT results and QOL (Table 1). Since islet transplantation is still considered experimental treatment in the US, the case number is limited. Further large-scale assessment is necessary to prove the usefulness of the SUITO index.
Summary of Correlation Between the SUITO Index and Clinical Features
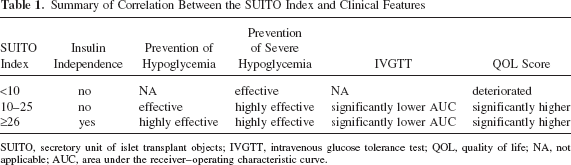
SUITO, secretory unit of islet transplant objects; IVGTT, intravenous glucose tolerance test; QOL, quality of life; NA, not applicable; AUC, area under the receiver–operating characteristic curve.
Footnotes
Acknowledgments
The authors thank Ms. Cynthia Orticio for her professional editing of this manuscript. This study was partially supported by grants from the National Institute of Diabetes and Digestive and Kidney Diseases (1R21DK090513-019), the Juvenile Diabetes Research Foundation (#3-2011-447), and the Baylor Health Care System Foundation. The authors declare no conflicts of interest.
