Abstract
Heart failure continues to be a major health care concern with relatively few options for severely advanced heart failure patients. The hallmark of heart failure is the progressive dilatation of the left ventricle, thinning of the left ventricular wall leading to increased wall stress and increased myocardial oxygen consumption. Applying Laplace's law to the failing dilated ventricle, left ventricular augmentation utilizes a tissue engineering strategy to increase wall thickness and reduce chamber diameter, resulting in a decrease in wall stress and improved left ventricular function. A review of the rationale for an in situ tissue engineering approach for this treatment of heart failure and early clinical results of the Algisyl-LVR™ program are presented.
Keywords
Introduction
Heart failure (HF) is a leading cause of morbidity and mortality with a 5-year mortality rate of 50%. There are approximately 6 million people in the United States diagnosed with congestive HF, with approximately 550,000 new cases of HF occurring annually (13,16). The incidence of advanced HF continues to increase despite considerable progress in the medical management of patients, which has occurred over the past two to three decades, and the more recent development of devices for the treatment of HF (1). For end-stage heart failure, heart transplantation or mechanical assist devices are the only viable options (7,11). The opportunity for heart transplantation is limited due to the scarcity of donor organs and the number of potential transplant recipients. Therefore, the search for new therapies is being investigated. Recently, the emerging fields of tissue engineering and biomaterials have begun to provide potential treatments (4).
An in situ tissue engineering approach has recently been advocated for the treatment of the failing myocardium. It was proposed that biopolymers could alter the material properties of the ventricle and prevent the adverse remodeling associated with a myocardial infarction (3). Biopolymers have also been shown to act as a scaffold and improve cell retention, thus acting as a platform for myocardial regeneration (5). More recently, the injection of biopolymers into a chronic aneurismal ventricle was shown to be able to reshape and restore left ventricle (LV) geometry, resulting in increased LV function (22). It has been suggested that the injection of biopolymers into the left ventricular wall of a failing heart will reduce myocardial wall stress, leading to improved myocardial function (6,19,20,22).
Regardless of the etiology, heart failure is a progressive disorder whereby the failing LV undergoes progressive dilation and wall thinning and progressive contractile dysfunction. Consistent with the Law of Laplace (Fig. 1), LV dilatation and wall thinning leads to increase wall stress and consequently increased myocardial oxygen consumption (MVO2) due to enhanced energy demands. The increased MVO2 can lead to the death of cardiomyocytes, leading to a vicious cycle of the deleterious molecular, cellular, and physiological responses that lead to LV dilation. Additionally, the maladaptive LV shape and size changes impair the ability of the weakened cardiomyocytes to shorten leading to continued deterioration of global LV function.

Law of Laplace. The Law of Laplace states that wall stress is directly related to the diameter and wall thickness of the left ventricle (LV) (left). With the development of heart failure, the LV diameter increases and the LV wall thickness decreases, leading to increased LV wall stress (right).
Based on the concepts of the Law of Laplace and the initial studies suggesting that intramyocardial biopolymer implants could increase wall thickness and improve LV function (3,22), a strategy was developed for LV wall augmentation and restoration of LV geometry, thus resulting in increased LV function (Fig. 2). Unlike previous surgical therapies that focused simply on modifying the overall size of the ventricle, LV augmentation with biopolymers attempts to decrease wall stress and improve regional and global LV function.

Restoration of the dilated ventricle. Applying Laplace's law to the failing dilated ventricle, LV augmentation utilizes a tissue engineering strategy by directly altering wall thickness (T) and chamber diameter (D). As wall thickness increases and LV diameter is decreased, wall stress (S) is reduced. Progressive remodeling may be stabilized or reversed.
Results presented by Sabbah et al. at the American Heart Association (AHA) Scientific Sessions in 2009 (17) demonstrated that Algisyl-LVR treatment was able to augment the LV wall in advanced heart failure dogs as compared to saline control treated dogs. Assessments of LV volumes and function pretreatment and at 17 weeks posttreatment were determined by ventriculography and by assessment of LV pressure–volume relationships. Systolic and diastolic volumes were significantly smaller, and the LV ejection fraction was higher in the Algisyl-LVR-treated group as compared to control. There was also significant improvement in both diastolic and systolic LV function as measured by press–volume (P-V) loop, end-diastolic pressure–volume relationship (EDPVR), and end-systolic pressure–volume relationship (ESPVR) values in the Algisyl-LVR-treated dogs. The Algisyl-LVR implants were found to be safe and nonarrhythmogenic in this chronic study. In a separate acute study presented by Ilsar et al. at the Heart Failure Society of America (HFSA) Meeting in 2010 (10), these efficacy trends were seen immediately, with the earliest measurement of LV function being 1 h after injection of the Algisyl-LVR.
The Algisyl-LVR program was initiated to transition the preclinical proof of concept studies into clinical trials with the objective to treat dilated cardiomyopathy by applying 10–15 Algisyl-LVR injections into the LV free wall in a circumferential pattern to thicken the LV wall and reshape the geometry of the LV with the hope of improving LV function in HF patients.
Materials and Methods
Clinical Study Design
The initial first-in-man safety and feasibility evaluation was initiated at the German Heart Center, Munich, Germany, by Dr. Robert Bauernschmitt and his colleagues in 2009 (clinicaltrials.gov identifier NCT00847964). This study was expanded based on the initial encouraging results and now includes the Dresden Heart Center, the German Heart Center Berlin, and the Sana Herzchirurgie Stuttgart.
The study is a pilot study to evaluate the safety and feasibility of Algisyl-LVR as a method of left ventricular restoration in patients with dilated cardiomyopathy undergoing open-heart surgery. The study is an open label, uncontrolled study to evaluate the safety of Algisyl-LVR, assess suitability of device design, and assess the feasibility of the procedure. The study is planned as an 18-patient feasibility study.
Patient eligibility includes (1) patients (aged 18–75 years) with dilated cardiomyopathy scheduled to undergo a coronary artery bypass graft (CABG) or a valve procedure; (2) New York Heart Association (NYHA) class of III or IV on stable, evidence-based therapy for HF; (3) ejection fraction (EF)≤40% with Left Ventricular End Diastolic Diameter index (LVEDDi) of 30–40 mm/m2 [LVEDD/body surface area (BSA)] as assessed via echocardiogram.
Patients will be treated with Algisyl-LVR during a planned CABG and/or valve replacement/repair procedure. Median sternotomy or thoracotomy is performed to expose the heart and the pericardium for the open-heart procedure. During the open-heart procedure, the left ventricular free wall is identified and Algisyl-LVR is delivered into the myocardium as 10–15 intramyocardial injections made under direct visualization at the LV midventricular level halfway between the LV apex and base starting at the anteroseptal groove and ending at the posteroseptal groove. Standardization of the placement of the implants is performed via the identification of standard anatomical features [atrioventricular (AV) groove and apex]. The injections of approximately 0.25–0.35 ml are made at the midwall depth of the myocardium.
Patients will undergo assessment of LV size and function, functional/physical status assessed via Kansas City Cardiomyopathy Questionnaire (KCCQ) and NYHA assessment. Safety is assessed via adverse events, blood chemistry, electrocardiogram (ECG), and Holter Monitor. Evaluations are performed prior to the procedure and then postprocedure at 3 days, 8 days, 3 months, 6 months, 12 months, 18 months, and 24 months.
Biopolymer Preparation
The two-component proprietary formulation of the self-gelling alginate hydrogel (Algisyl-LVR) is composed of a sterile aqueous solution of Na+-alginate at 2% w/v in 4.6% mannitol and a Ca2+-alginate component consisting of water-insoluble particles suspended in a sterile 4.6% mannitol solution. The two components are mixed by transferring the solutions back and forth between two syringes. The alginate hydrogel has a gelling halftime of 3–4 min and achieves a material strength of 3–5 kPa.
Results
Clinical Observations
The initial observations from this clinical study were presented by Dr. Bauernschmitt's group at the 2010 AHA Scientific Sessions. Six patients with ischemic (n = 4) and nonischemic (n = 2) dilated cardiomyopathy, NYHA class III or I V, received left ventricular restoration with 10–15 implants (injections) of Algisyl-LVR concomitant with either coronary artery bypass or valve surgery. The implants were placed into the LV wall along the circumference of the LV free wall halfway between the apex and base. Perioperative complications were minimal and typical for cardiac surgery. There were no implant-related complications. Improvements in LV size and function were observed as early as 3 days following surgery for all patients (Table 1). At the time of this report, all six patients had completed 3 months follow-up and demonstrated sustained improvements in LV size and function that were accompanied by statistically significant improvements in clinical status and quality of life (NYHA functional class and KCCQ score). All six patients enrolled in the study are alive, and none required an additional major cardiac procedure [no procedures for coronary revascularization, valve replacement, left ventricular assist device (LVAD), or transplant].
First-in-Man Experience With Left Ventricular Restoration in Patients With Systolic Heart Failure
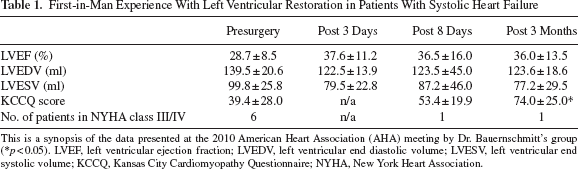
This is a synopsis of the data presented at the 2010 American Heart Association (AHA) meeting by Dr. Bauernschmitt's group (∗p < 0.05). LVEF, left ventricular ejection fraction; LVEDV, left ventricular end diastolic volume; LVESV, left ventricular end systolic volume; KCCQ, Kansas City Cardiomyopathy Questionnaire; NYHA, New York Heart Association.
Discussion
The study indicates a satisfactory tolerability and safety profile for this novel approach for LV augmentation and restoration of LV geometry. Algisyl-LVR treatment is feasible and can be safely performed in patients with advanced HF.
Although definitive conclusions are difficult to ascertain with an uncontrolled, nonrandomized concomitant study, the initial results of reduced LV size and improved LV function are encouraging. Additionally, preclinical and early clinical observations show no evidence that this approach impairs diastolic function. Unlike other cell-based, pharmacological, or passive mechanical-restraint device treatments for heart failure, the response to the Algisyl-LVR treatment had immediate affects in reduction of LV volume and improvement of LV function. These acute beneficial effects were sustained at the 3-month follow-up period and correlated with clinical improvement of heart failure symptoms. The mechanism of LV augmentation resulting in improved LV function most certainly includes an improvement in contractility due to reduced wall stress, which would result in a decrease in MVO2, better functioning myocytes leading to improved contractility; at least regionally if not globally.
In order to better define the effects of Algisyl-LVR therapy, the AUGMENT-HF study, a randomized, controlled study to evaluate Algisyl-LVR as a method of left ventricular augmentation for in patients with “severe heart failure” who have a dilated left ventricle and who are highly symptomatic (clinicaltrials.gov identifier NCT 01311791) is currently being initiated. The planned 76-patient randomized trial of 1:1 active (Algisyl-LVR implants) to control (standard medical therapy) study will be conducted at 10–12 centers in Europe.
The demonstration that biopolymers can be used to augment the LV wall and restore LV geometry also provides the basis for an in situ tissue engineering approach for myocardial regeneration. Several laboratories have demonstrated the utility of biopolymer scaffolds for the controlled delivery of angiogenic factors (15,21), cell transplantation (2,4,12,14,18), and modulation of transplanted stem cells (8,9). Altering the biological properties of the polymer by the addition of an integrin binding peptide can increase cell migration, proliferation, matrix deposition, and stimulate neovascularization (23) highlighting the possibility of developing biopolymers that can induce one's own body to act as a bioreactor and drive myocardial regeneration.
Footnotes
Acknowledgments
The study was partially supported by LoneStar Heart, Inc. R. J. Lee and H. N. Sabbah are consultants to LoneStar Heart, Inc. The authors declare no confict of interest.
