Abstract
This study was designed to observe the functional changes after fetal olfactory ensheathing glia transplantation (OEGT) into the spinal cord of patients with chronic spinal cord injury (SCI). Patients whose recovery had plateaued for longer than 6 months were enrolled. Six thoracic patients were tested for safety and five cervical patients for efficacy. OEGT was performed according to the method developed by Huang. Average follow-up was 14 months (range 1.0–1.5 years). Sensation improved moderately (light touch 14.2, pin prick 13.6); as did spasticity (1–2 modified Ashworth scale down). Locomotion recovery was minimal (1.6). Useful reticular formation functions were observed, but due to a lack of appropriate outcome measure, they were not recorded and reported.
Introduction
Spinal cord injury (SCI) is a devastating and debilitating lesion. For complete SCI, so far there is no method of cure or restoring lost functions substantially. Since the success of olfactory ensheathing glia transplantation (OEGT) in animal experiments (10,11,13,15, 16), recent attempts have been made to use the method to improve neurological outcome in humans. Only eight clinical or clinically related reports have been published on the procedure (1–3,6,7,9,12,14). No fatality has been reported despite criticism against some reports (3).
One study by Lima et al. reports minor deterioration of the neurological outcome (12) but the overwhelming majority of reports claim the method is safe. Hence, the result of the Phase 1 clinical trial to prove safety could be considered positive, unless new evidence proves otherwise. Now, from a clinical perspective, the scientific communities focus their attention on Phase 2 clinical trials. None of the above-mentioned reports have been universally accepted as a valid and complete Phase 2 clinical trial. After 10 years of trial and clinical application, there is no global consensus about the standards and complete outcome measures. The journal Spinal Cord has published a series of guidelines for international consumption [e.g., (17)]. Even these comprehensive guidelines compiled by world-class experts are yet to be tested for its feasibility, practicality, completeness, and reliability in the real world. The troubling missing piece of the jigsaw in these guidelines is the outcome measures for the functions of reticular formation (RF). According to an observer's account, the improvement of functions after OEGT mainly involves the RF (18). Therefore, in almost all cases of clinical use of OEGT, the functions of RF cannot be tested or reported even if important and useful improvements have been observed. It is worth noting that for life and health RF functions are more important than somatic functions. Until important and reliable outcome measures of these functions are in place, no clinical trial could be considered complete. Obviously, this simple study is not designed to resolve these long-lasting extremely complex and difficult issues that may take years, if not decades, to complete. However, it adds more clinical experience in OEGT that may facilitate a global standard for clinical trial in the future.
This is not a randomized double-blind control clinical trial, but is instead a preliminary self-control Phase 1/2 trial.
Materials and Methods
Culturing of Olfactory Ensheathing Glia (OEG)
The isolation of OEG from the olfactory bulb nerve layer has been described previously (8). Briefly, the olfactory bulbs from 16-week-old or older aborted human fetuses following strict ethical guidelines were harvested (17). The olfactory bulbs were dissected and rapidly placed into a petri dish with ice-cold Hanks balanced salt solution (HBSS). The bulbs were rinsed twice with ice-cold HBSS and then minced with a sterile scalpel blade. The minced bulb tissue was then suspended in 5 ml of HBSS with 1 mg/ml trypsin and incubated at 37°C for 15 min with continual shaking. DMEM (Sigma) and Ham's F-12 (DMEM/F-12; 1:1 mixture; Sigma) supplemented with 10% fetal bovine serum (FBS; DF-10S; Invitrogen) was added to stop the trypsinization and the specimen centrifuged briefly. The supernatant was aspirated and the cells were rinsed with multiple washes of DF-10S. DF-10S culture medium was supplemented with L-glutamine (20 mM). Penicillin/streptomycin (10,000 μg/ml) was added and the cells passed through a 70-μm mesh filter. The cell suspension was cultured in DF-10S for 3–4 days at 37°C with 5% CO2. Then OEG were purified from contaminated cells by using serum-free medium (SFM) instead of DF-10S for an additional 2–3 weeks with culture medium changed every 2 days. The SFM consists of DMEM, biotin, insulin, transferrin, sodium selenate, hydrocortisone, and glutamine. Consequently, the cells were detached with trypsin-EDTA, resuspended in DMEM at a density of 1 × 107/ml, as confirmed by immunostaining with anti-glial fibrillary acidic protein (GFAP) and anti-S100 antibodies, and prepared for clinical use.
Patient Selection
All patients had their neurological conditions stabilized for longer than 6 months (6–42 months). The outcome of neurological evaluation at the end point of the plateau was used as control for comparing with the results after OEGT.
Two stages were planned. In the first stage we tested the safety of our method of treatment although no major adverse effects or fatality have been reported elsewhere. For this purpose, six patients with complete injury of mid to lower thoracic level were enrolled. The reason for choosing this level for safety test was obvious as minor deterioration would not affect vital functions or important locomotion. However, this level is definitely not ideal for Phase 2 clinical trial, where testing efficacy is the main purpose, particularly when the changes are minor. Hence, five other cervical patients were enrolled for this purpose, assuming that the cervical cord would be less vulnerable than the thoracic one as it is much larger in size and hence a larger safety margin. Regardless of the level of injury, cervical or thoracic, all patients had complete injury according to Frankel scale so that minor damage would not cause further clinical adverse effects on the density of the lesion whereas any recovery would be more likely associated with the procedure. Altogether there were nine men and two women enrolled consecutively from January to December in 2004. Their ages ranged from 25 to 55 years. The causes of SCI consisted of traffic accidents, falls, and being hit by objects. The interval between accident and transplantation was 2.0–5.5 years. Apart from the level and density of injury, inclusion criteria were: no compression, tethering, or disruption of spinal cord. Exclusion criteria were age over 60 years or under 18 years or complications that made surgery potentially risky.
The Ethics Committee of Beijing Army General Hospital approved the clinical trial protocols in accordance with the guidelines of the Ministry of Health of the People's Republic of China. It is legal in China to use a 16-week-old fetus, aborted for other legitimate reasons, but not if intended specifically for OEGT. All the subjects gave their written informed consent to participate in this clinical trial.
Surgery
Following general anesthesia, the patients were placed prone on a spinal frame. A midline incision was made and the spinal cord was exposed with the standard laminectomy. The laminectomy encompassed the entire lesion. A suspension (0.05 ml) of cultured OEG (approximately 5 × 105 cells) was injected into the spinal cord adjacent to rostral and caudal ends of the lesions using a tuberculin syringe. Monitoring of motor and somatosensory evoked potentials provided instantaneous intraoperative assessment of the patients' electrophysiological responses. Hemostatics and prophylactic antibiotics were administered for 3 days after the procedure.
Clinical Assessment
We performed a precise neurological functional evaluation on the patients 1 week before surgery, 2 weeks postoperatively, and at final follow-up. Average follow-up was 14 months (range 12–18 months). The spinal cord function assessment, including motor score, light touch score, and pin prick score, was conducted according to the International Standards for Neurological classification of spinal cord injury (ASIA/ISCoS).
The patients' spasticity was evaluated according to a modified Ashworth scale (MAS).
MRI Evaluation
MRI for spinal cord in all patients was assessed before transplantation and 12 months after the procedure.
Statistical Analysis
The data were expressed as means. The SPSS for Windows, paired samples t-test was performed for the comparisons of the sensory and motor scores before and after transplantation. A value of p < 0.05 was considered statistically significant.
Results
All patients had no complication or deterioration of neurological conditions. Three cases with low cervical cord injury had improvement in motor components of ASIA scoring, presenting improvement of key muscle strength below the level of injury. The mean increase in five patients with cervical injury was 1.6. Sensation (light touch and pin prick scores) improved significantly in all patients by 3–10 segments. In five patients with cervical injury, the mean increase was 14.2 for light touch and 13.6 for pinprick. For five patients with thoracic injury, the mean increase was 14.3 for light touch and 15.0 for pinprick. Seven patients (three cervical and four thoracic) began to feel pain below injury level within 3 months postoperatively. The pain relieved gradually and finally disappeared. Spasticity in all patients was to a certain extent relieved. ASIA scores and MAS of 11 patients are presented in Tables 1 and 2 subject to their injury at cervical or thoracic level.
Characteristics of Patients With Cervical Injury
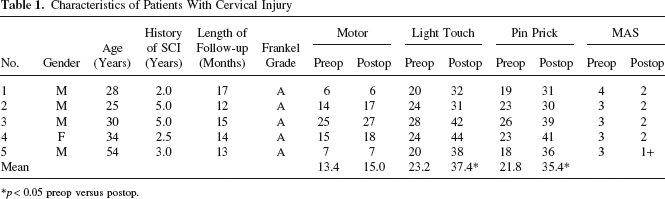
p < 0.05 preop versus postop.
Characteristics of Patients With Thoracic Injury

p < 0.05 preop versus postop.
The follow-up MRI scans revealed no mass, new hemorrhage, newly developed edema, intra- or extramedullary cyst at the transplantation site and surrounding areas (Fig. 1).

Preoperative (A) and postoperative (B) MRI showed no mass, new hemorrhage, newly developed edema, intra- or extramedullary cyst at the transplantation site and surrounding areas.
Discussion
OEGT for SCI Repair
OEG has been proven to stimulate axonal regeneration and functional recovery of injured spinal cord in animal models (10,11,13,15,16). These positive results have led to the idea of using it to repair spinal cord injury in humans.
Adult OEG for autograft transplantation can be obtained from either the olfactory bulb or the nasal mucosa of the patients. Recent efforts have been focused on auto nasal olfactory mucosa for ethical reasons. However, OEG from this source is difficult to culture, whereas fetal OEG is robust and proliferates rapidly. Moreover, removal of the olfactory bulb is an invasive procedure. If abortion just happens anyway in local culture and is not intended for OEGT, it may not be less justifiable on ethical ground than autograft. Harvesting OEG from the olfactory bulb provides an aseptic source of the cells, thus minimizing the risk of infection.
Our study further confirmed that fetal OEGT was a safe treatment for patients with SCI. The procedure is minimally invasive and free of complications. Adverse effects, such as glial proliferation or tumor, and damage or disruption of neural structures did not develop confirmed by MRI during initial follow-up, although longer follow-up is needed.
At final follow-up, sensation and spasticity had made moderate improvement, whereas improvement of locomotion was hardly palpable. The clinical order of recovery was spasticity, sensation, and locomotion. This follows the phylogenic order of evolution from ancient reticular formation to recent spinothalamic and palliospinal connections. Unfortunately, motor functions recovery that most patients wish to achieve first after cell therapy is rare and thin. All these suggest that full success of spinal cord repair with OEGT or other cells remains distant. More intensive and extensive researches in the light of evolution are needed to elucidate the phylogenic barrier of human CNS regrowth (4).
Timing of Treatment
The timing of cell therapy is of some controversy. Most prefer to use therapy after the natural recovery has ceased because after this time any recovery must be attributed to the therapy. Statistically speaking, there is no natural recovery observed after 28 months postinjury (5). Suggestion was made to delay the therapy at least later than 18 months postinjury (5). However, statistics is for group analysis whereas individual choice may not and should not follow statistical maximum. Unless there is long period of natural recovery, say longer than 6 months, the timing is better set from termination of natural recovery instead of injury as most studies do (6–9,12).
Under the pressure of patient and media demands, some began to do cell therapy before natural recovery had ceased or even in the acute stage. They may have difficulty in convincing the scientific community that the recovery is not a natural process but the benefit of the therapy. However, once the therapy has proven effective, then it can be used at any stage provided it is safe in individual patient.
Previous Study Results
One report recorded impairment of sensation after OEGT but we did not observe any deterioration associated with the procedure (12).
Other important signs of recovery observed included moderate to excessive sweating hours after OEGT with lasting increased warmth of skin, improved micturition, reflex penile erection, and change of pain. Unfortunately, there are no established or universally recognized outcome measures for these changes and hence they could not be recorded objectively, graphically, or quantitatively, nor scientifically reported. It is by no means to say that they do not exist and are less important than somatic functions. On the contrary, they are more important for life and health. The improved micturition and excessive sweating could be extremely important for sufferers of SCI in the developing world because they are the most common causes of death. Intensive and extensive efforts and resources must be put in as soon as possible to establish a series of standard outcome measures of these more important and realistic signs of recovery.
As to whether the moderate positive outcome was due to axonal regeneration or neural plasticity, it is impossible to tell in living humans. Only when advanced imaging technology can offer the opportunity to visualize minute details like nerve fibers can we give the answer. So far only changes of major structures like nuclei can be visualized in living humans but the technology is advancing in this direction quite rapidly and successfully. It may be possible in a few decades that new technology can help scientists to identify growing new nerve fibers in a living body. At the current level of science, judging from the early appearance of sweating within hours after the procedure, functional recovery based on neural plasticity seems more likely than structural regeneration.
In summary, it can be said that OEGT is safe. Sensation and spasticity improved moderately whereas somatomotor recovery was minimal. Much more efforts are needed in developing scientific outcome measures for RF functions that are essential for life and health.
Footnotes
Acknowledgment
The authors declare no conflicts of interest.
