Abstract
Dermal papilla (DP) cells are associated with the development of hair follicles (HFs) and regulation of the hair cycle. However, primary DP cells prepared from cultured HFs are known to lose their ability to induce HF after culturing in standard media, for example, fibroblast growth conditions. We explored a new culture condition by which DP cells maintained their HF induction ability. The addition of Wnt-10b to the first culture of primary DP cells promoted their proliferation and maintained their Wnt responsiveness and HF induction ability. Furthermore, DP cells in Wnt-10b-containing medium sustained those characteristics after 10 passages (100 days), which encompassed the entire experimental period. These results suggest that Wnt-10b plays a pivotal role in proliferation and maintenance of DP cells in vitro.
Introduction
Dermal papilla (DP) cells are specialized fibroblasts located in hair follicles (HFs) and deeply associated with the development of HFs and regulation of the hair cycle (1,5,24). Therefore, DP cells are an important target for elucidating the mechanism of HF induction and hair growth. Technical advances in isolation of DP cells from HFs and subsequent in vitro culture have made it possible to investigate the ability of these cells to induce HFs and promote hair growth (15,17,22,26).
It is well known that DP cells lose their ability for HF induction and do not promote hair growth after culturing in standard media for fibroblasts [e.g., Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal bovine serum (FBS)] (12,16,26). Therefore, it is considered important to establish an effective culture method by which the functions of DP cells are maintained. Although the effectiveness of conditioned medium obtained from cultures of skin epithelial cells has been reported (12), the medium components were not chemically elucidated in that study. As a defined chemical substance, fibroblast growth factor-2 (FGF-2) has been reported to promote DP cell proliferation along with an ability of HF induction (26). Furthermore, DP cells cocultured with Wnt-3a-producing chick embryonic fibroblasts were demonstrated to sustain their ability to induce HFs (17), suggesting that canonical Wnts may be important for DP cells. Another study provided supportive evidence concerning the role of Wnts, as it reported that a glycogen synthase kinase (GSK) inhibitor [(2′Z,3′E)-6-bromoindirubin-3′-oxime (BIO)] acted favorably for maintaining DP function (43).
Wnts are expressed in HFs, which are composed of epithelial cells and DP cells (10), throughout life from embryo to adult (32). We previously investigated the effects of Wnts (Wnt-3a, -5a, -10b, -11) on the differentiation of mouse primary skin epithelial cells and found that only Wnt-10b promoted differentiation of cultured epithelial cells and shaft growth of cultured hair follicles (27–29). However, there are no reports documenting the direct effects of Wnt proteins on DP cells. In the present study, we investigated the effects of Wnt-10b on primary DP cells prepared from DP cultures and DP cells after serial passages.
Materials and Methods
Animals
Inbred 4-week-old C3H/HeN and 8-week-old Balb/c nude (nu/nu) mice were purchased from Japan SLC (Hamamatsu, Japan). Adult C3H/HeN mice were used for preparation of DP cells, while postnatal day 2 (P2) C3H/HeN mice were used for preparation of primary skin epithelial cells and dermal fibroblasts, and the Balb/c nude mice were used for hair reconstitution experiments. All animal procedures were conducted in accordance with our institutional guidelines as well as those of the National Institutes of Health.
Primary Cultures of DPs After Isolation From Vibrissa (Fig. 1)

Experimental design and procedures. (A) Outline of the experiments. Dermal papilla (DP) cultures: DPs were isolated from vibrissa of C3H/HeN mice using a microdissection method, then placed in 6-cm dishes, and quietly cultured in Dulbecco's modified eagle's medium (DMEM) containing 10% fetal bovine serum (FBS). DP outgrowths were harvested with 0.25% trypsin-EDTA on day 10 and then used for DP cell cultures. DP cell cultures: Primary DP cells (1 × 103) harvested from DP cultures were seeded into 6-cm culture dishes and cultured in DMEM containing 10% FBS, COS supernatant (mock-COS sup), or Wnt-COS supernatant (Wnt-COS sup). DP cells were harvested on day 10 and used for RT-PCR and TOPFLASH assays or transplantation. DP cells cultured in Wnt-COS sup were passaged every 10 days until the end of the 10th culture. (B) Isolation and cultivation of DP cells. (a) Vibrissa follicles were isolated from 4-week-old C3H/HeN mice. (b) The collagen capsule surrounding each follicle was removed to expose the follicle base, and then the DP (enclosed by dotted line) was dissected. Scale bars: 250 μm (in a and b). (c) DP cells appeared only around the DP (arrow) on day 4. (d) During the following 2 days, DP cells showed rapid outgrowth. (e, f) By day 10, DP cells had spread to all areas of the dishes and showed alkaline phosphatase (ALP) activity. Scale bars: 100 μm (in c–f). (C) Experimental procedures for hair reconstitution assay. Epidermal keratinocytes from P2 C3H/HeN mice (2.5 × 106) and DP cells (2.5 × 106) were used for transplantation into Balb/c nude mice. (a) Silicon transplantation chambers were implanted into dorsal sites of Balb/c nude mice, and then a mixture of cells was injected into each of the chambers. (b) One week after transplantation, the roof of the chamber was cut off. (c) The chamber was removed 2 weeks after transplantation. (d) Two weeks after removing the chambers, hair follicle formation in the reconstituted skin was assessed.
DPs were isolated from mouse vibrissa of 4-week-old C3H/HeN mice using a microdissection method, as previously reported (26). Briefly, the mystacal pad was cut open, the skin was inverted, and follicles were removed with microscissors (Fig. 1B-a). The collagen capsules surrounding vibrissae follicles were removed to expose the follicle base, and DPs were dissected out using fine forceps (Fig. 1B-b). Each DP was placed on a 6-cm dish and quietly cultured for 4 days in DMEM containing 10% FBS (Fig. 1B-c). DP cells appeared only around the DPs and began to show rapid outgrowth over the following 2 days (Fig. 1B-d). By day 10, cells had spread throughout all areas of the dishes (Fig. 1B-e) and a high level of alkaline phosphatase (ALP) activity was expressed in nearly all of the cultured DP cells (Fig. 1B-f). At the end of 10 days, DP outgrowths were harvested with 0.25% trypsin-EDTA and used for culturing of DP cells after washing with saline.
Preparation of rWnt-10b
We previously established a Wnt-10b-secreting COS [CV-1 (simian) in Origin and carrying the SV40 genetic material] cell line (Wnt-COS cells) by introducing the Wnt-10b cDNA gene (29). mock-COS and Wnt-COS cells were seeded into 10-cm dishes and then cultured for 48 h in DMEM containing 10% FBS. After 48 h, the supernatants were collected and used as culture media for DP cells after filtration with a 0.22-μm filter membrane. It has been reported that the culture supernatant of Wnt-COS cells (Wnt-COS supernatant) contains bio active Wnt-10b protein, whereas the culture supernatant of mock-COS cells (mock-COS supernatant) does not (29).
Culture of DP Cells
Outlines of the DP and DP cell culture protocols are shown in Figure 1A. One thousand harvested primary DP cells from DP cultures were seeded into 6-cm culture dishes and cultured in DMEM containing 10% FBS, COS supernatant, or Wnt-COS supernatant, with the medium changed every 4 days. DP cells were harvested on day 10 using 0.25% trypsin-EDTA and used for RT-PCR experiments, TOPFLASH assays, or transplantation. DP cells cultured in Wnt-COS supernatant were passaged every 10 days until the end of the 10th culture.
Alkaline Phosphatase (ALP) Activity
The expression of ALP was observed using an alkaline phosphatase kit (Sigma), according to the manufacturer's protocol. Briefly, the cells were washed twice in saline, fixed in fixation solution at room temperature, and washed with water. Dye solution was added, and incubation was performed at room temperature for 30 min. The cells were then rinsed well with water for three to four times and observed under a light microscope (2,20).
Proliferation Assay
DP cells were plated at a density of 50 cells per well in flat-bottom 96-well plates, and cultured with or without recombinant Wnt-10b. The culture plates were subjected to cell proliferation assays with a CyQUANT® Cell Proliferation Assay Kit (Molecular Probes, CA).
TOPFLASH Assay
Involvement of the canonical Wnt signaling pathway was examined using reporter assays. Two reporter plasmids, pTOPFLASH, carrying the T-cell factor (TCF)-binding consensus sequence followed by the luciferase gene, and pFOPFLASH, carrying the dominant-negative TCF-binding sequence instead of the wild-type sequence in pTOPFLASH, were kindly supplied by Dr. B. Vogelstein (19). DP cells were transfected with the reporter plasmids, and then 4 h later, the cell culture medium was removed and replaced with medium with or without Wnt-10b. After 48 h of incubation, the cells were lysed and luciferase activity was quantified using a luciferase reporter assay kit (Clontech, Worcester, MA), as recommended by the manufacturer, and normalized using the level of β-galactosidase (β-gal) as the internal control.
RT-PCR
Total RNA was purified using Trizol (Invitrogen, Carlsbad, CA) following the protocol of the manufacturer. One microgram of DNase-treated total RNA was used for first-strand cDNA. This reaction was performed using a random primer (Invitrogen) and Moloney murine leukemia virus (M-MLV) reverse transcriptase (Promega, Madison, WI). For PCR analysis, 0.5 μg of cDNA was used as a template, and amplification was performed using the primer sequences shown in Table 1. The general PCR conditions were 25–30 cycles at 94°C for 2 min, 94°C for 30 s, 52–62°C for 30 s, and 72°C for 1 min. The PCR products were run on 1.5% agarose gels.
Gene-Specific Primers Used in the Present Study
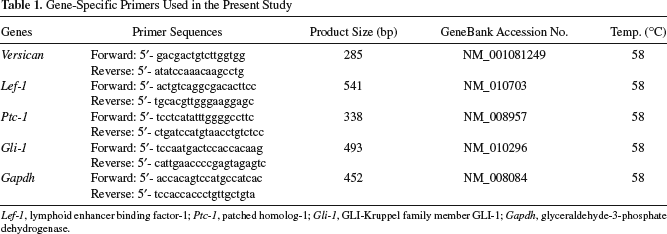
Lef-1, lymphoid enhancer binding factor-1; Ptc-1, patched homolog-1; Gli-1, GLI-Kruppel family member GLI-1; Gapdh, glyceraldehyde-3-phosphate dehydrogenase.
Hair Reconstitution Assay
A hair reconstitution experiment was performed as previously reported (18,28). Briefly, epidermal keratinocytes were isolated from the dorsal skin areas of C3H/HeN mice on postnatal day 2. The isolated skin samples were washed several times with PBS and incubated for 2 h at 37°C in 0.25% trypsin. Detachment of the dermis from the epidermis was performed gently using forceps, after which cells were scrubbed from the epidermis-facing surface of the dermis using the belly of the forceps. The keratinocytes thus obtained were suspended in Epilife™ serum-free culture medium and plated in 10-cm dishes coated with collagen type I and incubated for 2 days and then used as keratinocytes for transplantation. Mixture solutions containing epidermal keratinocytes (2.5 × 107/ml) and DP cells (2.5 × 107/ml) passaged with DMEM, mock-COS supernatant, or Wnt-COS supernatant were used for transplantation into Balb/c nude mice (n = 3 or 5) (Fig. 1C). Silicon transplantation chambers, kindly supplied by Dr. J. Kishimoto, were implanted into dorsal sites of the Balb/c nude mice (Fig. 1C-a) where the skin had been removed, and then 100 μl of mixture was injected into each of the chambers. One week after transplantation, the roof of the chamber was cut off to facilitate drying of the injured site (Fig. 1C-b), and the chamber was removed 2 weeks after transplantation (Fig. 1C-c). Two weeks after removing the chambers, skin tissues were harvested, embedded in OCT compound, and stained with hematoxylin–eosin, and then hair follicle formation in the reconstituted skin was assessed (Fig. 1C-d).
Immunohistochemistry
On day 28, grafted mice were terminally anesthetized using an overdose of pentobarbital IP, after which the grafted skin tissues were removed and postfixed for 24 h in 4% paraformaldehyde in PBS at 4°C and then sectioned. The sections were equilibrated in 10% sucrose in PBS for 4 h at 4°C, then 15% sucrose in PBS for 4 h at 4°C, and finally in 20% sucrose in PBS overnight at 4°C. Next, they were embedded in OCT compound (Tissue-TEK, Miles, Elkhart, IN) and frozen in liquid nitrogen. The sections were cut into 10-μm thick slices using a cryostat and placed on 3-aminopropyltriethoxysilane-coated slides. After rinsing in PBS, they were permeabilized in methanol at −20°C for 5 min. The sections were then blocked with HISTOMOUSETM blocking solution (Zymed Lab., Carlsbad, CA) and exposed to primary antibodies, followed by secondary antibodies. The following antibodies and dilutions were used: AE13 and AE15 (mouse, diluted 1:100, kindly provided by Dr. T. T. Sun) (3) and Alexa 546-conjugated antimouse antibodies (diluted 1:200, Molecular Probes). AE13 and AE15 are known to recognize the cortex (cytokeratin) and inner root sheath (trichohyalin), respectively, of mouse hair follicle tissue (21). Nuclei were stained with DAPI (4′6′-diamidino-2-phenylindole). Fluorescence was examined using a FLUOVIEW FV1000 confocal analyzer (OLYMPUS, Japan) or Zeiss Axiovert 200 microscope.
Statistical Analysis
Data are expressed as the mean ± SD of five independent experiments. Statistical significance was tested using Tukey's test. Results were considered significant at p < 0.05.
Results
Wnt-10b Promotes Proliferation of Primary DP Cells
DP cells were harvested from primary DP cultures on day 10, and 1 × 103 cells were cultured in 6-cm culture dishes in DMEM containing 10% FBS (control medium: none), mock-COS supernatant, or Wnt-COS supernatant (Fig. 2A). The proliferation of DP cells was prominently promoted in the culture with Wnt-COS supernatant, and the cell number after 10 days of culture showed an approximately fivefold increase, as compared to the control medium and mock-COS supernatant. Chemical proliferation assay findings on day 10 similarly showed promoted proliferation of DP cells cultured in Wnt-COS supernatant (Fig. 2B). The time course results of DP cell proliferation confirmed the promoted proliferation by Wnt-COS supernatant (Fig. 2C, D). In contrast, the proliferation of DP cells was scarcely promoted in control medium and mock-COS supernatant. The appearance of representative DP cells cultured in Wnt-COS supernatant is shown in Figure 2E. On day 2, DP cells were sparsely distributed in the culture dishes. However, the cells prominently proliferated and formed multilayered clumps in areas of proliferation by day 6. ALP activity, one of the characteristics of DP cells, was retained in nearly all (>90%) of the cultured DP cells on day 10 (Fig. 2F).

Wnt-10b promotes proliferation of primary DP cells. (A) The proliferation of DP cells was prominently promoted in cultures with Wnt-COS supernatant, with an approximately fivefold increase after 10 days. ∗p < 0.05. (B) Chemical proliferation assay findings on day 10 showed similarly promoted proliferation of DP cells in Wnt-COS supernatant. ∗p < 0.05. (C, D) The time course results of DP cell proliferation confirmed the promoted proliferation by Wnt-COS supernatant. (E) On day 2, DP cells were sparsely distributed in the culture dishes. Thereafter, they prominently proliferated and formed multilayered clumps in areas of proliferation by day 6. (F) ALP activity was retained in nearly all of the cultured DP cells (>90%). Scale bars: 50 μm (in E and F).
Wnt-10b Sustains Wnt Responsiveness in Cultured DP Cells
We next examined whether primary DP cells maintained Wnt responsiveness after 10 days of culture using a pTOPFLASH reporter assay. DP cells cultured in Wnt-COS supernatant had remarkable reporter activities (Fig. 3A). On the other hand, there were negligible activities in DP cells cultured in control medium or mock-COS supernatant.

Wnt-10b sustains Wnt responsiveness of cultured DP cells. (A) TOPFLASH assay findings revealed that DP cells cultured in Wnt-COS supernatant had remarkable reporter activities. ∗p < 0.05. (B) RT-PCR analysis showed that DP cells cultured with Wnt-10b distinctly expressed versican and Lef-1, while Ptc-1 was faintly expressed and Gli-1 was not expressed.
We also examined the gene expressions of versican, lymphoid enhancer-binding factor-1 (Lef-1), patched homolog-1 (Ptc-1), and glioma (GLI)-Kruppel family member GLI-1 (Gli-1) after 10 days of culture (Fig. 3B). Versican is known to be expressed in DP cells, beginning in developing follicular papilla (5). Lef-1 is a representative downstream gene for canonical Wnt signal (41), whereas Ptc-1 and Gli-1 are the receptor for hedgehog (Hh) and a downstream gene for Hh signaling, respectively (8). In the present study, isolated DPs expressed all of these genes (Fig. 3B, fresh DP lane). DP cells cultured with Wnt-COS supernatant distinctly expressed versican, Lef-1, and faintly Ptc-1, but not Gli-1 (Fig. 3B, lane Wnt-COS). On the other hand, none of those were expressed in DP cells cultured in control medium or mock-COS supernatant (Fig. 3B, none and mock-COS lanes). These results suggest that Wnt-10b-treated DP cells had sustained Wnt responsiveness and versican expression.
Sustained HF Induction Activity in DP Cells Cultured with Wnt-10b
It is well known that DP cells play a pivotal role in HF induction and hair growth (1,24,33). We examined whether primary DP cells retained their ability to induce HFs after 10-day cultures using in vivo skin reconstitution assays (Fig. 1C). DP cells cultured in control medium, mock-COS supernatant, or Wnt-COS supernatant were cotransplanted with epithelial cells, obtained from the dorsal skin of P2 C3H/HeN mice, into silicon chambers set in the dorsal sites of Balb/c nude mice. Hair growth became visible within 3 weeks and was distinct at 4 weeks after transplantation in the reconstituted skin-grafted Wnt-COS supernatant-treated DP cells, whereas no hair growth was seen in skin-grafted DP cells cultured in either control medium or mock-COS supernatant (Fig. 4A and Table 2). At 4 weeks, numerous HFs were observed histologically in reconstituted skin transplanted with Wnt-COS supernatant-treated DP cells (Fig. 4B). However, there were no or few HFs in reconstituted skin with untreated or mock-COS supernatant-treated DP cells.
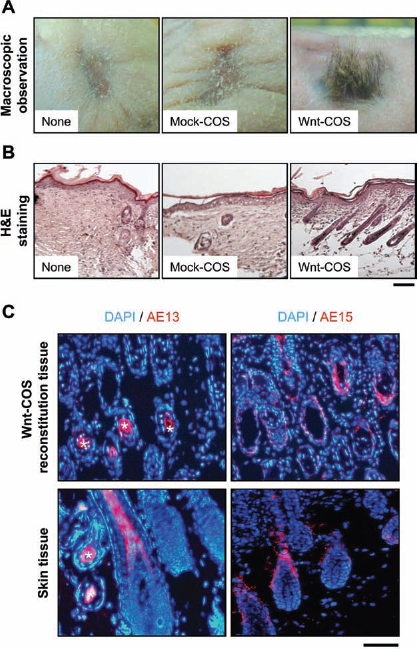
Hair follicle (HF) induction activity sustained in DP cells cultured with Wnt-10b. (A) Following transplantations of mixtures of epithelial cells and DP cells cultured in control medium or mock-COS supernatant, hair growth was not observed (none and mock-COS, respectively). In contrast, cotransplantation with epithelial cells and Wnt-COS supernatant-treated DP cells resulted in distinct hair growth (Wnt-COS, see Table 2). (B) Numerous HFs were observed histologically in reconstituted skin transplanted with Wnt-COS supernatant-treated DP cells (Wnt-COS). However, there were no or few HFs in reconstituted skin with untreated or mock-COS supernatant-treated DP cells (none and mock-COS, respectively). Scale bar: 200 μm. (C) Among skin-grafted epithelial cells and Wnt-COS supernatant-treated DP cells, both AE13- and AE15-immunopositive cells were observed, the same as hair follicles in normal skin (skin tissue). ∗Hair cortex. Scale bar: 100 μm.
Results of Hair Reconstitution Assay of Cultured Dermal Papilla (DP) Cells

Epidermal keratinocytes (Epi) were prepared from P2 C3H/HeN mice. Epi (+, 2.5 × 106; -, none) were transplanted into Balb/c nude mice with or without DP cells.
DP cells (2.5 × 106) after the 2nd or 10th passage (DP-2p, DP-10p, respectively) were used for transplantation. Words in parentheses indicate DP cells cultured in control medium (none), mock-COS supernatant (mock-COS), or Wnt-COS supernatant (Wnt-COS).
Four weeks after transplantation, hair induction was assessed. The numerators and denominators indicate numbers of mice that grew hair and received grafts, respectively. Numbers in parentheses indicate the percentage of hair induction.
HFs are composed of DP cells and epithelial cells, and hair growth involves the differentiation of epithelial cells. We investigated epithelial cell differentiation in reconstituted skin using AE13 and AE15 antibodies, which are known to react to the hair shaft and inner root sheath, respectively (21). An immunostaining study demonstrated that HF-like structures in reconstituted skin with Wnt-COS supernatant-treated DP cells were AE13- and AE15-immunopositive and stained in a manner similar to hair follicles in normal skin (Fig. 4C, D).
Properties Maintained in Serially Passaged DP Cells by Wnt-10b
We examined DP cells after 10 passages in Wnt-COS supernatant (DP-10p cells). DP-10p cells proliferated in a manner similar to DP cells after two passages in Wnt-COS supernatant (DP-2p) (Fig. 5A–C) and retained ALP activity (Fig. 5D, E). In addition, pTOPFLASH reporter assay findings revealed that Wnt responsiveness was maintained in DP-10p cells (Fig. 5F), and RT-PCR results showed clear expressions of Lef-1 and versican, with a trace of Ptc-1 expression (Fig. 5G). Furthermore, DP-10p cells exhibited HF induction activity in a skin reconstitution assay (Fig. 5H, I). HF induction was observed only in skin that was reconstituted with epithelial cells and in Wnt-COS supernatant-treated DP cells (Table 2), and the HF structures contained AE13-and AE15-immunopositive differentiated epithelial cells (Fig. 5J, K). Our findings for DP-10p cells were consistent with those for DP-2p cells.

Properties maintained in serially passaged DP cells by Wnt-10b. (A, B) DP cells after 10 passages in Wnt-COS supernatant (DP-10p cells) were compared with those after two passages in Wnt-COS supernatant (DP-2p). DP-10p cells proliferated in a manner similar to DP-2p cells in regard to cell number and chemical proliferation assay findings. (C) DP-10p and DP-2p cells showed similar time-course proliferation. (D, E) DP-10p cells formed multilayered clumps on day 6 and retained ALP activity. Scale bar: 100 μm. (F) pTOPFLASH reporter assay findings revealed DP-10p cells maintained Wnt responsiveness, similar to DP-2p cells. (G) RT-PCR findings showed clear expressions of Lef-1 and versican, with a trace expression of Ptc-1. (H) Hair growth was observed in skin reconstituted with DP-10p cells. (I) HFs were observed histologically in reconstituted skin with DP-10p cells at 4 weeks after transplantation. Scale bar: 200 μm. (J, K) HF structures contained AE13- and AE15-immunopositive differentiated epithelial cells. Scale bar: 100 μm.
Discussion
The DP is formed from mesenchymal cells via epithelial–mesenchymal interactions during embryonic HF formation (10,22,24). DPs are located at the base of the follicle and communicate with epithelial matrix progenitors (matrix cells), which differentiate into the inner root sheath (IRS) and hair shaft (30). In mammals, the DP is known to express molecular signals involved in HF development and cycling (13,31). Therefore, DPs are extremely important for evaluating the mechanisms of HF induction and cycling.
Although DP cells can be obtained from a DP culture, they lose their ability to induce HFs during prolonged cultures (12,16,26). To prevent the loss of HF induction ability, some improvements in culture conditions have been reported (12,26). For example, use of conditioned medium harvested from epithelial cell cultures allowed DP cells to retain their follicle induction ability even after long-term cultivation, although such conditioned media may contain a variety of undetermined factors (12). FGF-2 was also reported to be effective for more than 30 passages when DP cells were serially cultured as a floating cell aggregation, so-called sphere formation (26). Furthermore, Kishimoto et al. reported that the HF induction ability of DP cells was maintained by coculturing with Wnt-3a-expressing feeder cells (17), suggesting that Wnt-3a signaling has favorable effects on the follicle induction ability of DP cells. However, there are no reports regarding humoral factors capable of maintaining fully functional DP cells in adhesive flat cultures without assistance from feeder cells.
Wnts are divided into two groups according to their signal transduction pathways (4); canonical Wnt signaling, in which β-catenin stabilization occurs, and noncanonical Wnt signaling, in which Ca2+ flux (noncanonical Wnt/Ca pathway) or activation of the Jun N-terminal (noncanonical planar cell polarity, PCP pathway) is involved. In experiments with transgenic mice possessing gain-of-function mutations of the β-catenin gene, promotion of epithelial cell differentiation and new HFs were observed (7). On the other hand, downregulation of LEF-1 resulted in a decreased number of HFs and disappearance of the cuticle and medulla (41). Moreover, inhibition of Wnt signaling was shown to abrogate folliculogenesis after reepithelialization induced by wounding, whereas overexpression of Wnt ligand in the epidermis increased the number of regenerated hair follicles (14). Taken together, it is strongly suggested that Wnt signals are important for HF formation and may have an influence on DP cells as well as epithelial cells.
In an extensive number of studies of Wnt gene expression in skin tissue, Wnt-10b has been shown to be expressed in developing HFs, with the earliest and most marked localization in placodes (32), suggesting that Wnt-10b promotes the differentiation of skin epithelial cells and development of HFs. Therefore, we previously examined the effects of Wnt-10b on epithelial cells and found that Wnt-10b promotes differentiation of immature epithelial cells (29), elongation of the hair shaft in HF cultures (27), and development of HFs in an in vivo skin reconstitution assay (28). In the present study, we investigated the effects of Wnt-10b on DP cells, which are counterparts to epithelial cells in the development of HFs, particularly to determine whether DP cells maintain their ability to induce HFs in vitro in the presence of Wnt-10b. Others have reported that matrix cells surrounding DP cells express Wnt-3a and Wnt-10b, both of which are canonical Wnts (25,40). In addition, Kishimoto et al. showed that coculturing with Wnt-3a-expressing cells sustained the HF induction ability of DP cells (17), although the effects of Wnt-10b on DP cells in vitro have not been reported.
Our findings are the first to show the effects of Wnt-10b on cultured DP cells, as they revealed that medium containing recombinant Wnt-10b protein promoted the proliferation of DP cells, which successfully maintained their ability to induce HFs. We also confirmed that DP cells cultured in medium containing Wnt-10 sustained their responsiveness to Wnt-10b and ability to induce HFs up to at least 10 serial passages. Although not shown in the present study, we further obtained results showing that Wnt-10b protein prepared via a purification method with an affinity column (29) promoted the proliferation of DP cells, which maintained their HF induction ability even after 10 passages. These findings strongly suggest that Wnt-10b is an efficient and effective agent to maintain DP cells with their functional activities in culture.
ALP activity is known to be detected in DP cells throughout the HF cycle and is used as a marker of DP cells in vivo and in vitro (9,23). HF induction activity is considered to be closely associated with ALP activity in DP cells. In mice, ALP activity in DP cells was found to reach its maximal level in early anagen and then decrease after the mid-anagen growth phase (11). Although the periodicity of ALP activities in cultured DP cells is unknown, sustained ALP activity in sphere DP cells in prolonged culture with FGF-2 has been reported (26). It was also recently shown that bone morphogenetic protein-6 (BMP-6) ablation results in failure of HF generation (38), while DP cells cultured with BMP-6 maintained their ALP activity (34). Herein, we present novel findings that Wnt-10b also has an ability to maintain ALP activity in DP cells in vitro.
As for other factors associated with the HF induction activity of DP cells, versican is reported to be deeply involved in DP function (17,18). Versican is a chondroitin sulfate proteoglycan (CSP) localized in DP cells of adult murine HFs (17), and its expression is known to be lost in miniaturized hair follicles of androgenetic alopecia (39). It was also shown that versican expression in DP cells is correlated with HF induction activity based on analysis of versican-green fluorescent protein (GFP)-transgenic (Tg) mice, in which the expression of GFP protein is driven by the versican promoter gene (18). In the present study, we demonstrated the gene expression of versican in DP cells cultured with Wnt-10b. Wnt/β-catenin signaling was also reported to play an important role in the generation of CSP expressing glial progenitors (NG2 cells) in the adult brain, which differentiate into astrocytes in association with the disappearance of CSP (42).
In our study, primary DP cells prepared from DP cultures showed ALP activity. However, after only 10 days in culture without Wnt-10b, they lacked ALP activity and nearly lost Wnt responsiveness and Lef-1 expression. On the other hand, DP cells cultured with Wnt-10b maintained their ALP activity, Wnt responsiveness, and Lef-1 expression, even after 10 passages. Thus, Wnt-10b is highly suspected to be effective for maintaining DP cells as primary DP cells in vitro. Indeed, the importance of Wnt/β-catenin signaling in hair follicle regeneration in vivo has been reported by many researchers (6,35–37). In the present study, Wnt-10b-treated DP cells continued to maintain HF induction and hair growth abilities, as shown by our skin reconstitution assay findings. Thus, we also consider that maintenance of the canonical Wnt signaling pathway may be indispensable for functional DP cells. The results of the present experiments are shown in schematic form in Figure 6.

Schematic diagram of effects of Wnt-10b on DP cells.
In conclusion, we investigated a new culture condition under which DP cells could maintain HF induction ability. We found that Wnt-10b promoted the proliferation of DP cells, and those cells continuously exposed to Wnt-10b maintained their Wnt responsiveness and HF induction ability even after 10 serial passages encompassing 100 days. We conclude that Wnt-10b plays a pivotal role in the proliferation and maintenance of functional DP cells in vitro.
Footnotes
Acknowledgments
We thank Dr. T. T. Sun (New York University School of Medicine, NY) for the AE13 and AE15 antibodies, Dr. J. Kishimoto (Shiseido Co., Ltd., Japan) for the transplantation chambers and explaining the grafting method technique, Dr. B. Vogelstein (Howard Hughes Medical Institute) for the pTOPFLASH and pFOPFLASH plasmids, and Mrs. S. Shimada for the technical assistance. This research was funded by a Grant-in-Aid for Young Scientists (B) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (KAKENHI) (to Y.O.). The authors declare no conflicts of interest.
