Abstract
In this Commentary, we echo the conclusions of a recent review titled “The promise of stem cell-derived islet replacement therapy,” which highlighted recent advances in producing glucose responsive “islets” from stem cells and the benefits of their use in islet transplant therapy in type 1 diabetes (T1D). The review also outlined the status of clinical islet transplantation and the challenges that have prevented it from reaching its full therapeutic promise. We agree with the conclusions of the review and suggest that the identified challenges may be overcome by using the eye anterior chamber as an islet transplant site. We anticipate that the combination of stem cell-derived islets and intraocular transplant could help this promising T1D therapy reach full fruition.
Keywords
We read with great interest the review by D. Melton recently published in Diabetologia journal, titled “The promise of stem cell-derived islet replacement therapy.” 1 This review elegantly outlined the problem of insulin insufficiency resulting from the autoimmune destruction of beta cells in type 1 diabetes (T1D) and the challenges and potential solutions in addressing such a problem. The review focused on the biological replacement of insulin through transplantation of beta cell-like clusters and possibly other endocrine cells derived from stem cells in vitro. It highlighted the latest achievements in producing such glucose responsive “islets” in an efficient and reproducible manner. The advent of producing fully functional stem cell-derived islets is undoubtedly exciting and encouraging for the treatment of T1D and, possibly, type 2 diabetes (T2D), through transplantation. The review also addressed the critical shortage of pancreatic islets obtained from cadaveric organ donors for clinical transplantation. Although there is room for further refinement, stem cells provide a virtually limitless source of uniform supply of insulin-producing cells for clinical transplantation and research applications.
The review also asserted that addressing the islet supply challenge is only half of the solution, as mitigating the immune responses against transplanted islets remains a significant challenge, despite substantial advancements in immune therapies. Notably, protecting the transplanted islets from both allogeneic (allo) and autoimmune responses is required in the diabetic recipients, regardless of whether cadaveric or stem cell-derived islets are used. Moreover, transplanted islets derived from autologous stem cells could also be subject to recurrent autoimmunity that caused the recipient’s T1D. Indeed, protecting the transplanted islets from recurrent autoimmunity may be more challenging, as evidence has shown recurrent T1D in some transplant recipients who are on continuous immune suppression adequate to avoid allorejection 2 .
The review further discussed important considerations in islet transplant therapy to ensure its durability on the long-term. These included
We agree with the conclusions of this review, namely, that a transplant approach that can help in addressing the challenge of a suitable transplant site while simultaneously imparting immune protection onto the graft would be instrumental for this promising therapeutic procedure to reach full fruition. However, does such an “ideal” transplant site exist in the human body? Immune privileged sites, where immune responses are dampened, could potentially offer such a site. However, this site should be easily accessible without causing significant harm to the recipient. There are several sites in the body that are immune privileged (e.g., testis, brain, placenta, and anterior chamber of the eye). While the testicular site has been used in animal studies 4 , clearly, not all these sites are suitable for clinical islet transplant. In 2008, we introduced islet transplant in the anterior chamber of the mouse eye to study the islet physiology in vivo 5,6 . Three years later, we reported that intraocular islet transplant is suitable for clinical application based on nonhuman primate studies that demonstrated its feasibility and potential efficacy 7 . Since then, we have further developed intraocular islet transplant to study immune reactions against pancreatic islets during the development of type 1 diabetes and following transplant 8,9 . Hence, we have been using intraocular islet transplant to study various aspects of the islet immunobiology in health and disease 10 –19 .
Our extensive pre-clinical studies with intraocular islet transplant have demonstrated its technical and physiological advantages (Fig. 1). This includes enabling high-resolution monitoring of the graft survival and function noninvasively and longitudinally 5,20 , high local oxygen tension conducive to the transplanted islets’ efficient engraftment, revascularization, and reinnervation 13,14,21 , and allowing direct access to the graft immediate microenvironment for diagnostic and therapeutic purposes 22 –25 . Our studies have also shown how timely therapeutic interventions are facilitated by intraocular transplant if/when needed to prevent rejection or recurrent autoimmunity based on direct information derived from the intraocular islet grafts, and how such interventions are feasible, not only systemically, but also locally via topical application (i.e., eye-drops). They further demonstrated that intraocular islet grafts are reliable reporters of the autoimmune attack against islets in the native pancreas and extra-pancreatic sites, and how information obtained from such reporter islets enabled the prediction of T1D onset 24 and the transplant outcome (i.e., rejection versus tolerance) in murine models 26 .
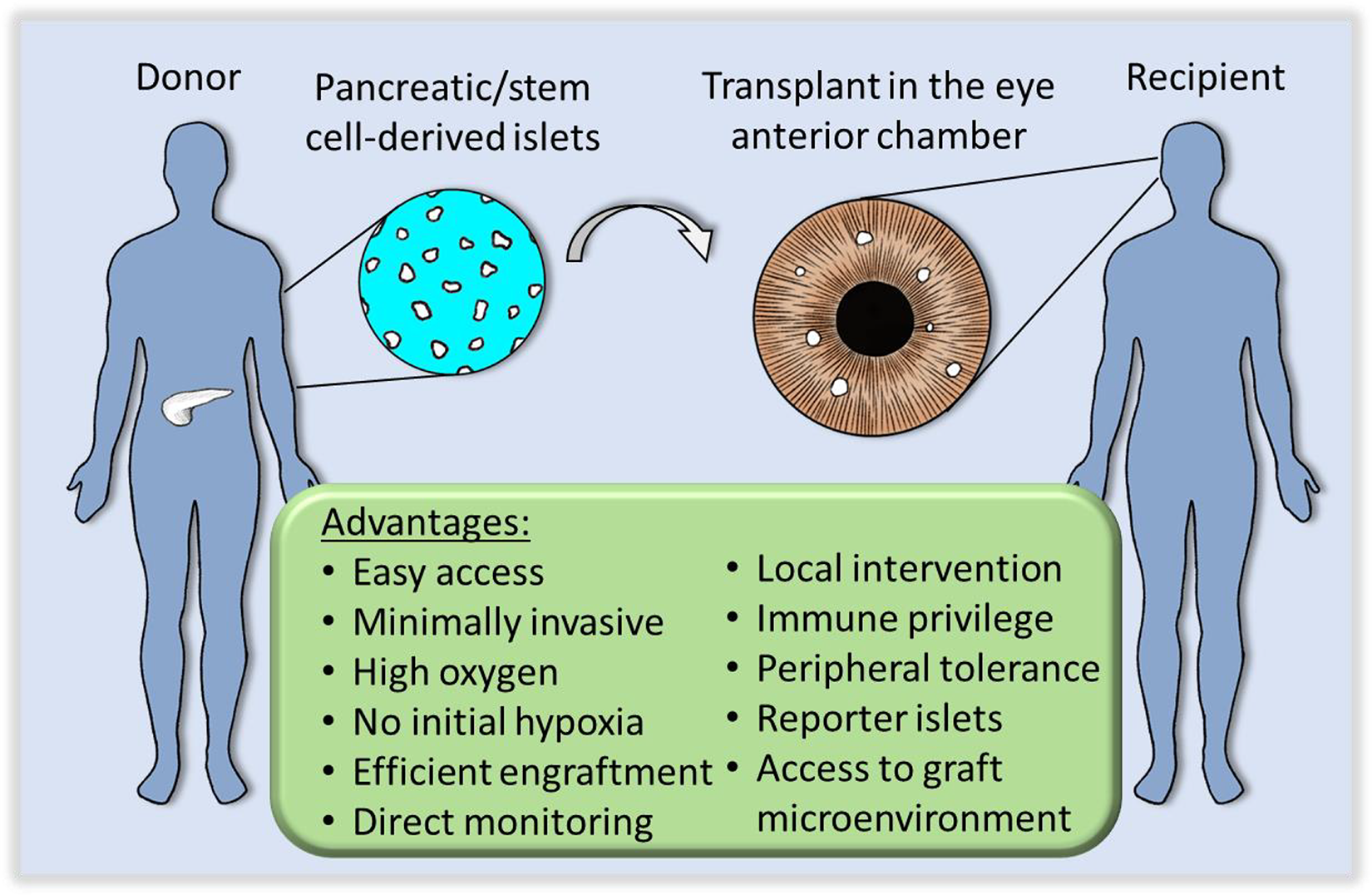
Islet transplant in the anterior chamber of the eye offers technical and physiological advantages that promote the graft survival on the short- and long-term. Schematic depiction of the transplant of pancreatic or stem cell-derived islets into the anterior chamber of the eye (credit: Zein M. Abdulreda and Tilo Moede).
Moreover, our studies demonstrated immunosuppression-free, long-term survival of intraocular islet allografts in mouse and nonhuman primate models 27,28 . These studies provided evidence for local and peripheral immune regulation consistent with donor-specific tolerance that promoted the survival of additional islet grafts from the same donors 27 . Importantly, this establishes a significant proof-of-concept for a potential approach of sequential islet transplants, whereby immune tolerance is induced through an initial intraocular islet transplant followed by an additional transplant of a curative islet dose (mass) in a preferred extra-pancreatic site. Notably, the recent advent of stem cell-derived islets is directly conducive to this sequential transplant approach since it provides “identical” islets from the same source (donor) for repeat transplants. This is significant when considering the poor islet recovery following cryopreservation and would be a great complementation to intraocular islet transplant in future clinical applications.
Encouraged by our pre-clinical studies, we recently initiated a pilot clinical trial to evaluate the safety and potential efficacy of intraocular islet transplant in select T1D patients with impaired hypoglycemia awareness and severe hypoglycemia (i.e., problematic hypoglycemia) 29 (ClinicalTrials.gov Identifier: NCT02846571). While we await the results from this trial, the pre-clinical and early clinical evidence thus far indicates that intraocular islet transplant will likely be safe in humans and may impact significantly on diabetes, as we previously discussed 30 . If, however, the human data show that complete insulin independence is not achieved through intraocular islet transplant alone, it is likely that the islet mass in the anterior chamber of one or both eyes will reduce hypoglycemic episodes consistently with previous trials, which will significantly improve the quality-of-life of the recipients 31,32 . Moreover, this could be further addressed by the sequential transplant approach discussed above, which is significantly facilitated by the advent of stem cell-derived islets.
In summary, the recent review by D. Melton highlighted the promise of stem cell-derived islets in clinical islet transplant but also pointed out the remaining challenges to solve. Using the eye anterior chamber as an islet transplant site may overcome these challenges. The eye anterior chamber is easily accessible, and the overwhelming evidence thus far supports its clinical utility where many of its technical and physiological advantages can be leveraged to promote the survival and function of islet transplants in the long-term. Thus, the combination of stem cell-derived islets and intraocular transplant could help islet transplant therapy reach full fruition.
Footnotes
Acknowledgments
Contribution statement
M.H.A: Writing and making critical revisions to the article, final approval of the article to be published. P-O.B: Writing and making critical revisions to the article, final approval of the article version to be published.
Declaration of Conflicting Interests
P-O.B is cofounder and CEO of Biocrine, an unlisted biotech company that is using the anterior chamber of the eye technique as a research tool. M.H.A is consultant for the same company. The funders were not involved in this work and the views expressed in it are solely those of the authors.
Ethical Approval
No animal studies are directly reported on in this commentary. All referenced animal studies were approved by the University of Miami’s Institutional Animal Care and Use Committee (IACUC).
Statement of Human and Animal Rights
All procedures and studies referenced in this article were conducted in accordance with the University of Miami’s Institutional Animal Care and Use Committee (IACUC) approved protocols (e.g., protocol # 20-133).
Statement of Informed Consent
There are no human subjects in this study and informed consent is not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research referenced in this article: Our work was supported in part through funds from the Diabetes Research Institute Foundation (DRIF) and the Diabetes Wellness Foundation and through grants from the National Institute of Allergy and Infectious Diseases (NIAID) – R56AI130330 – the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) – F32DK083226 and K01DK097194. It was also supported in part by the Swedish Research Council, the Family Erling-Persson Foundation, the Novo Nordisk Foundation, the Stichting af Jochnick Foundation, the Swedish Diabetes Association, the Berth von Kantzow’s Foundation, the Strategic Research Program in Diabetes at Karolinska Institutet, the European Research Council grant ERC-2018-AdG834860EYELETS, the Swedish Foundation for Strategic Research, and the Knut and Alice Wallenberg Foundation.
