Abstract
The initial success of islet transplantation (ITx) is followed by graft dysfunction (GDF) and insulin reintroduction. Exenatide, a GLP-1 agonist, increases insulin and decreases glucagon secretion and has potential for β-cell regeneration. To improve functional islet mass, exenatide treatment was given to ITx recipients with GDF. The objective of this study was to assess metabolic and hormonal effects of exenatide in GDF. In this prospective, single-arm, nonrandomized study, 11 type 1 diabetes recipients of ITx with GDF had HbA1c, weight, insulin requirements, and 5-h mixed meal tolerance test (MMTT; with/without exenatide given before test) at baseline, 3, 6, and 12 months after initiating exenatide treatment. Baseline MMTT showed postprandial hyperglycemia and hyperglucagonemia. Daily exenatide treatment resulted in improved glucose, increased amylin/insulin ratio, and decreased proinsulin/insulin ratio as assessed by MMTT. Glucagon responses remained unchanged. Exenatide administration 1 h before MMTT showed decreased glucagon and glucose at 0 min and attenuation in their postprandial rise. Time-to-peak glucose was delayed, followed by insulin, proinsulin, amylin, and C-peptide, indicating glucose-driven insulin secretion. Five subjects completed 12-month follow-up. Glucose and glucagon suppression responses after MMTT with exenatide were no longer observed. Retrospective 3-month analysis of these subjects revealed higher and sustained glucagon levels that did not suppress as profoundly with exenatide administration, associated with higher glucose levels and increased C-peptide responses. In conclusion, Exenatide suppresses the abnormal postprandial hyperglucagonemia and hyperglycemia observed in GDF. Changes in amylin and proinsulin secretion may reflect more efficient insulin processing. Different degrees of responsiveness to exenatide were identified. These may help guide the clinical management of ITx recipients.
Keywords
Introduction
Islet transplantation under steroid-free immunosuppression is being evaluated as potential treatment for C-peptide-negative patients with type 1 diabetes mellitus (T1DM) with severe hypoglycemia and hypoglycemia unawareness (18,24,32,34). Proven benefits are significant reduction of hypoglycemic events (6) and improved quality of life (29). Unfortunately, the initial success of islet transplantation, defined as achievement of insulin independence, is generally followed by progressive graft dysfunction requiring reintroduction of exogenous insulin (6,18,32,35). By 5-year follow-up, even though ~80% of the patients remain C-peptide positive, only ~10% maintain insulin independence (32).
Exenatide, a synthetic form of exendin-4 (a polypeptide produced by the salivary glands of the gila monster), is a glucagon-like peptide (GLP)-1 receptor agonist (9,10) with longer half-life than endogenous GLP-1. Activation of GLP-1 receptors results in enhanced glucose-stimulated insulin secretion, inhibition of glucagon secretion, delayed gastric emptying, and increased satiety (10,26,27). In rodent models, effects include promotion of pancreatic progenitor cell differentiation, improve β-cell function, and life span (13,37,42). Additionally, in T2DM management, exenatide has been shown to improve glycemic control and cause weight reduction (5,8,14).
To improve islet graft function and possibly increase islet mass, exenatide treatment was started in recipients of islet transplantation alone (IA) or islet after kidney (IAK) protocols (18,22,28) with a history of graft dysfunction. The objective of this study was to assess the metabolic and hormonal effects of exenatide treatment acutely during a mixed meal stimulation test and to see if these effects are sustained during prolonged exenatide treatment (20).
Materials and Methods
Study Subjects
Demographic Characteristics of Study Subjects at the Time of Enrollment in the Exenatide Protocol

Time of follow-up after starting exenatide where there are data available for analysis. ITx, islet transplantation; IA, islet alone; IAK, islet after kidney.
Subjects who underwent a supplemental islet infusion after 3 or 6 months of exenatide treatment.
No MMTT data available at 3 months post-ITX completion.
Definitions
Islet transplant completion is defined as the achievement of insulin independence after sequential islet transplantation, usually observed after two islet infusions. At 3 months postcompletion the best function attained after islet transplantation is typically demonstrated in insulin-independent subjects with stable glycemic control (2).
Graft dysfunction is defined as C-peptide-positive subjects (>0.09 nmol/L) with fasting and/or 2-h postprandial capillary glucose >7.8 and >10 mmol/L, respectively, on three or more occasions over a week, or who had two sequential monthly HbA1c values >6.5% (11). Any subject with graft dysfunction was considered eligible for reintroduction of exogenous insulin and study enrollment.
Study Design (Fig. 1)
HbA1c, weight, insulin requirements, and 5-h mixed meal tolerance test (MMTT) without exenatide were assessed at 3 months postcompletion, immediately before, and at 3, 6, and 12 months after starting exenatide treatment. Following commencement of exenatide treatment, MMTT with and without exenatide was performed at each follow-up visit. Subjects underwent a 5-h MMTT with exenatide administration 1 h before MMTT on the first visit day, then discontinued all exenatide until a 5-h MMTT without exenatide a day or two later.
To assess the acute metabolic effects of exenatide, the MMTT results with and without exenatide within the same follow-up visit (3, 6, or 12 months) were compared.
To assess the chronic metabolic effects of exenatide treatment, the results of the 3, 6, and 12 month MMTT performed without exenatide were compared to the baseline (pre-exenatide) MMTT.
Exenatide Treatment Regimen
Exenatide (Byetta®, Amylin Inc.) treatment was started at 5 μg subcutaneously twice daily and increased as tolerated up to a maximum dose of 30 μg/day (usually divided in three doses with each meal). Total daily insulin dose was reduced 30–40%; including cessation of all meal coverage coincident with exenatide administration and variable reduction in basal insulin coverage to avoid hypoglycemia yet maintain euglycemia. The insulin dose was then adjusted to achieve fasting and pre-prandial glucose levels <7.8 mmol/L and postprandial glucose levels <10 mmol/L. If insulin independence was not achieved after 3 months, and the subject was eligible, another islet infusion was offered.
Metabolic Studies
MMTT was performed after an overnight fast (8–12 h) with samples obtained at 0, 15, 30, 60, 90, 120, 180, and 300 min after ingestion of 360 ml Boost® High-protein (Novartis, Hanover NJ; 360 calories, 9 g fat, 49.5 g carbohydrate, 22.5 g protein), administered immediately after obtaining the 0-min sample. At each time point, glucose, C-peptide, insulin, proinsulin, amylin, and glucagon were measured.
For the MMTT with exenatide administration, an additional blood sample, followed immediately by exenatide injection, was performed at −60 min. Exenatide was administered at this time to obtain its maximal concentration (Cmax occurring 1–3 h after administration) and peak action (1) coinciding with the meal.
Glucose and C-peptide results from MMTT performed 3-months postcompletion were available in seven subjects and were used for comparison.
Biochemical Determinations
Serum glucose concentrations (mmol/L) were determined by hexokinase method. Plasma C-peptide was measured by double antibody radioimmunoassay [Diagnostics Products Corp., Los Angeles, CA; 0.03–1.66nmol/L detection limit; <10% inter- and intra-assay coefficient of variation (CV), 20% cross-reactivity with proinsulin]. Serum insulin was measured by solid phase 125I radioimmunoassay (Diagnostics Products Corp; 20.8 pmol/L detection limit; 4.5% intra- and 7.6% interassay CVs, respectively).
Amylin (pmol/L) was measured by ELISA, proinsulin (pmol/L), and glucagon (ng/L) by radioimmunoassay using reagents from Linco Research, Inc (St. Charles, MO). Respectively, detection limits (and intra- and interassay CVs) were: amylin 1 pmol/L (4.5% and 11.7%), proinsulin 2 pmol/L (4.1% and 7.4%), and glucagon 20 ng/L (3.8% and 9.1%).
Calculations
Area under the curve (AUC) of MMTT from 0–300 min was calculated for all the variables (AUCglucose, AUCC-peptide, AUCinsulin, AUCamylin, AUCproinsulin, AUCglucagon) by trapezoidal approximation. The amylin/insulin and the proinsulin/insulin ratios were assessed for all time points. The mixed meal stimulation index (MMSI) was calculated using the formula (33):

Statistical Analysis
For each outcome measure, a linear mixed model regression was fit to the data to estimate and compare mean response values corresponding to all acute and chronic assessments of interest. This method of analysis generalizes linear regression techniques allowing for repeated observations by taking into account correlation within observations from the same subject to more appropriately estimate variances used in tests of significance. Using this approach, we can simultaneously estimate differences comparing the variables obtained from the baseline pre-exenatide MMTT with values at each time point postinitiation of exenatide as well as between 3, 6, and 12 months postinitiation of exenatide while appropriately accounting for within-subject correlation. Similarly, we can estimate differences between MMTT performed with and without exenatide at each follow-up visit. Results are expressed as means ± SE; a value of p < 0.05 was considered statistically significant. Analysis was performed using SAS 9.1 software (SAS Institute Inc., Cary, NC, USA).
Results
Clinical Outcomes in Islet Transplant Recipients with Graft Dysfunction Treated with Exenatide
Of the 11 subjects followed, four subjects underwent a supplemental islet infusion under exenatide treatment (three after 3 months and one after 6 months of exenatide therapy; therefore, only eight patients completed 6-month follow-up). The outcomes of these four subjects after supplemental infusion are not included in this report. Exenatide was discontinued in two subjects; one developed pneumonia requiring hospitalization at 12 months, and the other had 15% weight loss by 9 months. There are no metabolic data available for these two subjects at 12 months. Five subjects completed the study up to 12 months (Fig. 1).
Study design. Three months after islet transplant (ITx) completion (C) subjects underwent a 5-h mixed meal tolerance test (MMTT). Graft dysfunction occurred at a variable time post-ITx-C. Before exenatide (Exn) initiation, subjects underwent a baseline visit (Pre-Exn) that included MMTT. At 3-, 6-, and 12-month follow-up visits, subjects underwent MMTT with (MMTT-Exn pos) and without (MMTT-Exn neg) exenatide administration before the test.
Sixty-four percent of patients developed nausea at any time, which was worse in the first 2 weeks of therapy, and were treated with dose adjustment and/or antiemetics as needed. No pancreatitis was observed.
Insulin independence with target A1c was achieved in 4 of 11 (36%) subjects. Two subjects were insulin independent prior to exenatide treatment and sustained this with a reduction in HbA1c to <6.5% (17). Two subjects were able to discontinue exogenous insulin while on exenatide for 67 and 386 days, respectively.
Metabolic and Hormonal Responses to MMTT
Three Months Post-Islet Transplant Completion
At 3-month postcompletion optimal improvement in metabolic and nutritional parameters is typically observed. All subjects were insulin independent with near-normalization of HbA1c levels (5.78 ± 0.12%, range 5.7–6.4). In addition, we observed near-normalization of glucose and C-peptide responses to MMTT (n = 7) (Table 2, Fig. 2).
Chronic metabolic and hormonal effects of exenatide treatment. Glucose (A), C-peptide (B), insulin (C), amylin (D), proinsulin (E), and glucagon (F) responses to a 5-h mixed meal tolerance test at 3 months post-islet transplant completion (3 mo Post-C; gray rhomboid, dashed gray lines), pre-exenatide (Pre-Exn; black circles, black lines), 3 month (3 mo; white squares, dashed black lines), 6 month (6 mo, black inverted triangles, black lines), and 12 month follow-up (black X, black line). Amylin/insulin ratio (G) and proinsulin/insulin ratio (H) calculated from these tests. Responses at 3, 6, and 12 months follow-up are those from MMTTs without exenatide administration before the test (MMTT-Exn-). At 3 months post-islet transplant completion there is only glucose and C-peptide data available. Clinical and Metabolic Outcomes at Baseline and at 3, 6, and 12 Months of Exenatide Treatment Clinical and metabolic outcomes at baseline (pre-exenatide) and at 3, 6, and 12 months of exenatide treatment. Metabolic outcomes expressed at 3, 6, and 12 months are obtained from the MMTT without exenatide administration before the test. Data at 3 months postislet transplant completion are shown for comparison. AUC: area under the curve. Exenatide dose is expressed as median and range. All other values are expressed as means ± SE and are compared to pre-exenatide. Values that showed statistically significant difference (p < 0.05) when compared to pre-exenatide. Significant differences estimated from linear mixed model regression.

Pre-Exenatide
After variable periods of time (678 ± 336 days), all subjects demonstrated islet graft dysfunction. Reintroduction of insulin had already occurred in 9 of 11 subjects (mean dose 0.15 ± 0.03 IU/kg/day; range 0–0.33) and elevation of HbA1c >6.5% (a criterion for reintroduction of insulin) in two subjects. Compared to 3-month postcompletion, there was a significant increase in HbA1c (6.5 ± 0.1, range 5.9–7.2%, p < 0.001, n = 11) and islet function had markedly declined, showing return to postprandial hyperglycemia, significant increase in 90-min glucose (p < 0.001), peak glucose (p < 0.001), and AUCglucose (p < 0.001). C-peptide responses to MMTT did not vary as much as the glucose levels, with a resultant decrease in the MMSI (p = 0.008) (Fig. 2, Table 2). Insulin, amylin, and proinsulin responses followed those of C-peptide. Abnormal postprandial hyperglucagonemia was observed.
Three Months Under Exenatide Treatment
Median exenatide dose was 15 μg/day (range 5–25). Weight and insulin requirement decreased significantly compared with pre-exenatide (decrement 2.69 ± 0.71 kg; p = 0.001 and 0.05 ± 0.02 IU/kg/day; p < 0.02 for weight and insulin requirement, respectively).
Chronic effects at 3 months (assessed by comparing the MMTT without exenatide at baseline and at 3 months) included a decrease in AUCglucose (p = 0.052), although overall the response remained abnormal (Fig. 2, Table 2). Other variables (C-peptide, insulin, and MMSI) were not significantly different to pre-exenatide. A significant increase in AUCamylin (p = 0.02) and an increase in the basal amylin/insulin ratio were observed. Postprandial hyperglucagonemia was still evident and essentially unchanged.
Acute effects of exenatide (assessed by comparing MMTT with and without exenatide at 3 months) were dramatic, demonstrating a significant suppression in the glucose response curve, and significantly increased time-to-peak (p < 0.01) (Fig. 3A, Table 3). The secretory responses followed those of glucose, with a shift-to-the-right in time-to-peak C-peptide (p < 0.001), amylin (p < 0.01), and proinsulin (p = 0.026). There was significant suppression of the glucagon responses after MMTT (decrease in AUCglucagon, p < 0.001).
Acute metabolic and hormonal effects of exenatide administration at 3 months. Glucose (A), C-peptide (B), insulin (C), amylin (D), proinsulin (E), and glucagon (F) responses to a 5-h mixed meal tolerance test with exenatide administration before the test (MMTT-Exn +; black squares, black lines) and without (MMTT-Exn -, white squares, black lines) exenatide administration before the test. Arrow shows time of subcutaneous exenatide administration. Acute Metabolic Effects of Exenatide Administration Before the Mixed Meal Tolerance Test Comparison of the acute metabolic effects of exenatide administration 1 h before the mixed meal tolerance test (MMTT-Exn +) versus no exenatide administration (MMTT-Exn -) at the same follow-up visit (3, 6, or 12 months). Values are expressed as means ± SE. AUC: area under the curve. Values where a significant difference (p < 0.05) was observed. Significant differences estimated from linear mixed model regression.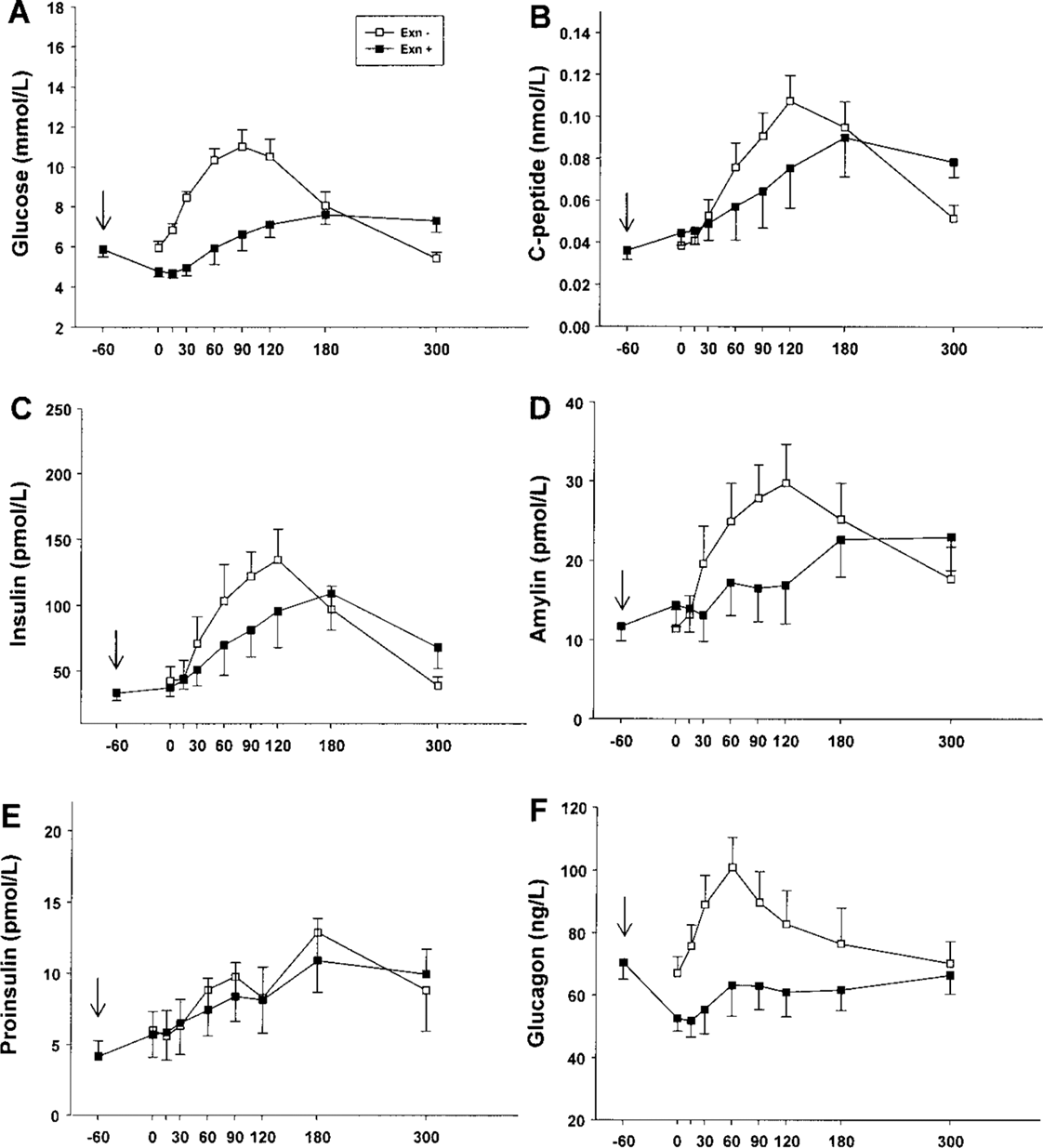

Six Months Under Exenatide Treatment
Median exenatide dose was 15 μg/day (range 7.5–25). Significantly decreased HbA1c to 6.26 ± 0.14% (range 5.5–7%, diff = 0.25 ± 0.12, p < 0.001) compared to pre-exenatide was observed.
Chronic effects at 6 months (assessed by comparing the MMTT without exenatide at baseline and at 6 months) included persistent marked improvement in glucose responses to MMTT; C-peptide levels were in creased but not significantly so; the combination of decreased glucose and higher C-peptide levels resulted in a significant increase in MMSI at this time point (p = 0.03) (Fig. 2). There was also a persistent marked increase in amylin secretion, both basal amylin (p = 0.016) and AUCamylin (p = 0.02), and significantly decreased AUCproinsulin (p = 0.012), greater than that seen after 3 months of treatment. Further increase in basal amylin/insulin ratio and decrease in proinsulin/insulin ratio were observed.
Acute effects of exenatide (assessed by comparing MMTT with and without exenatide at 6 months) included persistent significant suppression of postprandial hyperglucagonemia (Table 3). Changes in MMSI, AUCglucose, AUCC-peptide, AUCinsulin, AUCamylin, and AUCproinsulin were no longer significant.
Twelve Months Under Exenatide Treatment
Median exenatide dose was 20 μg/day (range 10–30). The mean body weight was stable. The mean insulin requirement was reduced compared to pre-exenatide (p = 0.019) accompanied with significantly increased HbA1c to 7.0 ± 0.1% (range 6.7–7.3, p < 0.001).
Chronic effects at 12 months (assessed by comparing the MMTT without exenatide at baseline and at 12 months) demonstrated an interval deterioration with recurrent elevation of glucose responses to MMTT, increased peak glucose (p = 0.015), and increased glucose AUC (Fig. 2). Significant increase in basal C-peptide (p = 0.01) persisted compared to pre-exenatide; AUCC-peptide remained increased, but did not reach significance at this time point either. The changes observed in amylin responses at 3 and 6 months were no longer seen. In contrast, the decrease in proinsulin/insulin ratio was still evident.
Acute effects of exenatide (assessed by comparing MMTT with and without exenatide at 12 months) were markedly diminished where compared with previous time points during exenatide treatment. The marked suppression of AUCglucose and AUCglucagon responses was not observed (Fig. 4, Table 3). In addition, no significant change in insulin, amylin, proinsulin, or glucagon responses compared to pre-exenatide was detected.
Acute metabolic and hormonal effects of exenatide administration at 12 months. Glucose (A), C-peptide (B), insulin (C), amylin (D), proinsulin (E), and glucagon (F) responses to a 5-h mixed meal tolerance test with exenatide administration before the test (MMTT-Exn +; black inverted triangles, black lines) and without (MMTT-Exn -, white inverted triangles, black lines) exenatide administration before the test. Arrow shows time of subcutaneous exenatide administration.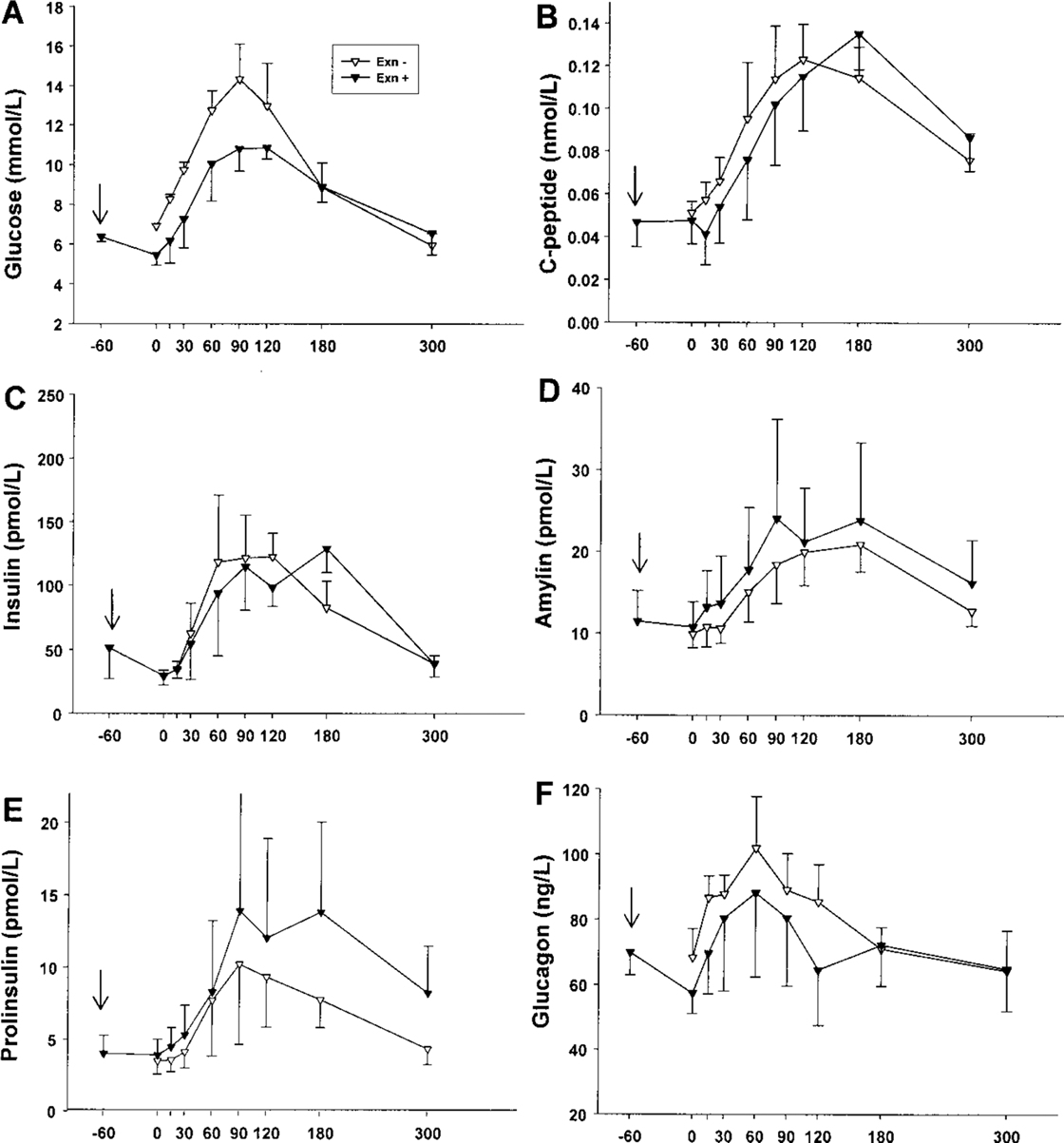
To see if there was a difference in response between subjects, we retrospectively compared the results at 3 months of five subjects that completed 12-month follow-up (subgroup 1) with six subjects who did not complete the 12-month follow-up (subgroup 2). Subgroup 2 included four subjects that received a supplemental infusion and two that developed adverse events that required cessation of exenatide. Baseline characteristics (age, diabetes duration, weight, BMI, islet equivalents/kg transplanted, time to graft dysfunction, baseline MMTT data) were similar for these subgroups. Results in subgroups 1 and 2 differed significantly in metabolic and hormonal responses to exenatide administration (Fig. 5). The MMTT performed without exenatide administration showed delayed and sustained hyperglucagonemia in subgroup 1. The statistically significant decrease in AUCglucagon and AUCglucose during MMTT with exenatide administration was similar in both subgroups, although in subgroup 1 there was less pronounced inhibitory response to exenatide administration. Subgroup 2 demonstrated a significantly more profound decrease in AUCamylin (p = 0.01). Peak C-peptide (p = 0.04) and AUCC-peptide (p = 0.03) were significantly higher in subgroup 1.
Acute metabolic and hormonal effects of exenatide administration at 3 months in subgroup 1 (left) and subgroup 2 (right). Glucose (A), C-peptide (B), glucagon (C), and amylin (D) responses to a 5-h mixed meal tolerance test with exenatide administration before the test (MMTT-Exn +; black squares, black lines) and without (MMTT-Exn -, white squares, black lines) exenatide administration before the test in the subgroup of five subjects that completed 12 month follow-up (subgroup 1). Glucose (E), C-peptide (F), glucagon (G), and amylin (H) responses to a 5-h mixed meal tolerance test with exenatide administration before the test (MMTT-Exn +; black squares, black lines) and without (MMTT-Exn -, white squares, black lines) exenatide administration before the test in the subgroup of six subjects that did not complete 12 month follow-up (subgroup 2).
Discussion
The peculiar pattern of islet allograft dysfunction in patients with T1DM, where C-peptide is produced but is inadequate to stabilize metabolic control, therefore requiring exogenous insulin, indicates a possible metabolic impairment of the marginal islet mass transplanted. There is increasing data on the incretin therapy efficacy in T2DM patients (5,8,14). This has generated great interest in the possible value of exenatide in preserving and/or improving the function of islet grafts showing progressive dysfunction in islet recipients (16,19). Ghofaili et al. (19), using hyperglycemic glucose clamps, demonstrated increased insulin secretory capacity, especially second phase insulin release, during exenatide administration. The current study examines both the acute and chronic metabolic and hormonal effects of 12 months of exenatide treatment as assessed by the physiologic stimulus of a mixed meal.
We show that chronic exenatide administration resulted in significant improvement in glucose responses during the MMTT at 3 and 6 months, when compared to baseline pre-exenatide. Additionally, there was significantly increased basal amylin/insulin ratio at 3 and 6 months and decreased proinsulin/insulin ratio at 6 months. These findings suggest improvement in islet function.
When comparing MMTT performed with and without exenatide at the same visit, the appearance of the glucose and glucagon response curves with exenatide was almost an inversion of those without exenatide. Interestingly, during the hour between exenatide administration and ingestion of the mixed meal, we observed an initial decrease in both glucagon and glucose concentrations (from −60 to 0 min); the former may be responsible for the latter in the absence of calorie ingestion. Even in the hour following the meal ingestion, the glucose levels remain below baseline and subsequently the glucose peak is very delayed, likely due to continued glucagon suppression combined with delayed gastric emptying effects. Prospective acetaminophen absorption studies (4) during MMTT are currently under way in an attempt to better elucidate the effect of exenatide on gastric emptying. The time-to-peak glucose significantly increased and was followed by that of insulin, proinsulin, amylin, and C-peptide. This observation suggests that insulin secretory response is glucose driven.
It has previously been demonstrated that glucagon levels remain unchanged in response to an oral glucose tolerance test in T1DM patients, as opposed to control subjects in whom glucagon levels decrease (21). Additionally, subjects with diabetes fail to suppress glucagon levels after carbohydrate meals and have an exaggerated glucagon response to protein meals (25) or arginine infusions (39). This is interesting, given that subjects with T1DM usually have normal GLP-1 and gastric inhibitory polypeptide (GIP) meal responses (40). In the present study, we demonstrated that islet transplant recipients with graft dysfunction have postprandial hyperglucagonemia before starting exenatide therapy. Acute exenatide administration suppressed the abnormal glucagon responses and prevented hyperglycemia after MMTT.
At 12 months, most of the beneficial changes observed at 3 and 6 months were no longer observed. Unfortunately, only five patients completed the 12-month follow-up; therefore, a small sample size could explain this lack of response. The retrospective analysis performed at 3 months to evaluate differences in patient responses demonstrated some marked differences in response to the medication. The tolerated doses of exenatide also ranged widely. This suggests that there are different degrees of responsiveness to exenatide within subjects that may be apparent early on. These differences may be multifactorial in nature, including islet composition, paracrine relationships between cells, specific responsiveness of each cell-type, as well as gastric emptying. The detection of responders versus nonresponders may help guide the clinical management of islet transplant recipients in the future.
Fineman et al. described that the infusion of the human amylin analog pramlintide can correct postprandial hyperglucagonemia observed in T1DM patients (15). Our patient population showed increased peripheral basal amylin/insulin ratio after exenatide treatment for 6 months, which may contribute to suppression of glucagon. The long-term significance of this increase in basal peripheral amylin remains to be defined. Constant stimulation by exenatide may lead to supraphysiological amylin secretion and this may be worse during hyperglycemia, as described by Rickels et al. (30). Our subjects demonstrated progressive increase in the amylin/insulin ratio when the chronic effects of exenatide were assessed. There is also the remote possibility that exenatide increases amylin secretion from different sites other than the islets (43). It is well known that intraislet amylin deposits (islet amyloid polypeptide-IAPP) are found in T2DM patients (7), but it is unclear if increased basal amylin peripheral concentrations are deleterious (36). Furthermore, the increased amylin and decreased proinsulin concentrations may reflect more efficient insulin processing and secretion by islet β-cells after exenatide treatment.
The changes observed in HbA1c levels up to 6 months are within acceptable range and are most likely related to adjustments in insulin and exenatide therapy. The significant increase in HbA1c levels observed at 12 months was likely secondary to the nonresponder nature of subgroup 1. In addition, the long interval in follow-up between 6 and 12 months did not allow for prompt intervention and adjustment of therapy.
There are several limitations of our study. There is no nondiabetic control group to assess exenatide effects on mixed meal response. While it would be interesting to see if changes are similar in healthy controls, it was not a necessity in this study where subjects acted as their own controls for the assessment of both the acute and chronic effects of exenatide. There is no control group of islet transplant recipients with graft dysfunction not treated with exenatide because there were insufficient subject numbers to perform a randomized controlled study with sufficient power to produce meaningful results. There is no control group of nondiabetic subjects on immunosuppression because these drugs could not be administered to this group in the long term due to potentially adverse effects on meal responses (38). In addition, the time from graft dysfunction to initiation of exenatide was variable in all study subjects, the reason being that the drug was not commercially available until FDA approval in 2005. Lastly, different responses in individual patients may have led to an unintended bias in subject selection. A larger randomized study may help clarify better the mechanisms leading to the different clinical and physiological exenatide responses in islet transplant recipients. Nevertheless, these differences were already apparent at 3 months of therapy. Perhaps performing MMTT with and without exenatide administration at the time of initiation of therapy may help identify those subjects that will be responders.
Measuring functional islet mass remains a major challenge in islet transplantation. Glucose potentiation of arginine-induced insulin secretion (GPAIS) was not performed in this group of patients (3,31,41) and therefore we cannot know whether increased β-cell reserve occurred in the subgroup of responders. If regeneration was to occur in islet allografts implanted ectopically (liver) despite multiple detrimental hits (i.e., high immunosuppressive levels and other toxins in the portal circulation, relative hyperglycemia in the liver, possible allorejection and autoimmunity), we postulate it would take much longer than the 6-month follow-up available. The reasons for the deterioration in some patients over time are unclear. The interpatient variability in both dose and effects may be one explanation. Another could be the formation of exenatide antibodies. These have been measured in the majority of subjects with type 2 diabetes clinical trials, but have been declared noninterfering. We were not able to measure antibody levels in these subjects. A third explanation could be the formation of islet amyloid as demonstrated in some islet transplant recipients and animal models (30). Regardless, exenatide demonstrated positive metabolic effects in islet transplant recipients with graft dysfunction and remain positive in several patients who continue on the medication (17).
Collectively, and based on the 12-month data collected, it appears that the effect of exenatide treatment in patients with islet allograft dysfunction is more metabolic than regenerative. Perhaps the use of exenatide (or other incretin mimetics) at all stages of islet preparation, transplantation, and follow-up (12,16,23) may have significant effects on engraftment and long-term islet allograft function and may even allow for more consistent achievement and preservation of insulin independence following transplantation of single donor islet preparation.
Footnotes
Acknowledgments
We express gratitude to the staff of the Clinical Islet Transplant Program, Islet Cell Processing Center, General Clinical Research Center and Informatics Core, for their continuous support. We also thank Drs. Jay S. Skyler, Luca Inverardi, Livio Luzi, and Daniel H. Mintz for valuable and stimulating discussions. This study was supported by National Institutes of Health (NIH)-National Center for Research Resources (U42-RR16603, GCRC-M01RR16587); NIDDK (R01-DK55347, R01-DK25802); JDRF International (4-2000-946 and 4-2004-361); State of Florida and Diabetes Research Institute Foundation. ClinicalTrials.gov Registration: NCT00306098; Islet Cell Transplantation Alone in Patients with Type 1 Diabetes Mellitus: Steroid Free Immunosuppresion. NCT00315588; Islet Cell Transplantation in Patients with Type 1 Diabetes with Previous Kidney Transplantation.
