Abstract
Improved functional recovery after spinal cord injury by transplantation of induced pluripotent stem cell-derived neural stem/progenitor cells (iPSC-NPCs) has been reported. However, beneficial effects of iPSC-based therapy have so far been produced mostly using genetically immunodeficient rodents. Because of the long time required for generation and characterization of iPSCs, the use of autologous iPSCs for treating patients with acute spinal cord injury (SCI) is not feasible. Therefore, it is of utmost importance to investigate the effect of iPSC-based therapy on functional recovery after SCI using pharmacologically immunosuppressed, immunocompetent animal models. Here we studied the functional outcome following subacute transplantation of human iPSC-derived NPCs into contused mouse spinal cord when tacrolimus was used as an immunosuppressive agent. We show that human iPSC-derived NPCs transplanted into pharmacologically immunosuppressed C57BL/6J mice exhibited poor long-term survival and failed to improve functional recovery after SCI as measured by Basso Mouse Scale (BMS) for locomotion and CatWalk gait analysis when compared to vehicle-treated animals. The scarce effect of iPSC-based therapy observed in the current study may be attributable to insufficient immunosuppressive effect, provided by monotherapy with tacrolimus in combination with immunogenicity of transplanted cells and complex microenvironment of the injured spinal cord. Our results highlight the importance of extensive preclinical studies of transplanted cells before the clinical application of iPSC-based cell therapy is achieved.
Keywords
Introduction
Spinal cord injury (SCI) destroys neural and glial elements and interrupts ascending and descending axon pathways connecting the body with the brain, which results in sensory loss, paralysis, and loss of autonomic function (35,38,42). Unfortunately, despite considerable progress over the past decades in the medical, surgical, and rehabilitative care of SCI patients, treatment options to improve function after injury are very limited (22,30). Thus, there is an urgent need to develop novel therapeutic strategies for SCI.
Neural stem/progenitor cells (NPCs) offer a great promise as a cell source for regenerative medicine in human SCI patients (21,32,35,37,51). They can be obtained from fetal and adult brains/spinal cords, pluripotent embryonic stem cells (ESCs), or induced pluripotent stem cells (iPSCs) (9,30,32,40). The beneficial effects of the cells transplanted into an injured spinal cord are their possible ability to promote functional recovery by replacing lost endogenous neuronal and/or glial cells, remyelination of spared axons, increased production of beneficial neurotrophins/cytokines, providing trophic support and/or enhancing intrinsic regenerative ability (21,30,43,45).
A number of studies have shown the ability of transplanted NPCs to survive in the lesioned spinal cord and differentiate into neurons and glial cells, making them a major candidate for cell replacement therapy in SCI (18,27,36,44). Thus, differentiated along neural lineage, NPCs derived from mouse ESCs and rat fetal stem cells gave rise to neurons, astrocytes, and oligodendrocytes, formed functional synapses with the host neurons and promoted functional recovery when transplanted into an injured spinal cord in a rat SCI model (27,36). In a nonhuman primate SCI model, subacutely transplanted human fetal forebrain-derived NPCs similarly exhibited trilineage neural differentiation and improved functional recovery compared with a control group (18). However, the clinical applications of ESC-derived or fetal NPCs are limited by ethical (challenging and limited source of the cells) and immunological (allogeneic nature of these cells requires immunosuppression) reasons, both of which might be solved by using pluripotent cells derived directly from a patient's own somatic cells (8,20,21,26,32,51). Indeed, in contrast to allogeneic ESCs, autologous iPSCs require theoretically less immunosuppression. This is of particular importance to SCI patients, as they are prone to opportunistic infections (33), and an immunosuppressive therapy would compromise their resistance to infections and interfere with healing processes (35).
To date only a few studies have investigated the effect of iPSC-based therapy in SCI models (21). In a study using mouse iPSC-derived NPCs, the transplanted cells, when injected subacutely into the injured mouse spinal cord, provided improved remyelination, formed astrocytes, expressed neurotrophic factors, and promoted serotonergic innervations of the distal spinal cord leading to improved locomotor recovery (51). Also human iPSC-derived neurospheres and long-term self-renewing neuroepithelial-like cells, derived from monolayer cultures, when transplanted into the injured spinal cord of immunodeficient nonobese diabetic-severe combined immunodeficient (NOD-SCID) mice were shown to differentiate into trineural lineage, express neurotrophic factors, stimulate angiogenesis and axonal regrowth, form synapses with surviving endogenous neurons, and improve functional recovery (10,34). In contrast, undifferentiated human iPSCs transplanted subacutely into nude rats did not show any significant difference in morphological or functional outcomes by 5 weeks after SCI compared to human fibroblast controls (21). Recently, human iPSC-derived NPCs were subacutely transplanted in the spinal cords of common marmoset with a cervical SCI model, which resulted in enhanced axonal sparing/regrowth and angiogenesis, prevented demyelination, thereby promoting functional recovery after SCI (20).
It is worth noticing, however, that the studies of iPSC-derived NPCs in the SCI field have been performed mostly utilizing genetically immunodeficient mice so far. For the first time, we here transplanted NPCs derived from human iPSCs into spinal cords of immunocompetent C57BL/6J mice with contusion SCI and showed that transplanted cells scarcely survived up to 35 days posttransplantation despite continuous immunosuppressive therapy according to standard protocols and regimen sufficient for long-term donor cell survival in brains of uninjured mice, transgenic APdE9 mice modeling Alzheimer's disease, and mice subjected to permanent focal brain ischemia. The transplantations of iPSC- derived NPCs neither statistically significantly promoted nor worsened the functional recovery after contusion SCI. We conclude that a more detailed preclinical evaluation is required before iPSC-based clinical trials, especially those that require immunosuppressive therapy, can be initiated.
Materials and Methods
Cell Culture
A human iPSC line (UEFhfiPS1.4), derived from skin fibroblasts of a healthy female volunteer and characterized in our laboratory (41), was used in this study. Pluripotent stem cells were maintained on the top of the mitotically inactivated human foreskin fibroblasts (CRL-2429; ATCC, Manassas, VA, USA), and neural differentiation was carried out in a suspension of cell spheres as described in detail in Puttonen et al. (40).
Prior to the transplantation, the disappearance of pluripotency and appearance of neural features of the cells were confirmed using Nanog and/or octamer-binding transcription factor 4 (OCT4) as markers of pluripotency (6,15,28) and paired box protein 6 (PAX-6), nestin, and doublecortin (DCX) as markers of neural development (15,28) as previously described (40). Briefly, RNA samples were collected every 10–14 days (two parallel samples at each time point), and immunocytochemistry was done for spheres attached on polyornithine–laminincoated wells (Greiner Bio-One, Frickenhausen, Germany) every 2 weeks. To confirm the loss of pluripotency both at the RNA and protein levels, the expression of Nanog was detected with RT-PCR, whereas OCT4 protein was assessed by immunocytochemistry. The neural differentiation process was followed by the detection of PAX-6, DCX, and microtubule-associated protein 2 (MAP2; detects mature neurons).
In RT-PCR, the primers for Nanog (Hs02387400_g1), PAX-6 (Hs00242217_m1), DCX (Hs01035496_m1), and MAP2 (Hs00258900_m1) genes were employed and purchased from Life Technologies (Carlsbad, CA, USA). In immunocytochemistry, primary antibodies were anti-OCT4 (1:400; Millipore, Billerica, MA, USA), anti-DCX (1:200; Cell Signaling Technology Inc., Danvers, MA, USA), and anti-MAP2 (1:200; Millipore). Goat anti-mouse Alexa Fluor conjugated 488 and goat anti-rabbit Alexa Fluor conjugated 568 (both from Life Technologies) were used as secondary antibodies. Cell nuclei were stained with 5 μg/ml Hoechst (Life Technologies). Imaging was done with an Olympus IXDP40 digital camera attached to an Olympus IX70 inverted epifluorescence microscope (Tokyo, Japan).
Labeling of NPCs
Seven to 9 weeks after initiation of differentiation, small and homogenous spheres were collected and single cells harvested by incubating with TrypLE Express (Life Technologies). Isolated NPCs were seeded on polyornithine (50 μg/ml) human laminin (10 μg/ml)-coated wells in neural proliferation medium that contained a 1:1 mix of DMEM/F12 and neurobasal media supplemented with 1× retinoic acid free B27, 1× N2, 2 mM Glutamax, 50 IU/ ml penicillin, and 50 μg/ml streptomycin (all from Life Technologies) and 25 ng/ml of basic fibroblast growth factor (bFGF; R&D Systems, Minneapolis, MN, USA). Two days later, the cells were transduced with lentiviruses encoding green fluorescent protein (GFP) (Biocenter Kuopio, National Virus Vector Laboratory, Kuopio, Finland) at multiplicity of infection 5 for 18 h, after which the medium was changed. Just prior to the transplantation (on days 5–8 postplating) GFP-positive NPCs were collected and suspended into phosphate-buffered saline (PBS; Life Technologies) at a concentration of 100,000 cells/μl.
Animals
Adult female C57BL/6J mice bred in National Animal Center, University of Eastern Finland, were used throughout this study. The mice were at the age of 8–10 weeks weighing 18–23 g at the time of injury. All animals were housed in the groups of three in cages under a 12-h light/ dark cycle with drinking water and standard rodent chow provided ad libitum. For the first days after SCI, additional drinking water, softened rodent chow, and regular pellets were placed directly in the cage.
All experimental procedures were performed according to the national regulation of the usage and welfare of laboratory animals and approved by the Animal Experiment Committee in State Provincial Office of Southern Finland.
Spinal Cord Contusion Injury
The anesthesia was induced through a nose mask by using 5% isoflurane (Baxter, Deerfield, IL, USA) in 30% O2/70% N2O, and surgical depth of anesthesia was maintained with 1–1.5% isoflurane. Surgery was done as described previously (39). Briefly, after exposure of the 10th thoracic vertebra lamina, it was removed, and moderate contusion SCI (60 kdyn) was induced by Infinite Horizons Impactor (Precision Scientific Instrumentation, Lexington, KY, USA). Mice having impact parameters out of the 400-to 600-μm displacement interval were excluded from the experiment. For analgesia, buprenorphine (0.3 mg/ml; Temgesic®, Schering-Plough, UK) was administered subcutaneously. The first dose (0.1 mg/kg) was given 30 min before surgery followed by the same dose every 12 h for 3 days. As mice with SCI are unable to maintain normal body temperature (54), for the first 24 h after surgery injured animals were housed in cages placed on heating pads at 37°C. Bladders were manually evacuated until the restoration of their normal function. Mice subjected to laminectomy without actual injury served as sham controls.
Cell Transplantations
Seven days after SCI, the mice were anesthetized as described above, and the injury site was reexposed. Two small dura incisions were made 0.5 mm from midline at both the rostral (left to midline) and caudal (right to midline) ends of the laminectomy window (Fig. 1A), and a total of two injections were stereotaxically performed for each mouse using a 5-μl Hamilton syringe and an injecting minipump (Nanomite Injector Syringe Pump; Harvard Apparatus, Holliston, MA, USA). A 33-gauge needle was lowered 1.3 mm beneath the dorsal surface of the spinal cord, and randomly, 2 μl of PBS with or without NPCs (100,000/μl) was injected at the rate of 0.5 μl per min. The needle was left in situ for 7 min after injection before being slowly raised, and the wound was sutured.

Transplantation of UEFhfiPS1.4-NPCs into the spinal cord and hippocampus. (A) A schematic drawing showing the injection sites for the cell transplantation. (B) Immunosuppression with tacrolimus is sufficient to provide survival of human induced pluripotent stem cell line-derived neural progenitor cells (UEFhfiPS1.4-NPCs) after transplantation into the brain of uninjured C57BL/6J mice. The images show excellent survival of UEFhfiPS1.4-NPCs grafts in the hippocampus 35 days after the transplantation as demonstrated by nuclear staining with 4′,6-diamidino-2-phenylindole (DAPI) and human nuclei (HuNu) immunostaining. n = 6. Scale bars: 200 μm. Scale bar in the high-power inset: 20 μm.
For immunosuppression, all mice, including nongrafted ones, received daily intraperitoneal injections of tacrolimus (2 mg/kg, PrografR; Astellas Pharma a/s, Glostrup, Denmark) until the end of the study. The dosing was selected as tacrolimus is typically administered intraperitoneally or subcutaneously with doses ranging from 0.05 to 3 mg/kg/24 h in cell transplantation studies that involve rodents or minipigs (5,25,47,52). The efficacy of the tacrolimus immunosuppression in a dose of 2–3 mg/kg was confirmed in our experiments where NPCs derived from human iPSCs and human ESCs were transplanted into the brains of uninjured mice (Fig. 1B), transgenic APdE9 mice, and mice subjected to permanent focal brain ischemia (our unpublished data). In all of these cases, a high number of transplanted cells were consistently observed in the brains at the site of transplantation (Fig. 1B) and adjacent to the ischemic lesion 5–12 weeks after grafting, indicating that daily intraperitoneal injections of tacrolimus at the dose used in the current study is sufficient to provide immunosuppression in the CNS under the basal and pathologic conditions.
Behavioral Testing
Motor function of the hindlimbs was assessed 24 h after injury and then weekly for up to 6 weeks after injury. Two independent raters who were blinded to the experimental conditions evaluated functional recovery using the Basso Mouse Scale (BMS) (4). Each mouse was observed separately for 4 min in each session, and hindlimb movements were assessed with the scale ranging from 0 (no ankle movement) to 9 (complete functional recovery) points. Averages of BMS scores for left and right hindlimbs were taken to obtain a single value per mouse. Mice with the BMS score higher than 1 at 24 h after injury were excluded from the future evaluation.
CatWalk-automated gait analysis system (Noldus, Wageningen, The Netherlands) was used to evaluate the precise hindlimb movements 5 weeks (35 days) after cell transplantation as described elsewhere (13). Prior to the testing day, the animals were trained during 3 consecutive days to run without interruption along a runway toward the end zone. At the testing day, the mouse was placed in front of the start zone of the CatWalk runway, and footprints of the animal crossing a glass walkway were recorded by the video camera positioned below. For data collection, three uninterrupted runs per animal were performed. The number of gait parameters such as base of support, intensity of the paws, print length, width and area, stand and swing speed and duration, max area and contact, stride length, and regularity index (RI) were examined with the CatWalk 7.1 software (Noldus). The values for every paw were taken, and averages between left and right hind paws were used for analysis. Only animals that showed at least occasional plantar stepping in the open field (BMS score ≥ 4) were tested on CatWalk as the system requires that mice are able to step along the walkway for the analysis.
Magnetic Resonance Imaging
Two weeks after cell transplantation, the mice were anesthetized as described above and imaged using a 9.4 Tesla Varian scanner (Varian Inc., Palo Alto, CA, USA) as we described previously (34). Imaging data were analyzed using in-house-built Matlab software (Aedes, Kuopio, Finland) and converted to micrometers (μm) by an observer who was blinded to the experimental conditions.
Immunohistochemistry
Animals were terminally anesthesized with tribromoethanol (Avertin; Sigma-Aldrich, St. Louis, MO, USA) and transcardially perfused with heparinized (2,500 IU/L) (Leo Pharma A/S, Ballerup, Denmark) saline (Baxter), followed by 4% paraformaldehyde (PFA) (Sigma-Aldrich). The spinal cords were dissected and postfixed in the same fixative overnight at 4°C. After fixation, tissues were cryoprotected in 10% sucrose (VWR International, Leuven, Belgium) overnight, followed by 30% sucrose overnight. A 6-mm length of the spinal cord containing the region of transplantation was dissected, embedded in Tissue-Tek O.C.T. Compound (Sakura Finetek, Zouterwoude, the Netherlands), frozen on dry ice, and stored at −70°C. Transversal 20-μm serial sections were cut on a cryostat (Leica Microsystems GmbH, Wetzlar Germany).
Frozen sections were processed for immunofluorescence staining with primary antibodies against glial fibrillary acidic protein (GFAP; 1:200; Dako, Glostrup, Denmark), ionized calcium-binding adapter molecule 1 (Iba-1; 1:250; Wako Pure Chemical Industries, Ltd., Tokyo, Japan), DCX (1:200; Cell Signaling Technology, Inc., Danvers, MA, USA), Ki-67 (1:500; Abcam, Cambridge, UK), class III β-tubulin (Tuj1; 1:300; Abcam), and Nanog (1:200; Abcam). All of these primary antibodies were made in rabbit. Primary antibody against GFP (1:200; Life Technologies) and human nuclei (HuNu; 1:250; Millipore) were made in mouse. For HuNu and GFP staining, mouse Ig blocking reagent (M.O.M.”; Vector Laboratories, Inc., Burlingame, CA, USA) was used to block endogenous mouse antibody in the tissue sections. On the following day, secondary antibody (goat antirabbit or goat anti-mouse Alexa Fluor conjugated 568, 1:200; both from Life Technologies) was applied. After air drying, the sections were mounted with Vectashield mounting medium with DAPI (Vector Laboratories).
Spinal cord sections were photographed using a digital camera (Color View 12 or F-view; Soft Imaging Systems, Münster, Germany) attached to an Olympus AX70 microscope. To assess colocalization, sections stained for DCX and Ki-67 were imaged using a Zeiss LSM 700 confocal microscope (Zeiss Inc., Maple Grove, MN, USA) with an attached digital camera (Color View 12 or F-View; Soft Imaging System) running Zen 2009 Image analysis Software (Zeiss Inc.).
Statistical Analyses
All data are shown as mean ± standard deviation (SD). An unpaired t-test was employed for comparing means of two treatment groups. For multiple comparisons, one-way ANOVA followed by Tukey's post hoc test was used. BMS data were analyzed using nonparametric Mann–Whitney test (for two group comparison) and Kruskal–Wallis test followed by Dunn's post hoc test (for multiply comparisons). Values of p < 0.05 were considered to be significant. All statistical analyses were performed in GraphPad Prism version 5.03 for Windows software (GraphPad Software, La Jolla, CA, USA).
Results
Characterization of UEFhfiPS1.4-NPCs In Vitro
To ensure that the donor cells lost their pluripotency prior to transplantations and were at the stage that would allow them to migrate and possibly integrate into the host tissue, we differentiated the cells for at least 6 weeks and, during this period, followed up the development of neural properties and loss of pluripotency at RNA and protein levels. Our characterization data show (Fig. 2) that most of the UEFhfiPS1.4-NPCs were positive for neuronal markers DCX and MAP2 (Fig. 2A, B). In addition, the RNA expression of DCX, MAP2, and PAX-6 increased during the time (Fig. 2C–E). Meanwhile, the disappearance of pluripotency was detected at the RNA level of Nanog (Fig. 2F) and by silencing of OCT4 in immunocytochemistry (data not shown).

UEFhfiPS1.4-NPCs lose their pluripotency and acquire neural properties prior to transplantation. (A) Doublecortin (DCX)-positive migrating progenitors already had morphology resembling that of neurons, that is, small soma (in blue: nuclei) and long projections (in green). (B) Also microtubule-associated protein 2 (MAP2; in green) immunopositivity for mature neurons confirmed that the neural differentiation was successful. NPCs located inside and in the vicinity of spheres seemed to coexpress (in yellow) DCX (in red) and MAP2 (in green). (A, B) The selected spheres were plated down on differentiation day 34, fixed 2 days later, and immunostained. At the RNA level, neuronal markers (C, DCX and D, MAP2) and early neuroectodermal [E, paired box protein 6 (PAX-6)] were upregulated, while the pluripotent gene (F, Nanog) was silenced, during the in vitro differentiation period prior transplantations. dd, differentiation day. The data are shown as mean ± SD.
Characterization of UEFhfiPS1.4-NPCs In Vivo
To estimate the efficacy of intraspinal transplantation of UEFhfiPS1.4-NPCs and characterize the grafts in vivo, we transplanted these cells into spinal cords of uninjured mice at the level of the 10th thoracic vertebra immediately after laminectomy. The mice were sacrificed 7 days after cell transplantation, and efficacy of cell grafting was assessed by GFP fluorescence. Transplanted GFP-positive UEFhfiPS1.4-NPCs were detected in both ventral horns and adjacent white matter (Fig. 3A) within the whole studied segment of the spinal cord.
To further characterize grafted UEFhfiPS1.4-NPCs, we labeled spinal cord sections with DCX, a microtubule-associated protein expressed by neuronal precursor cells and immature neurons, and Ki-67, a marker of cell proliferation. Many of the grafted GFP-positive cells were also positive for DCX and Ki-67 (Fig. 3B, C) indicating that at 7 days after grafting most of the UEFhfiPS1.4-NPCs were still actively proliferating immature neuronal cells. In order to determine whether undifferentiated pluripotent cells were still present in the grafts and, therefore, could give rise to teratoma formation, we applied Nanog labeling. We did not observe any Nanog-positive cells in the grafts (Fig. 3D), which is in line with the in vitro characterization data. Next, we labeled spinal cord sections with Tuj1 and GFAP to clarify whether UEFhfiPS1.4-NPCs were able to differentiate already 7 days after grafting into neurons and astrocytes, respectively. We did not observe NPCs expressing either GFAP or class III β-tubulin in the grafts (data not shown). Finally, Iba-1 labeling revealed accumulation of activated microglia around the grafts (Fig. 3E).
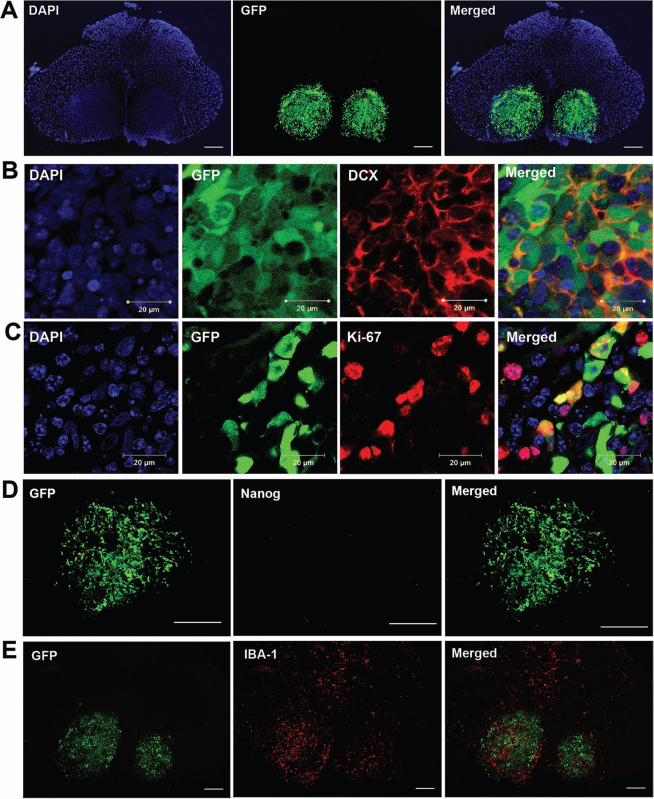
UEFhfiPS1.4-NPCs are proliferating immature neuronal cells without signs of pluripotency after transplantation into intact spinal cord. (A) UEFhfiPS1.4-NPCs labeled with green fluorescent protein (GFP) are found in spinal cord 7 days after intraspinal transplantation to laminectomized mice. (B, C) Confocal images showing colocalization of GFP and DCX (B) and GFP and Ki-67 (C), indicating neuronal lineage commitment and high mitotic activity of transplanted cells. Scale bars: 20 μm. (D) Transplanted cells do not express Nanog, a marker of pluripotency. (E) Accumulation of ionized calcium-binding adapter molecule 1 (Iba-1)-positive microglia/macrophages are found around the grafts. n = 3. Scale bars: 200 μm.
To assess the safety of intraspinal transplantation of UEFhfiPS1.4-NPCs, we performed laminectomy at the level of the 10th thoracic vertebra of mouse spinal cords. Seven days later, the laminectomy site was reopened, and the mice received intraspinal injections of UEFhfiPS1.4-NPCs or vehicle. The tumor formation after human cell transplantation into the spinal cord results in rapid decline in BMS score within 10 days after injection, and finally the BMS score reached zero levels (53). Therefore, we next compared BMS scores of the transplanted and vehicle-treated animal groups to the group of laminectomized mice without injection weekly for the next 5 weeks. Neither intraspinal delivery of vehicle alone nor UEFhfiPS1.4-NPCs resulted in behavioral impairment within the whole observation period. Thus, the BMS scores in all studied groups were between 8 and 9 indicating only instances of mild trunk instability in some of the operated mice (Fig. 4). Starting from day 28 after transplantation, all animals attained the maximal BMS score of 9, indicating the full functional competence in all studied groups. In addition, during postmortem dissection, no tumor formation was observed in the spinal cord at the end of observation period.

UEFhfiPS1.4-NPCs do not impair functional competence when transplanted into intact spinal cord. Hindlimb motor function was assessed by Basso Mouse Scale (BMS) at 1, 7, 14, 21, 35, and 42 days after laminectomy. The data are shown as mean ± SD. n = 3–6, p > 0.05 between the study groups.
Transplanted UEFhfiPS1.4-NPCs Did Not Affect Lesion Size After Contusion SCI
To assess whether the donor cells alter lesion size, we performed magnetic resonance imaging (MRI) at 14 days after cell transplantation. However, transplantation of UEFhfiPS1.4-NPCs did not affect the lesion volume compared to vehicle-treated controls (p > 0.05), which was consistent with BMS scores at this time point (Fig. 5).

Transplantation of UEFhfiPS1.4-NPCs does not affect lesion volume measured by MRI. Lesion volume was measured by magnetic resonance imaging (MRI) at 42 days after spinal cord injury (SCI). The data are shown as mean ± SD. n = 11. p > 0.05 between the study groups. the study groups. At the end point, the vehicle-treated mice attained BMS scores of 5.09 ± 1.11 (Fig. 6), which on average corresponds to frequent or consistent plantar stepping without coordination or with some coordination but with paw rotation at initial contact and lift off. Mice transplanted with UEFhfiPS1.4-NPCs attained a BMS score of 4.18 ± 0.84 (Fig. 6), which on average corresponds to occasional plantar stepping. Of note, the BMS scores in mice transplanted with UEFhfiPS1.4-NPCs were constantly lower than in the vehicle group, even though statistical significance was not achieved at any time point (p > 0.05). Importantly, at the time of dissection, no signs of tumorigenesis were detected in the spinal cords in the group transplanted with UEFhfiPS1.4-NPCs.
Transplanted UEFhfiPS1.4-NPCs Did Not Promote Behavioral Recovery After Contusion SCI
To further estimate the therapeutic potential of UEFhfiPS1.4-NPCs in a contusion SCI model, we assessed the effects of these cells on functional recovery when compared to vehicle control. Sixty-kilodyne contusion injury was applied at the level of the 10th thoracic vertebra of mouse spinal cords. Twenty-four hours after contusion SCI, most of the mice exhibited complete paralysis of hindlimbs (BMS score 0) (Fig. 6). Seven days later, we injected vehicle or UEFhfiPS1.4-NPCs into the spinal cord caudally and rostrally to the injury epicenter. At the day of the cell transplantation, the mice injected with vehicle attained an average BMS score of 2.09 ± 0.94, and the mice transplanted with UEFhfiPS1.4-NPCs had a score of 2.00 ± 0.87 (p > 0.05 between these two groups), which corresponds to extensive ankle movements (Fig. 6). The equal BMS score observed in both studied groups confirms the injury homogeneity immediately prior to cell transplantation. Within the next 35 days after transplantation, we observed slow recovery of hindlimb function without significant difference in BMS scores between Thirty-five days after cell transplantation, we also took advantage of CatWalk to evaluate precise hindlimb movements. In the CatWalk system, the RI reflects the degree of interlimb coordination during the gait cycle (13). The RI value for uninjured mice was close to 100%, which was significantly higher compared to both injured groups (Fig. 7). RI of the mice transplanted with UEFhfiPS1.4-NPCs (65.7%) was not significantly different from RI of the vehicle-treated mice (78.8%).

Transplantation of UEFhfiPS1.4-NPCs does not promote functional recovery after contusion SCI. Hindlimb motor function was assessed by BMS at 1, 7, 14, 21, 35, and 42 days after SCI. The data are shown as mean ± SD. n = 11, p > 0.05 between the study groups.
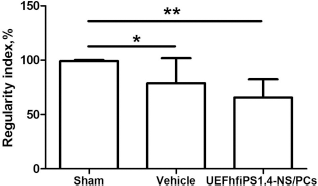
Transplantation of UEFhfiPS1.4-NPCs does not improve interlimb coordination after contusion SCI. Interlimb coordinations were measured by CatWalk at 42 days after SCI. The data are shown as mean ± SD. n = 6–11. *p < 0.05, **p < 0.01.
Similar to RI, a) hind paw intensity, an indication of paw pressure applied at the moment of floor contact and therefore a measure of weight support, b) max contact, the time at which the largest part of a paw contacts the ground, c) swing speed, reflecting the velocity of the moving limb during the swing phase, and d) stride length, the distance between successive placements of the same paw, were all significantly higher in the uninjured group when compared to injured groups (Fig. 8). However, we did not observe any difference in these gait parameters between groups treated with vehicle or transplanted with UEFhfiPS1.4-NPCs. No significant differences were detected in other CatWalk gait parameters (e.g., base of support, print area, width and length, stand and swing duration) between any of the study groups (data not shown).

Transplantation of UEFhfiPS1.4-NPCs does not restore gait parameters after contusion SCI. Hind paws (A) intensity, (B) max contact, (C) swing speed, and (D) stride length were measured by CatWalk at 42 days after SCI. The data are shown as mean ± SD. n = 6–11. *p < 0.05, **p < 0.01, ***p < 0.01.
Transplanted UEFhfiPS1.4-NPCs Exhibited Limited Long-Term Survival After Contusion SCI
Two studies where human iPSC-derived progenitors were transplanted into the injured spinal cord of immunodeficient mice report a survival rate of 18–20%, suggesting that this amount of surviving donor cells is sufficient to promote functional recovery (10,51). However, the degree of donor cell survival after the transplantation of human iPSC-derived progenitors into immunocompetent mice with SCI has not yet been studied. Therefore, in order to assess the donor cell survival 42 days after transplantation into the spinal cord of contused mice immunosuppressed with tacrolimus, we evaluated GFP expression within a 5.6-cm-long spinal cord segment in six mice transplanted with UEFhfiPS1.4-NPCs. Very few cells positive for GFP were observed in only one out of six spinal cords (Fig. 9), whereas the other five examined spinal cord samples were devoid of surviving donor cells, suggesting very limited survival of cell grafts in transplanted immunocompetent mice immunosuppressed with tacrolimus. These findings were further confirmed by anti-GFP and human-specific anti-HuNu immunostaining (data not shown).

UEFhfiPS1.4-NPCs exhibit very limited survival after the transplantation into the spinal cord of immunocompetent mice with SCI when immunosuppression with tacrolimus was used. Only one out of six SCI mice transplanted with NPCs showed any surviving donor cells. Scale bars: 20 μm.
Discussion
The beginning of the 21st century was marked by the impressive number of experimental cell transplantation studies aiming at improving the treatment of SCI (7). The doubtless therapeutic potential of fetal and embryonic-derived NPCs (18,27,36) as well as oligodendrocyte progenitor cells (19,48) led to ongoing clinical studies involving fetal brain and spinal cord-derived neural stem cells (clinical trial identifiers: NCT01321333 and NCT01772810). Despite the great promise, the wide clinical use of these cells is limited by their restricted availability, considerable ethical and practical challenges, and concerns imposed by their origins and derivation (9).
In contrast, autologous iPSC-derived NPCs, generated from the somatic cells of an adult patient, avoid ethical concerns and theoretically can be used for autologous transplantations with a reduced need for immunosuppression (35). Unfortunately, the time required to generate appropriately characterized and safe autologous iPSC lines in sufficient quantities make personalized iPSC-based treatment impractical, at least for acute and subacute SCI (8,9). Instead, generation and testing clinical-grade set of well-characterized allogeneic iPSC lines intended for subsequent administration into SCI patients is likely more feasible (8,9,21). In this situation, immunogenicity of iPSCs is a major obstacle that needs to be addressed (8,9). Therefore, the demand of immunosuppression is obvious, and most likely the first clinical trial with iPSCs in SCI would still require immunosuppressive therapy.
Recently, mouse and human iPSC-derived NPCs have been shown to provide some benefits in experimental SCI (10,20,34,51). Importantly, most of these studies utilized NOD-SCID mice and did not include the immunosuppressants in the treatment paradigm (10,34,51). NOD-SCID mice have compromised innate and adaptive immunity, as they lack complement hemolytic activity and both mature B- and T-lymphocytes (11,49). Although this strain is considered to be an ideal model to test the potential of transplanted cells, the recent study shows that these mice are different from C57BL/6J strains at least in terms of cellular inflammation and functional recovery (24). Importantly, an immune response to transplanted cells may not only result in graft rejection, but may also alter the cell fate (50). In line with this assumption, interleukin-6 has been shown to enhance glial fate in ESC-derived NPCs (17). Therefore, while the autologous iPSC transplantations for acute and subacute SCI are still questionable, there is an urgent need for studies not only of immunodeficient but especially of pharmacologically immunosuppressed animals.
The aim of the present study was to analyze the degree of therapeutic effect of human iPSC-derived NPCs once grafted into the spinal cord of immunosuppressed C57BL/6J mice with a subacute contusion SCI. To our knowledge, this is the first study to examine the functional outcomes of human iPSC-derived NPCs transplanted into immunocompetent C57BL/6J mice with SCI, involving immunosuppressive treatment. For transplantations, we used neural progenitors derived from the UEFhfiPS1.4 cell line, which was generated and characterized in our laboratory (41). Our data show that, prior to grafting, the transplanted cells had lost their pluripotency as indicated by the expression of the Nanog gene and the fact that the OCT4 protein was silenced. Instead, these cells expressed the early neuroectodermal marker PAX-6 and neuronal markers DCX and MAP2, suggesting predominant neuronal differentiation. When the UEFhfiPS1.4-NPCs were transplanted into the spinal cords of laminectomized mice, most of them expressed DCX and Ki-67 7 days after grafting, indicating that transplanted cells have the potential to differentiate into neuronal lineage in vivo. Importantly, transplanted cells did not impair the functional outcome of laminectomized mice and did not form tumors in the spinal cord during the whole observation period. However, transplantation of UEFhfiPS1.4-NPCs into the injured mice, despite a similar immunosuppressive regimen as in laminectomized mice, resulted in very limited donor cell survival in the recipient spinal cords, failed to improve functional recovery as measured by BMS and CatWalk, and, finally, did not reduce the lesion size as measured by MRI when compared to the vehicle-treated mice. Of note, BMS data are ordinal, and the magnitude of behavioral change between ranks may not be consistent (4). While interpreting BMS results, it is important to consider the biological significance in addition to statistical significance (4,16). Given this, the sustained impairment in functional recovery in the group transplanted with UEFhfiPS1.4-NPCs, which appeared already 1 week after grafting, persisted during the whole observation period, and resulted in occasional plantar stepping at the end of the observation period, whereas vehicle-treated mice were able to perform frequent or constant stepping. Altogether, these results suggest that transplanted cells may even have worsened the behavioral outcome in our study setup.
It is now becoming increasingly clear that ESCs are capable of inducing a strong response of both T cells and NK cells (21). The immunogenicity of iPSCs is studied to a much lesser extent and still remains controversial (3,12,29,56). In the present study, we observed accumulation of Iba-1-positive cells around the grafts in noninjured mice already 7 days after transplantation, indicating the triggering of an immune response toward the transplanted allogeneic cells.
Achievement of effective, safe, and long-term immunosuppression in experimental allogeneic cell transplantation studies is challenging as in the majority of such studies immunosuppression has been shown to be insufficient (1,24). Recently, Kobayashi et al. (20) reported a beneficial effect of human iPSC-NPCs transplanted into a contused common marmoset spinal cord when cyclosporine was used as an immunosuppressive drug. Although cyclosporine and tacrolimus share the common mechanism of action, there is substantial evidence that tacrolimus is more effective than cyclosporine in preventing graft rejection (46) and has a lower nephrotoxicity potential (31). Therefore, in the present study for immunosuppressive therapy, we used the calcineurin inhibitor, tacrolimus, in a dosage of 2 mg/kg/ 24 h, which is within the interval of 0.05–3 mg/kg/24 h typically used in allogeneic cell transplantation studies that use rodents (mice, rats) or minipigs as recipients (5,25,47,52). However, none of these studies included contusion SCI. It is also worth noticing that although immunosuppression was associated with increased efficacy in a focal cerebral ischemia (23), in SCI, in contrast, immunosuppressants were associated with reduced efficacy (2). In our experiments, we found that either hiPSC- or hESC-derived NPCs transplanted into the brains of uninjured mice, transgenic APdE9 mice, and mice subjected to permanent focal brain ischemia immunosuppressed with tacrolimus (2–3 mg/kg) were able to survive up to 5–12 weeks after transplantation. This indicates that the beneficial effect of immunosuppressants in promoting transplanted cell survival observed in intact animals or other models is canceled out in SCI by the complex and diverse microenvironment of the injured spinal cord. Interestingly, while tacrolimus has been successfully used as a monotherapy for spinal transplantation of human cells in a spinal ischemic paraplegia model (5,25), it similarly with our results failed in the studies involving G93A SOD1 animal models of ALS (14,55). Increased efficiency of combined immunosuppression (tacrolimus + rapamycin, tacrolimus + mycophenolate, or tacrolimus + anti-CD4 antibody) in comparison with tacrolimus monotherapy has been reported (14,50,55). However, to our knowledge, our study is the first one where monotherapy with tacrolimus was used for human cell transplantation in an acute SCI model. Whether combined immunosuppression would be more efficient also in acute SCI conditions may be a subject of follow-up studies.
Overall, this study demonstrates that human iPSC-derived NPCs transplanted into an injured mouse spinal cord environment do not provide functional recovery when immunosuppressive monotherapy with tacrolimus is used, warranting additional studies involving combined immunosuppression or/and reduction of immunogenicity of transplanted cells.
Footnotes
Acknowledgments
This work was supported by Academy of Finland, Sigrid Juselius Foundation, Finnish Cultural Foundation, and Tekes funding agency. We thank Dr. Piia Valonen for technical assistance with MRI data analysis, Mrs. Mirka Tikkanen for technical assistance with immunohistochemical staining, and Ms. Riitta Kauppinen for her help in preparation and maintenance of cell cultures. We also thank Dr. Erik Sundström for helpful comments on the manuscript. The authors declare no conflicts of interest.
